ISSN: 0973-7510
E-ISSN: 2581-690X
The increasing prevalence of antibiotic-resistant pathogens has drawn the attention of the researchers in the pharmaceutical field towards studies on the potential antimicrobial activity of plant-derived substances. This study was conducted to investigate the antibacterial and antifungal activity of leaf extracts of Lawsonia inermis (Henna) on some nosocomial infection pathogens. Leaf samples of the plant were collected from Yanbu city, Saudi Arabia. Methanol, ethanol, and hexane extracts were obtained by maceration method. The pathogens were isolated from the hospital environment and identified based on cultural, morphological and biochemical characteristics as Escherichia coli, Klebsiella pneumoniae, Enterobacter cloacae, Pseudomonas aeruginosa, Staphyloccocus aureus, Staphylococcus epidermidis, Enterococcus faecalis, and the yeast Candida albicans. The methanol extract was shown high antibacterial and antifungal activity compared with Hexane and Chloroform extracts. Methanol extract expressed highest and broad- spectrum antibacterial activity against pathogenic strains. It exhibited high activity against Gram- positive and Gram- negative bacteria and fungi. The results of this study support the use of Lawsonia inermis plant in traditional medicine to treat bacterial and fungal diseases and this plant could be a source of new antibiotic compounds.
Lawsonia inermis; Henna; Antibacterial; Antifungal; Human pathogens; Nosocomial infections.
The increasing prevalence of antibiotic-resistant pathogens may decrease the efficacy of existing antibiotics, making the treatment of patients costly and difficult1. The emergence of resistant microorganisms is a result of the misuse and overuse of antibiotics and the use of antimicrobial compounds in food animal production to improve animal health and higher production2. In 2004, more than 70% of pathogenic bacteria were estimated to be resistant to at least one of the currently available antibiotics3. Methicillin resistant S. aureus (MRSA) strains are the most common cause of nosocomial infections and cause high mortality and morbidity rate4. Enterococcal infections have emerged as one of the most common nosocomial infections in the past decade. They are an important cause of infection in organ transplant recipients and other seriously ill patients. Members of these genus, are resistant to a large number of antimicrobial agents5.There are also other examples of Gram-negative pathogens: Klebsiella, Escherichia, and Pseudomonas. More than 60% of sepsis cases in hospitals are caused by Gram-negative bacteria2. Among them, Pseudomonas aeruginosa accounts for almost 80% of these opportunistic infections. Furthermore, approximately 40% of death from nosocomial infections are caused by fungi, and 80% of these are caused by Candida and Aspergillus6.Therefore, there is a continuous and urgent need to explore new antibiotics to overcome the emerging resistance to currently available antibiotics. The herbal medicine still plays a vital role to treat infectious diseases in developing countries. Among the known plant species on earth (estimated at 250,000–500,000) only a small fraction have been investigated for the presence of antimicrobial compounds and only 1–10% of plants are used by humans7. In the past, plants have been used in traditional herbal medicine. The antimicrobial activity of several plant-derived compounds has been previously reported8,9, and a wide range of active components have been identified10. Several studies revealed that bioactive compounds from some medicinal plants exhibit anti-carcinogenic, antitumor, antibacterial, antifungal, antiviral, antioxidant and anti-inflammatory effects, as well as antioxidant properties11. Henna (Lawsonia inermis) the 2-6 m height flowering plant, is the unique species in the genus Lawsonia in the family Lythraceae12. Henna plant is used in traditional medicine to treat a variety of diseases such as a headache, rheumatoid arthritis, ulcers, diarrhea, leprosy, fever, diabetes, and cardiac disease13. In addition, this plant is used for controlling infectious diseases. The inhibitory activity of the plant against gram negative, gram positive bacteria, and fungi was described14,12. This activity is attributed to the presence of anthraquinones (lawsone) (2 hydroxynaphthoquinones) as major components of the plant, representing about 0.5-1.5% of henna15. Lawson is a natural dye used in the Middle East, Saudi Arabia, Asia and parts of Africa to color hand, finger, nails, and hair, having a wide spectrum of biological activities, such as anti-fungal and antibacterial for the skin and hair, antitumor, antibacterial, anti-inflammatory, anti-parasitic, cytotoxic activities. Many reports cite the antibacterial effects of henna plant especially for gram positive bacteria and antifungal activity against dermatophytes, and wound healing16, 17, 18, however few researches described its effect on nosocomial pathogens . Accordingly, this study reports the evaluation of the antagonistic potential of Saudi Henna plant against pathogenic microorganisms contributing to nosocomial infections.
