ISSN: 0973-7510
E-ISSN: 2581-690X
Natural compounds derived from higher plants can serve as new sources of antibacterial drugs with potentially novel mechanisms of action. Traditional medicine uses plant extracts to treat a variety of infectious disorders, including those caused by bacteria and fungi. Currently, there is a great demand for plant extracts with significant antibacterial activity. In this study, extracts from four plants—Lantana camara, Withania somnifera, Cetrariais landica, and Tribulus terrestris—were subjected to phytochemical screening, and in vitro antibacterial activity was evaluated. Phytochemical investigations revealed the presence of saponins, alkaloids, phenolics, flavonoids, carbohydrates, proteins, amino acids, glycosides, tannins, and terpenoids. The antimicrobial activities of the plant extracts were assessed using the disc diffusion method. They exhibited varied antimicrobial activities against gram-negative bacteria (E. coli and P. aeruginosa), gram-positive bacteria (B. subtilis and S. aureus), and fungi (C. albicans, A. niger, F. oxisporium, and F. solani). Lantana camara extract showed the highest phytochemical content and antimicrobial activity. These findings can be used in the pharmaceutical and alternative medicine industries to create natural bioactive compounds that are beneficial to human health.
Antimicrobial, Plant Extracts, Lantana camara, Tabuk
Antimicrobial resistance is a global problem that affects human health and economic development. Therefore, the discovery of novel antimicrobial agents is necessary. Antibiotics, which are secondary metabolites produced by a variety of bacteria, actinomycetes, and fungi, have impressive antibacterial properties; however, they also have severe side effects in the human body and unavoidably lead to resistance.1 Thus, extensive research has been conducted to find compounds with significant antibacterial activity to reduce the risk of infectious diseases caused by pathogenic bacteria, fungi, viruses, and parasites in humans.2 Moreover, efforts have been focused on developing potentially effective natural products.
Approximately 50,000 plant species have been evaluated for their medicinal properties and are used to cure a variety of human diseases by 80% of the world’s population.3 Plants possess a set of effective defense mechanisms, specifically secondary metabolite synthesis, that allow them to resist pests and pathogens before they cause major damage.4 Plant extracts continue to be a major source of medicinal compounds, particularly antimicrobial medicines, for the treatment of infectious illnesses.2 According to numerous studies, medicinal plants contain many phytochemical compounds, such as coumarins, flavonoids, phenolics, alkaloids, terpenoids, tannins, essential oils, lectins, polypeptides, and polyacetylenes. The composition of these biologically active compounds varies based on the plant species, soil type, and microbial association. These bioactive substances may have bacteriostatic or bactericidal effects against pathogenic bacteria with multidrug resistance (MDR).5 Antimicrobials and other drugs produced from plants are becoming increasingly amenable to mainstream medicine because traditional antibiotics (microorganisms or their synthetic derivatives) become ineffective and new diseases, particularly viral diseases, remain untreatable by this type of drug. The fast rate of (plant) species extinction has been another motivating factor for the new emphasis on plant antimicrobials in recent decades.6
Lantana camara is a tropical American evergreen shrub (Verbenaceae) with a low-erect rugged hairy appearance. It is a prominent weed with over 650 variants in over 60 countries and island groups. It is used for a variety of purposes, including herbal medicine, firewood, and mulch. It is prescribed for conditions such as cancer, chicken pox, measles, asthma, ulcers, swelling, eczema, tumors, high blood pressure, bilious fever, catarrhal infections, tetanus, rheumatism, malaria, and atoxy of the abdominal viscera.7 L. camara leaf extracts have antimicrobial, insecticidal, and nematocidal properties, and contain verbascoside, which has antimicrobial, immunosuppressive, and anticancer properties.8 Tribulus terrestris L. belongs to the Zygophyllaceae family and is found in warm climates worldwide. It is important in folk medicine because it is used as a tonic, astringent, antihypertensive, aphrodisiac, analgesic, diuretic, stomachic, and urinary anti-infective.9 Withania somnifera (L.) Dunal, often known as “Ashwaganda”, is a member of the Solanaceae family and is widely utilized in Ayurvedic medicine. It is used as a general tonic to boost energy, promote overall health and lifespan, and prevent disease in athletes and the elderly.10 Recently, several studies have investigated the antimicrobial activity of W. somnifera against various bacterial pathogens.11,-14 Cetraria isandica (Iceland Moss) is a fruticose or shrub-like plant with a bushy appearance, and is an effective antibiotic and expectorant. It calms inflamed tissues, particularly mucous membranes, and is frequently used in cough medications.15 Recently, Iceland lichen has been used to treat tuberculosis and other illnesses, and many other lichen extracts have been shown to kill gram-positive bacteria.16
The purpose of this study was to evaluate the antimicrobial potential of extracts of L. camara, W. somnifera, C. islandica, and T. terrestris against bacterial and fungal pathogens.
