ISSN: 0973-7510
E-ISSN: 2581-690X
To prepare and characterize silver nanoparticles from Guava leaves (Psidium guajava), extracts (chloroform and ethanol). The fabricated silver nanoparticles were tested on resistant fungal strains, Aspergillus niger and Candida glabrata. Psidium guajava leaf extracts were obtained by cold maceration method, by using Chloroform and Ethanol. To one mL of extract 2mM silver nitrate solution was added. The mixture was kept overnight in a dark chamber at room temperature. The solution was centrifuged at 10,000 rpm for 15 minutes and filtered by wattman filter paper after centrifugation. Psidium guajava leaf extract based silver nanoparticles were prepared and evaluated for anti-fungal activity by microtitre well plate method on resistant strains Aspergillus niger and Candida glabrata. The silver nanoparticles showed significant activity at 120 µL, Nystatin was used as standard.
Psidium guajava, Silver nanoparticles, Zeta potential, SEM analysis, Resistant strains, Antifungal activity
The preparation of silver nanoparticles (AgNPs) employing plant extracts is an evolving field since phytochemicals acts as reducing and capping agents and are free from harmful chemicals. Silver possess bactericidal activity. The present research work focused on fabrication of silver nanoparticles (AgNPs) by Guava (Psidium guajava) leaf extracts from green chemistry view and to assess the antifungal potency of fabricated AgNPs on resistant fungal strains
Candida species are harmful to humans and cause superficial as well as innate mycoses in humans and are distributed worldwide. Candida albicans and glabrata are prevalent fungi which cause Candidiasis. It is an opportunistic pathogen. It causes infections of urinogenital tract and Candidaemia, mostly seen in less immune patients3. It is highly resistant to antifungal agents. The opportunistic fungi are found in normal body flora and become pathogenic and intrusive in less immune individuals. Inspite of improvements in antifungal remedy, the last two to three decades have perceived major contaminations involving Candida albicans, Candida glabrata. Aspergillus niger produces mycotoxins which affects liver and kidney4. Aspergillus niger is a pathogenic allergen commonly linked with respiratory infections in individuals with frail immune system. Therefore, new compounds should be researched as an alternative to combat such resistance. Azole resistance is reported in many Aspergillus species5.
In immunocompromised individuals the infections due to bacteria and fungi may occur concurrently. The management for such infections is inadequate as anti-fungal agents show adverse effects, so the AgNPs may be an alternative antimicrobial agent.
Collection and authentication of plant part
Psidium guajava leaves were collected from medicinal garden in RIPER College, Ananthapuramu, India, authenticated with voucher no. 1834. The leaves were shade dried at room temperature for 14 days and blended into powder. The powder was kept in air tight container for further use.
Microorganism and culture conditions
Voriconazole resistant Aspergillus niger M046 (Clinical Isolate) and Fluconazole resistant Candida glabrata (ATCC200918) strains were used for present study. The organisms were maintained, grown, and sub cultured at 37°C on Peptone, Dextrose and Yeast extract. (PDA) plates 48 h prior to use in Biosafety level 1 laboratory.
Preparation of plant material
The powdered material was macerated in 200 mL of chloroform and ethanol, for four days with occasional stirring. The extract was filtered by using whatmann filter paper and dried under vacuum using rota-evaporator. The extracts were refrigerated and used further.
Chemicals and reagents
Silver nitrate (AgNO3) was bought from Sigma Aldrich. The other reagents and chemicals used were of analytical quality and procured from Sigma Aldrich, India. During the experiment double distilled water was used.
Phytochemical screening of plant extracts
Phytochemical screening of Psidium guajava leaf extracts of ethanol and chloroform was performed to investigate the phytochemicals present6,7.
Thin Layer Chromatographic (TLC) studies
The Rf values were found by one dimensional TLC using silica gel Gas immobile phase. The mobile phase used was n-hexane and ethyl acetate in ratio 7:3.
Biosynthesis of Silver nanoparticles (Ag NPs)
One ml of leaf extracts (chloroform and ethanol) was mixed with 10 ml of 2mM AgNO3 solution, in a 50 ml beaker. The preparation was kept in dark, overnight at room temperature. The change of chloroform light yellow to brown determines formation of AgNPs. The obtained nanoparticles were parted by centrifugation at 10000 rpm for 15 minutes8. The solution was filtered after centrifugation process by whatmann filter paper and kept in refrigerator for future use.