Plant material
Henna (Lawsonia inermis Linn) leaves were harvested from different regions of Yanbu city, Saudi Arabia in April 2016. The plant material was thoroughly washed with distilled water, dried in the shade and grinded to powder. The powder was stored in airtight containers at room temperature in the dark until use.
Chemicals and culture media
Gentamicin and nystatin were purchased from Himedia (India).The solvents ethanol, methanol, and n-hexane and all culture media were obtained from Merck (Germany).
Preparation of L. inermis extracts
The powdered samples of henna leaves were subjected to extraction according to the method of Gupta et al. (2009)19. Briefly, 150 g of air dried powder was soaked in 300 ml of ethanol, methanol, and n-hexane. The mixtures were stirred for 3 days at room temperature, then filtered through Whatman filter No 1. The filtrates were dried and concentrated using Rotary evaporator at 40°C. The crude extracts were weighted and stock solutions were prepared to obtain a final concentration of 300 mg/ml. The extracts kept in the dark at 4°C until use.
Isolation of pathogenic microorganisms
Microorganisms used in this study (Escherichia coli, Klebsiella pneumoniae, Enterobacter cloacae, Pseudomonas aeruginosa, Staphyloccocus aureus, Staphylococcus epidermidis, Enterococcus faecalis, and the yeast Candida albicans) were isolated from the hospital environment (staff nurses hands, sinks, tubs, ground (floors), walls, beds, blankets, doors, doors handle, nurse tables, chairs, electronic equipment’s, etc) at some hospitals in Yanbu city, Saudi Arabia. Strains were identified using standard microbiological procedures using Bergey’s Manual of Determinative Bacteriology20. These nosocomial pathogens were identified by colony characteristics, microscopic morphology, catalase and oxidase reactions, API20E test).
Antimicrobial Activity Assay (Disc Diffusion method)
The antibacterial and antifungal activities of ethanol, methanol and hexane extracts of henna plant were evaluated by disc diffusion method (Kirby and Bauer, 1966). Test microorganisms suspensions (prepared in sterile saline) , with a turbidity equivalent to that of 0.5 McFarland standard, were uniformly seeded with sterile swabs onto Muller Hinton Agar (MHA) for bacteria and Sabouraud dextrose agar (SDA) for yeast. The filter paper discs (6 mm) were impregnated with 20 µl of the plant extracts (100 mg/ml), dried and carefully laid on the surface of the agar plates inoculated with test microorganisms. The plates were left at 4 °C for 12-16 hours then incubated overnight at 37°C. Inhibition zone of test microorganisms around the paper disks was measured. Disks with ethanol, methanol and hexane solvents were used as a negative control. Antibacterial disks with Gentamicin (10 µg/disc) and nystatin (100 µg/disk) were used as positive control. All assays were carried out in duplicate.
Minimum Inhibitory Concentration (MIC) Determination
The MIC of the methanol extract against tested microorganisms was determined by the dilution method. Serial two-fold dilutions in Nutrient broth for bacteria and Sabouraud dextrose broth for yeast (300 mg/ml – 1.56 mg/ml) were prepared from stock solution of the extract. The tubes were then inoculated with 50µl of microorganism cultures. Un-inoculated tubes containing growth medium and extract were used as negative controls. The tubes were then incubated overnight at appropriate temperatures. The MIC was defined as the lowest concentration that completely inhibited the growth of the organism.
