ISSN: 0973-7510
E-ISSN: 2581-690X
Phytase (myo-inositol-hexakisphosphate phosphohydrolase, (EC 3.1.3.8) has been purified from a novel isolated Bacillus cereus EME 48, from environmental samples based on their ability. The enzymes were purified and characterized, using a three-step purification procedure with 12.3-fold. The optimum pH for phytase activity was in the range of 4.0 to 5.0 and the optimum temperature was 40 to 60°C for 10 min. The metal ions MgCl2, CoCl2, NiCl2 and KCl2 did not show any inhibitory effect on the phytase activity of Bacillus cereus EME 48. Strongly inhibited by FeCl2, Na Florida, EDTA, DTT, AlCl3 and ZnCl2 but significantly stimulated by MgCl2, BaCl2, MnCl2 and CuCl2. The enzymes were active in the pH range of 5.0 to 6.5 with pH optima at 5.5. The enzyme from Bacillus cereus EME 48 retained about 80% activity up to 75°C. It was highly specific to sodium phytate as the substrate. Km for phytate was estimated to be 0.23 mmol and especially for penta- and tri-phosphate esters of myo-inositol. Due to their relatively high specific activity, substrate specificity, good pH profile and thermostability, the enzymes could be interesting candidate for agricultural and feed application.
Microbial phytase, Isolate, Bacillus cereus purified and characterized.
Phytate is of great interest in human nutrition, feed technology, food and medical science1. Researchers, as this interest has generated a wealth of relevant information. The negative effect of phytate on the availability of Ca and zinc, in food stuffs has been extensively investigated. Phytate in human diets is also claimed to have benefits, such as anti-carcinogenic effect2. Animal nutritionists have long regarded phytate as both indigestible and an anti-nutritional factor for non-ruminant animals3. Phytate is a polyanionic molecule with the potential to chelate positively charged nutrients, which is almost certainly fundamental to the anti-nutritive properties of phytate. These anti-nutritive properties require further investigation, but phytate probably compromises the utilization of protein/amino acids, energy, calcium and trace minerals. Phytase, which occurs widely throughout nature, is the requisite enzyme to degrade and release inorganic P4 If the practical acceptance of microbial phytase in poultry diets continues, it is likely that phytase feed enzymes will re-define nutrient requirements for sustainable poultry production in the future. Nevertheless, three decades elapsed before an Aspergillus niger-derived phytase feed enzyme, with the capacity to liberate phytate-bound P and reduce P excretion, was commercially introduced in 19985.
Phytic acid (myo-inositol 1,2,3,4,5,6 -hexakisdihydrogenphosphate) and mixed cation salts of phytic acid, designated as phytate, are a group of organic phosphorus (P) compounds found widely in nature especially in legumes, cereals, and oil seed crops6. Phytic acid, which is the main constituent of animal diet, is not digested by monogastric animals and hence, create problem in the availability of phosphorus in their diet. Various reported phytase-producing isolates and showed that the phytases with broader substrate specificity generally had low specific activities. Despite the considerable economic interest, low yield and high cost of enzyme production are the limiting factors in using this enzyme in animal diet. It also causes environmental pollution by extra supplemented phosphorus in animal’s diet7. Phytases are the primary enzymes responsible for the hydrolysis of phytic acid8. Hence, phytases are considered to be potential candidate for use as an enzyme that have great value in enhancing the nutritional quality of phytate-rich foods and feed9.
In recent years, bacterial phytases have been isolated, characterized and proposed as potential tools in biotechnology. The highest frequency of phytase-like proteins has been reported in members of the gamma-proteobacteria group. Pure culture studies of ruminal bacteria have demonstrated phytate- degrading activity in various strains belonging to the Enterobacteriaeceae family, such as in Enterobacter coli, Klebsiella, Citrobacter, Yersinia etc. Here, we report a comparative study on isolation, purification and characterization of phytases from two enteric bacteria 10.
Two big goals for this study, first is to produce the microbial phytase from new isolate microorganisms. Second, study carried out to purify and characterize phytase from Bacillus cereus EME 48. To our knowledge, this is a first report on the purification and prpoperties of phytase from Bacillus cereus EME 48.
