ISSN: 0973-7510
E-ISSN: 2581-690X
The preparation of spawn or inoculum and its subsequent cultivation form the first step in the oyster mushroom supply chain, the preparation of the inoculum is related to quality parameters in the edible fungi production, where the use of grains of different cereals is essential to obtain the spawn The aim of this study was to determine the quality of different formulations of spawn using grains as wheat, corn, rice and sorghum furthermore, the growth of the strain of the CP-50 of Pleurotus ostreatus for oyster mushroom production on barley straw, under rustic conditions. In the spawn colonization and the percentage of contamination statistically significant differences were observed (P <0.05), being the treatments based on sorghum in combination with wheat, corn and rice the first to complete this process without presenting an incidence of competing organisms. Regarding yield and biological efficiency, no statistically significant differences (P <0.05) were found when using different formulations of spawn, values ranging from 84.17% were observed for fructification with the wheat + rice formulation, up to 71.45% when the formulation of spawn based on sorghum + corn was used. The production cycles had a variation of up to ten days between treatments, obtaining a more cut cycle when the sorghum + corn formulation was used. The results suggest the feasibility of using lower cost grains such as sorghum which impact in this process translates into a better cost benefit relation for small producers.
Substrates, Biological Efficiency, Spawn Production, Cereal Grains.
Edible mushrooms are a traditional food consumed in Mexico and the world since ancient times, however, mushroom cultivation in the country dates back to the third decade of the twentieth century with the cultivation of Agaricus bisporus, followed by Pleurotus ostreatus. Currently this activity has spread throughout the country given the commercial interest and to its due to the high nutritional value of the cultivated species (Jimenez et al., 2013). Also to be a natural source of protein with a similar or higher content than some legumes, fiber, carbohydrates, antioxidants, polyphenols and essential minerals such as K, P, Na and Mg mainly (Suarez and Holguin, 2012; Maftoun et al., 2015).
The production of edible fungi in Latin America is represented mainly by countries such as Mexico (80.8%), Brazil (7.7%) and Colombia (5.2%), among others, whose estimated production is 62, 374 tons per year (Martinez, 2012), with most of it being sold fresh and with a lower proportion processed. Nowadays, the genus Pleurotus occupies the second place in the commercial production of edible fungi worldwide (Mora and Martinez-Carrera, 2007) due to the diversity of cultivable species that are adaptable to a wide range of temperatures and diverse materials, rich in lignin and cellulose (Ohaco et al., 2016).
The biotechnology of the cultivation of edible fungi involves essential practices such as isolation, maintenance of the strains, elaboration of inoculum, selection of substrates for planting and production of fruiting bodies (Gaitan et al., 2006). The term inoculum refers to the fungal mycelium propagated massively in grains such as sorghum or wheat, previously sterilized (Royse, 2003). The preparation of inoculum is related to quality parameters within the production of edible fungi; therefore, it has an important role in this process. Romero et al. (2007) mention that the inoculum preparation process has not been adopted by the majority of mushroom producers, due to the lack of knowledge of laboratory techniques required for its production. Within this first link in the mushroom production chain there are two types of seed: a) Mother or master seed; obtained directly from the fungal mycelium developed in culture medium and b) Commercial seed, where the substrate is inoculated through the mother seed (Romero et al., 2010a).
The cultivation of oyster mushrooms is developed on a small scale in the northeastern region of the state of Puebla, mainly in rustic modules; production spaces, where the process of obtaining fruiting bodies is carried out, which is the next step in the productive chain and refers to the treatment of the substrate (chopping, cleaning, hydrated, neutralized and pasteurized), and inoculation of the substrate with seed commercial in aseptic conditions, to start the fruiting and finally the harvest (Albores and Alvarez, 2015). The generation of technologies through research is one of the main aspects that contribute in the resolution of problems of different nature, however, to demonstrate their viability, these must be transferred to the productive systems (Garcia, 2016).
The transfer of technology that this study proposes is the production of secondary inoculum or commercial seed of the CP-50 of Pleurotus ostreatus in different formulations based on cereals; in search of obtaining the best characteristics in the mycelium; through this we can guarantee an adequate production under rustic conditions, achieving a positive impact on the nutritional quality of the basidiomas.
