ISSN: 0973-7510
E-ISSN: 2581-690X
Mineral ores are the non-renewable resources of various metals. The extraction of metals is done only from the Ist and IInd grade ore particles and rest is left as such because of low metal content. Bacteria are the miraculous organisms as it can be used for the complete extraction of metals. In the present experiment, we have used the bacterium Acidithiobacillus ferrooxidans and Pseudomonas fluorescens as a bioleaching agent for iron and aluminum attribution from low grade bauxite ore collected from Radhanagari, Kolhapur India. Pulp density of the mineral ore is the appropriate concentration required for completion of the reaction. At specific pulp density, there is the maximum output of the extracted metals was found. In the present investigation, various pulp densities of the bauxite ore have been tried for the extraction of aluminum and iron. The maximum extraction of iron and aluminum was found 589.67mg (80.99%) and 286.45 mg (79.02%) at the pulp density of 5 and 4 g respectively by Acidithiobacillus ferrooxidans. In the case of Pseudomonas fluorescens the maximum yield of aluminum was exhibited 288.34mg (39.60%) and iron was 242.6 mg (73.98%) at 4 g bauxite Ore. In the present study, we have also found that the increase in the pulp density of the ore decreased the process of extraction. Our results also strengthen the potential of these bacteria’s for extraction of aluminum and iron from bauxite ore by an eco-friendly way.
Substrate Concentration, Bioleaching of Metals, Acidithiobacillus ferrooxidans, Pseudomonas fluorescens, Aluminum, Iron
Some physical factors such as temperature, pH, humidity, aeration and particle size affect the metal extraction. It has been experimentally shown that if the amount of the enzyme is kept constant and the substrate concentration is then gradually increased, the reaction rate increases until it reaches a maximum. In bioleaching of metals, the pH of the solution is a critical factor in metal extraction because metals dissolve in acids (Karavaiko et al., 1980). The metals are extracted by using microbes which secrets acids (Shaikh and Ade, 2012). These microbes can thrive well even in the extreme conditions like low or high temperature or pH thus known as extremophiles (Hebert, R. A., 1992). Earlier reports suggest that microbes have immense potential in the bioleaching process. Therefore it will essential to explore the bacterial communities which are useful in enhancing the bioleaching process. The bacterium, Pseudomonas fluorescens has a lot of significant function for bioremediation. It can degrade the pesticide (Huerta et al., 2007), oil (Zahang et al., 2005), dyes (Barragan et al., 2007) and helps in bioremediation. Along with these properties, it is also able to extract the metals from the mineral ores. Brandl et al. (2008) shown that metal can be extracted from the solid waste material using Pseudomonas fluorescens. Maria et al. (2009), Cheng et al. (2009), Ansari and Malik (2007) used Pseudomonas species for the metal extraction from different sources. There was a need to observe the effect of pH on the extraction of metals as it affects the solubility of the metals in the medium in which bioleaching experiment was carried out by Shaikh et al., 2010.
Acidithiobacillus ferrooxidans is an acidophile, living in environments with an optimal pH range of 1.5 to 2.5 (Shaikh et al., 2010) and is also thermophilic, preferring temperatures of 40 to 45°C (Karavaiko et al., 1998). The high-temperature tolerance of the bacteria may be due in part to its high G+C content of the DNA 55 to 65-mole per cent (Karavaiko et al., 1998). As the Acidithiobacillus ferrooxidans is a metal extracting bacterium usually grown on 9K medium (Silverman and Lundgren, 1959) because the bacterium needs ferrous sulphate as a major constituent from which sulphuric acid is produced and the metals are dissolved in this acid. In addition, this bacterium has the ability to oxidize the Fe2+ to Fe3+ which results in the reduction of other metal ions (Karavaiko et al., 1998). It is also reported that Acidithiobacillus ferrooxidans have the iron-reducing activity, therefore, it can extract iron and other metals too (Sand, W., 1989). Previous studies focused on the bioleaching of Pb and Zn (Cheng et al., 2009), Cu, As, Ag, Mn and Zn (Nguyen. et al., 2015), Zn and Fe (Bayat et al., 2009), Ni and Co (Cwalina et al. 2000) and the effect of pH on metal extraction (Shaikh et al., 2010) by the bacterium is also studied. The work on the bioextraction of aluminum and iron from bauxite ore has not been much focused earlier.
