ISSN: 0973-7510
E-ISSN: 2581-690X
An aqueous extract of the shade-dried parts of Asparagus aethiopicus L. and Asparagus densiflorus (Kunth) Jessop was used to synthesize copper (CuO) nanoparticles (Cu NPs). UV-visible spectroscopy was performed to assess the formation of Cu NPs. Maximum absorbance was obtained between 310–360 nm, confirming the formation of Cu NPs. CuO NPs were effective against Pseudomonas aeruginosa (ATCC 27853), a gram-negative human pathogenic bacterium. The bacterial growth curve showed that the NPs inhibited bacterial growth. This study was conducted using different parts of the plant, such as the roots, shoots, fruits, and seeds of A. aethiopicus and the roots and shoots of A. densiflorus. The fruit and seeds of A. aethiopicus showed better results than the roots and shoots. Therefore, employing green synthetic processes inhibits the release of harmful by-products. Compared to traditional techniques of NP synthesis, the transition to the extracellular production of CuO NPs utilizing dried biomass appears to be economical and environmentally beneficial.
Green synthesis, CuO, nanoparticles, Asparagus aethiopicus, Asparagus densiflorus, Pseudomonas aeruginosa
Particles 1–100 nm in size are referred to as nanoparticles (NPs). These NPs may be formed from carbon, metal, metal oxides, or organic components. NPs show physical, chemical, and biological properties that are different from those of their parent particles on a large scale.1 In the contemporary research, nanotechnology plays a dominant role in exhibiting anticancer and antimicrobial properties against bacteria. The development of environmentally acceptable processes for the synthesis of NPs without the use of hazardous chemicals is becoming increasingly important. Compared to larger particles, NPs display entirely new and better features. Biological techniques involving microorganisms and plant extracts have been proposed as viable alternatives to chemical methods of metal NP production. Because complex procedures such as intracellular synthesis are avoided, using plant resources in several purification processes and upkeep of microbial cell cultures for the creation of NPs may be more beneficial.2
Biological techniques use microorganisms and medicinal plants to create NPs. The benefit of using medicinal plants is that they impart therapeutic characteristics to NPs during manufacturing. Plant phytocompounds give the NPs antioxidant properties, whereas plants with antibacterial activity give them additional antioxidant and antibacterial properties.2 Metal NPs are among the types of NPs that are beneficial in biomedical applications, including imaging the delivery of biological activity, drug transport, catalysis and photo dynamics, and photothermal treatment.3
In recent years, the green production of metallic NPs has emerged as a new and exciting area of study. Creation of NPs using green methods has become more popular in recent years owing to its many benefits, including ease of use, low cost, high stability, quick production, lack of harmful by-products, and capacity to be scaled up for large-scale synthesis.4 Green synthesis to produce different metal and metal oxide NPs has drawn attention because chemical synthesis techniques result in the presence of harmful chemical species absorbed on the surface of NPs. These metal NPs are shown to be more efficient and dependable when produced using green synthesis methods.4
Because of their unique features and use in numerous domains of science and technology, metallic NPs synthesized using green methods have attracted enormous interest over the past decades. It is superior to chemical and physical syntheses because it is more affordable, environmentally friendly, and easily scaled-up for large-scale syntheses. It also eliminates need for toxic chemical, high temperature, energy-intensive processes, and synthetic production; is inexpensive; and have numerous scientific uses.5 The purpose of the study is to describe the benefit of various types of biomolecules as dependable, long-lasting, and environmentally acceptable building blocks for the synthesis of metal and metal oxide NPs.4 For medical and biological applications, where the purity of NPs is crucial, biogenic reduction in metal precursors to create matching NPs is more cost effective, chemically free, and environmentally benign. NPs show effective antibacterial activity against harmful bacteria, including Klebsiella aerogenes and Staphylococcus aureus.6
Nanomaterials produced via green syntheses are crucial for the use of nanotechnology in various industries. Green nanoproducts, and their use in promoting sustainable development, are referred to as “green technologies.” Chemical processes are most commonly used for creating NPs. However, chemical techniques cannot prevent the presence of harmful compounds in the synthetic pathway. Because metal NPs are frequently used in areas where people come in contact with them, there is an increasing need to develop ecologically safe NPs synthesis techniques that do not include hazardous chemicals. Environmentally friendly alternatives to chemical and physical processes for creating NPs have been proposed, including microbes, enzymes, and plant extracts.4