Culture conditions and sample preparation
The following liquid media was used to cultivate the isolate bacteria Bacillus cereus EME 48contained (% w/v): Dextrose 0.5; peptone 1; yeast extract 0.5; MgSO4 0.1; CaCl2 0.1, sodium phytate 0.1 (pH 7.0) Kim et al11. The media contents were completed to one liter, then the pH was adjusted at 7.0, the bacteria was incubated at 30ºC for 5 days
Purification of crud extra cellular phytase produced by Bacillus cereus EME 48
Partial purification of phytase
The present experiment included the purification of extra cellular phytase using precipitation with ammonium sulphate, ethanol, and acetone or using ultra filtration methods according to the method of Greiner and Alminger (12)
Gel filtration of extra cellular phytase using sephadex G-100
Sephadex G-100 was used as gel filtration for the enzyme purification. 7 ml of extra cellular phytase obtained from ultra filtration contain 200 mg/ml protein was applied to a sephadex G-100 column chromatography (108 x 2.6 cm) which had been equilibrated with 0.1 M Tris buffer at pH 7.0 at 11°C. Elution was carried out with the same buffer. Five ml fractions were collected at flow rate of 33 ml/h.
Anion exchange of extra cellular phytase using DEAE- sephadex A50
Cellulose DEAE-52 was used as anion exchange for enzyme purification. Active Fraction (22-30) was obtained from Sephadex G-100 column chromatography while contained (8.8mg protein) was lyophilized and dissolved in 1 ml of 0.1 M tris buffer then applied to the top of DEAE Sephadex -A52 column chromatography (35 cm × 2.5 cm) previously equilibrated with 0.1 M tris buffer at pH 7.0 at 11°C.
At first, the unbounded protein was removed by washing with the same buffer, and then enzyme was eluted by gradient elution using sodium chloride (0.0 to 1.0 M) in 0.1M Tris HCl buffer at pH 7.0. The flow rate was adjusted to 50 ml/h and 5 ml fractions were collected from the effluent.
Phytase and protein assay
0.1 ml of the phytase sample was mixed with 1.24 ml acetate buffer containing 0.2% sodium phytate and incubated at 39°C for 15 min. The reaction was terminated by adding 1.25 ml 5% trichloroacetic acid (TCA)13. A unit of phytase activity is defined as the amount of enzyme that liberates 1ìmol P/min under the given assay conditions. Protein concentration was measured by the method of Bradford14 using a protein assay kit (Bio-Rad Lab., Richmond, CA) with bovine serum albumin as the standard.
Characterization of phytase activity
The purified phytase was used for phytase activity characterization. All tests were repeated three times, each with triplicates.
Effect of temperature on the stability of phytase enzyme
The thermal stability of the extra cellular purified enzyme was determined by storage the enzyme suspensions at different temperature in a water bath set (20-80 °C) for different times (0 minuts-24 hours), then the enzymes activity were determined in each case.
Effect of pH on the phytase stability
In this experiment, 1 ml of 0.15 M acetate buffer at pH values ranged between 3.5-6 were stored at room temperature with 1 ml of purified phytase enzyme solutions. The storag time was between 0 min. – 48 hours. The remains enzyme activity was then determined at 55 °C and pH 5.5. Control was carried out using pH 5.5.
Effects of ions on phytase activity
In this experiment, the metal ions used in the experiment were MgCl2, MnCl2, ZnCl2, BaCl2, CuCl2, AlCl3, CaCl2, CoCl2, FeCl2, NiCl2, KCl2, MgCl2, Na Florida, EDTA and DTT were added in concentrations 1 and 2 mM to the reaction mixture to study their effect on the phytase activity. The control was carried out without adding activators or inhibitors
Effect of different concentrations of substrate on the activity of purified extra cellular phytase
The effect of substrate concentrations on the purified extra cellular phytase was studied. Different concentrations of Na phytate ranging from 1.5 to 5 mM in 0.15 M acetate buffer at pH 5.5, the reaction mixture was added to 150 ml of enzyme solution then it was incubated for 40 min. at 55°C and stopped by 0.75 ml of 5% trichloroacetic acid then the enzyme activity was measured as mentioned before
Fractional precipitation with ammonium sulphate
It is clear from the data presented in table (1) that, ammonium sulphate at concentration (30-60) gave activity for phytase enzyme 316 U/ml and protein 3 mg/ ml with specific activity 105
Precipitation with ethanol and acetone
Precipitation using acetone and ethanol also mad and the results is shown in table (1). It is clear that ethanol (0-30) gave high specific activity than acetone to precipitation phytase enzyme.