Considering these facts the effect of substrate concentration (pulp density) on the metal extraction was studied in this investigation. Different substrate concentration was tried (2, 4, 6, 8, 10 and 12 w/v %), keeping other parameters such as the agitation time, agitation speed, temperature constant. Further, in the present work, the effect of pulp density of bauxite ore on the extraction of aluminum and iron was carried out employing the Pseudomonas fluorescens and Acidithiobacillus ferrooxidans.
Sample Collection
The low-grade bauxite ore sample was collected (0.5 – 0.8 Kg) from different sites and various depths of Bauxite mines Radhnagari, Kolhapur, India and immediately sealed with sterile polythene bags and brought to the laboratory. The collected sample was crushed and ground into fine particles and stored in the refrigerator at 40C until use.
Experimental Set Up For Acidithiobacillus ferrooxidans
To assess the effect of substrate concentration on the bioleaching of aluminum and iron the experiment was carried out in triplicate by shake flask method employed by Chen and Lin, (2001). Acidithiobacillus ferrooxidans was grown in 9K medium (Silverman and Lundgren, 1959) [composition g/l ammonium sulphate (NH4)2SO4 – 3.0, magnesium sulphate MgSO47H2O -0.5, potassium hydrogen phosphate K2HPO4 – 0.5, potassium chloride KCl- 0.1, calcium nitrate Ca (NO3)2 -0.01, ferrous sulphate FeSO4.7H2O- 21.00] and pH was adjusted to 2.0 with 10N H2SO4 and the medium was inoculated with 24 hours grown cells-approximately 5 x 106 cells/ml maximum cell biomass concentration. The culture was grown under a sterile condition in 250 ml Erlenmeyer flasks containing 100 ml of 9K medium on a rotary shaker at the constant agitation of 140 rpm and incubated at 36 0C ± 2 0C. After incubation of four days pre-weighed bauxite ore was added in Erlenmeyer flasks as1.0, 2.0, 3.0, 4.0, 5.0, 6.0, 7.0, 8.0, 9.0 and 10 gm/100 ml respectively. For control-the flask was containing the 9K medium only to find out whether this bacteria is capable to secret either aluminum or iron and both. All the experiments were run for one month. At the end of the experiment, the final pH was measured. The contents of each Erlenmeyer flasks were filtered through Whatman filter paper no.41. The extraction of aluminum and iron measured as per the methods described by Raquel et al. (2008) for aluminum and Demirhan and Fikriye (2007) for iron.
Experimental Set Up for Pseudomonas fluorescens
To study the effect of substrate concentration on the bioleaching of metals, the experiment was carried out by the shake flask method employed by Chen and Lin (2001). The medium was inoculated with 24 hours grown bacterial cells-approximately 5 x 106 cells/ml (maximum cell biomass concentration). The culture was grown under a sterile condition in 250 ml Erlenmeyer flasks containing 100 ml Kings Medium (1954) [composition g/l protease peptone-20, Glycerin -15ml, Magnesium sulphate MgSO47H2O -5.0, Di-Potassium hydrogen phosphate K2HPO4 – 2.5, Distilled water – 1000ml pH – 7.2] on a rotary shaker at the continuous agitation of 140 rpm. Further, it was incubated at 32 ± 2 0C and pre-weighed bauxite ore was added to Erlenmeyer flasks such as 2.0, 4.0, 6.0, 8.0, 10, and 12 g/100 ml. For control-the flask was containing the Kings Medium only to find out whether this bacteria is capable to secret either aluminum or iron and both the metals. All the experiments were run for 21 days. At the end of each experiment, the final pH was measured. The content of each Erlenmeyer flask was filtered through Whatman filter paper no.41. The extraction of aluminum and iron measured as per the methods described by Raquel et al. (2008) for aluminum and Demirhan and Fikriye (2007) for iron.
