ISSN: 0973-7510
E-ISSN: 2581-690X
Mycoplasmas and Ureaplasmas inhabit the genitourinary tract of sexually active males and females. Mycoplasma genitalium infection as well as Ureaplasma urealyticum are related to many reproductive health problems such as cervicitis, urethritis and pelvic inflammatory disease (PID). the aim of this study was detection of Mycoplasma genitalium, Mycoplasma hominis and Ureaplasma urealyticum cervical colonization among childbearing age females attending Gynecology and Obstetrics clinics at Ain Shams University Hospital by using polymerase chain reaction. This study was conducted on 145 women attending Gynecology and Obstetrics clinics at Ain Shams University Hospital. The patients were divided into three groups according to symptoms. Cervical samples were collected using a sterile swab and placed in a liquid-based transport medium. The detection of Mycoplasma genitalium, Mycoplasma hominis and Ureaplasma urealyticum was performed by using polymerase chain reaction. This study was conducted during the period from January 2018 till August 2018 on145 women attending Gynecology and obstetrics clinic at Ain Shams University Hospital. Ninety females (62%)were positively colonized with Mycoplasma spp and U. urealyticum. M. genitalium was detected in 25 patients (28%), 10 patients (11%) were positive for M. hominis and 55 patients (61%) were positive for U. urealyticum. Group (1): 60 patients attending STD clinic. Twenty patients (33.3%) were positive for U. urealyticum, five (8.3%) were positive for M. hominis and seventeen (28.3%)were positive for M. genitalium. Group (2): 50patientswith premature rupture of membranes. Twenty eight patients(56%)were positive for U. urealyticum, two patients (4%) were positive for M. hominis and seven patients (14%) were positive for M. genitalium. Group (3): 35 patients with pelvic inflammatory disease. Seven patients(20%) were positive for U. urealyticum, three patients (8.6%) were positive for M. hominis and one patient (2.9%) was positive for M. genitalium. The highest prevalence of U. urealyticum was associated with premature rupture of membranes. The highest prevalence of M. genitalium was determined in STD clinic patients. This study shows a high prevalence of genitourinary infections due to U. urealyticum which was considerably higher when compared to Mycoplasma spp. and is significantly associated with premature rupture of membranes. M. genitalium was confirmed as an important cause of STD. The use of PCR for identification of Mycoplasmas and U. urealyticum on cervical samples should be recommended. Further studies are needed to definitely associates with Spontaneous rupture of membranes and U. urealyticum colonization.
Mycoplasma genitalium, Mycoplasma hominis, Ureaplasma urealyticum, childbearing age females
Genus Mycoplasmas and Genus Ureaplasmas are included in class Mollicutes. Different Mycoplasma species ( Mycoplasma hominis, Mycoplasma genitalium) and Ureaplasma urealyticum inhabit the genitourinary tract of many sexually active men and women . The percentage of women with vaginal colonization by Mycoplasma spp, and Ureaplasma spp increases after puberty1.
By adulthood, approximately 80 percent of healthy females acquire Ureaplasma spp and up to 50 percent acquire M. hominis in their cervical or vaginal secretions2.
M. hominis and U. urealyticum are found frequently in the urogenital tract in both healthy persons and symptomatic patients3. M. genitalium is a ‘true’ pathogen and causes sexually transmitted infection (STI) as urethritis in males. Furthermore, there is increasing evidence that M. genitalium infection is related to adverse outcomes in reproductive health in women, including urethritis, cervicitis, endometritis, pelvic inflammatory disease (PID) and infertility4,5.
There is no adequate evidence that M. hominis, U. parvum or U. urealyticum, can cause cervicitis, vulvovaginitis, urethritis, PID or infertility, but some studies showed an association between the colonization by one of these microorganisms and a disease condition6,7.
However, there is limitation on availability of data regarding Mycoplasmas and Ureaplasmas infections, so studies are needed to understand the contribution of these organisms to unfavorable reproductive health outcomes in women and to improve guidelines for both screening and treatment.
Aim of the study
The aim of the current study is to detect the frequency of M. genitalium, M. hominis and U. urealyticum in child bearing age females attending Gynecology and Obstetrics clinics at Ain Shams University Hospital by using polymerase chain reaction.
This study was conducted during the period from January 2018 till August 2018. 145 women attending Gynecology and Obstetrics clinics at Ain Shams University Hospital and fulfilling inclusion criteria were included.