Table (1):
Partial purification of phytase enzyme produced by Bacillus cereus EME 48.
| Purification steps | volume (ml) | Total protein | Protein recovery (%) | Total units | Units recovery (%) | Specific activity | Purification fold | |
|---|---|---|---|---|---|---|---|---|
| Culture filtrate | 50 | 390 | 100 | 38300 | 100 | 98 | 1 | |
| Acetone | 0-30 | 50 | 100 | 25.6 | 9050 | 23 | 90 | 0.9 |
| 30-60 | 50 | 190 | 48.7 | 20050 | 52 | 105 | 1.07 | |
| 60-90 | 50 | 22.2 | 5.5 | 821.4 | 2.1 | 37 | 0.37 | |
| Ethanol | 0-30 | 50 | 109 | 28 | 12400 | 32 | 113 | 1.1 |
| 30-60 | 50 | 200 | 51 | 17600 | 46 | 88 | 0.9 | |
| 60-90 | 50 | 00 | 00 | 00 | 00 | 00 | 0.0 | |
| Ammonim sulphate | 0-30 | 50 | 195 | 50 | 17600 | 46 | 90 | 0.92 |
| 30-60 | 50 | 150 | 38 | 15800 | 41 | 105 | 1.07 | |
| 60-90 | 50 | 00 | 00 | 00 | 00 | 00 | 00 | |
| Culture filtrate | 2000 | 15600 | 100 | 1532000 | 100 | 98.20 | 1 | |
| Ultra filtration | 50 | 10000 | 64.10 | 1550000 | 101 | 155 | 1.58 | |
Precipitation using ultra filtration
The present series of experiment were conducted to prepare partially purified phytase. These include isolation of active enzymes by ultrafilteration. The culture filtrate of about 2 liter was passed through Pellicon Cassette system with a membrane PLGC Cassette 10.000 NMWL. Low protein-binding.
The two liter was concentrated to about 50 ml which contain 10000 mg of protein was recovered which represent about 64.10 % of the total protein while 1550000 U of phytase enzyme was recovered which represent about 101 % of the total activity.
The results in fig (1) illustrated that, two peak for phytase activity (I) and (II) of protein components were present with different molecular weight. The first peak was at fraction22-30 and the second one was at36-41. The enzyme activity was detected in the first peak. The total enzyme activity was 14440 U/min at 40 ml of fraction22-30 the total protein was 8.8 mg. the specific activity reached to 1640.9 U/mg proteins. The total activity of the second peak was 7350 U/min at 25 ml of fraction36-41 and the total protein was 4.84 mg the specific activity reached to 1518 U/min.
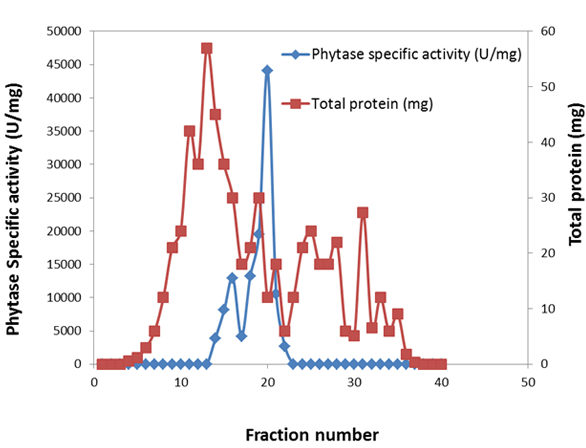
Fig. 1. Purification of extra cellular phytase by using sephadex G-100.
The results in fig (2) showed that, the enzyme activity was 138 U/min at 25 ml of fraction31-35 the total protein was 0.05 mg. the specific activity reached to 2760 U/mg protein. Fractions31-35 were chosen for check their purity on polyacrylamide gel disc electrophoresis phytase where the total activity was U/ml and the specific activity was 2760 U/mg protein.