Estimation of Aluminum and Iron
The contents of the flasks were filtered and biomass of the bacterial cells and bauxite ore remained on the filter paper was weighed. At the termination of the experiment, analysis of bioextracted metals was carried out by the spectrophotometric method developed by Raquel et al. (2008) for aluminum and Demirhan and Fikriye (2007) for iron.
Aluminum was analyzed by taking 20 µl leached samples from the filtrate and added with 20 µl of 10 N H2SO4. The volume was raised up to 3ml with doubled distilled water. 500 µl of 15 % sodium acetate was added to it for adjusting reaction pH. 200 µl of 0.1 % ascorbic acid was added to overcome the interference of iron. Lastly, 200 µl of chrome azurol-s (0.04 % working solution diluted from a stock solution of 0.1 %) was added to it. The whole assembly was incubated for 10 min. for violate colour development, the absorbance was measured at 545 nm wavelength and for the standard curve- aluminum potassium sulphate was used.
The iron was detected from the filtrate by taking 20 µl leached sample and digested with 20 µl 10N H2SO4. The volume was raised up to 3 ml with doubled distilled water. 1ml of Hydroxylamine hydrochloride (10 %) and 8ml of sodium acetate (10%) and 10 ml of 1, 10 phenanthroline solution (0.1 %) were added to it. The whole mixture was diluted to 100 ml with distilled water and allowed to stand for 10 min. for developing a brick red colour. The absorbance was measured at 510 nm wavelength. The standard curve was prepared by taking ferrous ammonium sulphate.
Metal Extraction through Acidithiobacillus ferrooxidans
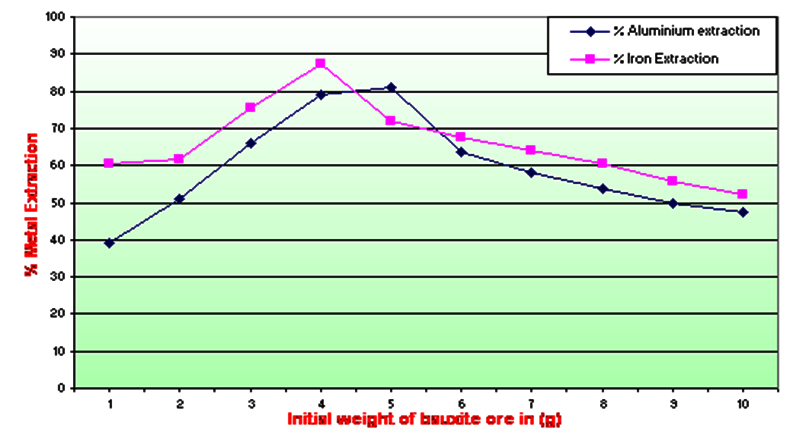
From the results depicted in Table 1 and Graph A, Acidithiobacillus ferrooxidans found suitable for extraction of iron and aluminum in presence of different concentrations of low-grade bauxite ore. 4 and 5 gm of the substrate (pulp) concentration was found to be better for the maximum iron and aluminum extraction and weighed 286.45±1.82 mg and 589.67±0.827 mg respectively. At the concentration of 1- 4 gm pulp density iron extraction was obtained in the range of 198.43±1.78 to 286.45±1.82 mg and percentage-wise it was 60.49% to 87.33%. Similarly, the pulp density of 1-5 gm showed the extraction of aluminum in the range of 284.66±0.981 to 589.67±0.827 mg and percentage-wise it was 39.10% to 80.99%. Further, it was noted that with the increase in the substrate concentration the extraction of metals was also got decreased. The final weight was noted with minimum 2.46 gm and maximum 2.90 gm. However, when both the aluminum and iron extraction efficiency was calculated, it was found between 45.74% – 81.60% at 1.0 gm to 4.0 gm substrate concentration respectively. The aluminum and iron content was compared with the control flask and it was found no aluminum and iron extracted using the bacteria. The pH of the medium also fell down to 0.7 and the bacterial growth was ceased.