Lawsonia inermis plant was used to evaluate their antimicrobial activity against eight nosocomial infection pathogens. Three solvents ethanol, methanol, and n-hexane were used for extraction of active compounds. The isolated pathogens were identified as Escherichia coli, Klebsiella pneumoniae, Enterobacter cloacae, Pseudomonas aeruginosa, Staphyloccocus aureus, Staphylococcus epidermidis, Enterococcus faecalis, and the yeast Candida albicans based on cultural, morphological and biochemical characteristics. Data on antimicrobial activity measured as a zone of inhibition of solvent extracts on test microorganisms are shown in tables 1 and represented by the figure 1. In the present study, the strong in vitro inhibition against isolated microorganisms was recorded in methanol extract of henna with inhibition zone of 30mm against S. aureus (table 1 & figure 1). Also strong inhibitions were recorded with methanol extract against C. albicans (26mm), E. cloacae (23mm), E. faecalis (20mm) and K. pneumonia (19mm). Ethanol extract showed moderate activity against S. aureus and K. pneumonia while there was no activity in hexane extract.
Table (1):
Activity of Lawsonia inermis leaf extracts and controls against pathogenic microorganisms.
| No | Organism | Diameter of zone of inhibition (mm) (Including 6mm of disc size) | ||||
|---|---|---|---|---|---|---|
| Hexane | Ethanol | Methanol | Gentamycin | Nystatin | ||
| 1 | S. aureus | 0 | 18 | 30 | 15 | – |
| 2 | S. epidermidis | 0 | 0 | 0 | 10 | – |
| 3 | K. pneumonia | 0 | 13 | 19 | 20 | – |
| 4 | E. faecalis | 0 | 0 | 20 | 0 | – |
| 5 | E. cloacae | 0 | 0 | 23 | 17 | – |
| 6 | E. coli | 0 | 0 | 16 | 22 | – |
| 7 | P. aeruginosa | 0 | 0 | 16 | 10 | – |
| 8 | C. albicans | 0 | 0 | 26 | – | 18±0 |
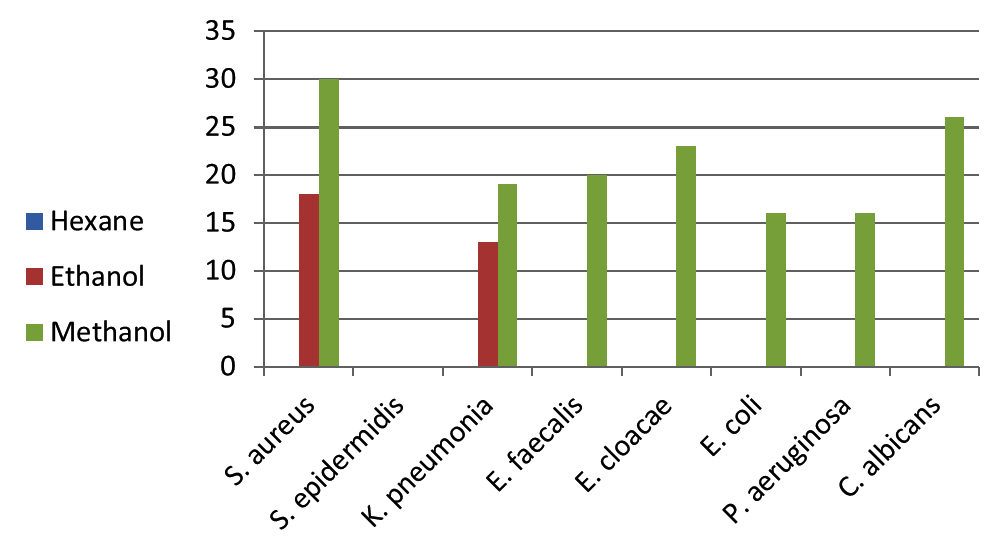
Fig. 1. Activity of Lawsonia inermis leaf extracts against pathogenic microorganisms
Significant antibacterial and antifungal effects were expressed as MIC of methanol extract against tested microorganisms are shown in
table 2. The strongest activity of methanol extract was against S. aureus with MIC value of 1.5 mg/ml followed by C. albicans (3.12 mg/ml).