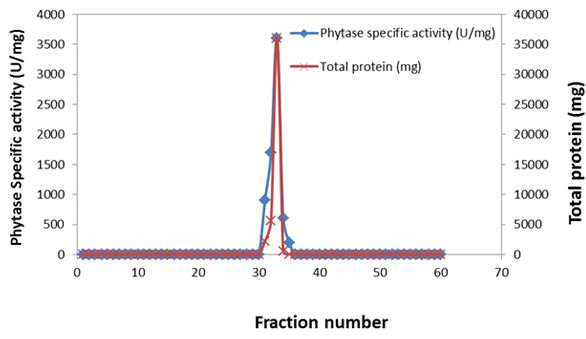
Fig. 2. Purification of extra cellular phytase by using DEAE- sephadex A50
Summarized purification steps of phytase
Typical purification steps of phytase from Bacillus cereus EME 48 are summarized in table (2). The cultural filtrate obtained after centrifugation of the fermentation broth was subjected to ultra filtration using Pellicon Cassette system with a membrane PLGC Cassette 10.000 NMWL, low protein-binding. This step yielded 1.58 fold of purification. It was it was chromatographer on sephadex G100 column and the enzymes were eluted with tris HCl buffer of pH 7.0.
On the other hand, the phytase was purified approximately 16 fold from the culture filtrate with a specific activity of about 1640 U/mg proteins. The active fractions from the Sephadex G100 Column22-30 were pooled and concentrated to one ml and it was then applied on DEAE-Sephadex A-50 column.
Table (2):
Purification steps of phytase.
| Purification steps | Volume (ml) | Total protein | Protein Recovery (%) | Total activity (U) | Unit’s recovery (%) | Specific activity | Purification fold |
|---|---|---|---|---|---|---|---|
| Culture filtrate | 40 | 312 | 100 | 30640 | 100 | 98.2 | 1 |
| Ultra filtration | 1 | 200 | 64.1 | 31000 | 101 | 98.2 | 1.58 |
| Sephadex G-100 | F(1-21) | 145.8 | 46.5 | 00 | 00 | 00 | 00 |
| F(22-30) | 267 | 85.5 | 11900 | 38.9 | 44.5 | 0.45 | |
| F(31-35) | 81 | 26 | 00 | 00 | 00 | 00 | |
| F(36-60) | 119.1 | 38 | 00 | 00 | 00 | 00 | |
| DEAE Sephadex A-50 | F(1-30) | 4.45 | 1.4 | 00 | 00 | 00 | 00 |
| F(31-35) | 5.8 | 1.9 | 7010 | 22.9 | 1209 | 12.3 | |
| F(36-62) | 9.1 | 2.9 | 00 | 00 | 00 | 00 |
By this procedure phytase was purified approximately 28 fold from the culture filtrate. A protein sample of each step of purification was examined by polyacrylamide gel electrophoresis Fig (3). Phytase resulted from sephadex G100 column chromatography was migrated with other proteins, but phytase resulted from DEAE sephadex column chromatography was also migrated as single protein band on polyacrylamide gel electrophoresis.
(1) Crude enzyme, (2) partially purified enzyme by ultra filtration, (3) the enzyme of phytase separated on sephadex G 100, (4) the enzyme of phytase separated on DEAE sephadex A 50.
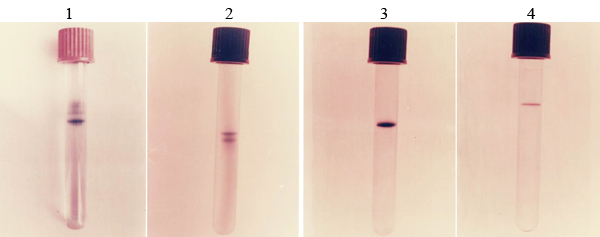
Fig. 3. Purification steps of phytase
Effect of temperature on the stability of phytase enzyme
The results in tables (3) showed that the stability of phytase depending on the temperature and the storage time. In most cases, the loss in activitiey was increased by increasing the temperature and the storage time.
The phytase enzyme was stable at temperature 20, 30, 40 C after 24 hours. At 50°C the enzyme lost about 8% of its activity after 90 min. At 65 °C and 70 °C the enzyme activity was decreased to 91% and 83% respectively after 5 min.