Graph A. Effect of pulp density on extraction of metals from bauxite ore by Thiobacillus ferrooxidans
Metal Extraction through Pseudomonas fluorescens
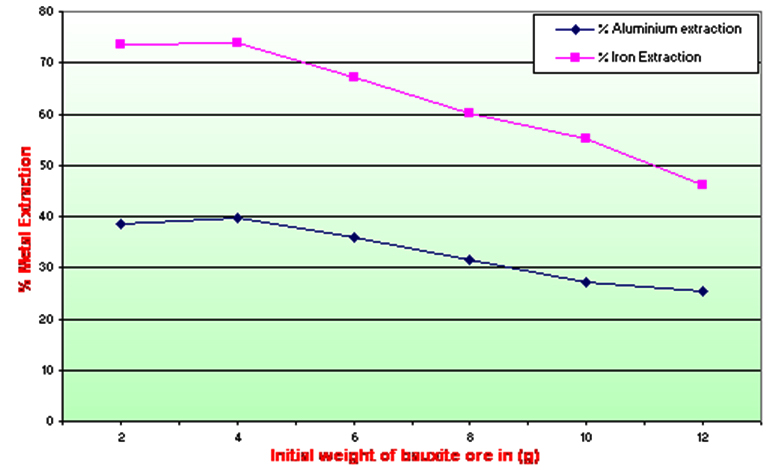
The results are shown in Table 2 and Graph B. The aluminum and iron extraction was higher with 288.34±1.23 and 242.66±1.42 mg respectively at 4 gm of bauxite ore concentration. The metal extraction percentage for aluminum and iron was found at 39.60% and 73.98% respectively. Other pulp density also showed aluminum extraction from 25.32% to 38.46 and iron extraction 46.23% to 73.57% at12 and 2 gm of bauxite ore concentrations respectively. These results also showed that the bioextraction process is affected by the increase in the substrate concentration. The minimum aluminum and iron extraction were found 184.33±1.47 and 151.64±1.23 mg at 12 gm with the metal extraction percentage of 25.32 and 46.23 respectively. The highest bioextraction efficiency of aluminum and iron was observed 50.28% at the 4 gm ore and also found to be most favourable for extraction of metals from low-grade bauxite ore. The pH of the medium decreased from 7.2 to 3.5. The amount of metal extracted employing the bacterium was significantly higher than the control flask. The statistical analysis was also done to find the credibility of results obtained i.e. significance values.
Table (1):
Effect of pulp density on extraction of metals from bauxite ore by Acidithiobacillus ferrooxidans.
| Initial wt of bauxite ore (g) | Bauxite ore (g) | Metal extracted (mg) |
Metal extracted (%) |
Bioextraction Efficiency of Al and Fe (%) | ||
|---|---|---|---|---|---|---|
| Final wt | Aluminum | Iron | Aluminum | Iron | ||
| 1 | 2.46 | 284.66±0.981 | 198.43±1.78 | 39.10 | 60.49 | 45.74 |
| 2 | 2.59 | 371.33±0.654 | 202.67±1.56 | 51.00 | 61.78 | 54.35 |
| 3 | 2.68 | 481.33±0.981 | 247.78±1.43 | 66.11 | 75.54 | 69.04 |
| 4 | 2.88 | 575.32±0.720 | 286.45±1.82 | 79.02 | 87.33 | 81.60 |
| 5 | 2.90 | 589.67±0.827 | 236.32±1.56 | 80.99 | 72.04 | 78.15 |
| 6 | 2.75 | 464.67±0.982 | 221.34±1.65 | 63.82 | 67.48 | 64.96 |
| 7 | 2.69 | 423.00±0.707 | 210.32±1.86 | 58.10 | 64.12 | 59.97 |
| 8 | 2.65 | 390.67±1.51 | 198.54±1.87 | 53.66 | 60.53 | 55.79 |
| 9 | 2.57 | 363.33±0.987 | 182.65±1.98 | 49.90 | 55.68 | 51.70 |
| 10 | 2.52 | 344.67±0.596 | 171.32±1.67 | 47.34 | 52.23 | 48.86 |
| C.D. (p=0.05) | 80.51 | 27.55 | ||||
Table (2):
Effect of pulp density on extraction of metals from bauxite ore by Pseudomonas fluorescens.