Compared to other biological processes, using plants for NP synthesis is advantageous because it does not require a laborious process to maintain cell cultures and can be appropriately scaled up for large-scale NP creation.4 Plant extracts are used in the quick, low-cost, and environmentally friendly biogenic production of metallic NPs. Because of their non-toxic nature and special qualities, copper NPs have been employed in several cutting-edge biomedical applications, such as in vitro and in vivo cancer treatment, gene transport, tissue engineering, and biological half-life investigations. In preclinical cancer models, nanomedicine has shown exceptional success, yet only a few anticancer nano-drugs have received an approval from regulatory agencies.3 Although just a few studies have examined the antibacterial abilities of copper NPs, they have great potential as bactericidal agents.4
Studies have shown that copper NPs outperform silver NPs in tests comparing their antibacterial properties using Escherichia coli and Bacillus subtilis. In this study, a straightforward and environmentally friendly method was used to create copper oxide NPs that included aqueous extracts of Asparagus aethiopicus and Asparagus densiflorus. Spectral and bioactivity studies were used to describe the products of CuO NPs.3
Chemical processes such as chemical reduction, electrochemical methods, microwave-aided processes, and green chemistry are currently used to create and stabilize metal NPs. The use of plants for NP synthesis is a novel approach. This method is superior to chemical and physical methods because it is economical and environmentally friendly and can be easily scaled up for large-scale synthesis. It also avoids the use of toxic chemicals, high pressure, high energy, and high temperatures. Although bacteria and fungi can be utilized to make NPs, using leaf extract lowers the cost and eliminates the need for specialized culture preparation and separation methods.3
Use of copper and copper oxide
Metallic NPs play an important role in plant health and nutrition as excellent antimicrobial agents and catalysts. Cu particles in plants stimulate the photosynthetic rate at low concentrations. DNA damage and lesions are treated with CuO NPs. The phytotoxic effect increases, which influences plant growth and plays a significant role in chlorophyll synthesis for the metabolism of carbohydrates and proteins. It is ubiquitously used as an antimicrobial agent cost-effective compared to other particles and is highly effective against microorganisms, such as Pseudomonas aeruginosa, S. aureus, and Candida albicans.7
Asparagus based on Asparagus officinalis L. was recognized as a genus by Linnaeus in “Species Plantarum” (1753). This genus comprises more than 200 species, but the use of Asparagus species in humans is limited. Some species of Asparagus are used as ornamental plants (Asparagus asparagoides, Asparagus scandens, Asparagus plumosus, and Asparagus falcatus). A. officinalis is an important vegetable cultivated worldwide. Asparagus racemosus (known as Shatavari in Sanskrit) and Asparagus cochinchinensis are extensively used in Ayurvedic and Oriental medicine. With more than 200 species and limited usage in humans, a large number of untapped genetic resources exist within this genus.8
Asparagus aethiopicus L. is a flowering perennial monocot herb that belongs to the Asparagaceae family. An ornamental plant that grows both indoors and outdoors has solid green aerial stems, partial spines with fasciculate clustered roots, bright green needle-like leaflets, and small white or pink flowers arranged in clusters on the stems. Its fruit is a berry, spherical, red in color with slight pungent smell; each fruit contains two to a few seeds that are black in color and tough.
Asparagus densiflorus (Kunth) Jessop, commonly called the foxtail fern, is a woody perennial herb with a fern-like habit that belongs to the Asparagaceae family. It has thick fasciculate roots; narrow, flat branches; and dark green, needle-like leaves. Its axillary, bell-shaped, yellow–green flowers droop. It forms green fruits mature to brilliant red berries contains one to three black seeds.
A. aethiopicus and A. densiflorus were differentiated and identified based on the morphological characteristics of their shoots and roots.
Shoot structure
Leaf spurs on the stem are cladode and leaves are spiny and highly reduced in both species. In A. aethiopicus, the leaf spur is mostly solitary, and the distance between the spurs is greater than that in A. densiflorus, where the leaf spur is in fascicles and the distance between the spurs is smaller. A. densiflorus stem is tapering towards the axils, which gives it a tail-like appearance, whereas the stems of A. aethiopicus are long and droopy in nature.
Roots
Thick, fibrous, and fascicular roots. Fascicular root structures are short, fleshy, watery, bulb-like structures. These roots are characteristic of both A. aethiopicus and A. densiflorus.