Plant collection
Different parts of L. camara, W. somnifera (leaves and flowers), and T. terrestris (shoot system) were collected from Tabuk Gardens (Tabuk, Saudi Arabia), whereas C. islandica (whole plant)was purchased from an herbal store at Tabuk. Plants were dried in the shade to inhibit degradation of any bioactive components, ground into a fine powder, and stored for further use (Figure 1).
Figure 1. Plants used in this study. (a) Lantana camara, (b) Withania somnifera, (c) Cetraria islandica, (d) Tribulus terrestris
Preparation of plant extract
Aqueous extraction
Fine shoot powder was dissolved in 10% (w/v)sterile distilled water separately in an Erlenmeyer flask to prepare the aqueous extract. The flasks were placed on an orbital shaker for 24 h to allow extraction and then evaporated using a rotary evaporator at 60°C.17 The final dried samples were stored in labeled sterile bottles and kept at 4°C.
Ethanol extraction
Plants were dried for 2 weeks at room temperature, pulverized to a powder, and passed through a No. 40 sieve. The plant powder (100 g) was weighed, transferred to a round-bottom flask, and treated with 95% ethanol using a Soxhlet apparatus. The process lasted 24 h and was maintained at 45–47°C. The extract was then collected and evaporated using a vacuum distillation unit and stored at”20°C.
Phytochemical screening
Qualitative phytochemical screening of T. terrestris, W. somnifera, C. islandica, and L. camara was performed to determine the presence of biologically active compounds or secondary metabolites, including saponins, alkaloids, phenolics, flavonoids, carbohydrates, proteins, amino acids, glycosides, tannins, and terpenoids. The ethanolic extract was subjected to phytochemical screening and gas chromatography-mass spectrometry (GC-MS) analysis.18 Saponins, alkaloids, phenols, and flavonoids were also quantitatively determined.19-21
Microorganisms
Microbial cultures were obtained from the American Type Culture Collection (ATCC, Rockville, MD, USA) and Northern Utilization Research and Development Division, United States Department of Agriculture, Peoria, Illinois, USA (NRRL). The gram-positive bacteria Bacillus subtilis (ATCC 6633) and Staphylococcus aureus (ATCC 6538), and gram-negative bacteria Escherichia coli (ATCC 25922(and Pseudomonas aeruginosa (ATCC 27853) were grown in fresh nutrient broth medium (HiMedia, India) at 37°C for 24 h before the test. Fungi Aspergillus niger NRRL-3, Fusarium oxisporium, Fusarium solani, and Candida albicans (ATCC102) were cultured on potato dextrose agar (PDA; HiMedia, India) for 7 d at 28°C before the experiment.