Table (3):
Effect of temperature on phytase enzyme stability.
| Time Temp.°C | Phytase Relative activity (%) | |||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Minutes | Hours | |||||||||||||
| 0 | 5 | 10 | 20 | 30 | 40 | 50 | 60 | 90 | 2 | 4 | 6 | 12 | 24 | |
| 20 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| 30 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| 40 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| 50 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 92 | 75 | 65 | 58 | 54 | 46 |
| 55 | 100 | 100 | 100 | 100 | 100 | 100 | 97 | 91 | 80 | 70 | 61 | 45 | 26 | 0 |
| 60 | 100 | 100 | 100 | 88 | 77 | 65 | 56 | 47 | 41 | 36 | 29 | 17 | 11 | 0 |
| 65 | 100 | 91 | 82 | 73 | 64 | 53 | 44 | 39 | 32 | 27 | 20 | 0 | 0 | 0 |
| 70 | 100 | 83 | 76 | 70 | 62 | 56 | 30 | 22 | 0 | 0 | 0 | 0 | 0 | 0 |
| 75 | 100 | 35 | 27 | 21 | 12 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 80 | 100 | 17 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
Effect of pH on the phytase
The results recorded in tables (4) indicated that the stability of phytase, in absence of substrate depended on the pH values. The phytase enzyme was stable at pH 5.5 to 6 for 48 hours. The enzyme was stable at pH 5.0 till 60 min and partially loss its activity with increasing the time of storage. At pH values 3.5, 4, 4.5 phytase enzymes lost its activity after 60 min., 8 hour and 16 hours respectively.
The phytase enzyme was stable at pH values ranged between 5.5 to 6.0.
Table (4):
Effect of temperature on phytase enzyme stability.
| Time/pH | Phytase Relative activity (%) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Minutes | Hours | ||||||||||
| 0 | 15 | 30 | 45 | 60 | 2 | 4 | 8 | 16 | 24 | 48 | |
| 3.5 | 69 | 48 | 24 | 11 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 4 | 78 | 62 | 49 | 31 | 20 | 9 | 7 | 0 | 0 | 0 | 0 |
| 4.5 | 91 | 85 | 76 | 64 | 48 | 30 | 22 | 15 | 0 | 0 | 0 |
| 5 | 100 | 100 | 100 | 100 | 100 | 76 | 53 | 24 | 13 | 9 | 0 |
| 5.5 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
| 6 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 | 100 |
Effect of different concentrations of different activators and inhibitors on the phytase activitie
The results recorded in table (5) showed that the enzymes activity was greatly inhibited by FeCl2, Na Florida, EDTA, DTT, AlCl3 and ZnCl2. On the other hand the addition of MgCl2, BaCl2, MnCl2 and CuCl2 showed moderately inhibited for the phytase, while, MgCl2, CoCl2, NiCl2 and KCl2 had no effect on the both enzymes. Moreover, CaCl2 made a slight increase in the activity of phytas when added at concentration in 1 mM, the activity was decreased with decreasing the concentration.
Table (5):
The effect of different concentrations of different activators and inhibitors on the phytase.
| Additive | Phytase Relative activity % | |
|---|---|---|
| 1mM | 2mM | |
| Control | 100 | 100 |
| CaCl2 | 101 | 100 |
| KCl2 | 99 | 98 |
| CoCl2 | 99 | 98 |
| NiCl2 | 97 | 98 |
| MgCl2 | 99 | 97 |
| CuCl2 | 96 | 95 |
| MnCl2 | 95 | 93 |
| BaCl2 | 88 | 82 |
| MgCl2 | 85 | 81 |
| AlCl3 | 83 | 79 |
| ZnCl2 | 79 | 77 |
| DTT | 80 | 73 |
| EDTA | 77 | 65 |
| Na fluoride | 71 | 62 |
| FeCl2 | 70 | 53 |
Effect of different concentrations of substrate on the activity of purified extra cellular phytase
The results recorded in fig (4) showed a parallel relationship existed between substrate concentration and phytase Specific activity up to 3 mM (3273 U/mg protein). Higher concentration had pronounced inhibition on enzyme activity.