| Initial wt of Bauxite ore (g) | Bauxite ore (g) | Metal extracted (mg) |
Metal extracted (%) |
Bioextraction Efficiency of Al and Fe (%) | ||
|---|---|---|---|---|---|---|
| Final wt | Aluminum | Iron | Aluminum | Iron | ||
| 2 | 2.96 | 280.00±1.47 | 241.33±1.78 | 38.46 | 73.57 | 49.36 |
| 4 | 2.98 | 288.34±1.23 | 242.66±1.42 | 39.60 | 73.98 | 50.28 |
| 6 | 2.82 | 262.43±1.45 | 220.66±1.89 | 36.04 | 67.27 | 45.74 |
| 8 | 2.80 | 230.00±1.56 | 197.33±1.46 | 31.59 | 60.16 | 40.46 |
| 10 | 2.70 | 197.23±1.47 | 181.45±1.47 | 27.10 | 55.32 | 35.86 |
| 12 | 2.68 | 184.33±1.47 | 151.64±1.23 | 25.32 | 46.23 | 31.81 |
| C.D. (p=0.05) | 45.68 | 37.65 | ||||
In the present investigation, the magnitude of the substrate concentration was studied and it was observed that 4-5 g of bauxite ore concentration was found to be better for aluminum and iron extraction by Acidithiobacillus ferrooxidans and 4 gm proved the best with the Pseudomonas fluorescens bacterium respectively. Our results showed that as the pulp density decreased the bioleaching of these metals was increased. Similar results are also documented by Ballester et al. (1989) in copper bioleaching. It is because of the adaptation period of bacteria (David et al. (2017). According to Haragobinda et al (2014), the pulp density increases the adaptation to that density and requires a long period due to that metal bioleaching decreases. These results can be explained by the fact that as the solid-liquid ratio decreases, the conditions of the hydrodynamics of the fluid involved in the process improved and thus the metal leaching is enhanced. The diffusion of O2 and CO2 has a limiting effect on the development of the microorganisms. Due to more solid material in the medium, the availability of oxygen to the bacterium decreased hence the process become attenuated.
Cwalina et al. (2000) in their experiment observed that the concentration of the leached metals was considerably higher in the leaching solutions with higher pulp densities, although the further increase in the pulp densities caused a decrease in the yields of bioleached metals. Further, Brandl et al. (2008) also postulated that the pulp density has a major influence on bacterial growth and activity, whereas increased pulp density more than 50 g/l significantly affect the metabolic activity of bacteria. Pradhan et al. (2008) have studied different pulp concentration and also documented that the increasing pulp density decreases the metal extraction capacity due to poor diffusion of oxygen in the medium (as the strain used as an aerobic bacteria). They have also explained that the decrease in leaching beyond a certain limit can be attributed to improper contact of the medium with the ore particles. Vachon et al. (1994) studied different pulp concentration on bioleaching of red mud sludge by Acidithiobacillus ferrooxidans and revealed that 10 % and 15 % of pulp concentration inhibited the bacterial growth. The results found in this experiment showed the maximum bioextraction of iron at 4% pulp concentration and for aluminum at 5% by Acidithiobacillus ferrooxidans as compared to earlier studies. The present investigation also shows that the pH of medium decreased to 0.7 when the bioextraction process was carried out by the Acidithiobacillus ferrooxidans indicating the low pH favours for bioleaching of above metals.