Table (2):
Activity of Lawsonia inermis leaf extracts against pathogenic microorganisms MIC (mg/ml)
S. aureus |
S. epidermidis |
K. pneumonia |
E. faecalis |
E. cloacae |
E. coli |
P. aeruginosa |
C. albicans |
|---|---|---|---|---|---|---|---|
1.5 mg/ml |
300
mg/ml |
6.25
mg/ml |
6.25
mg/ml |
6.25
mg/ml |
12.5
mg/ml |
12.5
mg/ml |
3.12
mg/ml |
Henna plant has been used over centuries in traditional herbal medicine and as preservative, and cosmetic in the Middle East, Asia and parts of Africa.
In this study, the methanol extract of L. inermis exhibited a higher degree of antimicrobial activity as compared with ethanol and hexane extracts. Methanol as a solvent is the most commonly used for preliminary studies of antimicrobial activities in plants21. This higher antibacterial and antifungal activities of methanol extract is due to the nature of biological active components (alkaloids, flavonoids, terpenoids, tannins etc.), which may be more soluble in methanol. The stronger extraction capacity of methanol that may have yielded a greater number of active constituents responsible for the activities 22. Several studies have been found that methanol is a best solvent for extraction of the active compounds23,24 as compared with ethanol, hexane and chloroform.The strains of S. aureus and C. albicans were the most susceptible to plant extracts. On the contrary, E. coli and P. aureginosa were the most resistant microorganisms. This result is in accord with formers studies that confirmed that gram positive bacteria are more sensitive to antimicrobial compounds than gram positive bacteria. The methanol extract of Lawsonia inermis showed broad activity against gram positive and gram negative bacteria, and yeast except S. epidermidis. The microorganism E.coli, which is already known to be multi-resistant to drugs, was inhibited by the methanol extracts of henna. On the other hand, Pseudomonas aeroginosa and other Pseudomans sp, have always been important causes of hospital infection because of their intrinsic resistance to most antibiotics and ability to survive and even multiply at low temperatures and in disinfectant solutions, were inhibited by the methanol extracts. Such results are very interesting, because E.coli and P. aeruginosa are important nosocomial pathogens, resistant to antibiotics and its control is very difficult by therapeutic means. The antifungal activity of L. inermis leaf extract was evaluated in vitro against fungi such as C. albicans, S. cerevisiae, and dermatophytes by several investigators21, 25. A study of Muhammad and Muhammad (2005)14 demonstrated the activity of L. inermis leaves against the hospital strains A. niger and F. oxysporum in addition to Streptococcus sp. and S. aureus. The aqueous and chloroform extracts of henna leaves were effective in inhibiting the growth of microorganisms that are involved in causing burn wound infections. On the other hand, some studies suggested that henna plant has a wide spectrum of antimicrobial activity including antibacterial, antiviral, antifungal and antiparasitic activities. Kannahi and Vinotha (2013)23 found that the ethanol extract was shown significant activity against yeasts (Candida albicans and Saccharomyces cerevisiae ) compared to petroleum ether extract. In study of Suleiman and Mohamed ( 2014 )26 the antifungal activity of L. inermis leaf extract was evaluated in vitro against C. albicans, S. cerevisiae, and some selected dermatophytes. Petroleum ether extract showed a wide antifungal spectrum compared to ethanol extract against tested yeasts. Many reports cite the inhibitory activity of henna plant against gram negative and gram positive organisms . The extracts of tested plants showed a great activity in inhibiting the growth of bacteria and fungi, probably due to the presence of active ingredients that inhibit bacterial and fungal growth. Henna plant contains Lawsone in about 0.5 to 1.5% of its ingredients. Lawsone (2- hydroxynapthoquinone) is the main constituent responsible for the dyeing properties of the plant. However, henna also contains mucilage, flavonoids, naphthalene derivatives, coumarins, tannic acid, luteolin and gallic acid12. Antimicrobial activity may be due to more than one component. Investigations showed that henna plant is effective against different microorganisms especially against P. aeruginosa. In nosocomial infection, Staphylococcus aureus is one of the most prevalent microorganisms worldwide. Methicillin resistant strains represent 15-45% of all Staphylococcus aureus isolates27. Inhibitory activity of henna was shown against both gram negative and gram positive microbes. In one study28 Lawsone, displayed inhibitory effects against common nosocomial urinary tract pathogens such as Escherichia coli, Proteus mirabilis, Klebsiella pneumoniae, Pseudomonas aeroginosa and Staphylococcus aureus at certain concentrations. Recent data indicate that E. coli is the most common etiologic gram-negative organism, followed by P. aeruginosa, Klebsiella species, and enterobacter species29.