Collection of plant samples
The plant samples were collected from the Sri Subramanya Swamy Nursery in Vidyaranyapura, Bengaluru, India. The identification of the collected plants was cross-verified and confirmed with the help of eflora.9,10 Procured plants were stored in a greenhouse until used in the experiment.
Chemicals used
High-purity analytic grade chemicals were employed for the green synthesis of NPs.
CuSO4.5H2O
Copper sulphate pentahydrate (99% purity) was purchased from Rankem Laboratory Reagents, RFCL Limited.
Cu(NO3)2.3H2O
Copper II nitrate trihydrate (99%) was purchased from Qualigens (Thermo Fisher Scientific).
Preparation of A. aethiopicus and A. densiflorus root and shoot extracts
The roots and shoots of A. aethiopicus and A. densiflorus were collected, surface-cleaned under running tap water, and then cleaned with distilled water several times. The plant parts were then dried in a shaded room for 21 d (A. aethiopicus) or 60 d (A. densiflorus). A. densiflorus root parts were placed in a hot air oven at 50°C for 72 h. The wet and dry weights of the plant parts were recorded. The dried shoots and roots were chopped using a secateur and powdered using a mixer grinder. Powdered samples were transferred and stored in polythene Ziplock pouches. The extraction was performed by adding 10 g of powdered root and shoot into a 500-mL beaker, followed by the addition of 200 mL of deionized water. The contents in the beaker were allowed to boil at 50°C for 1 h on a magnetic stirrer, after which it was allowed to cool down to room temperature overnight. The mixture of plant samples and water was filtered using Whatman No.1 filter paper to obtain clear solutions.11 This extract was then subjected to green synthesis.
Green synthesis of CuO NPs from root and shoot extracts
Aqueous solution of 0.2 M Cu(NO3)2.3H2O was prepared and stored in brown bottles. Shoot and root extract (50 mL) was mixed with 200 mL of 0.2 M Cu(NO3)2.3H2O solution (1:4) dropwise with constant stirring. A brown precipitate of CuO NPs was obtained at the end of this procedure. The NP product obtained in the aqueous form was subjected to further studies.12
Preparation of A. aethiopicus fruit and seed extract
A. aethiopicus fruits were collected from St. Joseph’s College Campus (SJCC) garden and dried in the shade for two weeks. The fruits were washed thoroughly under running tap water, followed by washing with deionized water. The washed fruits were then squashed to separate the seeds from the fruit pulp. The separated seeds and pulp were stored in different containers. The obtained seeds were thoroughly washed to remove excess fruit pulp and dried over blotting sheets. The weights of the fruits, pulp, and seeds were recorded at each step.
Fruit pulp was boiled in 100 mL distilled water at 80°C using water bath for 10 min and was allowed to cool to room temperature. The extract mixture was filtered through Whatman No.1 filter paper and stored at room temperature until further use.11
The seeds were ground using a mortar and pestle. The seed powder (1 g) was boiled in 100 mL of deionized water for 5 min and allowed to cool to room temperature. The extract mixture was filtered through a filter paper (Whatman No.1) and stored at room temperature.11
Green synthesis of CuO NPs from A. aethiopicus fruit and seed extract
Aqueous fruit extract (100 mL) was mixed with 4 g of CuSO4.5H2O using magnetic stirrer at room temperature for 4 h. Aqueous seed extract (20 mL) was mixed with 80 mL CuSO4.5H2O with constant stirring using magnetic stirrer at 45–50°C for 6–7 h. At the end of this step, a brownish-black product was obtained (Figure 1). The obtained product was subjected to further studies.11
Figure 1. (1) Habit of Asparagus densiflorus. (2) Habit of A. aethiopicus. (3) Shoots of A. densiflorus. (4) Shoots of A. aethiopicus. (5) Roots of A. densiflorus. (6) Roots of A. aethiopicus. (7) Nanoparticle samples of A. densiflorus shoot. (8) Nanoparticle samples of A. densiflorus root. (9) Nanoparticle samples of A. aethiopicus shoot. (10) Nanoparticle samples of A. aethiopicus root. (11) Nanoparticle samples of A. aethiopicus fruits. (12) Nanoparticle samples of A. aethiopicus seeds
UV–Visible Spectrophotometer studies
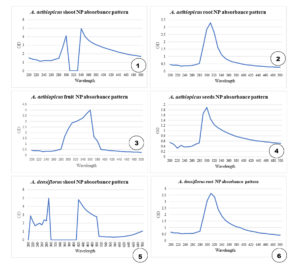
The NP end samples obtained were subjected to UV–Visible Spectrophotometer (Thermo Scientific UV–Visible Spectrophotometer Genesys 10S) and readings of the samples were obtained.13 The readings were taken in the range of 200–500 nm. UV Visible spectroscopy was also used to confirm the formation of Cu NPs based on their optical properties (Figure 2).