The inclusion criteria were: females in childbearing period (from 20 to 47 years), and complaint of only one of the following: symptoms of vaginitis/cervicitis, (vaginal discharge, pain, itching, burning sensation and dyspareunia), history of premature child delivering or premature rupture of membranes and diagnosis of PID.
Exclusion criteria was administration of antimicrobial therapy within the last 30 days before the evaluation.
Sample collection
Cervical samples were collected using a sterile swab and placed in a liquid-based transport medium. Informed consent was obtained from patients according to the regulations of Scientific research Ethical Committee Faculty of Medicine – Ain Shams University). The samples were stored in -20 until testing.
All cervical samples were subjected to DNA extraction using QIAGEN DNA extraction Kit® (QIAGEN, USA), for DNA purification according to manufacturer’s instructions.
Amplification of M. genitalium, M. hominis and U. urealyticum genes was done using primers shown in Table (1).
Table (1):
Primer sequences used in the polymerase chain reaction.
| Organism | Direction | Primer sequence | Product size |
|---|---|---|---|
| Mycoplasma Genitalis (8) | MgPa1 | 5′ AGT TGA TGA AAC CTT AAC CCC TTG G 3′ | 500 bp |
| MgPa3 | 5′ CCG TTG AGG GGT TTT CCA TTT TTG C 3′ | ||
| Ureaplasmaurealyticum | UU AS | 5′ ACT ATA TTT CTA TAG CGT CGC AA 3′ | 900 bp |
| UU S | 5′ TCA CCT TAA GTT GGG GAT AA 3′ | ||
| Mycoplasma Hominis | Mh s | 5′ ACC CAT TGG AAA CAA TGG CTA ATG CCG GAT ACG 3′ | 600 bp |
| MhAs | 5′ ATA GAC CCA GTA AGC TGC CTT CGC CT 3′ | ||
| Beta Globin | Bglobin F | 5′ TGA GTC TAT GGG ACG CTT GA 3′ | 250 bp |
| B.globin R | 5′ AAA AAT TGC GGA GAA GAA AAA 3′ |
PCR amplification conditions were 95°C for 10 min for denaturation, followed by 35 cycles at 65°C for Mycoplasma genitalis, 58°C for Ureaplasma urealyticum and Mycoplasma hominis for annealing and a final extension period of 15 min at 72°C.
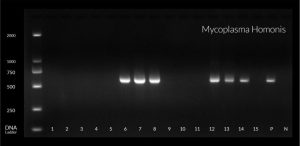
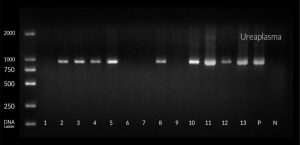
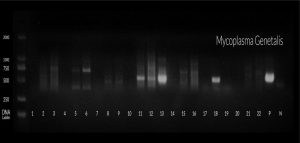
Beta globin was used as internal control , amplification conditions were 95°C for 10 min for denaturation , followed by 35 cycles at 55°C for annealing and then final extension at 72°C for 15 min. Then the detection of amplified products were done using 2% agarose gel containing ethidium bromide. Then the products were visualized on a UV transilluminator (365 wavelength). Qiagen gel pilot 250 bp Plus was used as a ladder for molecular weight as shown in figure (1 , 2 , and 3).
Data entry and statistical analyses were performed using SPSS (statistical package of social sciences) version 16.0 (SPSS Inc., Chicago, IL, USA).
This study was conducted on 145 women attending Gynecology and Obstetrics clinics at Ain Shams University Hospital. Their age ranged from 20 to 47.
These patients were divided into three groups according to the clinical presentations as shown in Table 2:
Table (2):
Distribution of the patients in studied groups.
| Groups | Patients N % | |
|---|---|---|
| Total 145 | STD Clinic | 60 (41.4%) |
| Premature rupture membrane | 50 (34.5%) | |
| PID | 24.1%))35 | |
Group (1): Sixty patients(41.4%) were attending Sexually Transmitted Diseases(STD) clinic with symptoms of vaginitis/cervicitis (mean age 31± 4)
Group (2): Fifty patients (34.5%)with diagnosis of premature rupture of membranes (mean age 32± 4)
Group (3):Thirty-five patients (24.1%)with diagnosis of PID (mean age 33± 3)
The distribution of M. genitalium, M. hominis and U. urealyticum detection among total positive patients is shown in Table 3. U. urealyticum was the most frequent microorganism detected.