Woznick and Huang (1982) indicated that the efficiency of metal dissolution is strongly influenced by the solid concentration of the leach slurry. Bayat et al. (2008) suggested that the increase in pulp density results in a decrease in per cent extraction of both Zn and Fe. The results obtained in the current study are similar to earlier studies. It might be due to increasing pulp density which created a condition with higher toxicity and shear stress and decreased the mass transferred that could result in the decreased per cent extraction of Al and Fe.
In the case of Pseudomonas fluorescens, we have not found any attempts made by the researchers for the bioextraction of aluminum and iron. In the present investigation pulp density, 2-4 % was found optimum for complete bioextraction of aluminum and iron from low-grade bauxite ore. The output was decreased gradually afterwards with an increase in the pulp density. The results are in agreement with the previous report of Park et al., 2006a. A large solid to liquid ratio increases the resistance for diffusion of fresh extractant to the surface of the catalyst (Wadood et al. 2011; Abhishek et al. 2012). Therefore, requires longer time and requires higher extractant concentration as concluded by Shariat et al. (2001). Duarte et al. (1990) in his experiment showed that pulp density may affect not only the hydrodynamic regime and reactor design but it also affects the microorganism’s behaviour. Hence the appropriate pulp density is required for enhancing metal extraction. The pH of the medium was also got down to 3.1 from 7.2.
The results of this study showed that the pulp density of the mineral ore influenced the extraction of metals. The 5 gm of bauxite ore concentration was found better for aluminum extraction by Acidithiobacillus ferrooxidans. Whereas, maximum iron extraction was obtained at 4 gm concentration. In the case of Pseudomonas fluorescens, aluminum extraction was higher at 4 gm bauxite ore concentration. Apart from the impact of pulp density the present study also showed that the bioextraction is an eco-friendly way for complete extraction of metals from low-grade mineral ores, as the ores are the nonrenewable source of different metals. The present results may also contribute to the extraction of these metals by using this appropriate concentration of bauxite ore for large-scale production in near future. We suggest that the results obtained in the current study have immense potential in the bioextraction of aluminum and iron from the low-grade bauxite ores.
ACKNOWLEDGMENTS
Authors are thankful to Professor and Head, Department of Botany, Dr. Babasaheb Ambedkar Marathwada University, Aurangabad for providing laboratory facilities.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
- Karavaiko, G. I., Z. A. Frutsko, E. O. Melnikova, Z. A. Avakyan and Y. I. Ostroushko. Role of microorganisms and some physico-chemical factors of the medium in quartz destruction. Microbiology-USSR, 1980; 49: 547- 549.
- Shaikh Shafikh and Avinash Ade Thiobacillus ferrooxidans: contribution of cells for metal extraction. International Journal of Biological and Biomedical Sciences, 2012; pp. 01-02.
- Hebert, R. A. A perspective on the biotechnological potential of extremophiles. Trends in Biotechnology, 1992; 10: 395-402. https://doi.org/10.1016/0167-7799(92)90282-Z.
- Huerta BEB, Perez CC, Cruz JP Cortes JB, Garcia FE and Vazquez RR.,. Biodegradation of organochlorine pesticides by bacteria grown in microniches of the porous structure of green bean coffee. Int Biodeterioration and Biodegradation, 2007; 59(3): 239-244. https://doi.org/10.1016/j.ibiod.2006.11.001.
- Zahang G L, Wu Y T, Qian X P and Meng Q. Biodegradation of crude oil by Pseudomonas aeruginosa in the presence of rhamnolipids. J. Zhejiang Univ.- Sci (B). 2005; 6(8): 725-730. https://doi.org/10.1007/BF02842430.
