Probiotic bacteria are producers of secretory products such as bacteriocins and polysaccharides. Both homopolymeric and heteropolymeric exopolysaccharides (EPS) present on the surface of microorganisms have shown beneficial properties. While the fact that they play a role in bacterial homeostasis is well established, studies exploring their health promoting effects have also gained traction. Some exopolysaccharides function by inducing immune tolerance, others act by evading immune responses such as those by B and T cells. The interaction between the EPS and the immune system helps protect the bacteria against an attack by the host immune system. Several exopolysaccharides also show tolerogenic properties by reducing the amount of pro-inflammatory cytokines and increasing IL-10 production. They are also associated with anti-bacterial activity, anti-biofilm activity and anti-tumour properties. This review highlights the different types of exopolysaccharides and their health benefiting potentials. Better understanding of these mechanisms will pave the way for harnessing their potential to improve our health.
Probiotic, Exopolysaccharide, Immunomodulation, Anti-biofilm, Adhesion, Colonization, Antimicrobial, Anti-inflammatory, Cytokines
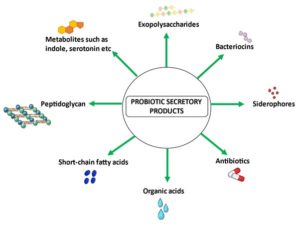
The word probiotic has its origin traced back to Latin and it means ‘for life’. They have existed for a long time in yoghurt, beer, bread, kefir and wine.1 Henry Tessler from the Pasteur Institute in Paris reported in 1899 that Bifidobacterium in the intestines of breast-fed infants resulted in fewer diarrheal episodes. In 1907 Eli Metchnikoff shed light on the health benefits of probiotics.2 The current description of probiotics is that they are “live microorganisms which when administered in adequate amounts confer a health benefit on the host”.3 The International Scientific Association for Probiotics and Prebiotics (ISAPP) state that any commensal organisms in the gut can’t be called as probiotics unless the characterized strains are proven to have health benefits.4 Several species of Lactobacillus and Bifidobacterium have been commonly identified as probiotics.5 Bifidobacterium bifidum, Bifidobacterium adolescentis, Bifidobacterium animalis, Lactobacillus acidophilus, Lactobacillus rhamnosus, Lactobacillus casei, and Lactobacillus salivarus are a few examples. Studies have also revealed potential next-generation probiotics (NGP) such as Bacteroides thetaiotaomicron, Christensenella minuta, Akkermansia muciniphila, Prevotella copri, and Parabacteroides goldsteinii.6 They have gained popularity mainly because they colonise the gut, competitively exclude pathogens and contribute to host defence mechanisms. But, the use of whole cells of probiotic bacteria has raised concerns in the scientific community due to the safety problems associated with them, especially among individuals with compromised immune systems, infants and patients with underlying conditions.7 This has led to a shift in focus from whole cells to cell components and metabolites such as exopolysaccharides,8 siderophores, antibiotics and bacteriocins9,10 organic acids11 and metabolites such as indoles, folate, secondary bile acids, serotonin and short-chain fatty acids (SCFA)12 as seen in Figure 1. Exopolysaccharides are associated with properties against phagocytosis, cell recognition, phage attack, antibiotics or toxic compounds, desiccation and osmotic stress,13 induction of autophagy and apoptosis in cancer cell lines, effects on blood pressure, antioxidant activity, influence on blood glucose levels and modulation of immune system response.14 The ability of EPS to interact with microorganisms in the gut and their ability to modulate the immune system will be the aspects explored in this review.
Figure 1. The secretory products such as exopolysaccharides, siderophores, antibiotics, bacteriocins, organic acids and metabolites such as indoles, folate, secondary bile acids, serotonin, and short-chain fatty acids (SCFA) are produced by Lactic acid bacteria
Characteristics of probiotics
Most of the probiotic bacteria are Gram-positive bacteria that do not form spores. They are catalase-negative and non-motile rods.15,16 They grow at an ideal temperature of 37°C and an optimum pH of 6.5-7.05. There are a few probiotics that are Gram negative in nature as well. For example, Escherichia coli Nissle 1917 (EcN), or Mutaflor is Gram negative and has been used for treating chronic constipation and colitis for several years.17 To be classified as a potential probiotic, the organism must be able to resist high pH conditions of the gastric environment and have a tolerance to intestinal enzymes and bile salts.18 The ability to adhere, grow and establish themselves in the mucus membrane and in the human epithelial cells /cell lines is also considered an important prerequisite.3 They should also have the capacity to fight against pathogenic microbes by competing with them for adhesion and by using other mechanisms. Other characteristics associated with a functional probiotic are proteolysis, probiotic stability and viability.3,19
Exopolysaccharides and their structure
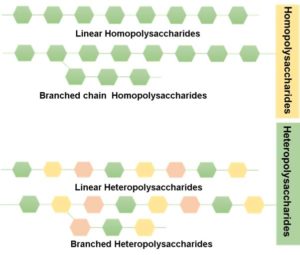
There are two types of polysaccharides associated with bacteria. The first category includes capsular polysaccharides that associate with the cell wall of bacteria and the second category includes exopolysaccharides(EPSs) that are secreted into the environment.14 Exopolysaccharides (EPS) are produced by algae, fungi, and bacteria and are composed of different monosaccharides.20 Their role in bacterial homeostasis has been well-known.21-23 Lately, studies exploring their health promoting effects are increasing in number. LAB strains such as Lactobacillus, Streptococcus, Lactococcus and Bifidobacterium are EPS producers. The main varieties of EPSs are homopolysaccharides and heteropolysaccharides.24 These EPSs are of 2 types namely homopolysaccharides(HoPS) and heteropolysaccharides(HePSs) depending on whether they are made of a solitary sugar such as glucose or a combination of sugars at different ratios24 as in Figure 2. Homopolysaccharides are made of glucose or fructose monosaccharides and are called β-D-glucans, fructans (Ex. inulin and levan) dextrans, mutan, alternan, or reuteran (a-D-glucans), with 3-8 repeating units and polygalactans.13 HoPS are classified depending on various factors such as the carbon atom position in the bond, the glycosyl type and the kind of linakge.25 HePSs are made of 2 or more different sugars such as glucose, galactose and rhamnose at different ratios.26 These different monosaccharides are arranged in linear or branched units in HePSs.27 Sugar derivatives such as N-acetylgalactosamine and N-acetylglucosamine are also used in the composition of the EPS.25 The structure and composition of EPS is influenced by the strains, the environment, culture conditions, medium composition and the rate of microbial growth.25,26 Most of the EPS are HePSs synthesized by mesophilic organisms such as the Lactococcus lactis subsp. lactis, Lactobacillus rhamnosus, Lactobacillus lactis subsp. cremoris, Lactobacillus sakei and thermophilic Lactobacillus delbrueckii subsp. bulgaricus, Lactobacillus acidophilus and Lactobacillus helveticus.27
Figure 2. Homopolysaccharides and Heteropolysaccharides are the 2 categories of exopolysaccharides based on the monomeric units that compose them and depending on whether they are made up of a solitary sugar such as glucose or a combination of sugars at different ratios
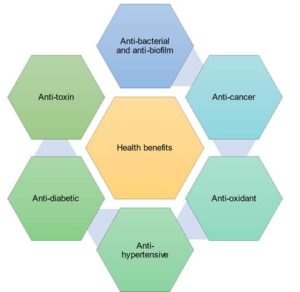
Figure 3. The exopolysaccharides produced by LAB are associated with various health benefits such as immunomodulation, anti-biofilm, anti-cancer, anti-oxidant, anti-hypertensive, anti-diabetic, anti-toxin and cholesterol lowering properties
Health benefits of EPSs
The exopolysaccharides associated with the LABs are linked to health benefits such as anti-ulcer property, maintenance of intestinal barrier, anti-biofilm property against pathogens, anti-cancer property,28 and immunomodulatory properties as observed in Figure 3.25 They influence the production of cytokines, lymphocytes and NK cells. This ability to modulate is influenced by the molecular weight of the polysaccharide and it’s monosaccharide composition.13,29 Some of the properties of exopolysaccharides in promoting health are elaborated below.