Table (3):
Distribution of Mycoplasma genitalium, Mycoplasma hominis and Ureaplasma urealyticum among total positive colonized patients n = 90.
Detected Pathogen |
Total N Positive cases(90) |
|---|---|
Mycoplasmagenitalium |
25 (28% ) |
Mycoplasma hominis |
10 (11%) |
Ureaplasma urealyticum |
55 (61%) |
Table 4 shows that in the group (1) there were 20 patients (33.3%) positive for U. urealyticum, five patients (8.3%) were positive for M. hominis and seventeen patients (28.3%) were positive for M. genitalium. A highly statistical association was observed between M. genitalium detection and patients attending STD clinic.
Table (4):
Distribution of Mycoplasma genitalium, Mycoplasma hominis and Ureaplasma urealyticum among different groups.
| Disease group | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| STD Clinic (60) | Premature rupture membrane (50) | Pelvic inflammatory disease (35) | ||||||||
| N | N % | P-value | N | N % | P-value | N | N % | P-value | ||
| Urea plasma | Negative | 40 | 66.7% | > 0.05 | 22 | 44.0% | > 0.05 | 28 | 80.0% | ≤ 0.05 |
| Positive | 20 | 33.3% | > 0.05 | 28 | 56.0% | ≤ 0.05 | 7 | 20.0% | > 0.05 | |
| M.hominis | Negative | 55 | 91.7% | > 0.05 | 48 | 96.0% | > 0.05 | 32 | 91.4% | > 0.05 |
| Positive | 5 | 8.3% | > 0.05 | 2 | 4.0% | > 0.05 | 3 | 8.6% | > 0.05 | |
| M. genitalium | Negative | 43 | 71.7% | > 0.05 | 43 | 86.0% | > 0.05 | 34 | 97.1% | ≤ 0.05 |
| Positive | 17 | 28.3% | ≤ 0.05 | 7 | 14.0% | > 0.05 | 1 | 2.9% | > 0.05 | |
- P> 0.05 Not significant
- P ≤ 0.05 Highly significant
In the Group 2, there were 28 patients (56.0%) positive for U. urealyticum, two patients (4.0%) were positive for M. hominis and seven patients (14.0%) were positive for M. genitalium. A highly statistical association was observed between U. urealyticum detection and premature rupture of membranes.
In the Group 3 , there were seven patients(20.0%) positive for U. urealyticum, three patients (8.6 %) positive for M. hominis and one patient (2.9%) positive for M. genitalium. No statistical association between diagnosis of PID and M. genitalium, M. hominis and U. urealyticum was observed.
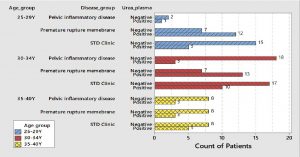
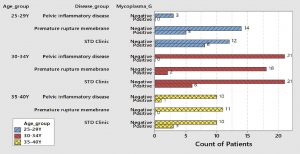
Fig. (4) shows that there was a high prevalence of Ureaplasma urealyticum among patients with premature rupture of membranes especially in the Age group from 25 to 29 years old and age group from 30 to 34 years old.
Fig. (5) shows that the Mycoplasma hominis was highly prevalent among female patients attending STD clinic especially among age group from 30-34 years.
Fig. (6) shows that the high prevalence of Mycoplasma genitals was in patients attending STD clinic especially among age group 25-29 years.
M. hominis and U. urealyticum are found frequently in the urogenital tract of humans in both patients and healthy individuals3. However, M. genitalium is a ‘true’ sexually transmitted pathogen causing urethritis in males and there is increasing evidence that M. genitalium infection could be related to unfavorable reproductive health outcomes in women, including urethritis, cervicitis, PID, endometritis and infertility and adverse birth outcomes4,5.
The aim of the current study was to detect the distribution of M. genitalium, M. hominis and U. urealyticum in child bearing age females attending Gynecology and Obstetrics clinics at Ain Shams University Hospital by using polymerase chain reaction.