Bioactivity tests
The bioactivity of the obtained NPs was assessed against bacterium Pseudomonas aeruginosa (ATCC 27853) by studying their growth curves. P. aeruginosa, a gram-negative bacterium, is an opportunistic pathogen. It causes nosocomial (hospital-borne) and community-acquired infections in humans.14
Preparation of stock bacterial culture: Stored-plated cultures of P. aeruginosa were used to obtain a stock liquid culture of bacteria. For which, 100 mL of nutrient broth was prepared, autoclaved, and cooled. To this broth, 200 µL fluconazole antifungal agent was added. The stored culture was inoculated into the broth using an inoculation loop under aseptic conditions in a laminar airflow (LAF) chamber. The inoculated bacteria were incubated at 37°C for 24 h.
The nutrient broth was prepared again for the bioactivity test. To a conical flask, 15 mL of nutrient broth and 1 mL of an aqueous NP sample were added. Conical flasks containing the nutrient broth and NP samples were prepared similarly. To these conical flasks, 1 mL of P. aeruginosa culture was added under LAF with aseptic conditions. UV–Visible spectrophotometer readings were obtained. After each hour, spectrometer readings were recorded for six generations. The reading was noted and a graph was plotted.
UV–Visible Spectrophotometer studies
Absorbance readings were obtained using UV-Vis spectroscopy. The spectra were plotted against wavelength to obtain graphs for all the NPs samples. The highest absorbance of the NP samples was observed in the range of 310–360 nm (Figure 2).
Bioactivity tests
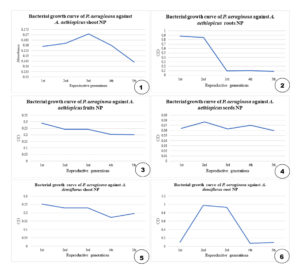
CuO NP obtained from the plant extracts were assessed for their antimicrobial properties against P. aeruginosa by studying the growth curve of the bacterium. Test samples with NP and bacteria were subjected to UV–Vis spectrophotometry at 660 nm to obtain absorbance readings at each generation. The absorbance values were plotted against their respective reproductive generations. The surface area of Cu NPs may be high, resulting in the high inhibitory activity of Cu NPs. The decline in the growth of P. aeruginosa in the graphs shows that the CuO NP had detrimental effects on the growth of P. aeruginosa, demonstrating their antimicrobial effects against microbes (Figure 3).
Green synthesis of Cu NPs was achieved using A. aethiopicus and A. densiflorus species using the root and shoot parts of both species, fruits, and seeds of A. aethiopicus. The NPs were in the aqueous extract form, and characterization was performed using a UV–visible spectrophotometer. The absorbance maxima were observed in the range of 310–360 nm, confirming the presence of CuO NP. According to Rajeshkumar and Rinitha,15 the maximum absorbance of the Cu NPs was observed at 357 nm (Figure 2). Thus, the presence of the NP in the extract was confirmed. These NPs inhibited the growth of P. aeruginosa, as confirmed by bacterial growth curve studies at 660 nm and thus proved to be a viable alternative to contemporary antimicrobial agents. Increased oxidative stress induced by NPs and lytic degradation are the reasons for bacterial inhibition. This may be the mechanism underlying the anti-microbial activity of NPs.16-18
Similarly, Cu NPs synthesized from Asparagus adscendens, a native species from Pakistan to western Himalayas and northwestern India, showed antimicrobial activity against.19 However, bimetallic alloy nanoparticles (Ag–Au alloy NPs) of A. racemosus, a wild tropical biome species, showed highest zone of inhibition against P. aeruginosa and S. aureus strains when compared to single metal nanoparticles and plant extract.20 Whereas the CuO nano-rods synthesized using root extract of A. racemosus showed effective antibacterial activity against tested bacterial pathogens like Bacillus subtilis, S. aureus, E. coli, Klebsiella pneumonia, Aeromonas hydrophila, Pseudomonas fluorescence, Flavobacterium branchiophilum, Edwardsiella tarda, and Yersinia rukeri.21