The production of the seed or inoculum was carried out within the laboratory 204 of the Agroecology Center (BUAP) Mexico, while the basidiomas production evaluation was carried out in a rustic module of the community of Xaltipan, population of the municipality of San Juan Xiutetelco, Puebla-Mexico. The dominant climate is humid temperate, with abundant rainfall in summer, has an average annual temperature of 15°C and a precipitation of 1 618 mm per year (Encyclopedia of the municipalities of Puebla, 2006).
Obtaining primary and secondary inoculum
The substrate used for the preparation of the primary inoculum or mother was wheat (Triticum aestivum L.), and broken corn (Zea mays L.), wheat (Triticum aestivum L.), sorghum (Sorghum bicolor L.) and rice (Oryza sativa L.) for obtaining the secondary inoculum.
Preparation of the mother seed
The procedure consisted of boiling 10 Kg of wheat (Triticum aestivum L.) in 15 L of water for 20 minutes and letting it stand for 10 minutes, then draining it and letting it cool at room temperature for 30 minutes, then adding the amount of 5 g of calcium hydroxide and 20 g of calcium sulfate in a homogeneous manner in order to alkalize the substrate and raise the pH to 9 (Romero, 2007). Later, it was packed in 20 bottles with a capacity of 1 L and sterilized for 60 min at 121°C, after 24 hours, when the bottles were completely cooled; each was inoculated with 22.5 cm2 of colonized agar from CP-50 of Pleurotus ostreatus. The incubation process after inoculation was performed at room temperature with indirect light and a photoperiod of approximately 10 hours for 25 days (Ríos et al., 2010).
Preparation of secondary or commercial seed
The grains of wheat, sorghum, corn and rice were hydrated through different procedures. The treatment of wheat and corn grains consisted of boiling the amount of 10 Kg respectively for 15 minutes, letting it stand for 10 minutes and then draining and cooling for 30 minutes, once the grains reached room temperature, the amount of was added 5 g of calcium hydroxide and 20 g of calcium sulfate. Sorghum grains, like rice grains, were washed and soaked for 12 hours in sterile water, then drained and added with 2.5 g of calcium hydroxide and 10 g of calcium sulfate for rice and 3 g of hydroxide of calcium and 12 g of calcium sulphate for the sorghum, per 10 kg of each. Once the substrates were impregnated, they were packed in polypropylene bags according to the proposed treatments, in quantities of 3 Kg (Table 1), obtaining a total of 3 repetitions for each treatment. When the bags were cooled after a period of 24 hours, they were inoculated with 100 g of seed in the laminar flow hood and were incubated for 21 days at 25°C.
Table (1):
Formulations in grains to be evaluated in the production phase of the secondary inoculum
| Code | Treatments | Description | Total formulation (%) | |||
|---|---|---|---|---|---|---|
| Wheat | Corn | Sorghum | Rice | |||
| ( Kg ) | ||||||
| T1 | Sorghum | 3 | 100 | |||
| T2 | Sorghum + Corn | 1.5 | 1.5 | 100 | ||
| T3 | Sorghum + Rice | 1.5 | 1.5 | 100 | ||
| T4 | Sorghum+ Wheat | 1.5 | 1.5 | 100 | ||
| T5 | Wheat + Rice | 1.5 | 1.5 | 100 | ||
| T6 | Wheat+ Corn | 1.5 | 1.5 | 100 | ||
| T7 | Wheat | 3 | 100 | |||
| T8 | Corn | 3 | 100 | |||
| T9 | Rice | 3 | 100 | |||
| T10 | Rice + Corn | 1.5 | 1.5 | 100 | ||
Obtaining production support for the production of basidiomas of the CP-50
For sowing it was used barley straw, the straw was mechanically fragmented into portions of 1 to 3 cm in length (Romero, 2010b) and were pasteurized in hot water at 80°C for 2 hours. (Bandopadhyay, 2013), after the pasteurization time, the substrate was transported to the sowing area to allow their cooling and the draining of excess moisture (around 30 min). Subsequently, the inoculation was carried out; 6 kg (wet weight) plastic bags of barley straw were prepared, homogenously inoculated with the previously prepared “seed” in a 1:10 ratio. The experimental design that was implemented is random blocks, which consists of six treatments with 20 repetitions each, resulting in 180 production units. The sown samples were incubated at room temperature (26 ± 2°C), when the mycelium of the fungus completely colonized the substrates and showed the appearance of primordia, the bags were transferred to the fructification room where appropriate humidity conditions were promoted (80%), temperature (28°C), indirect daylight and air extraction for 1 h, every 8 h (Garzon and Cuervo, 2008; Romero, 2010b).
