ISSN: 0973-7510
E-ISSN: 2581-690X
Sugarcane is an important cash crop in India, as well as a key raw material in the production of sugar, jaggery, and bioethanol. Sugarcane yellow leaf disease incited by Sugarcane yellow leaf virus (SCYLV) is emerging as a major threat to sugarcane cultivation and the canes infected by SCYLV were found to be frequently invaded by fungal pathogens. SCYLV infected canes when used as seed material recorded low germination and seedling vigour. Hence, the present investigation was undertaken to test the efficacy of thermotherapy followed by carbendazim treatment for enhancing germination of SCYLV infected seed setts and seedling vigour. Single node setts of SCYLV infected canes were subjected to thermotherapy at 50°C, 51°C and 52°C for 10, 20 and 30 minutes in a hot water bath. Single node setts dipped in water or carbendazim (0.1%) solution served as control. Results of the study revealed that sett treatment with hot water at 50°C for 20 min followed by dipping in carbendazim (0.1%) solution for 30 minutes enhanced the sett germination and vigour of virus infected single noded setts. The presence of SCYLV in the seedlings that had emerged was determined using RT-PCR. The virus was found in germinated seedlings even after thermotherapy, suggesting that at the temperatures tested, thermotherapy was ineffective in eliminating the SCYLV sett borne inoculum. Thermotherapy and fungicide treatment, on the other hand, improved the germinability of infected single noded setts, probably due to a reduction in secondary fungal infection.
Hot Water Treatment, RT-PCR, Saccharum officinarum, Sett Germination, Yellow Leaf Disease
Sugarcane (Saccharum officinarum L.) is one of the most important cash crops grown in Andhra Pradesh providing raw material for sugarcane and bio-ethanol. Due to the occurrence of many pests and diseases, the area under sugarcane cultivation is currently being reduced. Red rot, smut, wilt and viral diseases are the major constraints leading to economic losses in commercially grown sugarcane varieties. Majority of the diseases are disseminated by infected setts leading to germination loss and reduced seedling vigour.
Yellow leaf disease (YLD), caused by the Sugarcane Yellow Leaf Virus (SCYLV), is one of the most destructive viral diseases, causing massive losses in sugarcane yield and quality1. Seed germination was found to be reduced, and different fungal pathogens such as Fusarium sacchari, Schizophyllum commune, and Marasmius sacchari were shown to be associated with germination problems2. Several researchers examined the efficacy of various fungicides, hot water, and moist hot air treatments for the removal of sett-borne inoculum of fungal pathogens, phytoplasma, and viruses infecting sugarcane. Grassy shoot disease (GSD) infection in sugarcane seed cane has been reported to be eradicated by moist hot air therapy (MHAT) at 52°C and 54°C for 4 and 2 hours, respectively, without affecting seed germination3.
Khaing et al.4 studied the effects of lime, fungicides, and hot water treatments on the germination of single node sugarcane setts, recommending that single node setts be dipped in lime (0.75 percent) or thiophanate methyl (0.05 percent) solution or hot water treatment (50°C) for 30 minutes to ensure uniform sett germination. Damayanti and Putra5 investigated the thermal inactivation point of Sugarcane streak mosaic virus (SCSMV) infected plant sap by heating it at 50, 55, 60, and 65 degrees Celsius. The viability of SCSMV was shown to be lowered as temperature and submersion period were increased. Similarly, demonstrated that when sett canes were treated with hot water at 50°C for 2 hours, germination was reduced, but there was a lower incidence of ratoon stunting disease.6
However, the majority of these treatments were developed using three or two sugarcane budded setts. Sugarcane single node seedlings grown from single node setts in portray are becoming increasingly popular. When SCYLV-infected setts were employed as seed material, germination of single node setts was reduced. The objective of the present study was to optimize the temperature and submersion times for hot water treatment of virus-infected single node sugarcane setts to improve their germination and seedling vigour.