Characterization of nanoparticles
To characterise the AgNPs absorption studies were carried out on a UV-visible spectrophotometer9 (LAB INDIA, UV-3092) for well-dispersed extract nanoparticles solution in between the wavelength range of 200-800 nm. The Size distribution of the prepared nanoparticles and zeta potential was determined by Zetasizer (Horiba SZ-100). The detailed morphology of nanoparticles was established by Scanning electron microscopic (SEM) images.
UV-Visible spectroscopy
Reduction of silver nanoparticles during exposure to seed extract could be detected by the color change. A color change from light yellow to brown was observed when the seed extracts containing silver nitrate solution was kept for overnight. It may be due to reducing and capping ability of phytoconstituents of seed extract with aqueous AgNO3 solution. The Ag+ ions were reduced to Ago.
Particle size and Zeta potential
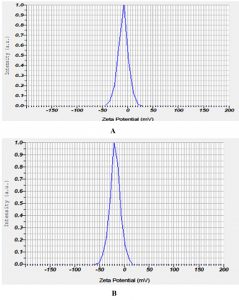
The size dispersal and poly-disperse property of the AgNPs in a suspension is determined by DLS10. The size and Zeta potential of the synthesised nanoparticles was determined by using Zetasizer (Horiba SZ-100 Ver 2.20). The size distribution of the AgNPs was measured by dynamic light scattering. Zeta potential values of nanoparticles in the range +30 mV or below –30 mV are considered electrostatically stable. The stabilization of nanoparticles is due to electrostatic interactions and steric hindrance provided by biomolecules.
Phytochemical Investigation
Phytochemical screening of the leaf extracts of Psidium guajava showed presence of phenols, tannins, terpenoids, flavonoids and glycosides as presented in (Table 1)
Table (1):
+ indicates presence of constituent (positive), – indicates absence of constituent (negative).
Extracts |
Phenols |
Tannins |
Terpenoids |
Flavonoids |
Glycosides |
|---|---|---|---|---|---|
Ethanol |
+ |
+ |
+ |
+ |
+ |
Chloroform |
+ |
+ |
+ |
+ |
+ |
Thin Layer Chromatographic (TLC) studies
The Rf values obtained from ethanol 0.10,0.14,0.35,0.45,0.60,0.91; and chloroform 0.14,0.37,0.58,0.89 serve as characteristic fingerprint of Psidium guajava leaf. This data would therefore be suitable for monitoring the identity and purity of the plant material as depicted in Fig. 1.
Table (2):
Zeta size and Zeta potential of Psidium guajava silver nanoparticles with Ethanol and Chloroform extract.
S. No |
Sample |
Zeta size(nm) |
Zeta potential(mv) |
|---|---|---|---|
1. |
GES |
158 |
-8.9 |
2. |
GCS |
195 |
-18.8 |
Fig. 1. TLC profile of the ethanol and chloroform extract of P. guajava leaf. Solvent system: Hexane : Ethyl acetate (7: 3).
UV-Visible spectroscopy
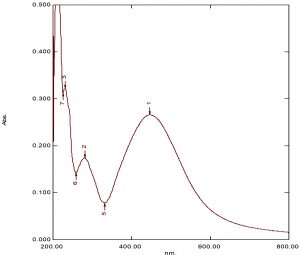
The fabricated silver nanoparticles revealed superior plasmon resonance absorbance at 432 nm, as shown in (Fig. 2).
Particle size and Zeta potential
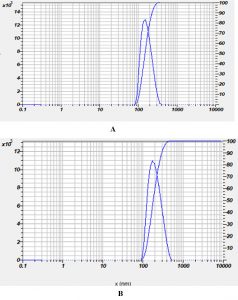
The AgNPs prepared from Nigella sativa chloroform extract, DLS analysis showed nanoparticles with an average diameter of 158nm, with a Polydispersity Index (PdI) of 0.309 for guava AgNPs (ethanol extract). The mean diameter 190 nm with a Polydispersity Index of 0.321 for guava AgNPs (chloroform extract) as shown in (Table 1) and (Fig. 5). The zeta potential for AgNPs prepared from Psidium guajava chloroform extract was -18.8 mV. In the case of AgNPs prepared from Psidium guajava ethanol extract, the zeta potential was -8.9 mv as shown in (Fig. 6).