- Barragan Blanka E, Carlos Costa and Carman, Marquez M. Biodegradation of azo dyes by bacteria inoculated on solid media. Dyes and pigments, 2007; 75(1): 73-81. https://doi.org/10.1016/j.dyepig.2006.05.014.
- Brandl, H., Lehmann, S., Faramarzi, M.A. and Martinelli, D. Biomobilization of silver, gold and platinum form solid waste materials by HCN-forming microorganisms. Hydrometallurgy, 2008; 94(1): 14-17. DOI: 10.1016/j.hydromet.2008.05.016.
- Maria R, Silva P, Rodriguez A A, Montes J M, Oca D and Moreno DC. Biosorption of chromium, copper, manganese and zinc by Pseudomonas aeruginosa AT18 isolated from a site contaminated with petroleum. Biores Technol, 2009; 100 (4): 1533-1538. https://doi.org/10.1016/j.biortech.2009.06.057.
- Cheng, Yi, Guo Z., Liu, X., Yin, H., Qiu, G., Pan F. and Liu, H. The bioleaching feasibility for Pb/Zn smelting slag and community characteristics of indigenous moderate thermophilic bacteria. Biores. Technol, 2009; 100 (10): 2737-40. doi: 10.1016/j.biortech.2008.12.038.
- Ansari M. I., and Malik A. Biosorption of nickel and cadmium by metal resistant bacterial isolates from agricultural soil irrigated with industrial wastewater. Biores Techno, 2007; 98: 3149-3153. https://doi.org/10.1016/j.biortech.2006.10.008
- Shaikh, S. M., Khan, Z. S. and. Ade A. B. Effect of pH on Metal Extraction from Bauxite Ore by Thiobacillus Ferrooxidans. J. Sci. Res, 2010; 2 (2): 403-406 DOI: 10.3329/jsr.v2i24020
- Karavaiko, G. I., T. A. Pivovarova, L. N. Muntyan and T. F. Kondratyeva. Physiological and genetic characterization of Thiobacillus ferooxidans strains used in Biohydrometallurgy. Mineral processing and Extractive Metallurgy Review, 1998; 19(1): 1677- 1682. https://doi.org/10.1080/08827509608962438
- Silverman MP and Lundgren DG. Studies on chemoautotrophiic iron bacteria, Thiobacillus ferrooxidans: an improved medium and a harvesting procedure for securing high cellular yields. J. Bacteriol, 1959; 10 (77): 642-647.
- Sand, W. Ferric iron reduction by Thiobacillus ferrooxidans at extremely low pH-values. Biogeochemistry, 1989; 7: 195-201.
- Nguyen, Van Khanh., Lee, Mu Hyun., Park, Hyung Jun., Lee, Jong-Un. Bioleaching of arsenic and heavy metals from mine tailings by pure and mixed cultures of Acidithiobacillus spp. Journal of Industrial and Engineering Chemistry, 2015; 21: 451-458. https://doi.org/10.1016/j.jiec.2014.03.004
- Bayat, O., Sever, E., Bayat, B., Arslan, V. and Poole, C. Bioleaching of Zinc and Iron from Steel Plant waste using Acidithiobacillus ferrooxidans. Appl. Biochem. Biotechnol, 2009; 152(1): 117-126. doi: 10.1007/s12010-008-8257-5.
- Cwalina, B., Fischer, H. and Ledakowicz, S. Bacterial leaching of nickel and cobalt from Pentlandite. Physicochemical Problems of Mineral Processing, 2000; 34(1): 17-24.
- Chen, S.Y. and Lin, J.G. Effect of substrate concentration on bioleaching of metal contaminated sediment. J. Hazard. Mater, 2001; 82(1): 77-89. https://doi.org/10.1016/S0304-3894(00)00357-5
- Raquel, B., Mesquita, R. Antonio O. S. and Rangel, S. Development of Sequential Injection Methodologies for the Spectrophotometric Direct and Kinetic Determination of Aluminum in Natural and Waste Waters. J. Braz. Chem. Soc, 2008; 19 (6): 1171-1179.