Antimicrobial assay
The in vitro antimicrobial activity of plant extracts was assessed using an agar disc diffusion technique,22 and each extract was sterilized with a 0.22 ìm bacterial filter before use. A 0.1 mL aliquot of 18 h broth culture of the above-mentioned bacteria that had been adjusted to a turbidity equivalent of 0.5 McFarland standards,23 was dispensed into sterile Petri dishes. Muller-Hinton agar (MHA; Lab M Limited, Bury, Lancashire, UK) was aseptically poured into the plates and gently rotated to ensure a homogeneous distribution of bacteria in the medium. The agar plates were allowed to solidify, then sterile blank antimicrobial susceptibility disks (6 mm) were placed on the agar plates, loaded with 30°L of sterile plant extracts, and allowed to diffuse into agar for 1 h at 4°C. All plates were incubated at 37°C for 24 h for bacterial strains and at 28–30°C for 48 h for fungal strains. The activity was determined by measuring the diameter of the inhibition zone (mm). Antibiotic disks standard Imipenem (10 µg) and Clotrimazole (50 µg) (Bioanalyse, Turkey) were used as positive controls for bacteria and fungi, respectively. The antimicrobial activity was reported from two independent assays.
Determination of minimum inhibitory concentrations (MICs)
Minimum inhibitory concentrations (MICs) were determined for L.camara extracts, which revealed the best antibacterial activity according to the Clinical and Laboratory Standards Institute.24 Sequential dilutions were prepared at concentrations ranging from 200–1000 (1–5), and dimethylsulfoxide (DMSO) was used as the negative control. Each concentration prepared in tubes was applied in MHA plates that were inoculated with 100 µL bacterial inoculum adjusted to a concentration of 106 CFU/mL and spore suspension from fungal strains adjusted to a final concentration of 106 spores/mL. The assay was performed either by agar well diffusion for aqueous solutions or by the disc method for solvent samples. The plates were incubated aerobically at 37°C (18–24 h) for bacterial strains, and 25°C (48 h) for fungal strains. MIC values were defined as the lowest concentration of an antimicrobial agent that inhibited the growth of a microorganism. The experiment was conducted using a triplicate.
Phytochemical screening
The results of the qualitative and quantitative phytochemical analyses of L. camara, W. somnifera, C. islandica, and T. terrestris extracts are presented in Table 1. These bioactive compounds occur naturally and possess bactericidal and fungicidal properties. The qualitative phytochemical analysis of the L. camara plant exhibited the highest tannins, coumarins, and phenols in aqueous extraction. However, T. terrestris had the lowest phytochemical content in both aqueous and ethanol extracts.
Table (1):
Qualitative phytochemical analysis of aqueous and ethanolic extracts for the studied plant species.
| Test | T. terrestris | W. somnifera | C. islandica | L. camara | ||||
|---|---|---|---|---|---|---|---|---|
| AquExt | EthaExt | AquExt | EthaExt | AquExt | EthaExt | AquExt | EthaExt | |
| Tannins | – | + | – | + | – | + | +++ | +++ |
| Saponins | – | + | + | ++ | ++ | + | + | + |
| Flavonoids | + | + | + | + | – | + | ++ | + |
| Terpenoids | – | – | – | + | + | ++ | + | ++ |
| Glycosides | – | + | + | + | + | + | + | + |
| Alkaloids | – | – | – | + | ++ | + | ++ | ++ |
| Coumarins | + | + | ++ | ++ | ++ | ++ | +++ | ++ |
| Anthraquinones | + | – | + | – | – | + | + | ++ |
| Phenols | – | + | + | ++ | ++ | ++ | +++ | ++ |
(+++) Appreciable amount; (++) Moderate amount; (+) Trace amount; (-) Not detected.
Quantitative phytochemical analysis of the ethanolic extracts of the studied plant species (Table 2) revealed that the L. camara extract had the highest total phenol (263.57 mg GAE/g), total flavonoid (143.89 mg QE/g), and total alkaloid contents (2.80%).
Table (2):
Quantitative phytochemical analysis of ethanolic extracts for the studied plant species.