Fig. 5. Size distribution of Nigella sativa silver nanoparticles (A) Ethanolic extract (B) Chloroform extract
Fig. 6. Zeta Potential of Psidium guajava silver nanoparticles (A) Ethanolic extract (B) Chloroform extract
SEM analysis
It confirms morphology and size particulars of AgNPs. The experimental result showed that the diameter of the prepared nanoparticle (chloroform extract) with average size of 158 nm as shown (Fig.-7).
In vitro antifungal activity
Minimum inhibitory and fungicidal concentrations (MIC/MFC)
The MIC is the least concentration of AgNPs which cause 90% decline in absorbance paralleled to control. MIC90 was measured in triad by broth micro-dilution method19 employing UV spectrophotometer. To determine MFC, aliquots were taken from each visually unblemished test tube after 48 hours incubation and positioned on newly prepared Yeast Extract Peptone Dextrose agar plates11. The plate entirely empty of any colony at last dilution gives Minimum Fungicidal Concentration value.
Table (3):
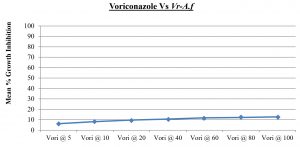
Validation of resistant fungal strains: The resistant fungal strain, Aspergillus niger and Candida glabrata was validated by using Voriconazole (vori) and Fluconazole (Flu) respectively. The mean percentage growth inhibition of fungi was measured at 5, 10, 20, 40, 60, 80 and 100 µg/ml.
Drug & Conc. in μg per mL |
Mean % growth inhibition of Vr-A.f (n=3) |
SD |
|---|---|---|
Vori @ 5 |
6.23 |
1.52 |
Vori @ 10 |
8.21 |
1.69 |
Vori @ 20 |
9.68 |
1.74 |
Vori @ 40 |
10.69 |
1.28 |
Vori @ 60 |
11.85 |
0.87 |
Vori @ 80 |
12.17 |
1.35 |
Vori @ 100 |
12.84 |
0.95 |
Table (4):
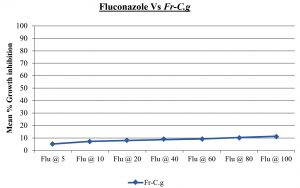
Fluconazole resistant Candida glabrata.
Drug & Conc. in μg per mL |
Mean % growth inhibition of Fr-C.g (n=3) |
SD |
|---|---|---|
Flu @ 5 |
5.22 |
1.23 |
Flu @ 10 |
7.31 |
1.68 |
Flu @ 20 |
8.03 |
1.46 |
Flu @ 40 |
8.76 |
1.09 |
Flu @ 60 |
9.36 |
0.84 |
Flu @ 80 |
10.45 |
0.63 |
Flu @ 100 |
11.38 |
0.72 |
Antifungal activity
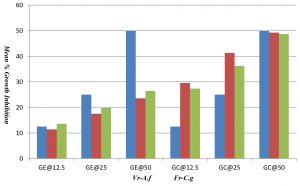
Solutions of each compound (GE, GC, GES and GCS) were used at appropriate concentrations. Each Source solution (GE, GC,) was diluted to attain the final concentrations of 12.5, 25 and 50 mg/mL. Each formulation (GES, GCS) was diluted to obtain the final concentrations – 10, 20, 40, 60, 80, 100 and 120 μg/mL. Two fungal resistant strains namely Voriconazole resistant Aspergillus niger and Fluconazole resistant Candida glabrata were cultured as per standard protocol. An aliquot of 80 μl of every dilution of compounds were poured on a 96-welled (12 x 8) microtitre plate, laterally with 100 μl of fungal broth, 20-μl of fungal inoculum and a 5-μl aliquot of 0.5% of 2,3,5-trifenyl tetrazolium chloride (TTC). All the above were poured on to a well and incubated at 37oC for 24 hrs in Orbital shaker. The appearance of pink color is owed to TTC which indicates fungal growth, and the absence of color represents mean inhibition of fungal growth. The first well of microplate is control without any additional molecules. And second well contains Nystatin as positive control. The MIC value was measured at the well, where no color was established. 90% of growth inhibition was considered as MIC of the compound as per standard protocol12.