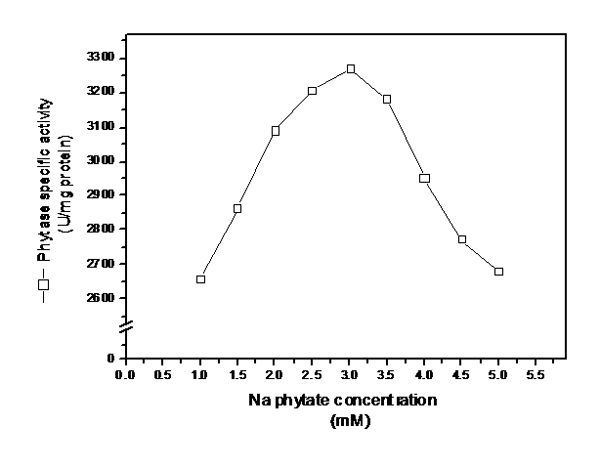
Fig. 4. Effect of different concentrations of substrate (Na phytate) on the activity of purified extra cellular phytase
Kinetic parameters (Km and Vmax values) of purified extra cellular phytase and acid phosphatase produced by Bacillus cereus EME 48…
These were determined for the purified extra cellular phytase and acid phosphatase by using the natural substrate (Na phytate) and then determine from Line weaver-Burk plot. Na phytate concentrations ranged from 1.5-5 mM dissolved in 0.15 M acetate buffer (pH 5.5) were used. The activities were assayed at 55 C for 40 min. and the reaction was stopped by 0.75 ml of 5% trichloroacetic acid. The results in fig (5) showed that the Km value was 0.23 mM and Vmax value was 3278 mM/mg protein.
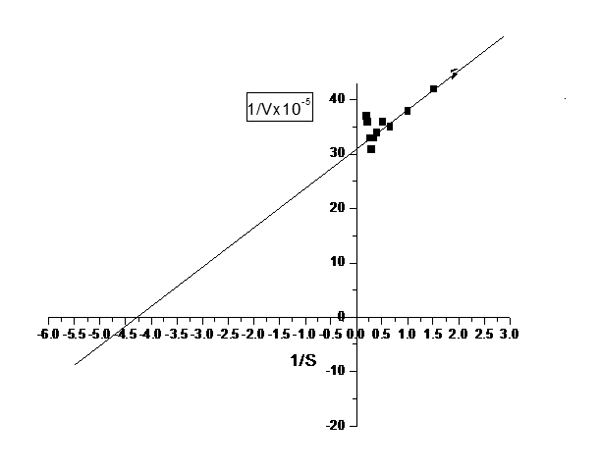
Fig. 5. Line weaver-Burk plot for purified exracellular phytase produced by Bacillus cereus EME 48 by using (Na phytate) as a substrate
This study showed that Bacillus cereus EME 48 could be a promising novel bacterial source of phytase. The properties of the phytase from Bacillus cereus EME 48 are favorable for it to be used as an enzyme for improving the availability of phytate phosphorus and minerals in feedstuff for non-ruminants. Phytase activities have been reported in a wide range of seeds, animal tissue and microbes.
First attempts to separate phytase activity from the major acid phosphatase activity by the one-step procedure described by Gibson and Ullah (2) for phytase purification were unsuccessful. The gel filtration methods are based on the principle that different molecules can be separated on the basis of differences in their size by passing them through a column containing swollen particles of a gel. This procedure was based on the difference of isoelectric points of enzyme. When chromatographed on a strong cationic-exchanger (SP-Trisacryl) column at pH 4.5, soybean phosphatase was retained on the column, whereas phytase was excluded. Under the same conditions, enzyme activities were retained and eluted at the same saline concentration. A three-step purification procedure is described, consisting of ultra-fractionation, gel filtration and anion-exchange chromatography. Phytase was completely separated from the major acid phosphatase activity after the third step.
The optimum temperature for phytase activity for Bacillus cereus EME 48 was 40 to 60°C. Although the optimum temperature for phytase activity of Selenomonas ruminantium JY35, an anaerobic rumen bacterium, is also 55°C, the enzyme activity declines dramatically at 60°C (15). The optimum temperatures of phytase for most micro-organisms are in the range of 40 to 60°C. High optimum temperatures for phytase activity have been observed in bacteria such as Klebsiella aerogenes (60 to 70°C) (16), and Bacillus sp. DS11 (70°C) (10). Among yeasts, Schwanniomyces castellii showed maximum phytase activity at 77°C (17), while Arxula adeninivorans and Pichia spartae at 75 to 80°C, and Pichia rhodanensis at 70 to 75°C (18). Phytase of Aerobacter aerogenes exhibited the lowest optimum temperature at 25°C (19).