- Demirhan, N. and Fikriye T. E. Spectrophotometric determination of iron (II) with 5-nitro, 6-amino -1, 10 phenathroline .Turk J. Chemistry, 2007; 27: 315-321.
- Ballester, A., Gonzalez, F., Blazquez, M.L. and Barril, M.A. Microbiological leaching of copper from lead mattes. Metallurgical Transactions, 1989; 20 B: 773-779. https://doi.org/10.1007/BF02670183
- David Lukumu Bampole, Patricia Luis and Emmanuel Lukumu Mulamba. Effect of substrates during the adaptation of indigenous bacteria in Bioleaching of Sulphide ores. American Scientific Research Journal for Engineering, Technology, and Sciences (ASRJETS), 2017; 32 (1): 200-214.
- Haragobinda Srichandan, Ashish Pathak, Dong Jin Kim, and Seoung-Won Lee. Effect of substrate concentration and pulp density on Bioleaching of metals from as received spent refinery catalyst World Academy of Science, Engineering and Technology. International Journal of Biotechnology and Bioengineering, 2014; 8 (8). 16th International Conference on Bioscience, Biotechnology and Biochemistry, Amsterdam, The Netherlands.
- Pradhan, D., Pal, S., Sukla, L.B., Chaudhury, G.R. and Das, T. Bioleaching of low-grade copper ore using indigenous microorganisms. Indian J. Chem. Technol, 2008; 15: 588-592.
- Vachon, P., Tyagi, R. D., Auchlalr, J. C. and Wilkinson, J. K. Chemical and biological leaching of aluminum from red mud. Environ. Sci. Technol, 1994; 28(1): 26-30. DOI: 10.1021/es00050a005
- Woznick, D. J. and Huang, J. Y. Variables affecting metal removal from sludge. J. Water. Poll. Contr. Fed, 1982; 54 (12): 1574-1580.
- Park, K.H., Mohapatra, D., Reddy, B.R., Nam, C.W. Hydrometallurgical processing and recovery of molybdenum trioxide from spent catalyst. International Journal of Mineral Processing, 2006; 80(2): 261–265. https://doi.org/10.1016/j.minpro.2006.05.002.
- Wadood T. Mohammed, Nada S. Ahmedzeki, Mariam F. Abdul Nabi. Extraction of Valuable Metals From Spent Hydrodesulfurization Catalyst By Two Stage Leaching Method. Iraqi Journal of Chemical and Petroleum Engineering, 2011; 12 (4): 21-35.
- Abhishek Tripathi, Manoj Kumar, D. C. Sau, Archana Agrawal, Sanchita Chakravarty and T. R. Mankhand. Leaching of Gold from the Waste Mobile Phone Printed Circuit Boards (PCBs) with Ammonium Thiosulphate. International Journal of Metallurgical Engineering, 2012; 1(2): 17-21. DOI: 10.5923/j.ijmee.20120102.02
- Shariat, M. H., Setoodeh, N., Atash Dehghan, R. Optimizing conditions for hydrometallurgical production of purified molybdenum trioxide from roasted molybdenite of Sarcheshmeh. Miner. Eng, 2001; 14: 815–820. https://doi.org/10.1016/S0892-6875(99)00000-X.
- Duarte, J. C., Estrada, P., Beaumont, H., Sitima, M. and Pereilra, P. Biotreatment of tailings for metal recovery. International Journal of Mine Water, 1990; 9(1-4): 193-206. https://doi.org/10.1007/BF02503692.
- Kings, E.O., Ward, M. K. M., and Raney, D. E., Two simple media for the demonstration of Pyocyanin and fluorescein. Lab. Clin. Med. 1954; 44: 301-307.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.