Antibacterial and anti-biofilm properties of exopolysaccharides
LAB-derived EPSs are found to have antimicrobial activity against pathogenic organisms. The following mechanisms relate to the antimicrobial activity of EPS under different conditions:
- Protection against host barrier disruption.
- Their capacity to interfere with biofilm formation
- The capacity to competitively hinder the proliferation of pathogenic organisms.27
Protection against host barrier disruption
HePS belonging to Bifidobacterium animalis, Bifidobacterium longum and Lactobacillus rhamnosus GG were examined for their ability to influence barrier integrity. All three reduced the cytotoxic effects of Bacillus cereus toxins on barrier integrity in a model using Caco-2 cells.30 In another study, EPS-1 produced by Streptococcus thermophilus MN-BM-A01 when used in Caco-2 cells demonstrated a protective property on intestinal barrier integrity when stimulated with lipopolysaccharides.31
Their capacity to interfere with biofilm formation
EPS obtained from Lactobacillus rhamnosus from human breast milk inhibited biofilm formation by the organisms Escherichia coli (E. coli), Staphylococcus petrasii subsp. Pragensis KY196531 and Salmonella typhimurium (S. typhimurium) in a concentration dependent manner. This was observed when there was a rise in the EPS concentration from 0.2 to 5mg/ml.32 The EPS of Lactobacillus casei NA-2 isolated from a variety of sauerkraut showed antibacterial and antibiofilm properties against Salmonella typhimurium, Escherichia coli O157:H7 and other organisms.27 In another study, exopolysaccharide (EPS) from Lactobacillus plantarum WLPL04 hindered the biofilm formation ability of E. coli O157:H7, Pseudomonas aeruginosa CMCC10104, Salmonella Typhimurium ATCC13311, and Staphylococcus aureus CMCC26003.33
The capacity to competitively hinder the proliferation of pathogenic organisms
Hetero-polymeric exopolysaccharide obtained from Lactobacillus gasseri was useful against pathogenic Listeria monocytogenes MTCC 657. EPS of Lactobacillus sp. Ca6 strain displayed antagonistic action against Salmonella enterica and Micrococcus luteus. Both these polysaccharides have also been studied for their wound healing properties.34 The EPS of Lactobacillus kefiranofaciens DN1 (EPS_DN1) was studied for its action against Salmonella Enteritidis and Listeria monocytogenes and it showed bactericidal effects against both the pathogens when used at a minimum concentration of 1%.35 The antibiofilm and antibacterial properties of Rhodotorula mucilaginosa coated with the EPS UANL-001L (rEPS-SNPs) were examined against clinically important pathogens such as Pseudomonas aeruginosa (P. aeruginosa), Staphylococcus aureus (S. aureus) and E. coli. The rEPS-SNPs proved to be effective against the pathogens.20 EPS of Lactobacillus brevis MSR104 showed effective antimicrobial activity against different organisms. They were also effective against free radicals of DPPH (2,2-diphenyl-1-picrylhydrazyl) and nitric oxide.32 The EPS produced by marine Bacillus subtilis SH1 was studied for its antibacterial and antiviral activity. The antibacterial activity was displayed majorly against Streptococcus faecalis followed by Pseudomonas aeruginosa and Aeromonas hydrophilas. It also showed antiviral action at 500 µg/ml.36
Immunomodulation by EPS
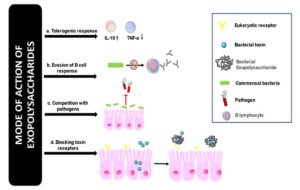
The first line of defense includes barriers that help defend the body from infection. Polysaccharides produced by microorganisms collectively called as microbially produced glycans(MPGs) are a crucial part of this defense in the gut.37 Depending on whether they are present in the commensals or in the pathogens, they may be a boon or a bane. Several researchers have shown that microbial exopolysaccharides have a protective effect against various defense mechanisms of the hosts and some of the proposed mechanisms are shown in Figure 4.
Figure 4. Proposed mode of action of exopolysaccharides
- Tolerogenic response that involves the upregulation of anti-inflammatory cytokines such as IL-10 and a decrease in pro-inflammatory cytokines such as TNF-a
- By evading responses from immune cells such as B and T lymphocytes
- By competing with pathogens for common niches making it harder for the pathogens to colonize
- The exopolysaccharides block toxin receptors and prevent the action of toxins in the gut
The gut is protected from pathogens by (a) The mucus layer38 (b)Commensals that compete with pathogens.39 One of the main reasons for infectious organisms to take over the gut is the absence of protective bacteria and overcrowding of pathogens. A thick mucus layer also prevents pathogens from colonizing. The thickness and properties of the mucus layer are also dependent on microbiota.38 Thus, the commensal organisms play an important role in protecting against pathogens, both directly and indirectly. Probiotic bacteria such as Bifidobacterium, have the ability to maintain a homeostatic balance in the composition of gut microbiota in neonates and adults as they are one of the first colonizers of the gut. Bifidobacterium shows immunomodulatory properties by inhibiting pathogenic microbes from adhering to the intestinal wall. They also interact with other beneficial microbes and aid in the digestion of otherwise resistant polysaccharides resulting in the production of antimicrobial compounds.40 There are other mechanisms involving B and T cells, that the immune system has in place to ensure that the pathogenic microorganisms are destroyed when they breach the enterocyte barrier. Probiotic bacteria function by improving the efficiency of these existing defence mechanisms. They are also associated with downregulating hypersensitive reactions and creating a balanced response instead.39
When a bacterium enters the gut, the intestinal epithelium produces antimicrobial peptides (AMPs) like cathelicidins and defensins.37 Among them human α-defensin 5 (HD-5) is an antimicrobial peptide that is seen in the small intestine in the highest proportion.62 MPGs have the ability to block these defensins and protect the bacteria. When HD-5 acts on enteropathogenic Escherichia coli (EPEC), a wild type EPEC can better withstand this attack compared to an unencapsulated EPEC owing to the presence of MPGs in the former category. Protection of the unencapsulated strain is made possible by the addition of an external polysaccharides that bind to the HD-5 and prevent entry into the bacterial membrane.37,62 Another study shows galactose-rich long exopolysaccharides (EPS) obtained from Lactobacillus rhamnosus GG (LGG) that form a shielding barrier against LL-37, which is a cationic protein that belongs to the cathelicidin family present in the gastrointestinal tract of humans.37 It was found that when concentrations of LL-37 are subinhibitory, there is increased production of EPS signifying that EPS making is adjusted to strike a balance between immune system evasion and adhesion.63 In another study, protective properties were also displayed by Lactococcus lactis cell wall polysaccharide pellicle. They show protective effects against murine macrophage induced phagocytosis.64 Some probiotic bacteria produce metabolites that improve the barrier function. Bifidobacterium longum subspecies infantis (B. longum) is an example as it has shown to improve barrier function by making short-chain fatty acid (SCFA) acetates which induce protection against lethal infections with enterohemorrhagic E. coli (EHEC). Some probiotics modulate immune responses by interacting with intestinal epithelial cells. An example for this is the K5 capsule