In fact, Gram- negative bacteria are frequently reported to have developed multi-drug resistance to many of the commercially available antibiotics of which E. coli and P. aeroginosa are the most prominent. However, methanol extract of L. inermis is of special interest for further investigation as it showed activity against both gram- negative bacteria E. coli and P. aeroginosa.
According to the present study, L. inermis extracts showed varying degrees of antimicrobial activity against pathogenic microorganisms contributing to nosocomial infections. L. inermis plant was effective to combat the pathogenic microorganisms studied especially, S. aureus and C. albicans. Thus, the plant could be used as alternative source of antibacterial and antifungal agents. Further work is needed to isolate the secondary metabolites from the methanol extract of L. inermis in order to test specific activities and toxicity against animal and human cells.
ACKNOWLEDGMENTS
The author would like to thank Taibah University for providing lab facilities to perform the research.
- Penesyan A, Gillings M, Paulsen IT. Antibiotic Discovery: combatting bacterial resistance in cells and in Biofilm Communities. Molecules 2015; 20 (4):5286-5298.
- Barriere SL. Clinical, economic and societal impact of antibiotic resistance. Expert Opin Pharmacother 2015; 16(2):151–153.
- Katz ML, Mueller LV, Polyakov M, Weinstock SF. Where have all the antibiotic patents gone? Nat Biotechnol 2006; 24:1529–1531.
- Hughes D. Exploiting genomics, genetics and chemistry to combat antibiotic resistance. Genetics 2003; 4: 433-441.
- Paterson, DL. Impact of Antibiotic resistance in gram-negative bacilli on empirical and definitive antibiotic therapy. Clin Infectious Dis 2008; 15(47) Suppl 1:S14–20.
- Demain AL, and Sanchez S. Microbial drug discovery: 80 years of progress. J Antibiot 2009; 62: 5–16
- Berdy J, Aszalos A, Bostian M, Mcnitt KL, CRC () Handbook of antibiotic compounds. Boca Raton, CRC Press, 2006; v. 8, Part 1.
- Burt S. () Essential oils: their antibacterial properties and potential applications in foods—a review. Int Journal Food Microbiol 2004; 94(3):223–253.
- Newman DJ, and Cragg GM. Natural products as sources of new drugs over the 30 years from 1981 to 2010.J Nat Prod 2012; 75(3):311–33.
- Dixon RA. Natural products and plant disease resistance. Nature 2001; 411(6839):843–847.
- Ozgová Š, HeYmánek J, Gut I. Different antioxidant effects of polyphenols on lipid peroxidation and hydroxyl radicals in the NADPH-, Fe-ascorbate- and Fe-microsomal systems. Biochem Pharmacol 2003; 66(7):1127–1137.
- Chaudhary, G., Goyal, S., Poonia, P. Lawonia inermis Linnaeus: A phytopharmaclogical review. Int J Pharm Sci Drug Res, 2010; 2(2) :91-98.