Table (5):
Anti-fungal activity of Guava leaf extracts ( GE- Guava leaf Ethanol extract, GC- Guava leaf Chloroform extract).
| Conc. in mg/mL | Vr-A.f | Fr-C.g |
|---|---|---|
| Mean % growth inhibition of Vr-A.f |
Mean % growth inhibition of Fr-C.g |
|
| GE @ 12.5 | 11.43 | 13.67 |
| GE @ 25 | 17.55 | 19.82 |
| GE @ 50 | 23.56 | 26.49 |
| GC @ 12.5 | 29.58 | 27.39 |
| GC @ 25 | 41.33 | 36.21 |
| GC @ 50 | 49.23 | 48.75 |
Table (6):
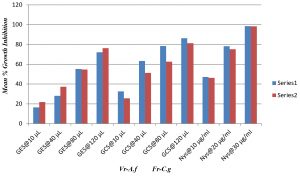
Antifungal activity of Silver nanoparticles synthesised from guava leaf ethanol and Chloroform extracts. (GES- Guava Leaf Ethanol extract silver Nanoparticles, GCS- Guava Leaf Chloroform extract silver Nanoparticle.
Volume spiked in μL |
Mean % growth inhibition of Vr-A.f |
Mean % growth inhibition of Fr-C.g |
|---|---|---|
GES @ 10 |
16.52 |
21.88 |
GES @ 20µL |
28.14 |
29.87 |
GES @ 40 |
42.36 |
37.27 |
GES @ 60 |
49.46 |
45.52 |
GES @ 80 |
55.27 |
54.63 |
GES @ 100 |
64.23 |
71.23 |
GES @ 120 |
72.12 |
76.23 |
GCS @ 10 µL |
32.65 |
25.63 |
GCS @ 20 |
49.72 |
38.27 |
GCS @ 40 |
63.32 |
51.32 |
GCS @ 60 |
71.19 |
58.47 |
GCS @ 80 |
78.35 |
62.63 |
GCS @ 100 |
82.18 |
71.56 |
GCS @ 120 |
86.33 |
81.34 |
Nys @ 5 μg/ml |
32.56 |
34.96 |
Nys @ 10 μg/ml |
47.23 |
46.22 |
Nys @ 15 μg/ml |
63.28 |
61.39 |
Nys @ 20 μg/ml |
78.25 |
75.14 |
Nys @ 25 μg/ml |
92.35 |
89.36 |
Nys @ 30 μg/ml |
98.56 |
98.36 |
Nys @ 35 μg/ml |
99.68 |
99.12 |
Candida glabrata is next to Candida albicans in causing candidiasis. These fungi show resistance to antifungal agents. C. glabrata is linked with high death rates in immune-compromised patients. Azole resistance is reported in many Aspergillus species. Contemporary antifungal drugs are not effective and there is an imperative necessity for novel anti-fungal drugs. The present work aims to synthesize silver nanoparticles by using leaf extracts of Psidium guajava with 2 mM AgNo3 solution. The guava leaf extract (ethanol) showed presence of phenols, terpenoids, glycosides and flavonoids. The chloroform leaf extract showed all the above constituents along with saponins. The Rf values from TLC suggests presence of flavonoids, terpenoids and glycosides. The color of the solution changes and gives surface plasmon resonance at 432 nm as presented in Fig.1. The particle size and zeta potential was found to be 158 nm, 195 nm and -8.9 and -18.8.The morphology of fabricated AgNPs was confirmed by SEM. The antifungal efficacy of AgNPs synthesized using plant extracts was evaluated against resistant Aspergillus niger and Candida glabrata, the strains were validated using Voriconazole and Fluconazole respectively. The guava leaf extracts showed minimal antifungal activity compared to AgNPs. The MIC value of synthesized nanoparticles against Aspergillus niger and C. albicans was 120 µL, shown in (Fig. 8) and (Fig. 9). Nystatin was used as standard drug.
Fig. 8. Mean percentage growth inhibition of Psidium guajava leaf extract (ethanol and chloroform) against Aspergillus niger and Candida glabrata
Fig. 9. Mean percentage growth inhibition of synthesized AgNPs (GES and GEC) against Aspergillus niger and Candida glabrata
The fungal organisms to be tested here have the potential to cause infections in humans, particularly immunosuppressed individuals. Thus, adequate biosafety practices need to be followed. For Aspergillus spp., all manipulations should be performed within a biological safety cabinet to prevent contamination of other laboratory surfaces.The antifungal property of AgNPs on the resistant Candida glabrata and Aspergillus niger, may be owed to disturbance of membrane structure which damages fungal cells. The phytoconstituents of Psidium guajava leaf have the ability to constrain the pathogens. Henceforth biologically fabricated AgNPs, alike the one used in the current study, can be considered as potent antifungal agents.