expressed by E. coli 1917. Escherichia coli strain Nissle 1917 has shown to have immunomodulatory effects. K5 capsule from E. coli Nissle, stimulates Caco-2 cells to produce chemokines and the same was observed ex vivo in the mouse intestine. The loss of capsule demonstrated a significant decrease in levels of immunoproteins in vitro and ex vivo.65 In a study performed on Litopenaeus vannamei which is a kind of white Shrimp, the EPS derived from Porphyridium cruentum (purpureum) was found to stimulate the immune response against Vibriosis by targeting the non-specific immune response. There was an increase in the phagocytotic activity (PA), respiratory burst (RB) and total hemocytes (THC) value proportional to the increase in the concentration of EPS. These results are indicative of EPS acting as an immunostimulator or modulator50 Se-ECZ-EPS-1 is an exopolysaccharide that is Selenium-enriched and is derived from bacterium Enterobacter cloacae Z0206. It was examined for its immunomodulatory properties. The EPS was made of glucose, mannose and galactose. The organism was grown in sodium selenite containing potato dextrose agar medium. When the Selenium containing Se-ECZ-EPS-1 was administered to animals that were exposed to cyclophosphamide (CP) there was an enhancement in the humoral and cellular immune responses. They also caused an increase in the relative weight of the thymus and the spleen which are significant indicators of nonspecific type of immunity.66
Another proposed mechanism for the action of surface EPS is by the evasion of B cell response. The surface EPS of Bifidobacterium breve UCC2003 helps evade a response from the adaptive B cells, helps colonize the host by being immunologically silent and also reduces pathogen colonization. When organisms with(EPS+) and without (EPS -) EPS were compared, it was found that EPS – organisms developed a strong immune response whereas EPS+ individuals developed a weak response due to the masking effect of the EPS on the bacteria.55
EPS as an anti-inflammatory agent in Tolerogenic responses
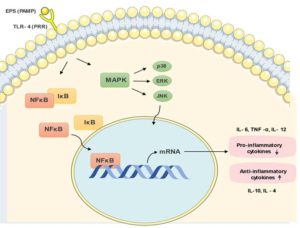
Inflammation is when the body responds to tissue injury or an infection by producing chemokines, cytokines, and different vasoactive amines.67 The innate immune system uses transmembrane receptors called toll-like receptor (TLRs) to interact with unique sites in the pathogen.68 This engagement drives the MyD88-dependent initiation of nuclear factor kB (NFkB) and other transcription factors that activate pro-inflammatory cytokine genes.69 For a brief period, there is an upregulation in the production of pro-inflammatory cytokines and a downregulation in the production of anti-inflammatory cytokines as shown in Figure 5. A prolongment in this can be a sign of disease13 and in such cases EPS produced by probiotics have been used in an attempt to induce immune tolerance.
Figure 5. Toll-like receptors are a type of PRRs that are transmembrane receptors expressed by cells of the innate immune system. They interact with unique microbial sites called pathogen-associated molecular patterns (PAMPs) now also referred to as microbe-associated molecular patterns (MAMPs). The engagement of the TLR with their respective ligand drives the MyD88-dependent activation of nuclear factor kB (NFkB) and several other transcription21,22,81 factors that in turn activates pro-inflammatory cytokine genes
As shown in the examples with as highlighted in table, many researchers have shown a corelation between EPS use and an increase in the levels of Interleukin-10 (IL-10) along with a decrease in the levels of pro-inflammatory cytokines. Bifidobacterium longum subsp. longum 35624 is linked with elevated IL-10 production and decreased production of proinflammatory biomarkers.51 EPS from L. rhamnosus RW-9595M showed an elevated level of IL-10 and a reduction in the amounts of TNF-a which caused a suppressive effect on macrophages.41 There was a decrease in the secretion of pro-inflammatory cytokines in porcine intestine by the action of Lactobacillus plantarum strain 14 secreted EPS.42 Lactobacillus gasseri strains (G10 and H15) found in human vagina under normal conditions were used for EPS extraction and it was found that there was an increase in IL-10 production and decrease in TNF-a production that resulted in anti-inflammatory properties displayed on HeLa cells.49 Other bacterial exopolysaccharides are also linked with alterations in the IL-10 concentration and the associated pro-inflammatory cytokines. The EPS produced by B. longum BCRC 14634 was used against murine J77A.1 macrophages. EPS exposure resulted in growth and an increase in cytokine IL10 production when compared to those with basal levels of EPS. EPS treatment also reduce TNF-a secretion and variations in the J774A.1 cell structure.52 The EPS from Bifidobacterium animalis subsp. lactis IPLA-R1 which is rich in rhamnose was also associated with increased IL10 production and decreased TNF α production.53 The EPS produced by Bifidobacterium breve UCC2003 was linked with an increase in proinflammatory TNF-a, IL12 and INF-g in murine models and this was associated with a protective action against Citrobacter rodentium infection.55 A water-soluble heteropolysaccharide (EPS-1) produced by Streptococcus thermophilus MN-BM-A01 was checked for its disease alleviating property on murine model of colitis. The EPS was made of galactose, glucose, rhamnose and mannose in a molar ratio of 12.9:26.0:60.9:0.25, and had a molecular weight of 4.23 × 105 Da. The disease severity in mouse was found to drop owing to the action of the administered EPS-1. This was indicated by a decrease in the disease activity index. A lowering in the levels of cytokines was seen along with an increase in the expression of tight junction protein.31
Table:
Immunological effects of exopolysaccharides produced by LABs
No |
Source organism |
Immunological Effect of EPS |
Reference |
|---|---|---|---|
1. |
L. rhamnosus RW-9595M |
Elevated anti-inflammatory IL10 cytokine production and decreased TNF-α, IL-6, and IL-12 secretion |
41 |
2. |
L. plantarum14 |
When challenged with enterotoxigenic Escherichia coli (E. coli) (ETEC) a reduction of IL-6, IL-8, and monocyte chemoattractant protein (MCP-1) was observed in porcine intestinal epithelial cells |
42 |
3. |
Lactobacillusdelbrueckii (L. delbrueckii) subssp. bulgaricus OLL1073R-1 |
Activation of interferon gamma (IFN-g ) along with NK cells in mice |
43 |
4. |
L. plantarum YW32
|
HT-29 cell inhibition |
44 |
5. |
Lactobacillus acidophilus 20079 strain |
Expression of TGF genes, IKba and P53 genes was upregulated and nuclear factor kappa B (NF-kB) associated inflammatory pathway and other apoptotic pathways human colon cancer were regulated |
45 |
6. |
L. casei X12, L.casei K11, L. casei SB27, L. casei M5 strains |
Induced the arrest of the G0/G1 cell which suppressed HT-29 cell and also induced apoptosis |
46 |
7. |
L.plantarum NCU116
|
TLR2 dependent apoptosis was induced in CT26 cells of mouse via Fas/Fasl-mediated apoptosis that is c-Jun dependent |
23 |
8. |
L. acidophilus |
Showed results in colon cancer cell lines by inhibiting tumor angiogenesis related genes in vitro |
47 |
9. |
L. acidophilus LA1
|
Showed results against Ehrlich ascites carcinoma cells. There was a reduction in the anti-tumor activity of the cancer cells by suppressing nitric oxide and malondialdehyde levels in the serum in vivo. |
48 |
10. |
EPS of Lactobacillus gasseri |
Immune response modulation by inhibiting cervical cancer cells from growing |
49 |
11. |
Lactobaciluus rhamnosus |