Test |
Unit |
T. terrestris |
W. somnifera |
C. islandica |
L. camara |
|---|---|---|---|---|---|
Total phenol content |
mg GAE/g |
44.44 |
62.37 |
34.21 |
263.57 |
Total flavonoid content |
mg QE/g |
71.33 |
67.10 |
83.40 |
143.89 |
Total alkaloid content |
% |
1.17 |
1.21 |
1.10 |
2.80 |
Antibacterial activity of plant extracts
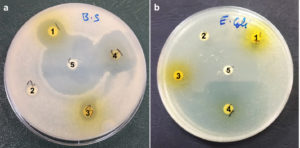
The disc diffusion method was used to qualitatively evaluate the antibacterial activity of the plant extracts against gram-negative bacteria (E. coli and P. aeruginosa) and gram-positive bacteria (B. subtilis and S. aureus). The plant extracts exhibited varying degrees of inhibition against the tested pathogenic bacteria (Figure 2; Table 3). These results revealed that the four extracts were effective against the tested pathogenic bacteria. L. camara ethanolic extract showed high activity against S. aureus (30.0 ± 0.04 mm) and B. subtilis (24.9 ± 0.02 mm). Moderate and lower activities were observed against both P. aeruginosa (19.5 ± 0.01 mm) and E. coli (10.9 ±0.01 mm), whereas their aqueous extracts showed moderate activity against P. aeruginosa (19.5 ± 0.0 mm) and E. coli. W. somnifera ethanolic extract showed high activity against S. aureus (30.0 ± 0.07 mm) and moderate effects against P. aeruginosa (13.5 ± 0.07 mm). However, it showed less activity against B. subtilis (10.9 ± 0.03 mm), and its aqueous extract showed less activity against E. coli (15.4 ± 0.01 mm) and P. aeruginosa(12.0 ± 0.03 mm). Both aqueous and ethanolic extracts of C. islandica were observed to have less activity against E. coli (11.9 ± 0.06, 10.0 ± 0.03, and 9.9 ± 0.07 mm). T. terrestris ethanolic extract exhibited moderate effects against B. subtilis (17.92 ± 0.07 mm) and its aqueous extract against E. coli (15.5 ± 0.02) mm.
Table (3):
Antibacterial assay of plant extracts against bacterial pathogens.
| Plant Extracts/Antibiotics | Diameter of inhibition zone (mm)* | |||
|---|---|---|---|---|
| Gram-positive | Gram-negative | |||
| B. subtilis | S. aureus | E. coli | P. aeruginosa | |
| T. terretris Ethanolic extract Aqueous extract |
17.9 ± 0.07 – |
– – |
– 15.5 ± 0.02 |
– – |
| C. islandica Ethanolic extract Aqueous extract |
9.9 ± 0.07 – |
– – |
10.0 ± 0.03 11.9 ± 0.06 |
– – |
| W. somnifera Ethanolic extract Aqueous extract |
10.9 ± 0.03 – |
30.0 ± 0.07 – |
– 15.4 ± 0.01 |
13.5 ± 0.07 12.0 ± 0.03 |
| L. camara Ethanolic extract Aqueous extract |
24.9 ± 0.02 – |
30.0 ± 0.04 – |
10.9 ± 0.01 12.7 ± 0.01 |
19.5 ± 0.01 19.5 ± 0.0 |
| Imipenem (10 μg) | 40.0 ± 0.0 | 25.0 ± 0.0 | 40.0 ± 0.0 | 20.0 ± 0.0 |
*The values represent mean of the inhibition zone ± standard deviation (SD); (-) No.
Inhibition. Antibiotic Imipenem (10 μg) was used as a positive control.