The AgNPs synthesized from Psidium guajava leaf extracts exhibited significant fungicidal activity against resistant strains like Aspergillus niger and Candida glabrata. The resistant fungi were validated by Voriconazole and Fluconazole. The synthesised AgNPs showed better anti-fungal activity than leaf extracts, the increased action is due to presence of Silver ions. The cell morphology is targeted to a great degree representing that these bio-fabricated AgNPs disrupts the integrity of fungal cells, making it friable, which leads to permeable and leaky fungal cells and eventually leads to death. The current work culminates the antifungal ability and method of action of phytogenically synthesized AgNPs. Future exploration on animal models will help to explore the invivo efficiency of these antifungal agents.
ACKNOWLEDGMENTS
We would like to express our heartfelt thanks to Principal YPR, Vice-Principal JRR and R&D Director PR of Raghavendra Institute of Pharmaceutical Education and Research for providing the necessary facilities.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
ETHICS STATEMENT
This article does not contain any studies with human participants or animals performed by any of the authors.
AVAILABILITY OF DATA
All datasets generated or analysed during this study are included in the manuscript and/or the Supplementary Files.
- Shekhawat MS, Manokari M, Kannan N, Revathi J, Latha R. Synthesis of silver nanoparticles using Cardiospermum halicacabum L. leaf extract and their characterization. J Phytopharmacol. 2013;2(5):15-20.
- Bose D, Chatterjee S. Biogenic synthesis of silver nanoparticles using guava (Psidium guajava) leaf extract and its antibacterial activity against Pseudomonas aeruginosa. Applied Nanoscience. 2016;6(6):895-901.
Crossref - Khatoon N, Sharma Y, Sardar M, Manzoor N. Mode of action and anti-Candida activity of Artemisia annua mediated-synthesized silver nanoparticles. J Mycol Med. 2019;29(3):201-219.
Crossref - Gautam AK, Sharma S, Avasthi S, Bhadauria R. Diversity, pathogenicity and toxicology of A. niger: an important spoilage fungi. Research Journal of Microbiology. 2011;6(3):270-280.
Crossref - Howard SJ, Harrison E, Bowyer P, Varga J, Denning DW. Cryptic species and azole resistance in the Aspergillus niger complex. Antimicrob Agents Chemother. 2011;55(10):4802-4809.
Crossref - Naseer S, Hussain S, Naeem N, Pervaiz M, Rahman M. The phytochemistry and medicinal value of Psidium guajava (guava). Clinical Phytoscience. 2018;4(1):1-8.
Crossref - Gayathri V, Kiruba D. Preliminary Phytochemical Analysis of Leaf Powder Extracts of Psidium guajava L. International Journal of Pharmacognosy and Phytochemical Research. 2014;6(2):332-334.
- Tripathi A, Chandrasekaran N, Raichur AM, Mukherjee A. Antibacterial applications of silver nanoparticles synthesized by aqueous extract of Azadirachta indica (Neem) leaves. J Biomed Nanotechnol. 2009;5(1):93-98.
Crossref - Mourdikoudis S, Pallares RM, Thanh NT. Characterization techniques for nanoparticles: comparison and complementarity upon studying nanoparticle properties. Nanoscale. 2018;10:12871-12934.
Crossref - Shekhawat MS, Manokari M, Kannan N, Revathi J, Latha R. Synthesis of silver nanoparticles using Cardiospermum halicacabum L. leaf extract and their characterization. J Phytopharmacol. 2013;2(5):15-20.
- Pierce CG, Uppuluri P, Tristan AR, et al. A simple and reproducible 96-well plate-based method for the formation of fungal biofilms and its application to antifungal susceptibility testing. Nature Protocols. 2008;3(9):1494.
Crossref - Panacek A, Kolar M, Vecerova R, et al. Antifungal activity of silver nanoparticles against Candida spp. Biomaterials. 2009;30(31):6333-6340.
Crossref - Fidel PL, Vazquez JA, Sobel JD. Candida glabrata: review of epidemiology, pathogenesis, and clinical disease with comparison to C. albicans. Clin Microbiol Rev. 1999;12(1):80-96.
Crossref - Cornistein W, Mora A, Orellana N, Capparelli FJ, del Castillo M. Candida: epidemiology and risk factors for non-albicans species. Enferm Infecc Microbiol Clin. 2012;31(6):380-384.
Crossref
© The Author(s) 2020. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.