Antimicrobial activity against E. coli, S. typhimurium, and S.petrasii subsp. Pragensis KY196531 |
32 |
12. |
Lactobacillus gasseri |
Antimicrobial activity against Listeria monocytogenes MTCC 657 |
34 |
13. |
Lactobacillus sp.Ca6 strain |
Antimicrobial activity against Salmonella enterica and Micrococcus luteus |
34 |
14. |
Lactobacillus kefiranofaciens DN1 (EPS_DN1) |
Antimicrobial activity against Listeria monocytogenes and Salmonella Enteritidis |
35 |
15. |
Rhodotorula mucilaginosa UANL-001L |
Antimicrobial activity against P. aeruginosa,S. aureus and E coli |
20 |
16. |
Lactobacillus casei NA-2 |
Antimicrobial activity against Salmonella typhimurium, Escherichia coli O157:H7, Bacillus cereus, Staphylococcus aureus |
27 |
17. |
Bacillus subtilis SH1 |
Antimicrobial activity against Pseudomonas aeruginosa, Streptococcus faecalis and Aeromonas hydrophila |
36 |
18. |
Lactobacillus rhamnosus GG (LGG) |
Shielding barrier against LL-37, a human cationic protein 18 in the GI tract |
37 |
19. |
Porphyridium cruentum (purpureum) |
As the EPS concentration changed, there was a proportional increase in the respiratory burst (RB), the total hemocytes (THC) value and phagocytotic activity (PA). |
50 |
20. |
Bifidobacterium longum subsp. longum 35624 |
A lowered production of proinflammatory markers, elevated levels of IL-10 and an induction of T regulatory (Treg) cells |
51 |
21. |
B. longum BCRC 14634 |
Elevated anti-inflammatory IL10 cytokine production and decreased TNF-α secretion |
52 |
22. |
B. animalis subsp. lactis IPLA-R1 |
Elevated anti-inflammatory IL10 cytokine production and decreased TNF-α secretion |
53 |
23. |
Streptococcus thermophilus MN-BM-A01 |
A lowered production of proinflammatory markers and an increase in expression of tight junction protein |
31 |
24. |
Bacteroides fragilis |
Suppressing pro-inflammatory IL-17 production to provide a protective effect against Helicobacter hepaticus associated colitis |
54 |
25. |
Bifidobacterium breve UCC2003 |
Enhancing the mucosal secretion of pro-inflammatory IL-12, TNFα and INFγ that resulted in a protective effect against Citrobacter rodentium infection |
55 |
26. |
Lactobacillus plantarum N14 (LP14) |
Reduction in MCP-1, IL-8 and IL-6, which are pro-inflammatory cytokines in porcine intestinal epitheliocyte cell lines (PIE) cells on ETEC challenge, activation NF-κB via RP105 and TLR2 toll-like receptors |
42 |
27. |
Lactobacillus delbrueckii subsp. delbrueckii TUA4408L |
Downregulation in the production of inflammatory cytokines and inhibition of MAPKs and NFkB by upregulating TLR negative regulators |
56 |
28. |
Lactobacillus johnsonnii 142 |
Dendritic cell differentiation and preferentially induced immune responses from Th-2 cells. |
57 |
29. |
Lactobacillus casei WXD030 |
Better secretion of IFN-γ and IL-4 in CD4+ T, increase in IL-6, TNF-α and IL-1β secretion in RAW264.7 macrophages and an increase in the production of CD40, CD86, CD80 and MHC II on dendritic cell surfaces |
58 |
30. |
Lactobacillus rhamnosus KL37
|
Collagen specific- IgG antibody production decreased due to the action of crude EPS. |
59 |
31. |
Lactobacillus rhamnosus KL37 |
Inhibition of in vitro growth of T cells and reduction in the secretion of interferon (IFN)-γ |
60 |
32. |
Leuconostoc mesenteroides strain NTM048(NTM048 EPS) |
Induction of Ig-A production , Th1 and Th2 mediated response in splenocytes. |
61 |
33. |
Lactobacillus plantarum WLPL04 |
Inhibition of HT-29 proliferation, inhibition of adhesion of Escherichia coli O157:H7 and prevention of biofilm formation by pathogenic bacteria |
33 |
Bacteroides fragilis produces a polysaccharide A, PSA which was examined for its ability to protect from colitis induced by Helicobacter hepaticus. In the absence of PSA, there was an increase in cytokines that are pro-inflammatory in function and administration of PSA was seen to suppress interleukin-17 production.54 Studies on porcine systems are of importance because the porcine gastrointestinal tract resembles the human system better than mice. Several studies have used PIE cells to observe the nature of probiotic exopolysaccharides on the porcine gut pattern recognition receptors (PRRs).70 EPS from Lactobacillus delbrueckii subsp. delbrueckii TUA4408L (Ld) and its interaction with TLR2, TLR4, and TLR negative regulators was studied in porcine intestinal epitheliocyte cell lines (PIE cells) and a downregulation in the secretion of inflammatory cytokines was observed.56
The use of EPS of Lactobacillus plantarum N14 (LP14) strain reduced the secretion of cytokines such as MCP-1, IL-6 and IL-8 and in PIE cells on ETEC challenge. The TLRs of Lactobacillus plantarum N14 (LP14) EPS called radioprotective 105 (RP105)/MD1 complex, along with 2 others namely, TLR4 and TLR2 were examined for their involvement in immunoregulation in HEKRP105/MD1 and HEKTLR2 cells. The results showed that NF-κB activation was through RP105 and TLR2. PIE cells with knocked out TLR4, TLR2 and RP105 showed a reduction in the ability to modulate pro-inflammatory cytokine expression indicating that the EPS of LP14 is capable of reducing inflammation in a RP105/MD1-dependend manner.42 The polysaccharide from Lactococcus lactis subsp. cremoris induced NF-kBs in human embryonic kidney (HEK) 293 cells constructed with swine TLR2 (sTLR2).71 Since NF-kB is a crucial player in array of diseases such as cancers and in coordinating inflammatory responses, EPSs could be harnessed for their beneficial properties.72
TLR4 dimerizes when activated and causes an initiation of a response that is pro-inflammatory in nature. Binding of a ligand to a TLR mediates signalling via a myeloid differentiation primary response gene 88 (MyD88)-dependent pathway or a MyD88-independent pathway. The MyD88-dependent signalling causes NFkB to activate transcription of pro-inflammatory cytokine genes. MyD88-dependent pathway also triggers the mitogen-activated protein kinase (MAPKs) such as JNK and p38 to get activated70. The Ld EPS inhibited activation of MAPKs and NFkB by upregulating TLR negative regulators.56
EPS influenced immune cell alterations
The EPS from Lactobacillus delbrueckii (L. delbrueckii) subssp. bulgaricus OLL1073R was capable of activating natural killer cells and inducing the production of interferon gamma (IFN-g) in mice.43 The immunomodulatory properties of exopolysaccharide of Lactobacillus johnsonnii 142 were tested on healthy mice and mice that were induced with inflammatory bowel disease (IBD). The study revealed that the composition of EPS differed in mice with IBD and without. The EPS promoted dendritic cell differentiation and preferentially induced immune responses from Th-2 cells.57 Due to the toxicity of existing adjuvants such as Freund’s complete adjuvant (FCA) and lipopolysaccharide (LPS), only adjuvants like alum that are partial adjuvants have gained clinical acceptance. EPS of probiotic Lactobacillus casei WXD030 (L-EPS) was assessed for its adjuvant properties. It induced better production of IL-4 and IFN-g in CD4+ T cells when compared to alum, thus improving cytokine secretions and helper T-cell responses 58.The L-EPS also caused an increase in the production of MHC II, CD80, CD86, and CD40 on dendritic cell surfaces and an elevation in RAW264.7 macrophage production of TNF-a, IL-6 and IL-1β when treated with the EPS.58 Collagen specific- IgG antibody production decreased due to the action of crude EPS of Lactobacillus rhamnosus KL.37 In mice there was an improvement in the condition of collagen-induced arthritis (CIA) when LPS was used as an adjuvant.59 They furthered their research with the use of highly purified form of the EPS to eliminate the contribution of other components in the crude extract to the beneficial effects of the EPS. The EPS from Leuconostoc mesenteroides strain NTM048(NTM048 EPS) has a molecular weight of 10-40kDA and is made of glucose and fructose. It has shown the ability to induce Ig-A production in the Peyer′s patch cells.61