Cu NPs play a major role in physiological function as copper is involved in immune system of animals via lymphocyte proliferation,22 also increases skin cell migration, multiplication, and neovascularization in rapid wound healing process.23 Since Cu NPs are involved in body functions, these particles can be used in antibiotics, drug delivery, and other pharmacological formulations.24 Cu NPs are proven to have high antimicrobial activity against E. coli, S. aureus, and C albicans.25,26 Cu NPs along with polypyrrole formed nanocomposite through in situ polymerization method in water and inhibited against E. coli and S. aureus.27 Green-synthesized NPs not only performs antimicrobial functions, but is also useful in environmental remediations.28
It was concluded that aqueous extracts of Asparagus densiflorus root and shoot, and A. aethiopicus leaves, shoots, roots, fruits, and seeds can produce CU NPs that are quite stable in solution. The green synthesis of Cu NPs was performed using aqueous extracts of A. densiflorus and A. aethiopicus. Cu NP synthesis using plant samples was confirmed by the color change of the reactions and UV spectra absorption in the range of 500–700 nm wavelength. supported the synthesis of the NPs. The bioactivity and usefulness of the systematics were ascertained. This is the first report of the synthesis of Cu NPs from A. densiflorus and A. aethiopicus. Further characterization of NPs and their potential in plant systematics are future prospects for this study. This study also opens a new avenue for the convenient synthesis Cu NPs using cultivated Asparagus spp., which could be useful for various potential applications, including drug formulation, drug delivery, and biomedical applications.
ACKNOWLEDGMENTS
The authors would like to thank Rev. Fr. Swebert D’silva SJ, Pro Chancellor, Rev. Fr. Dr. Victor Lobo SJ, Vice Chancellor, Dr. Jayarama Reddy, Ex Head, and Dr. V.J. Jacob Paul, HoD, Department of Botany, St. Joseph’s University, Bengaluru for providing all the necessary facilities, encouragement and congenial environment for research and teaching at SJU.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
AS conceived the study and identified the plants. AS, SRMS, NP and SA developed and implemented the methods and compiled the data. AS wrote the manuscript with contributions from SRMS, NP and SA. All authors read and approved the final manuscript for publication.
FUNDING
None.
DATA AVAILABILITY
The datasets generated and/or analysed during the current study are available from the corresponding author on reasonable request.
ETHICS STATEMENT
This article does not contain any studies on human participants or animals performed by any of the authors.
- Ealias AM, Saravanakumar MP. A review on the classification, characterisation, synthesis of nanoparticles and their application. IOP Conf Ser: Mater Sci Eng. 2017;263(3):032019.
Crossref - Manikandan D, Prakash DG, Gandhi NN. A Rapid and Green Route to Synthesis Of Silver Nanoparticles from Plectranthus barbatus (Coleus Forskohlii) Root Extract for Antimicrobial Activity. Int J Chemtech Res. 2014;6(9):4391-4396.
- Vinayagam R, Santhoshkumar M, Lee KE, David E, Kang SG. Bioengineered gold nanoparticles using Cynodon dactylon extract and its cytotoxicity and antibacterial activities. Bioprocess Biosyst Eng. 2021;44(6):1253-1262.
Crossref - Lee HJ, Lee G, Jang NR, Yun JH, Song JY, Kim BS. Biological synthesis of copper nanoparticles using plant extract. Technical Proceedings of the 2011 NSTI Nanotechnology Conference and Expo, NSTI-Nanotech. 2011;1:371-374.
- Naseem T, Farrukh MA. Antibacterial Activity of Green Synthesis of Iron Nanoparticles Using Lawsonia inermis and Gardenia jasminoides Leaves Extract. J Chem. 2015;2015:912342.
Crossref - Hussain I, Singh NB, Singh A, Singh H, Singh SC. Green synthesis of nanoparticles and its potential application. Biotechnol Lett. 2016;38(4):545-560.
Crossref - Rastogi A, Zivcak M, Sytar O, et al. Impact of Metal and Metal Oxide Nanoparticles on Plant: A Critical Review. Front Chem. 2017;5:78.
Crossref - Kanno A, Yokoyama J. Asparagus. In: Kole C (eds), Wild Crop Relatives: Genomic and Breeding Resources. Springer, Berlin, Heidelberg. 2011:23-42.
Crossref - WFO. Asparagus aethiopicus L. 2022a. http://www.worldfloraonline.org/taxon/wfo-0000631225. Accessed on: 03 Aug 2022.