Biological efficiency was evaluated and described (EB = weight of fresh fungi / weight of dry substrate used x 100 (Salmones et al., 1997), production rate (TP = EB / elapsed time from inoculation to the last harvest and the rate of biodegradation (TB = [initial substrate dry weight – final substrate dry weight / initial substrate dry weight] * 100) Productivity was expressed in terms of grams of fresh fungi per the number of total harvests (Chairez-Aquino et al., 2015).
Proximal chemical analysis of the basidiomas of strain CP-50
A proximal chemical analysis of the fruiting body obtained from treatment evaluated was performed, the basidiomas were dried at a temperature of 35°C for 36 h, then ground to a particle size of 0.2 m (AOAC, 2006). Soluble carbohydrates (CHS) were determined by the Antrona method (Koehler, 1952), for analysis of dry matter, ash, and crude protein, the Kjeldahl method was used, the nitrogen conversion factor used for the protein was 4.38. (Yang et al., 2001), for organic matter (MO = MS-Cen) and carbon (C = MO / 1.72) (Van Soest et al.,1991). The energy value was calculated with the following factors: Kcal / 100 g = protein X 2.62 + ethereal extract X 8.37 + carbohydrates X 3.48 (Matila et al., 2002).
The data obtained were processed in the statistical program SPSS Statistics version 17 (Stadistical Package for the Social Sciences) for Windows, performing an analysis of variance (ANOVA) and subsequently Tukey’s multiple comparisons test (P< 0.05) was applied to determine the differences between treatments.
Percentage of colonization and contamination
For the variables colonization percentage and percentage of contamination were found statistically significant differences (ANOVA, F10,4=2.7, P<0.041), observing better results when using to 100% sorghum formulations and 50% in combination with the other grains, as a basis for the production of commercial inoculum , whose colonization process was completed in an average time of 18 days, followed by treatments based on wheat and in combination with rice which completed their colonization process 21 days after being inoculated, these treatments being those with the lowest incidence of contaminating agents (Table 2) except for the T7 treatment in which the incidence of contaminants was 25%. Alemu (2013), reported a range of 15-20 days in the colonization of the fungus in sorghum grains incubated at 28°C in dark conditions and attributed the rapid invasion of the grain to the optimum temperature maintained in the incubation process. Pathmashini et al. (2008) reported a good development of the mycelium of P. ostreatus using sorghum as a substrate for the production of inoculum, this being, the second best after of African millet (Eleusine coracana L.). The grains attribute the possible variation in the colonization rate to the size of the grain used, since small grains have a higher number of inoculation points per kilogram than larger grains (Mamiro and Royse, 2008).
Table (2):
Mycelium growth in the different substrate formulations after 21 days of incubation
Treatments |
Colonization (%) |
Pollution (%) |
|---|---|---|
Sorghum |
100 a |
0a |
Sorghum + Corn |
100 a |
0a |
Sorghum + Rice |
100 a |
0a |
Wheat + Sorghum |
100 a |
5a |
Wheat + Rice |
100 a |
0a |
Wheat + Corn |
80 ab |
10 ab |
Wheat |
100 a |
15 b |
Corn |
70 b |
20 b |
Rice |
70 b |
20 b |
Rice + Corn |
70 b |
25 b |
Unequal lowercase letters indicate statistically significant differences between treatments (P< 0.05, Tukey).
For the case of the treatments with formulations of 100% corn, 100% rice and 50% corn + 50% rice, the colonization was very slow and was affected by the high incidence of contaminants, so they were not considered viable for the sowing in to barley straw.
Total production
In the comparison regarding the yields obtained in each treatment, no statistically significant differences were found (Fig. 1). The results obtained within this variable range from 1 078.3 g in Sorghum + Corn to 1 262.5 g when using the wheat + rice formulation for the inoculum. Bautista et al. (2005) report a range of 764.4 to 1 351 g obtained in 4 cuts in a rustic production module. Bermudez et al. (2001) report a yield of 897 g when cultivating a strain (P-184) of P. ostreatus in coffee pulp (Coffea arabica) using 0.5 Kg dry weight of substrate within a rustic production module. Romero et al. (2010b) report a production of 2 000 to 2 250 g when using 6 kg of barley straw substrate (wet weight) with the strain CP-50 of Pleurotus ostreatus.