Cell type specific anti-tumor properties of exopolysaccharides
Cancer is a condition when cells undergo rapid division and deprive healthy tissues of nutrition. Breast, lung, prostate, colon, rectum, and skin cancers are some of most frequently occurring cancers in the world.73 The potential for EPS from probiotics to be used as anti-cancer agents revolves around the prevention of tumor formation, apoptosis of cancer cells and immunity improvement. The intrinsic apoptotic pathway involves BCl-2, BAX, caspase-9 and caspase-3. The extrinsic pathway has caspase-10 and caspase-8. Caspase-3 activation generally demonstrates nuclear fragmentation, cell shrinkage and chromatin condensation without causing damage to healthy tissues.13 The action of exopolysaccharides on different cancer cell lines has been examined and some researchers have shown a positive link between EPS action and the inhibition of cancer cells. The activity of EPS116, the EPS from Lactobacillus plantarum (L. plantarum) NCU116 and its role in cancer cells was studied in CT26 cells which are a type of mouse intestinal epithelial cancer cells. The EPS116 inhibited the growth and proliferation of the CT26 cells by inducing the apoptotic pathway. This was indicated by a marked increase in the activity of genes that are pro-apoptotic in nature, such as Fasl, Fas, and c-Jun and an upregulation of TLR2 (Toll like receptor 2). The anti-cancer activity of EPS116 may be due to the c-Jun dependent Fas/Fasl-mediated apoptosis via TLR2 23(p116). The EPSs produced by G10 and H15 which are 2 strains of L. gasseri, isolated from a healthy human vagina when examined for their ability to prevent cervical cancer cell (HeLa) growth were found to initiate apoptosis in HeLa cells with an increase of Caspase 3 and Bax. The monosaccharide composition of the EPS played a role in apoptosis induction.49 The EPS from L. plantarum YW32 showed repressive activity against the colon cancer cell line, HT-29 (44(p32)63). A suppression in HT-29 cell growth was also observed when EPS from L. casei M5, L. casei SB27, L.casei K11 and L. casei X12 strains were used. This was owing to the induction of G0/G1 cell cycle arrest and apoptosis46(p29). The tumor suppressive effects of EPS from Lactobacillus acidophilus DSMZ 20079 was observed in Human colon cancer (CaCo-2) and Human breast cancer (MCF7) cell lines. It was found that there was an increase in the production of IKba, P53 and TGF genes due to the action of the EPS.45 The oligosaccharide showed regulatory effects on the apoptotic and NF-kB inflammatory pathways45. The action of EPS from Lactobacillus acidophilus was observed on colon cancer cell lines namely HCT15 and CaCo2. It was found that EPS inhibits the expression of genes needed for tumor angiogenesis and survival by downregulating the expression of vascular endothelial growth factor (VEGF), hypoxia-inducible factor-1a (HIF-1a) and upregulating the expression of tissue inhibitor of metalloproteinases-3 and other proteins associated with angiogenesis.47 Along with antibacterial and antiviral properties, the previously mentioned EPS obtained from marine Bacillus subtilis SH1 also displayed antitumor property against HCT-116, HepG2 and MCF-7.36 Acidic EPS of marine Bacillus amyloliquefaciens 3MS 2017 (BAEPS), showed anti-breast cancer (MCF-7) activity in female rats via mechanisms such as inhibition of cyclooxygenase-2 and by influencing cancer-growth-rate-limiting enzymes such as aromatase and Na+/K+.28
Several findings indicate that EPS as an anti-cancer agent, inhibits the cell proliferation in a cell-type specific manner resulting in non-identical cellular responses to EPS treatment. Bacterial polysaccharides examined for their anticancer activity against human cancer cell lines showed that the EPS works efficiently as a therapeutic agent against breast cancer MCF7 cells by displaying cytotoxicity towards MCF7 at low concentrations without showing any cytotoxicity against normal cells.74 In another study, Lactobacillus fermentum NCIMB 5221 when used against colon cancer cells supresses colon cancer cells and promotes normal epithelial colon cell growth.75 These findings indicate that the probiotic bacteria selectively target cancer cells. This may be due to the presence of propionate and butyrate, as histone deacetylase inhibitors (HDACs) stimulate proliferation in healthy colon cells but kill cancer cells.76 A study reported that Enterococcus lactis IW5 byproducts reduced the viability of different cancer cells, such as MCF7, HeLa, AGS, and HT-29. They reported that the primary mechanism used by exopolysaccharides to combat cancer cells is via the activation of apoptosis in cancer cells.77
Exopolysaccharides as modulators of the intestinal microbiota
Studies on immunomodulation have shown a strong corelation between reduced levels of Bifidobacterium in the gut and increased incidence of atopic diseases in children between 3 months and 3 years. This is because these organisms are able to regulate immune responses by altering of the gut microbiota.53 Probiotic strains of Lactobacilli and Bifidobacterium are found to influence the immune system by controlling the production of proinflammatory and anti-inflammatory cytokines and T helper (Th) cells.78 A study found that Lactobacillus-exposed human myeloid dendritic cells (MDCs)up-regulated the expression of different clusters of differentiation and secreted high levels of IL-18 and IL-12.79 Lactobacillus kefiranofaciens present in the dairy product kefir produces an EPS called kefiran that has shown the capacity to interfere with pathogen growth.80 EPS interacts with toxins produced by bacteria and interferes with their adhesion onto eukaryotic cells. Some EPS act as scavengers that chelate bacterial toxins and others block the toxin receptors on cell surfaces.30 Wild type Lactobacillus rhamnosus GG (LGG) containing long galactose-rich exopolysaccharides (EPS) were found to perform better in the lower parts of the intestine when compared to the isogenic EPS mutant CMPG5351.63 The EPS of Bacillus amyloliquefaciens called EPS-K4 has shown the ability to secrete mucin that helps maintain the integrity of the gut barrier and reduce production of endotoxins in the serum.21
In the past few years, researchers have shown a lot of interest in using exopolysaccharides. This is owing to the various health benefits conferred by them. They are preferred over whole probiotic cells due to the risks involved with the use of whole cells among immunocompromised individuals. There are various different types of exopolysaccharides that are classified based on structure, function and their location within the cell. Although they are associated with several industries such as food and cosmetics, their role as health promoters has garnered maximum attention. Exopolysaccharides influence the way microorganisms interact in the host gut and the way the host immune system responds to these organisms. These exopolysaccharides have proven to be useful as modulators of the immune system, as anti-biofilm forming agents and have shown to be effective against cholesterol, cancer and viruses too. Some EPS evade exaggerated immune responses and others aid the host organism to compete with the pathogens for common niches.