Figure 2. Antibacterial activity of (1) T. terrestris, (2) C. islandica, (3) C. islandica, (4) L. camara ethanolic extract, and (5) Imipenem (10 µg) as a positive control. Visible clear zones (mm) were observed against pathogenic bacteria B. subtilis and E. coli (a) &(b), respectively
Antifungal activity of plant extracts
The antifungal activities of both the aqueous and ethanolic plant extracts were evaluated against sporogenous A.niger, C. albicans, F. oxisporium, and F. solani. The aqueous extract of L. camara revealed the highest activity against unicellular C. albicans (23.5 ± 0.06 mm) compared to the other extracts. Generally, C. albicans was the fungal strain most affected by plant extract treatment (Table 4). On the other hand, the extracts exhibited a moderate activity against filamentous A. niger, W. somnifera ethanolic and aqueous extracts (19.9 ± 0.06 mm), and T. terrestris ethanolic extract (19.9 ± 0.04 mm). The aqueous extract revealed an inhibition zone of 15.2 ± 0.02 mm. C. islandica aqueous extract showed an activity of 19.9 ± 0.03 mm, while the ethanolic extract showed an inhibition zone of 14.8 ± 0.08 mm. The inhibition zone diameter of L. camara aqueous extract was 14.7 ± 0.1 mm, and that of the ethanolic extract was 11.9 ± 0.3 mm. Plant extracts were less active against F. oxisporium. The ethanolic extracts of W. somnifera and L. camara revealed inhibition zones of 12 ± 0.3 mm and 9.0 ± 0.3 mm, respectively. Additionally, less activity was observed against F. solani, with W. somnifera and L. camara ethanolic extracts showing inhibition zones of 11 ± 0.7 mm and 8.5 ± 0.2 mm, respectively.
Table (4):
Antifungal assay of plant extracts against selected fungi.
| Plant Extracts/Antibiotics | Diameter of inhibition zone (mm)* | |||
|---|---|---|---|---|
| C. albicans | A. niger | F. oxisporium | F. solani | |
| T. terrestris Ethanolic extract Aqueous extract |
14.9 ± 0.3 15.1 ± 0.5 |
19.9 ± 0.04 |
– – |
– – |
| C. islandica Ethanolic extract Aqueous extract |
14.9 ± 0.05 14.9 ± 0.03 |
14.8 ± 0.08 |
– – |
– – |
| W. somnifera Ethanolic extract Aqueous extract |
19.9 ± 0.3 15.9 ± 0.7 |
19.9 ± 0.06 |
12.0 ± 0.3 – |
11.0 ± 0.7 – |
| L. camara Ethanolic extract Aqueous extract |
19.9 ± 0.03 |
11.9 ± 0.3 |
9.0 ± 0.3 |
8.5 ± 0.2 |
| Clotrimazole (50 μg) | 20.0 ± 0.0 | 20.0 ± 0.0 | 30.0 ± 0.0 | 25.0 ± 0.0 |
*The values represent mean of the inhibition zone ± standard deviation (SD); (-) No. Inhibition. Antibiotic Clotrimazole (50 μg) was used as a positive control.
Minimum inhibitory concentrations (MICs)
The maximum antimicrobial activities obtained from L. camara against B. subtilis and C. albicans were used to investigate the MIC values against each strain (Figure 3). The MIC results showed that B. subtilis was highly susceptible to the minimum inhibitory concentration of L. camara ethanolic extract of 12.5 mg/mL, and C. albicans was also susceptible to the minimum inhibitory concentration of L. camara aqueous extract (25 mg/mL). Thus, L. camara is a promising antimicrobial and curative plant extract.
Antibiotic resistance is a severe socio-economic obstacle affecting both developed and developing nations.25 Poverty, poor sanitation, easily available antibiotics, and clinical malpractice are factors that aid the spread of multidrug resistant (MDR) microbial strains.26 Alternatives to traditional antibiotics are highly desirable because of the shortage of new antibiotics that could potentially compensate for the increase in resistance to current antibiotics.27 Medicinal plant extracts are affordable antimicrobials considered a potential source of drugs for preventing and curing human diseases, especially in developing countries. Consequently, the search for secondary metabolites with antimicrobial activities in medicinal plants is highly recommended.28
In this study, Lantana camara, Withania somnifera, Cetrariais landica, and Tribulus terrestris plant extracts showed potential antimicrobial activity against various clinically relevant gram-negative and gram-positive bacterial species and fungi. Moreover, L. camara extract had the best antibacterial activity compared to the other plant extracts used in the current study. The ethanolic extract exhibited high activity against B. subtilis and S. aureus. This finding agrees with that of Barre et al., who used the extract of L. camara to inhibit the growth of pathogen S. aureus owing to the presence of active phytocompounds possessing bactericidal effects. Gram-positive bacteria were more sensitive to L. camara ethanolic extract than gram-negative bacteria (E. coli and P. aeruginosa). Unlike the results of this study on E. coli, many reports have shown promising results, as medicinal plants are rich in various bioactive compounds that potentially inhibit the growth of bacterial human pathogens.29
The aqueous extracts of L. camara exhibited antifungal activity against A. niger and C. albicans, which agreed with several reports.30 F. solani and F. oxisporium revealed tolerance to T. terrestris and C. islandica extracts. Phytochemical analysis of L. camara includes several iridoid glycosides, furano naphthoquinones, flavonoids (3-methoxyquercetin, 3,7 dimethoxyquercetin, and 3,7,49-trimethoxyquercetin), a flavone glycoside and camaraside, and three new pentacyclic triterpenoids, camarin, lantacin, and camarinin.