The optimum pH of phytase activity of M. jalaludinii was in the range of 4.0 to 5.0. The activity declined significantly above pH 5.5. The moderately acidic pH optimum of M. jalaludinii phytase indicates that this enzyme belongs to the acidic phytases, as are most of the so far characterized phytases of microorganisms: Selenomonas ruminantium JY35, pH 4.0 to 5.5 (15); E. coli, pH 4.5 (20) and all yeast strains studied by Nakamura (19), pH 3 to 5.5. These pH optima are different from those of other bacterial phytases, such as pH 6.5 for Bacillus subtilis (natto) N-77 (21), pH 7.0 for Bacillus subtilis VTT E-68013 (5) and Bacillus sp. DS11 (10). When the enzyme was incubated in more acidic buffers of pH 3.0 or less, about 34 to 96% of activity was lost.
The results from this study showed that phytase from Bacillus cereus EME 48 belongs to the second class since it is highly specific to sodium phytate and has very little or no activity on other phosphate esters under the given assay conditions, indicates that the phytase is different from the general acid phosphatase. The metal ion chelating agents, namely MgCl2, CoCl2, NiCl2 and KCl2 did not show any inhibitory effect on the phytase activity of M. jalaludinii. Therefore, this enzyme, like many other phytases is not a metallo-enzyme, the study of the effect of metal ions on enzyme activity revealed that phytase from M. jalaludinii displayed a pattern of cation sensitivity similar to those of E. coli (20), Klebsiella terrigena (22) and Selenomonas ruminantium JY35 (15). The most significant inhibitory effect was by iron cation. This has also been commonly observed in many phytases from various sources.
However, the study with Selenomonas ruminantium JY35 phytase, Yanke (15) found that precipitates were also obtained with Ba2+ and Pb2+, but Ba2+ did not inhibit the phytase activity and Pb 2+ significantly stimulated the activity.
In the present study, it was found that 62 and 99% of phytase activity of Bacillus cereus EME 48 was inhibited by 1 and 2 mmol/l chloride supplemented to the assay mixture, respectively. However, Kim (23) reported that the phytase activity of Bacillus amyloliquefaciens was not inhibited by 5 mmol/l phosphate in assay mixture. The results from this study support the suggestion of Yanke (13) that phytate is readily hydrolysed by bacteria in the rumen, even though the inorganic phosphate concentration in the rumen fluid can be as high as 14 mmol/l when the animal is fed with concentrate diet.
Similar results have been reported by Wyss (24) pointed out that on the basis of substrate specificity, phytases could be classified into two classes: (i) phytases with broad substrate specificity such as those from A. fumigatus, Emericella nidulans, Myceliophthora thermophila (24), canola seed (25), germinated oat (26), rye (27) and barley (26); and (ii) phytases with narrow substrate specificity, which are very specific for phytate, such as those from A. niger, A. terreus, E. coli (Wyss et al., 1999), Bacillus sp. DS11 (11) and Bacillus subtilis (natto) N-77 (21).
- Hara A., Ebina S., Kondo A. and Funaguma T. A new type of phytase from pollen of Typha latifolia L. Agric Biol Chem, 1985; 49: 3539-44.
- Ullah A.H. and Gibson D.M. Extracellular phytase (E.C.3.1.3.8) from Aspergillus ficuum NRRL 3135: purification and characterization. Prep Biochem. 1987; 17: 63–91.
- Sandberg A.S. and Anderson R. A high performance liquid chromatographic method for determination of inositol triphosphates, tetraphosphates, pentaphosphates and hexaphosphates in foods and intestinal contents. J Food Sci, 1986; 51:547-550.
- Dvorakova J. Phytase: Sources, preparation and exploitation. Folia Microbiol. 1998; 43: 323-338.
- Kerovuo J., Lauraeus M., Nurminen P., Kalkkinen N. and Apajalahti J. Isolation, characterization, molecular gene cloning and sequencing of a novel phytase from Bacillus subtilis. Appl Environ Microbiol. 1998; 64: 2079–2085.
- Hill J. E., Kysela D. and Elimelech M. Isolation and assessment of phytate-hydrolysing bacteria from the DelMarVa peninsula. Environ. Microbiol. 2007; 9: 3100-3107.