- Reddy K. Folk medicine from Chittoor District, Andhra Pradesh, India used in the treatment of jaundice. Int J Crude Drug Res 1988; 26(3): 137-140.
- Muhammad HS. Muhammad S. The use of Lawsonia inermis linn. (Henna) in the management of burn wound infections. Afr J Biotechnol 2005; 4(9): 934- 937.
- Ramya A, Vijayakumar N, and Renuka M. Antiarthritic effect of aqueous extarct of lawsonia inermis.l – an in-vitro study, Int J Mod Res Revs 2015; 3(8): 744-747.
- Chung Y, Yoo J, Park S, Kim BH, Chen X, Zhan C and Cho H. “Dependence of antitumor activity on the electrophilicity of 2-substituted 1,4-naphthoquinone derivatives”, Bull Korean Chem Soc 2007; 28(4): 691-694.
- Rahmoun NM, Boucherit-Otmani Z, Boucherit K, Benabdallah M, Villemin D and Choukchou-Braham N. “Antibacterial and antifungal activity of lawsone and novel naphthoquinone derivatives,” Med Mal Infec 2012; 42(6): 270-275.
- Sharma KK, Saikia R, Kotoky JC, Kalita J, Devi R. Antifungal activity of Solanum melongena L, Lawsonia inermis L. and Justicia gendarussa B. against Dermatophytes. Int J PharmTech Res 2011; 3(3)1635-1640.
- Gupta, Nishant K, Dixit, Vinod K. Hepatoprotective activity of Cleome viscosa Linn. extract against thioacetamide-induced hepatotoxicity in rats. Nat Prod Res 2009; 23(14): 1289–1297.
- Holt, J G, Krieg N R, et al. “Bergey’s manual of determinative bacteriology” 1994.
- Habbal O, Hasson S, El-Hag A, Al- Mahrooqi Z, Al-Hashmi N, Al- Bimani Z, Al-Balushi M, Al-Jabri A. Antibacterial activity of Lawsonia inermis Linn (Henna) against Pseudomonas aeruginosa. Asian Pac J Trop Biomed 2011; 1(3): 173-176.
- Tabassum H, Ali MN, Al-Jameil N and Khan FA. Evaluation of Antibacterial Potential of Selected Plant Extracts on Bacterial Pathogens Isolated from Urinary Tract Infections. Int J Curr Microbiol App Sci 2013; 2(10): 353-368.
- Kannahi M and Vinotha K. Antimicrobial activity of Lawsonia inermis leaf extracts against some human pathogens, Int J Curr Microbiol App Sci 2013; 2(5): 342-349.
- Rayavarapu A, Kaladhar D and Kumar S. Evaluation of antimicrobial activity of Lawsonia Inermis (Henna) on aquapathogens. J Pharm Biomed Anal. JPBMS 2011; 7(02).
- Rahmoun N, Zahia Boucherit-Otmani, Kebir Boucherit, Mohammed Benabdallah2, and Noureddine Choukchou-Braham Antifungal activity of the Algerian Lawsonia inermis (henna), Agric Food chem 2013; 51(1): 131–135.
- Suleiman E A and Mohamed E A. Antifungal activity of the Algerian Lawsonia inermis (henna) J Mycol 2014; Article ID 375932, 5 pages.
- Singh R, Chidambara K, Jayaprakasha G. Studies on the antioxidant activity of pomegranate (Punica granatum) peel and seed extracts using in vitro models. Journal of agricultural and food chemistry 2002; 50(1): 81-86.
- Bhuvaneswari, K S, Gnana Poongathai A, Kuruvilla and Appala Raju A. Inhibitory concentrations of Lawsonia inermis dry powder for urinary pathogens. Indian J. Pharmacol 2002; 34: 260-3.
- Peleg A Y and Hooper D C. Hospital-Acquired Infections Due to Gram-Negative Bacteria, N Engl J Med. 2010; 362(19):1804–1813.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