Hot Water Treatment of Setts
The study was carried out over two years at the Regional Agricultural Research Station in Anakapalli, utilising the Sugarcane Yellow Leaf Virus (SCYLV) susceptible variety 2001A 63. Virus-infected sugarcane plants were identified using RT-PCR and chosen as seed material for hot water treatment. Single node setts were excised using a single node cutting machine and subjected to hot water treatments in a water bath at three temperatures (50, 51, and 52°C) for three submersion times (10, 20 and 30 minutes), followed by 30 minutes dip in carbendazim solution (0.1 percent). Setts soaked in water and carbendazim (0.1%) served as controls. The setts were sown in portrays filled with a potting mixture of cocopeat and vermicompost (1:1). Fifty setts were sown in each portray and three replications were maintained per each treatment. The treatments were arranged in a Completely Randomized Design (CRD) and incubated in a greenhouse for 30 days. Per cent sett germination and seedling length were recorded at 30 days after sowing and seedling vigour was computed. The data were analyzed using R software. Leaf samples were excised from 30 days old seedlings and presence of SCYLV was determined using RT-PCR.
Detection of SCYLV through RT-PCR
Total plant RNA was isolated from 30 days old single node seedlings developed from hot water treated setts and diseased seed material using trizol method using standard protocols.7 RNA was converted to cDNA using Thermo Fisher first strand cDNA synthesis kit. SCYLV coat protein gene specific primers, SCYLV 615F and SCYLV 615R, were used for the study. The amplified gene products were separated by gel electrophoresis and visualized in a gel documentation unit.
Hot water treatment (HWT) of Sugarcane Yellow Leaf Virus (SCYLV)-infected single node sugarcane setts, followed by 30 minutes of dipping in carbendazim (0.1 percent) solution was found to enhance the germination ability of the setts compared to control (Table 1). Germination of the virus infected setts was found to be high (85%) when they were subjected to HWT at 50°C for 20 minutes which was followed by HWT at 52°C for10 minutes when compared to control (30%). At a submersion time of 30 minutes, all temperatures tested showed a reduction in sett germination. When the setts were submerged for 20 minutes at 50°C, germination and vigour were shown to be superior to 10 and 30 minutes. At 51°C, a similar trend was found. However, when submerged for 10 minutes at 52°C, germination percentage and seedling vigour were higher than at other submersion durations. At submersion times of 20 and 30 minutes at 52°C, the germination of single node setts gradually decreased.
Table (1):
Effect of hot water treatment on the regeneration of SCYLV infected single node setts of sugarcane.
| Treatment | 2020-21 | 2021-22 | Mean | |||
|---|---|---|---|---|---|---|
| Per cent germination** | Seedling vigour Index*** | Per cent germination** | Seedling vigour Index*** | Per cent germination | Seedling vigour Index | |
| HWT* at 50OC for 10 min | 60.00 (50.77)e | 2460 (3.39)e | 45.00 (42.13)c | 2002 (3.30)b | 52.5 | 2231 |
| HWT at 50OC for 20 min | 95.00 (77.09)a | 4750 (3.68)a | 75.00 (60.00)a | 3075 (3.49)a | 85.0 | 3912.5 |
| HWT at 50OC for 30 min | 65.00 (53.73)d | 1820 (3.26)f | 30.00 (33.21)f | 774 (2.89)e | 47.5 | 1297 |
| HWT at 51OC for 10 min | 70.00 (56.80)c | 3640 (3.56)b | 40.00 (39.23)d | 1730 (3.24)bc | 65.0 | 2685 |
| HWT at 51OC for 20 min | 65.00 (53.73)d | 3380 (3.53)c | 65.00 (53.73)b | 3185 (3.50)a | 65.0 | 3282.5 |
| HWT at 51OC for 30 min | 55.00 (47.87)f | 2640 (3.42)d | 40.00 (39.23)d | 1745 (3.24)bc | 47.5 | 2192.5 |
| HWT at 52OC for 10 min | 75.00 (60.00)b | 3450 (3.54)bc | 75.00 (60.00)a | 3431 (3.54)a | 75.0 | 3440.5 |
| HWT at 52OC for 20 min | 65.00 (53.73)d | 2470 (3.39)e | 35.00 (36.27)e | 1330 (3.12)d | 50.0 | 1900 |