The results of our study showed that out of 145 patients,90 patients (62%)were positive for Mycoplasma spp and U. urealyticum. 28% were positive for M. genitalium, 11% were positive for M. hominis and 61% were positive for U. urealyticum. These results are similar to those described by Bayraktar et al.9, who reported U. urealyticum detection in 44% of patients and M. hominis in 4% of patients. Moreover, the prevalence of M. genitalium was similar to that reported by Balkus et al.4
In this study the prevalence of M. genitalium was 28% among studied group, most of cases were detected in age group ≤30 years. Lokken and coworkers reported lower prevalence of Mycoplasma genitalium in Uganda (14%) and Kenya (12.9%–16%) than the prevalence in this study10. Also ,Getman et al., reported prevalence of M. genitalium infections in all females (14 to 70 years of age) was 16.3%. M. genitalium infections were significantly more prevalent in subjects ≤30 years of age than in subjects >30 years of age11. Furthermore, Campos et al. who studied the prevalence of Mycoplasma genitalium and Mycoplasma hominis in urogenital tract of Brazilian women reported that the prevalence of M. hominis and M. genitalium were detected in 31.8% (96/302) and 28.1% (85/302), respectively and also reported that women under age 25 represent a higher risk for M. hominisinfection12. As for Mycoplasma hominis infections are found to be less common than Ureaplasma infections. In a study performed by Sarier et al. reported that Mycoplasma hominis infections occur mainly in severely immuno suppressed patients13.
Furthermore, patients were divided into three groups: Group (1): patients attending STD clinic, Group (2): patients with diagnosis of Premature rupture of membranes and Group (3): patients with diagnosis of PID.
There was a high prevalence of U. urealyticum in the three groups, where it represents 33% in patients attending STD clinic, 56.5% in females with premature rupture of membrane and 20% in PID.
Furthermore, Bayraktar and coworkers reported that there was a high prevalence of U. urealyticum among pregnant women in age group18—34 years. They concluded also that U. urealyticum was more frequently detected than M. hominisin both the controls and patients9.
There is an association between the presence of genital mycoplasmas and increased risk of obstetrical complications, such as, preterm labor,spontaneous abortion and delivering low birth weight14. Bayraktar and coworkers stated that there is an association between U. urealyticum and preterm labor9.
Luton et al. performed a study on 218 pregnant women of different gestational age and followed them. He reported that there is a significant increase in the prevalence of Mycoplasmas as a cervical colonizer during whole pregnancy15. Moreover, Lillis et al. reported that 70 women (17.5%) were positive for M. genitalium among 400 women attending a STD clinic. M. genitalium was significantly associated with age < 25 years16.
There are different methods that can be used for detection of Mycoplasmas, like culture, antigen detection,antibody detection and molecular methods. Moreover, the molecular methods allowing simultaneous detection of different species of genital Mycoplasmasis very useful in a clinical setting. Quantitative PCR analysis plays an important role in their diagnosis as it also can show microbial load17,18. Furthermore, detection of U. urealyticum in culture is difficult so PCR is a gold standard method for its detection. Recent study performed by Sarier et al., they stated that using quantitative PCR in diagnosis of U. urealyticum is a valuable test for demonstrating microbial load and avoiding false-positive results. Thus, quantitative PCR analysis plays an important role in the diagnosis since it also can show microbial load18.
This study shows a high prevalence of U. urealyticum and M. genitalium in comparison to M. hominis as genitourinary infections. There is a significant association between prevalence of U. urealyticum and age. U.urealyticum is more prevalent in patients with premature rupture of membrane in the current study so there may be an association between U. urealyticum infection and obstetrical complications. So testing the presence of Mycoplasma species and U. urealyticum during pregnancy may be necessary for better pregnancy outcomes and can be included in the routine follow up of pregnancy. The use of molecular techniques like PCR for identification of Mycoplasma spp and U. urealyticum on cervical samples can be beneficial in comparison to culture methods. Further studies are recommended to prove the association between spontaneous abortion and premature rupture of membranes and U. urealyticum colonization.
ACKNOWLEDGMENTS
The authors are grateful to members of obstetrics and gynecology of Ain Shams University hospitals, for their support to carry out this study.
CONFLICT OF INTEREST
The authors declare that there is no conflicts of interest
AUTHORS’ CONTRIBUTION
All authors designed the study. MMF collected samples from patients. SAAS and MAK performed the molecular work of the study performed PCR on the samples. All authors analyzed the data. All authors wrote the manuscript. All authors read and approved the manuscript.
FUNDING
None.
ETHICS STATEMENT
Informed consent was taken from all patients according to the Ethical Committee regulation of Faculty of Medicine Ain Shams University.