This study showed that ethanolic and aqueous extracts of Lantana camara, Withania somnifera, Cetrariais landica, and Tribulus terrestris have the potential to inhibit the growth of various gram-negative and gram-positive bacterial species, and fungi. The above-mentioned findings could be a potential topic for further in vivo studies to develop therapeutic plant extracts from Lantana camara and avoid the health hazards and antimicrobial resistance associated with the use of chemically synthesized antimicrobial agents.
ACKNOWLEDGMENTS
None.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
Not applicable.
- Yuan G, Guan Y, Yi H, Lai S, Sun Y, Cao S. Antibacterial activity and mechanism of plant flavonoids to gram-positive bacteria predicted from their lipophilicities. Sci Rep. 2021;11(1):10471.
Crossref - Mohamed EAA, MuddathirAM, Osman MA. Antimicrobial activity, phytochemical screening of crude extracts, and essential oils constituents of two Pulicaria spp. growing in Sudan. Sci Rep. 2020;10(1):17148.
Crossref - Egamberdieva D, Wirth S, Behrendt U, Ahmad P, Berg G. Antimicrobial Activity of Medicinal Plants Correlates with the Proportion of Antagonistic Endophytes. Front Microbiol. 2017;8:199.
Crossref - Chassagne F, Samarakoon T, Porras G, et al. A Systematic Review of Plants with Antibacterial Activities: A Taxonomic and Phylogenetic Perspective. Front Pharmacol. 2021;11:586548.
Crossref - Gadisa E, Tadesse E. Antimicrobial activity of medicinal plants used for urinary tract infections in pastoralist community in Ethiopia. BMC Complement Med Ther. 2021;21(1):74.
Crossref - Lewis WH and Elvin-lewis MP. Medicinal Plants as Sources of New Therapeutics. JSTOR. 1995;82(1):16-24.
Crossref - Epps J. Preparation of Extracts. Lancet. 1833;20(513):423-424.
Crossref - Adiguzel A, Gulluce M, Utcu HO, Fikrettin N. Antimicrobial Effects of Ocimum basilicum (Labiatae) Extract. Turkish J Biol. 2005;29(3):155-160.
- Soleimanpour S, Sedighinia FS, SafipourAfshar A, Zarif R, Ghazvini K. Antibacterial activity of Tribuluster restris and its synergistic effect with Capsella bursa-pastoris and Glycyrrhi zaglabra against oral pathogens: an in-vitro study. Avicenna J Phytomed. 2015;5(3):210-217.
- Bisht P, Rawat V. Antibacterial activity of Withania somnifera against Gram-positive isolates from pus samples. Ayu. 2014;35(3):330-332.
Crossref - Mahesh B, Satish S. Antimicrobial Activity of Some Important Medicinal Plant Against Plant and Human Pathogens. World J Agric Sci. 2008;4:839-843. http://idosi.org/wjas/wjas4(s)/7.pdf
- Arora S, Dhillon S, Rani G, Nagpal A. The in vitro antibacterial/synergistic activities of Withania somnifera extracts. Fitoterapia. 2004;75(3-4):385-388.