| HWT at 52OC for 30 min | 45.00 (42.13)g | 1834 (3.26)f | 35.00 (36.27)e | 1400 (3.15)cd | 40.0 | 1617 |
| Untreated control (Sett treatment with carbendazim) | 65.00 (53.73)d | 845 (2.93)g | 45.00 (42.13)c | 540 (2.73)f | 55.0 | 692.5 |
| Untreated control (Without carbendazim sett treatment) | 35.00 (36.27)h |
394 (2.60)h | 25.00 (30.00)g | 266 (2.40)g | 30.0 | 330 |
| CD (0.05) | 1.12 | 0.03 | 1.04 | 0.11 | ||
| CV (%) | 1.25 | 1.49 | 1.43 | 2.02 | ||
HWT*= Hot Water Treatment, **Values in parenthesis are arcsine transformation, ***Values in parenthesis are log transformation
The findings are consistent with Yulianti et al.6, who found that treating sett cane with hot water at 50°C for 2 hours reduced germination. In the present study, hot water treatment significantly enhanced the proportion of setts that germinated when compared to control. Trippi8 observed that hot water treatment at 50°C stimulated bud germination in sugarcane seed cuttings. Thermotherapy has been utilised to treat sugarcane sett transmitted infections in a practical way. Sugarcane mosaic virus strains were eradicated from sugarcane setts after four days of repeated hot water treatments at 54.8, 57.3, 57.3 and 57.3 degrees Celsius for seven minutes each day.9
Balamuralikrishnan et al.10 investigated the effect of serial thermotherapy on SCMV titre and regeneration in two sugarcane cultivars, Co 740 and CoC 671. None of the thermal treatments generated virus-free seedlings, and some of the treatments inhibited sett germination, according to the researchers. The viral titre was observed to be reduced in thermo-treated setts at an early stage in field-planted setts, up to four weeks after planting. In the current study, there was a difference in sett germination between the first and second years of evaluation, which could be related to the viral titre in the seed material used. Although the trend in terms of germination was virtually the same, the first year of evaluation yielded higher germination percentages and seedling vigour than the second.
Detection of SCYLV through RT-PCR
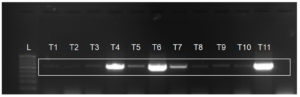
RT-PCR was used to assess the presence of SCYLV in the canes that showed visual symptoms of the disease. The SCYLV-infected canes were used to see if thermotherapy could help eliminate SCYLV from single node setts. The presence of SCYLV was determined in thirty-day-old seedlings regenerated from single node setts after thermotherapy using coat protein gene specific primers SCYLV 615F and SCYLV 615R (Figure 1). The study found that at the temperatures and submersion timeframes studied, the virus could not be removed from SCYLV-infected single node setts. Reduction of virus titre, but not virus eradication, was frequently found in the plants following virus eradication treatments11. High temperature treatments were found to prevent virus movement towards meristematic cells, inhibit virus replication or caused RNA degradation or promoted RNA silencing, thus decreasing virus titre in the infected plant parts. However, the eradication efficiency of temperature treatments and exposure times varied with the type of virus. Karan et al.12 studied the efficacy of chemotherapy, thermotherapy and their combinations for the eradication of PVX, PVY, PVS, PVM and PLRV viruses from potato and reported the efficacy of chemotherapy and thermotherapy in elimination of PVM and PLRV, respectively. Combination of thermotherapy and chemotherapy was found effective in the elimination of PVY and PLRV. Mirza et al.13 found that Sugarcane mosaic virus could only be eliminated by thermotherapy from bud chips of sugarcane in few cultivars tested. Repeated hot water treatments for 4 days at 55, 56.5 and 57.50C with submersion times of 7 min, 10 min with an interval of 24 hours between treatments could eliminate SCMV from only 3 cultivars out of 19 cultivars tested.