Several beneficial aspects of EPS have been highlighted in this review. But, for the full potential of EPS to be explored there is a need for more research related to their structure and function. They have tremendous potential to be used in the medical field as their immunomodulatory properties can be of therapeutic benefit to humans. A better understanding of their mechanism of modulation will help in utilizing them efficiently. But for future research there is a need to shed more light on their structural composition as the structure directly influences function. Even though several researchers have elucidated the structure of different polysaccharides, the information is insufficient to determine the exact structure–function relationships. One of the reasons why there hasn’t been enough data on the structure of EPS is due to the low EPS yield. Scientists can also explore the possibility of synthesizing EPS artificially as modifications to create specific EPS may enhance specific functions of EPS. The procedures used for extracting EPS also need to be precise as impurities such as lipopolysaccharides may hinder the extraction process or alter the properties of the EPS. Failing to do this, the information on chemical composition, molecular weight and the structure-function associations will be far from accurate. If we intend to see a greater use of exopolysaccharides in the health sector, it is necessary to have more insights on their positive effects.
ACKNOWLEDGMENTS
The authors would like to thank Department of Life Sciences and Centre for Research, CHRIST (Deemed to be University), India. Graphical illustrations were made using images from Servier Medical Art (https://smart.servier.com/), reproduced under Creative Commons License attribution 3.0 Unported License.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
This study was supported by CHRIST (Deemed to be University), Bengaluru, India. Under the Funding Number MRP-DSC_1936.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
Not applicable.
- Ozen M, Dinleyici EC. The history of probiotics: the untold story. Benef Microbes. 2015;6(2):159-165.
Crossref - Islam SU. Clinical Uses of Probiotics. Medicine (Baltimore). 2016;95(5):e2658.
Crossref - Kim JA, Bayo J, Cha J, et al. Investigating the probiotic characteristics of four microbial strains with potential application in feed industry. PLoS One. 2019;14(6):e0218922.
Crossref - Hill C, Guarner F, Reid G, et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. 2014;11(8):506-514.
Crossref - Song D, Ibrahim S, Hayek S. Recent Application of Probiotics in Food and Agricultural Science. 2012.
Crossref - Shenoy A, Gottlieb A. Probiotics for oral and vulvovaginal candidiasis: A review. Dermatol Ther. 2019;32(4):e12970.
Crossref - Doron S, Snydman DR. Risk and Safety of Probiotics. Clin Infect Dis. 2015; 60 (suppl_2): S129-S134.
Crossref - Abdalla AK, Ayyash MM, Olaimat AN, et al. Exopolysaccharides as Antimicrobial Agents: Mechanism and Spectrum of Activity. Front Microbiol. 2021;12:664395.
Crossref - Gaspar C, Donders GG, Palmeira-de-Oliveira R, et al. Bacteriocin production of the probiotic Lactobacillus acidophilus KS400. AMB Express. 2018;8(1):153.
Crossref - Hoseinifar SH, Sun YZ, Wang A, Zhou Z. Probiotics as Means of Diseases Control in Aquaculture, a Review of Current Knowledge and Future Perspectives. Front Microbiol. 2018.
Crossref - Pavlova AS, Ozhegov GD, Arapidi GP, et al. Identification of Antimicrobial Peptides from Novel Lactobacillus fermentum Strain. Protein J. 2020;39(1):73-84.
Crossref - Chang CJ, Lin TL, Tsai YL, et al. Next generation probiotics in disease amelioration. J Food Drug Anal. 2019;27(3):615-622.
Crossref - Angelin J, Kavitha M. Exopolysaccharides from probiotic bacteria and their health potential. Int J Biol Macromol. 2020;162:853-865.
Crossref - Dinic M, Pecikoza U, Djokic J, et al. Exopolysaccharide Produced by Probiotic Strain Lactobacillus paraplantarum BGCG11 Reduces Inflammatory Hyperalgesia in Rats. Front Pharmacol. 2018;9:1.
Crossref - Yasmin I, Saeed M, Khan WA, et al. In Vitro Probiotic Potential and Safety Evaluation (Hemolytic, Cytotoxic Activity) of Bifidobacterium Strains Isolated from Raw Camel Milk. Microorganisms. 2020;8(3):354.
Crossref - Somashekaraiah R, Shruthi B, Deepthi BV, Sreenivasa MY. Probiotic Properties of Lactic Acid Bacteria Isolated From Neera: A Naturally Fermenting Coconut Palm Nectar. Front Microbiol. 2019.
Crossref - Behnsen J, Deriu E, Sassone-Corsi M, Raffatellu M. Probiotics: Properties, Examples, and Specific Applications. Cold Spring Harb Perspect Med. 2013;3(3):a010074.
Crossref - Plaza-Diaz J, Ruiz-Ojeda FJ, Gil-Campos M, Gil A. Mechanisms of Action of Probiotics. Adv Nutr. 2019;10(suppl_1):S49-S66.
Crossref - Fijan S. Microorganisms with Claimed Probiotic Properties: An Overview of Recent Literature. Int J Environ Res Public Health. 2014;11(5):4745-4767.
Crossref - Vazquez-Rodriguez A, Vasto-Anzaldo XG, Leon-Buitimea A, Zarate X, Morones-Ramirez JR. Antibacterial and Antibiofilm Activity of Biosynthesized Silver Nanoparticles Coated With Exopolysaccharides Obtained From Rhodotorula mucilaginosa. IEEE Trans NanoBioscience. 2020;19(3):498-503.
Crossref - hua KJ, Yan HY, Shuang HJ, Jia YJ, Yu ZQ, Mei LD. Exopolysaccharides from Bacillus amyloliquefaciens DMBA-K4 ameliorate dextran sodium sulfate-induced colitis via gut microbiota modulation. J Funct Foods. 2020;75:104212.
Crossref - Xie Z, Bai Y, Chen G, et al. Modulation of gut homeostasis by exopolysaccharides from Aspergillus cristatus (MK346334), a strain of fungus isolated from Fuzhuan brick tea, contributes to immunomodulatory activity in cyclophosphamide-treated mice. Food Funct. 2020;11(12):10397-10412.
Crossref - Zhou X, Hong T, Yu Q, et al. Exopolysaccharides from Lactobacillus plantarum NCU116 induce c-Jun dependent Fas/Fasl-mediated apoptosis via TLR2 in mouse intestinal epithelial cancer cells. Sci Rep. 2017;7(1):14247.
Crossref - Ciszek-Lenda M. Biological functions of exopolysaccharides from probiotic bacteria. Cent Eur J Immunol. 2011:36(1):51-55.