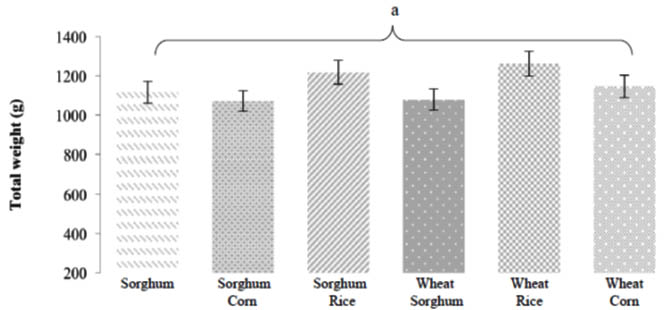
Fig. 1. Comparison of the yield obtained for each one of the inoculum formulations used
The shortest total production period was 52 days where the sorghum + rice formulation was used as the basis for the inoculum, followed by the sorghum + corn formulation whose production period was 54 days, being the largest production period in the treatment of sowing with the formulation of wheat + corn with a total of 66 days, obtaining three harvests in each treatment. In all the treatments, was obtained more than 50% of the total harvested during the first cut, observing that as the cuts went on, yield decreased.
Biological efficiency and production rate
Regarding the biological efficiency, no statistically significant differences were found in the sowing when using different formulations of secondary inoculum, values ranging from 84.17% were observed for the sowing done with the wheat + rice formulation, up to 71.45% in the sowing where the inoculum formulation based on sorghum + corn was used, these values are higher than those reported by Salmones et al. (1997) when inoculating P. ostreatus in barley straw ranging from 25.8 to 75.6%. Bautista et al. (2005) report values from 61.17 to 84.07% for the sowing of the HEMIM-50 strain of Pleurotus ostreatus in a rustic module, while Romero et al. (2010b) report values of 82.91% in corn stubble and 67.77% in straw of beans for the same strain used in this work. Bermudez et al. (2001) observed an EB of 179.4% when using coffee pulp as a growing medium for the P-184 strain of P. ostreatus. Girmay et al. (2016) used sorghum grains as a substrate for the inoculum, and reported a range of 9.73 to 74.17% when inoculating Pleurotus ostreatus on substrates such as sawdust, wheat straw and cottonseed.
With respect to the production rate the values oscillate between 0.89 in the sowing made with the formulation of inoculum based on sorghum, to 1.07 in the sowing made with the formulation sorghum + rice (Table 3), these values are within the range reported by Salmones et al. (1997) for P. ostreatus from 0.48 to 1.38, on the other hand, Romero et al. (2010b) report a TP of 1.77 and 1.90 when using the strain CP-50 of P.ostreatus in barley straw, and mention that a high production rate indicates a high biological efficiency within short production cycles. Within the parameter of the cultivation cycle there was a variation of up to 8 days between treatments and different statistical ranges were observed, with the lowest cycle being that of the sowing performed with the inoculum formulation based on sorghum + rice, while the cycle of The longest crop was obtained by using the wheat + corn formulation.
Table (3):
Biological efficiency, cycle of production and production rate
Formulación |
EB (%) |
CC (%) |
PR (%) |
|---|---|---|---|
Sorghum |
74.39 a |
83 a |
0.89 a |
Sorghum + Corn |
71.45 a |
77 dc |
0.93 a |
Sorghum +Rice |
81.17 a |
76 d |
1.07 a |
Sorghum + Wheat |
71.89 a |
79 bc |
0.91 a |
Wheat + Rice |
84.17 a |
81 ba |
1.04 a |
Wheat + Corn |
76.39 a |
84 ba |
0.94 a |
EB = Biological efficiency, CC = Culture cycle (days from the inoculation of the substrate with the different for-mulations until the end of the third harvest), PR = Production rate. Unequal lowercase letters indicate statistically significant differences between treatments (P< 0.05, Tukey).