Figure 1. Detection of SCYLV in single node seedlings of sugarcane after thermotherapy.
[L=100bp ladder; T1= 50°C for 10 min; T2= 50°C for 20 min; T3= 50°C for 30 min; T4= 51°C for 10 min; T5 = 51°C for 20 min; T6= 51°C for 30 min; T7= 52°C for 10 min; T8= 52°C for 20 min; T9= 52°C for 30 min; T10= Untreated control; T11= Untreated control (without Carbendazim)].
In conclusion, the present study clearly indicates that SCYLV could not be eliminated from infected setts by thermotherapy at temperatures of 50, 51 and 52°C at submersion times of 10, 20 and 30 minutes. The germination of single node setts of sugarcane infected with SCYLV could be enhanced by combination of thermotherapy and chemotherapy.
ACKNOWLEDGMENTS
The authors would like to thank Director of Research, ANGRAU, Lam, Guntur, India for providing laboratory facilities for conducting this research.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
PKV performed planning, execution of experiment. VCS assisted in execution of the trial. GVK performed molecular detection of SCYLV. KKC performed data analysis. PKV and MB wrote the manuscript. All authors read and approved the final manuscript for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
Not applicable.
- Lehrer AT, Wu KK, Komor E, Impact of Sugarcane yellow leaf virus (SCYLV) on growth and sugar yield of sugarcane. J Gen Plant Pathol. 2009; 75(4): 288-296.
Crossref - Varma PK, Vani CS, Kumar PBP, Chandrasekhar V, Bharathalakshmi M. Survey for sugarcane diseases in major sugarcane growing areas of Andhra Pradesh, India. Int J Curr Microbiol. 2020; 9(8): 1865-1873.
Crossref - Singh K, Misra SR, Shukla US, Singh RP. Moist hot air therapy of sugarcane control of sett-borne infections of GSD, smut and red rot. Int Sugar J. 1980; 43(5): 26-28.
- Khaing TZO, Aung MT, Kyaw KW, Kyi T, Ei EK. Evaluation of seed cane treatments on sugarcane germination in two planting methods. Int J Environ Rural Dev. 2018; 9-2.
- Damayanti TA, Putra LK. Hot water treatment of cutting-cane infected with sugarcane streak mosaic virus (SCSMV). J Int Soc Southeast Asian Agric Sci. 2010; 16(2): 17-25.
- Yulianti T, Wijayanti, Supriyono. Management of ratoon stunting disease by hot water to provide healthy cane seed. In: Proceedings of 1st International conference on sustainable plantation. IOP Conf.Series: Earth and Environmental Science. 2020; 418:012063.
Crossref - Vega I, Seagliusi SM, Ulian EC. Sugarcane yellow leaf disease in Brazil: Evidence of association with a luteovirus. Plant Dis. 1997; 81:21-26.
Crossref - Trippi VS. Action of hot and cold water on the germination of cane. Sugar Azucar. 1961; 56(10): 38.
- Benda GTA, Ricaud C. The use of heat treatment for sugarcane disease control. Proceedings of the International Society of Sugar Cane Technologists 1978; 26: 1-9
- Balamuralikrishnan M, Sabitha D, Ganapathy T, Viswanathan R. Impact of serial thermotherapy on Sugarcane mosaic virus titre and regeneration in sugarcane. Archives Phytopathology Plant Protection. 2003; 36(3-4): 173-178.
Crossref - Wang MR, Cui ZH, Li JW, et al. In vitro thermotherapy-based methods for plant virus eradication. Plant Methods. 2018; 14:87.
Crossref - Karan Y, Scheuring DC, Chappell AL. Eradication of PVX, PVY, PVS, PVM and PLRV from potato by chemotherapy, thermotherapy and their combinations. Journal of Agricultural faculty of Gaziosmanpasa University 2021; 38(3): 117-122.
Crossref - Mirza MS, Ahmad M, Anwar MS. Elimination of sugarcane mosaic virus from diseases sugarcane buds with thermotherapy. Journal of Agricultural Research. 1986; 24(3): 207-210.
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.