- Saadat YR, Khosroushahi AY, Gargari BP. A comprehensive review of anticancer, immunomodulatory and health beneficial effects of the lactic acid bacteria exopolysaccharides. Carbohydr Polym. 2019;217:79-89.
Crossref - Silva LA, Lopes Neto JHP, Cardarelli HR. Exopolysaccharides produced by Lactobacillus plantarum: technological properties, biological activity, and potential application in the food industry. Ann Microbiol. 2019;69(4):321-328.
Crossref - Xu X, Peng Q, Zhang Y, et al. Antibacterial potential of a novel Lactobacillus casei strain isolated from Chinese northeast sauerkraut and the antibiofilm activity of its exopolysaccharides. Food Funct. 2020;11(5):4697-4706.
Crossref - Ibrahim AY, Youness ER, Mahmoud MG, Asker MS, El-Newary SA. Acidic Exopolysaccharide Produced from Marine Bacillus amyloliquefaciens 3MS 2017 for the Protection and Treatment of Breast Cancer. Breast Cancer Basic Clin Res. 2020;14:1178223420902075.
Crossref - Juraskova D, Ribeiro SC, Silva CCG. Exopolysaccharides Produced by Lactic Acid Bacteria: From Biosynthesis to Health-Promoting Properties. Foods Basel Switz. 2022;11(2):156.
Crossref - Ruas-Madiedo P. Biosynthesis and Bioactivity of Exopolysaccharides Produced by Probiotic Bacteria. Food Oligosaccharides. 2014:118-133.
Crossref - Chen Y, Zhang M, Ren F. A Role of Exopolysaccharide Produced by Streptococcus thermophilus in the Intestinal Inflammation and Mucosal Barrier in Caco-2 Monolayer and Dextran Sulphate Sodium-Induced Experimental Murine Colitis. Molecules. 2019;24(3):513.
Crossref - Riaz Rajoka MS, Jin M, Haobin Z, et al. Functional characterization and biotechnological potential of exopolysaccharide produced by Lactobacillus rhamnosus strains isolated from human breast milk. LWT. 2018;89:638-647.
Crossref - Liu Z, Zhang Z, Qiu L, et al. Characterization and bioactivities of the exopolysaccharide from a probiotic strain of Lactobacillus plantarum WLPL04. J Dairy Sci. 2017;100(9):6895-6905.
Crossref - Trabelsi I, Ktari N, Ben Slima S, et al. Evaluation of dermal wound healing activity and in vitro antibacterial and antioxidant activities of a new exopolysaccharide produced by Lactobacillus sp.Ca6. Int J Biol Macromol. 2017;103:194-201.
Crossref - Jeong D, Kim DH, Kang IB, et al. Characterization and antibacterial activity of a novel exopolysaccharide produced by Lactobacillus kefiranofaciens DN1 isolated from kefir. Food Control. 2017;78:436-442.
Crossref - Hassan SWM, Ibrahim HAH. Production, Characterization and Valuable Applications of Exopolysaccharides from Marine Bacillus subtilis SH1. Pol J Microbiol. 2017;66(4):449-461.
Crossref - Hsieh SA, Allen PM. Immunomodulatory Roles of Polysaccharide Capsules in the Intestine. Front Immunol. 2020;11:690.
Crossref - Sassone-Corsi M, Raffatellu M. No vacancy: how beneficial microbes cooperate with immunity to provide colonization resistance to pathogens. J Immunol Baltim Md. 2015;194(9):4081-4087.
Crossref - Chen CC, Walker WA. Probiotics and Prebiotics: Role in Clinical Disease States. Adv Pediatr. 2005;52:77-113.
Crossref - Pyclik M, Srutkova D, Schwarzer M, Gorska S. Bifidobacteria cell wall-derived exo-polysaccharides, lipoteichoic acids, peptidoglycans, polar lipids and proteins – their chemical structure and biological attributes. Int J Biol Macromol. 2020;147:333-349.
Crossref - Bleau C, Monges A, Rashidan K, et al. Intermediate chains of exopolysaccharides from Lactobacillus rhamnosus RW-9595M increase IL-10 production by macrophages. J Appl Microbiol. 2010;108(2):666-675.
Crossref - Murofushi Y, Villena J, Morie K, et al. The toll-like receptor family protein RP105/MD1 complex is involved in the immunoregulatory effect of exopolysaccharides from Lactobacillus plantarum N14. Mol Immunol. 2015;64(1):63-75.
Crossref - Makino S, Sato A, Goto A, et al. Enhanced natural killer cell activation by exopolysaccharides derived from yogurt fermented with Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1. J Dairy Sci. 2016;99(2):915-923.
Crossref - Wang J, Zhao X, Yang Y, Zhao A, Yang Z. Characterization and bioactivities of an exopolysaccharide produced by Lactobacillus plantarum YW32. Int J Biol Macromol. 2015;74:119-126.
Crossref - El-Deeb NM, Yassin AM, Al-Madboly LA, El-Hawiet A. A novel purified Lactobacillus acidophilus 20079 exopolysaccharide, LA-EPS-20079, molecularly regulates both apoptotic and NF-kB inflammatory pathways in human colon cancer. Microb Cell Factories. 2018;17(1):29.
Crossref - Di W, Zhang L, Yi H, Han X, Zhang Y, Xin L. Exopolysaccharides produced by Lactobacillus strains suppress HT 29 cell growth via induction of G0/G1 cell cycle arrest and apoptosis. Oncol Lett. 2018;16(3):3577-3586.
Crossref - Deepak V, Ramachandran S, Balahmar RM, et al. In vitro evaluation of anticancer properties of exopolysaccharides from Lactobacillus acidophilus in colon cancer cell lines. Vitro Cell Dev Biol – Anim. 2016;52(2):163-173.
Crossref - Ghany KA, Elhafez EA, Hamouda R, Mahrous H, Ahmed F, Hamza H. Evaluation of Antioxidant and Antitumor Activities of Lactobacillus acidophilus Bacteria Isolated from Egyptian Infants. Int J Pharmacol. 2014;10(5):282-288.
Crossref - Sungur T, Aslim B, Karaaslan C, Aktas B. Impact of Exopolysaccharides (EPSs) of Lactobacillus gasseri strains isolated from human vagina on cervical tumor cells (HeLa). Anaerobe. 2017;47:137-144.
Crossref - Risjani Y, Mutmainnah N, Manurung P, Wulan SN, Yunianta null. Exopolysaccharide from Porphyridium cruentum (purpureum) is Not Toxic and Stimulates Immune Response against Vibriosis: The Assessment Using Zebrafish and White Shrimp Litopenaeus vannamei. Mar Drugs. 2021;19(3):133.
Crossref - Schiavi E, Gleinser M, Molloy E, et al. The Surface-Associated Exopolysaccharide of Bifidobacterium longum 35624 Plays an Essential Role in Dampening Host Proinflammatory Responses and Repressing Local TH17 Responses. Appl Environ Microbiol. 2016;82(24):7185-7196.
Crossref - Wu MH, Pan TM, Wu YJ, Chang SJ, Chang MS, Hu CY. Exopolysaccharide activities from probiotic bifidobacterium: Immunomodulatory effects (on J774A.1 macrophages) and antimicrobial properties. Int J Food Microbiol. 2010;144(1):104-110.