AVAILABILITY OF DATA
All Datasets generated or analyzed during this study is available.
- Baum SG, Edwards MS. Mycoplasma hominis and Ureaplasma urealyticum infections. Up-ToDate, Waltham, MA. 2017;1-46.
- Waites KB, Schelonka RL, Xiao L, Grigsby PL, Novy MJ. Congenital and opportunistic infections: Ureaplasma species and Mycoplasma hominis. In Seminars in Fetal and Neonatal Medicine. 2009;14(4):190-199. WB Saunders.
Crossref - Taylor-Robinson D. Mollicutes in vaginal microbiology: Mycoplasma hominis, Ureaplasma urealyticum, Ureaplasma parvum and Mycoplasma genitalium. Res Microbiol. 2017;168:875-881.
Crossref - Balkus JE, Manhart LE, Jensen JS, et al. Mycoplasma genitalium infection in Kenyan and US women. Sexually Transmitted Diseases. 2018;45(8):514-21.
Crossref - Unemo M, Jensen JS. Antimicrobial-resistant sexually transmitted infections: gonorrhoea and Mycoplasma genitalium. Nat Rev Urol. 2017;14:139-152.
Crossref - Kyndel A, Elmer C, Kallman O, Altman D. Mycoplasmataceae colonizationsin women with urethral pain syndrome: a case-control study. J Low Genit Tract Dis. 2016;20:272-274.
Crossref - Michou IV, Constantoulakis P, Makarounis K, Georgoulias G, Kapetanios V, Tsilivakos V. Molecular investigation of menstrual tissue for the presence of Chlamydia trachomatis, Ureaplasma urealyticum and Mycoplasma hominis collected by women with a history of infertility. J ObstetGynaecol Res. 2014;40:237-242.
Crossref - Jensen JS, Uldum SA, Sondergard-Andersen J, Vuust J, Lind K. Polymerase chain reaction for detection of Mycoplasma genitalium in clinical samples. J Clin Microbiol. 1991;29(1):46-50.
Crossref - Bayraktar MR, Ozerol IH, Gucluer N, Celik O. Prevalence and antibiotic susceptibility of Mycoplasma hominis and Ureaplasma urealyticum in pregnant women. Int J Infect Dis. 2010;14(2):e90-5.
Crossref - Lokken EM, Balkus JE, Kiarie J, et al. Association of recent bacterial vaginosis with acquisition of Mycoplasma genitalium. Am J Epidemiol. 2017;186(2):194-201.
Crossref - Getman D, Jiang A, O’Donnell M, Cohen S. Mycoplasma genitalium prevalence, coinfection, and macrolide antibiotic resistance frequency in a multicenter clinical study cohort in the United States. J Clin Microbiol. 2016;54(9):2278-83.
Crossref - Campos GB, Lobao TN, Selis NN, et al. Prevalence of Mycoplasma genitalium and Mycoplasma hominis in urogenital tract of Brazilian women. BMC Infectious Diseases. 2015;15(1):60.
Crossref - Sarier M, Sepin N, Guler H, et al. Prevalence of Sexually Transmitted Disease in Asymptomatic Renal Transplant Recipients. Exp Clin Transplant. 2018.
Crossref - Clegg A, Passey M, Yoannes M and Michael A. High rates of genital Mycoplasma infection in the highlands of Papua New Guinea determined both by culture and by a commercial detection kit. J Clin Microbiol. 1997;35:197-200.
Crossref - Luton D, Ville Y, Luton-Sigy A, et al. Prevalence and influence of Mycoplasma hominis and Ureaplasma urealyticum in 218 African pregnant women and their infants. Eur J Obstet Gynecol Reprod Biol. 1994;56:95-101.
Crossref - Lillis RA, Martin DH, Nsuami MJ. Mycoplasma genitalium infections in women attending a sexually transmitted disease clinic in new orleans. Clin Infect Dis. 2019;69(3):459-65.
Crossref - Bao T, Chen R, Zhang J, et al. Simultaneous detection of Ureaplasma parvum, Ureaplasma urealyticum, Mycoplasma genitalium and Mycoplasma hominis by fluorescence polarization. J Biotechnol. 2010;150(1): 41-43.
Crossref - Sarier M, Kukul E. Classification of non-gonococcal urethritis: a review. Int Urol Nephrol., 2019.
Crossref
© The Author(s) 2020. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.