Crossref - Owais M, Sharad KS, Shehbaz A, Saleemuddin M. Antibacterial efficacy of Withania somnifera (ashwagandha) an indigenous medicinal plant against experimental murine salmonellosis. Phytomedicine. 2005;12(3):229-235.
Crossref - Mathur V, Vats S, Jain M, Bhojak J, Kamal R. Antimicrobial Activity of Bioacive Metabolites Isolated from Selected Introduction. Asian Journal of Experimental Sciences. 2007;21(2):2-7.
- Grujieic D, Stosic I, Kosanic M, Stanojkovic T, Rankovic B, Milosevic-Djordjevic O. Evaluation of in vitro antioxidant, antimicrobial, genotoxic and anticancer activities of lichen Cetrariais landica. Cytotechnology. 2014;66(5):803-813.
Crossref - Kotan E, Alpsoy L, Anar M, Aslan A, Agar G. Protective role of methanol extract of Cetraria islandica (L.) against oxidative stress and genotoxic effects of AFB1 in human lymphocytes in vitro. ToxicolInd Health. 2011;27(7):599-605.
Crossref - Kandil O, Radwan NM, Hassan AB, Amer AM, el-Banna HA, Amer WM. Extracts and fractions of Thymus capitatus exhibit antimicrobial activities. J Ethnopharmacol. 1994;44(1):19-24.
Crossref - Ramya GL, Vasanth PM, Prasad PV, SarathBabu V. Qualitative Phytochemical Screening Tests of Alpinia. World J Pharm Res. 2019;8(5):1064-1077.
Crossref - Majinda RR. Extraction and isolation of saponins. Methods Mol Biol. 2012;864:415-26.
Crossref - Khalifa A, Ahmad H, Antar M, Laoui T, Khayet M. Experimental and theoretical investigations on water desalination using direct contact membrane distillation. Desalination. 2017;404:22-34.
Crossref - Hussain J, Khan FU, Ullah R, et al. Nutrient evaluation and elemental analysis of four selected medicinal plants of Khyber PakhtoonKhwa, Pakistan. Pak J Bot. 2011;43(1):427-434.
- Perez C, Paul M, Bazerque P. An Antibiotic assay by the agar well diffusion method. Acta Bio Med Exp. 1990;15:113-115.
- Lahuerta Zamora L, Perez-Gracia MT. Using digital photography to implement the McFarland method. J R Soc Interface. 2012;9(73):1892-7.
Crossref - CLSI. Performance Standards for Antimicrobial Susceptibility Testing; Twentieth Informational Supplement. CLSI document M100-S20; Wayne, PA: Clinical and Laboratory Standards Institute. 2010.
- Sirinavin S, Dowell SF. Antimicrobial resistance in countries with limited resources: unique challenges and limited alternatives. Semin Pediatr Infect Dis. 2004;15(2):94-98.
Crossref - Byarugaba DK. A view on antimicrobial resistance in developing countries and responsible risk factors. Int J Antimicrob Agents. 2004;24(2):105-110.
Crossref - Cooper MA, Shlaes D. Fix the antibiotics pipeline. Nature. 2011;472:32.
Crossref - Zuo GY, Wang GC, Zhao YB, et al. Screening of Chinese medicinal plants for inhibition against clinical isolates of methicillin-resistant Staphylococcus aureus (MRSA). J Ethnopharmacol. 2008;120(2):287-290.
Crossref - Valli M, Pivatto M, Danuello A, et al. Tropical biodiversity: Has it been a potential source of secondary metabolites useful for medicinal chemistry? Trop Biodivers. 2012;35(11):2278-87.
Crossref - Sule WF, Okonkwo IO, Omo-Ogun S, et al. Phytochemical properties and in-vitro antifungal activity of Senna alata Linn. crude stem bark extract. J Med Plants Res. 2011;5(2):176-183.
Crossref
© The Author(s) 2023. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.