Crossref - Ruiz L, Delgado S, Ruas-Madiedo P, Sanchez B, Margolles A. Bifidobacteria and Their Molecular Communication with the Immune System. Front Microbiol. 2017;8:2345.
Crossref - Mazmanian SK, Round JL, Kasper DL. A microbial symbiosis factor prevents intestinal inflammatory disease. Nature. 2008;453:620-625.
Crossref - Fanning S, Hall LJ, Cronin M, et al. Bifidobacterial surface-exopolysaccharide facilitates commensal-host interaction through immune modulation and pathogen protection. Proc Natl Acad Sci U S A. 2012;109(6):2108-2113.
Crossref - Wachi S, Kanmani P, Tomosada Y, et al. Lactobacillus delbrueckii TUA4408L and its extracellular polysaccharides attenuate enterotoxigenic Escherichia coli-induced inflammatory response in porcine intestinal epitheliocytes via Toll-like receptor-2 and 4. Mol Nutr Food Res. 2014;58(10):2080-2093.
Crossref - Gorska S, Sandstrom C, Wojas-Turek J, et al. Structural and immunomodulatory differences among lactobacilli exopolysaccharides isolated from intestines of mice with experimentally induced inflammatory bowel disease. Sci Rep. 2016;6(1):37613.
Crossref - Xiu L, Zhang H, Hu Z, et al. Immunostimulatory activity of exopolysaccharides from probiotic Lactobacillus casei WXD030 strain as a novel adjuvant in vitro and in vivo. Food Agric Immunol. 2018;29(1):1086-1105.
Crossref - Nowak B, Ciszek-Lenda M, Srottek M, et al. Lactobacillus rhamnosus exopolysaccharide ameliorates arthritis induced by the systemic injection of collagen and lipopolysaccharide in DBA/1 mice. Arch Immunol Ther Exp Comment Instituti Immunol Ther Exp. 2012;60(3):211-220.
Crossref - Nowak B, Srottek M, Ciszek-Lenda M, et al. Exopolysaccharide from Lactobacillus rhamnosus KL37 Inhibits T Cell-dependent Immune Response in Mice. Arch Immunol Ther Exp. 2020;68(3):17.
Crossref - Matsuzaki C, Hayakawa A, Matsumoto K, Katoh T, Yamamoto K, Hisa K. Exopolysaccharides Produced by Leuconostoc mesenteroides Strain NTM048 as an Immunostimulant To Enhance the Mucosal Barrier and Influence the Systemic Immune Response. J Agric Food Chem. 2015;63(31):7009-7015.
Crossref - Thomassin JL, Lee MJ, Brannon JR, Sheppard DC, Gruenheid S, Moual HL. Both Group 4 Capsule and Lipopolysaccharide O-Antigen Contribute to Enteropathogenic Escherichia coli Resistance to Human a-Defensin 5. PLOS ONE. 2013;8(12):e82475.
Crossref - Lebeer S, Claes IJJ, Verhoeven TLA, Vanderleyden J, De Keersmaecker SCJ. Exopolysaccharides of Lactobacillus rhamnosus GG form a protective shield against innate immune factors in the intestine. Microb Biotechnol. 2011;4(3):368-374.
Crossref - Caggianiello G, Kleerebezem M, Spano G. Exopolysaccharides produced by lactic acid bacteria: from health-promoting benefits to stress tolerance mechanisms. Appl Microbiol Biotechnol. 2016;100(9):3877-3886.
Crossref - Hafez M, Hayes K, Goldrick M, Warhurst G, Grencis R, Roberts IS. The K5 Capsule of Escherichia coli Strain Nissle 1917 Is Important in Mediating Interactions with Intestinal Epithelial Cells and Chemokine Induction. Infect Immun. 2009;77(7):2995-3003.
Crossref - Xu CL, Wang YZ, Jin ML, Yang XQ. Preparation, characterization and immunomodulatory activity of selenium-enriched exopolysaccharide produced by bacterium Enterobacter cloacae Z0206. Bioresour Technol. 2009;100(6):2095-2097.
Crossref - Delves PJ, Martin SJ, Burton DR, Roitt IM. Roitt’s Essential Immunology. John Wiley & Sons; 2017.
- Sherwood L, Willey JM, Woolverton C. Prescott’s Microbiology. McGraw-Hill; 2011.
- Martin SJ, Burton DR, Roitt IM, Delves PJ. Roitt’s Essential Immunology. John Wiley & Sons; 2016.
- Laino J, Villena J, Kanmani P, Kitazawa H. Immunoregulatory Effects Triggered by Lactic Acid Bacteria Exopolysaccharides: New Insights into Molecular Interactions with Host Cells. Microorganisms. 2016;4(3):27.
Crossref - Tohno M, Shimosato T, Kawai Y, et al. Advanced molecular immunoassay system for immunobiotic lactic acid bacteria using a transfectant of Toll-like receptor 2. Anim Sci J. 2007;78(2):195-205.
Crossref - Mitchell S, Vargas J, Hoffmann A. Signaling via the NF-kB system. Wiley Interdiscip Rev Syst Biol Med. 2016;8(3):227-241.
Crossref - Pierce BA. Genetics Essentials: Concepts and Connections. WH Freeman; 2018.
- Vidhyalakshmi R, Vallinachiyar C. Apoptosis of Human Breast Cancer Cells (MCF-7) Induced by Polysacccharides Produced by Bacteria. J Cancer Sci Ther. 2013;5(2):031-034.
Crossref - Kahouli I, Malhotra M, Alaoui-Jamali M, Prakash S. In-Vitro Characterization of the Anti-Cancer Activity of th Probiotic Bacterium Lactobacillus Fermentum NCIMB 5221 and Potential against Colorectal Cancer Cells. J Cancer Sci Ther. 2015;7: 224-235.
Crossref - Khalil MA, Sonbol FI, Al-Madboly LA, Aboshady TA, Alqurashi AS, Ali SS. Exploring the Therapeutic Potentials of Exopolysaccharides Derived From Lactic Acid Bacteria and Bifidobacteria: Antioxidant, Antitumor, and Periodontal Regeneration. Front Microbiol. 2022;13:803688.
Crossref - Nami Y, Haghshenas B, Haghshenas M, Abdullah N, Yari Khosroushahi A. The Prophylactic Effect of Probiotic Enterococcus lactis IW5 against Different Human Cancer Cells. Front Microbiol. 2015;6:1317.
Crossref - Nicola S, Amoruso A, Deidda F, et al. Searching for the Perfect Homeostasis: Five Strains of Bifidobacterium longum From Centenarians Have a Similar Behavior in the Production of Cytokines. J Clin Gastroenterol. 2015;S126-S130.
Crossref - Mohamadzadeh M, Olson S, Kalina WV, et al. Lactobacilli activate human dendritic cells that skew T cells toward T helper 1 polarization. Proc Natl Acad Sci U S A. 2005;102(8):2880-2885.
Crossref - Rodrigues KL, Caputo LRG, Carvalho JCT, Evangelista J, Schneedorf JM. Antimicrobial and healing activity of kefir and kefiran extract. Int J Antimicrob Agents. 2005;25(5):404-408.
Crossref - Zhou X, Zhang D, Qi W, et al. Exopolysaccharides from Lactobacillus plantarum NCU116 Facilitate Intestinal Homeostasis by Modulating Intestinal Epithelial Regeneration and Microbiota. J Agric Food Chem. 2021;69(28):7863-7873.
Crossref
© The Author(s) 2023. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.