- WFO. Asparagus densiflorus (Kunth) Jessop. 2022b. http://www.worldfloraonline.org/taxon/wfo-0000632151. Accessed on: 03 Aug 2022.
- Amer MW, Awwad AM. Green synthesis of copper nanoparticles by Citrus limon fruits extract, characterization and antibacterial activity. Chemistry International. 2021;7(1):1-8.
Crossref - Murthy HCA, Desalegn T, Kassa M, Abebe B, Assefa T. Synthesis of Green Copper Nanoparticles Using Medicinal Plant Hagenia abyssinica (Brace) JF. Gmel. Leaf Extract: Antimicrobial Properties. J Nanomater. 2020;3924081
Crossref - Abbas Q. Understanding the UV-Vis Spectroscopy for Nanoparticles. J Nanomater Mol Nanotechnol. 2019;8(3).
Crossref - Labauve AE, Wargo MJ. Growth and Laboratory Maintenance of Pseudomonas aeruginosa. Curr Protoc Microbiol. 2012;25(1):6E.1.1-6E.1.8.
Crossref - Rajeshkumar S, Rinitha G. Nanostructural characterization of antimicrobial and antioxidant copper nanoparticles synthesized using novel Persea americana seeds. Open Nano. 2018;3:18-27.
Crossref - Xie Y, He Y, Irwin PL, Jin T, Shi X. Antibacterial activity and mechanism of action of zinc oxide nanoparticles against Campylobacter jejuni. Appl Environ Microbiol. 2011;77(7):2325-2331.
Crossref - Cioffi N, Rai M. Nano-antimicrobials: progress and prospects’, Springer-Verlag, Berlin, Heidelberg, 1st edn., 2012.
Crossref - Seil JT, Webster TJ. Antimicrobial applications of nanotechnology: methods and literature. Int J Nanomed. 2012;7:2767-2781.
Crossref - Thakur S, Sharma S, Thakur S, Rai R. Green Synthesis of Copper Nano-Particles Using Asparagus adscendens Roxb. Root and Leaf Extract and Their Antimicrobial Activities. Int J Curr Microbiol App Sci. 2018;7(4): 683-694.
Crossref - Amina M, Al Musayeib NM, Alarfaj NA, El-Tohamy MF, Al-Hamoud GA. Antibacterial and Immunomodulatory Potentials of Biosynthesized Ag, Au, Ag-Au Bimetallic Alloy Nanoparticles Using the Asparagus racemosus Root Extract. Nanomaterials. 2020;10(12):2453.
Crossref - Pallela PNVK, Ummey S, Ruddaraju LK, Kollu P, Khan S, Pammi SVN. Antibacterial activity assessment and characterization of green synthesized CuO nanorods using Asparagus racemosus roots extract. SN Appl Sci. 2019;1:421.
Crossref - Pocino M, Baute L, Malave I. Influence of the oral administration of excess copper on the immune response. Fundam Appl Toxicol. 1991;16(2):249-256.
Crossref - Alizadeh S, Seyedalipour B, Shafieyan S, Kheime A, Mohammadi P, Aghdami N. Copper nanoparticles promote rapid wound healing in acute full thickness defect via acceleration of skin cell migration, proliferation, and neovascularization. Biochem Biophys Res Commun. 2019;517(4):684-690.
Crossref - Subha V, Thulasimuthu E, Ilangovan R. Bactericidal action of copper nanoparticles synthesized from methanolic root extract of Asparagus racemosus. Materials Today: Proceedings. 2022;64(21):1761-1767.
Crossref - Bogdanovic U, Lazic V, Vodnik V, Budimir M, Markovic Z, Dimitrijevic S. Copper nanoparticles with high antimicrobial activity. Materials Letters. 2014;128:75-78.
Crossref - Ramyadevi J, Jeyasubramanian K, Marikani A, Rajakumar G, Rahuman AA. Synthesis and antimicrobial activity of copper nanoparticles. Materials Letters. 2012;71:114-116.
Crossref - Masojevic D, Stamenovic U, Otonicar M, et al. Evaluation of the antibacterial effectiveness of novel copper/polypyrrole nanocomposite. Materials Letters. 2023;338:134051.
Crossref - Singh J, Dutta T, Kim KH, Rawat M, Samddar P, Kumar P. ‘Green’ synthesis of metals and their oxide nanoparticles: applications for environmental remediation. J Nanobiotechnol. 2018;16:84.
Crossref
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.