- Scott J.J. Alkaline phytase activity in nonionic detergent extracts of legume seeds. Plant Physiol. 95:1298-3018-Pizzolon L. 1996. Importance of Cyanobacteria as potential factor of toxicity in continental waters. Interciencia. 1991; 21: 239-245.
- Pizzolon L. Importance of Cyanobacteria as potential factor of toxicity in continental waters. Interciencia. 1996; 21: 239-245.
- Lan G. Q., Abdullah N., Jalaludinand S. Ho Y. W. Purification and characterization of a phytase from Mitsuokella jalaludinii, a bovine rumen bacterium. African Journal of Biotechnology, 2011; 10(59), 12766-12776
- Palaciosa M.C., Harosa M., Sanzb Y. and Rosella CM. Selection of lactic acid bacteria with high phytate degrading activity for application in whole 2007.
- Kim YO, Kim HK, Bae KS, Yu JH, Oh TK. Purification and properties of a thermostable phytase from Bacillus sp. DS11. Enzyme Microbial. Technol., 1998; 22: 2-7.
- Greiner R, Jany KD, Alminger ML. Identification and properties of a phytate-degrading enzymes from barley (Hordeum vulgare). J.Cereal Sci., 1999; 30: 11-17.
- Yanke LJ, Bae HD, Selinger LB, Cheng K-J. Phytase activity of anaerobic ruminal bacteria. Microbiology, 1998; 144: 1565-1573.
- Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein using the principle of protein-dye binding. Analy. Biochem., 1976; 72: 248-252.
- Yanke LJ, Selinger LB, Cheng KJ. Phytase activity of Selenomonas ruminantium: a preliminary characterization. Lett. Appl. Microbiol., 1999; 29: 20-25.
- Tambe SM, Kaklij GS, Kelkar SM, Parekh LJ. Two distinct molecular forms of phytase from Klebsiella aerogenes: Evidence for unusually small active enzyme peptide. J. Ferm. Bioeng., 1994; 77: 23-27.
- Segueilha L, Lambrechts C, Boze H, Moulin G, Galzy P. Purification and properties of the phytase from Schwanniomycescastellii. J. Ferm. Bioeng., 1992; 74: 7-11.
- Greaves MP, Anderson G, Webley DM. The hydrolysis of inositol phosphates by Aerobacter aerogenes. Biochim. Biophys. Acta., 1967; 132: 412-418.
- Nakamura Y, Fukuhara H, Sano K. Secreted phytase activities of yeasts. Biosci. Biotechnol. Biochem., 2000; 64: 841-844.
- Greiner R, Konietzny U, Jany KD. Purification and characterization of two phytases from Escherichia coli. Arch. Biochem. Biophys., 1993; 301: 107-113.
- Shimizu M. Purification and characterization of phytase from Bacillus subtilis (natto) N-77. Biosci. Biotechnol. Biochem., 1992; 56: 1266-1269.
- Greiner R, Haller E, Konietzny U, Jany KD. Purification and characterization of a phytases from Klebsiella terrigena. Arch.Biochem. Biophys., 1997; 341: 201-206.
- Kim DH, Oh BC, Choi WC, Lee JK, Oh TK. Enzymatic evaluation of Bacillus amyloliquefaciens phytase as a feed additive. Biotechnol. Lett., 1999; 2: 925-927.
- Wyss M, Brugger R, Kronenberger A, Rémy R, Fimbel R, Oesterhelt G, Lehmann M, van Loon APGM. Biochemical characterization of fungal phytases (Myo-inositol hexakisphosphate phosphohydrolases): Catalytic properties. Appl. Environ. Microbiol., 1999; 65: 367-373.
- Houde RL, Alli I, Kermasha S. Purification and characterization of canola seed (Brassica sp.) phytase. J. Food Biochem., 1990; 14: 331-351.
- Greiner R, Alminger ML. Purification and characterization of a phytate-degrading enzyme from germinated oat (Avena sativa). J. Sci. Food Agri., 1999; 79: 1453-1460.
- Greiner R, Konietzny U, Jany KD. Purification and properties of a phytase from rye. J. Food Biochem., 1998; 22: 143-161.
© The Author(s) 2016. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