The relative humidity within the module was maintained between 53.8 and 88.38% with an average of 71.09% in the incubation period, while during the fruiting period the relative humidity oscillated between 60% and 88% with an average of 74% until the last harvest The weekly temperature in the incubation period of the bags oscillated between 10.82°C (minimum) and 23.08°C (maximum) with an average of 17°C. While in the fruiting period, the average weekly temperature remained between 12°C (minimum) and 24.5°C (maximum) during the months of August and September with an average of 17.3°C. These data were compared with historical data about the weather in San Juan Xiutetelco.
Diameter of carpophores
During the fructification evaluation stage, the treatments required an average period of 22 days of incubation to colonize the substrates, where good mycelial growth was observed in all the treatments evaluated. 24 days after sowing, the appearance of primordia was observed in the bags inoculated with the different formulations whose development occurred in a range of 3 to 5 days in which they reached a commercial, size in which they were collected in the first cut. The sizes of the piles were greater in the sowing with the formulation of sorghum with a variation between 4.5 to 11.4 cm (Table 4) nevertheless no statistically significant differences with regard to this variable were found, reason why the use of different grains in the Inoculum elaboration did not affect the size of the carpophores, Romero et al. (2010b) report a variation of 4.7 to 8.5 cm in the fungal diameter of the CP-50 strain inoculated in banana residues and from 5.1 to 10.2 cm in wheat straw.
Table (4):
Diameters of fruiting bodies obtained in the three harvests in the different formulations
| Formulations* | ||||||
|---|---|---|---|---|---|---|
| Diameter (cm) | Sorghum | Sorghum Corn | Sorghum Rice | Sorghum Wheat | Wheat Rice | Wheat Corn |
| Ø higher | 11.4 a | 10.2 a | 10.6 a | 9.4 a | 10.5 a | 9.4 a |
| Ø minor | 4.5 a | 5.2 a | 4.8 a | 4.2 a | 4.6 a | 4.7 a |
* Unequal lowercase letters indicate statistically significant differences between treatments (P< 0.05, Tukey).
Proximal composition of the fruiting bodies produced
The conversion factor used from N to protein was 4.38 reported by Miles and Chang (1997), which excludes the non-protein nitrogen from the cell wall chitin of the fungi, instead of 6.25, which is used for the most of the food, in order not to overestimate the protein content (Sales et al., 2011). The protein content found in the fruiting bodies from the six formulations evaluated (Table 5) is within the range reported by Sales et al. (2011) from 11.96 to 21.16% and higher than those reported by Fernandes et al. (2015) who report a range of protein content from 9.29 to 14.7%.
Table (5):
Nutritional composition of the fruiting bodies from the evaluated formulations
Formulations |
Crude Protein (N x 4.38) |
Lipids |
Ashes |
Carbohydrates Totals a* |
Energy Kcal/100 g a |
|---|---|---|---|---|---|
Sorghum |
17.90 |
1.40 |
7.60 |
73.10b |
28.62b |
Sorghum+Corn |
17.30 |
2.20 |
7.00 |
73.50a |
29.26a |
Sorghum+Rice |
17.20 |
1.80 |
8.10 |
72.90b |
28.72b |
Sorghum+Wheat |
17.20 |
2.20 |
6.70 |
73.90a |
29.38a |
Wheat+Rice |
17.90 |
1.80 |
8.10 |
72.20c |
28.18c |
Wheat+Corn |
16.70 |
2.00 |
8.30 |
73.00b |
28.81b |
N = Nitrogen content, FDN = Neutral detergent fiber, FDA = Acid detergent fiber. * Unequal lowercase letters indicate statistically significant differences between treatments (α = 0.05, Tukey). Note: all data are presented as a percentage of dry weight with the ex-ception of humidity (percentage of fresh weight) and energy content. a: Calculated values.
The content of ashes found was 6.7 to 8.3%, the content found in the basidiomas coming from the sorghum + wheat formulation being lower. Sales et al. (2011) obtained content with a variation of 6.71 to 8.97%, as well as those reported by Fernandes et al. (2015) from 5.69 to 15.9%.
The content of crude lipids was higher in the fruiting bodies from the sowing with the formulation of sorghum + corn and sorghum + wheat, with values similar to those observed by Sales et al. (2011) with a range of 1.27 to 2.14%. The observed carbohydrate range was between 72.2 and 73.9%, which is higher compared to the value reported by Hung and Nhi (2012), of 61.3% for a commercial strain of Pleurotus ostreatus.
According to the results obtained, it is concluded that the best substrate for the elaboration of secondary inoculum due to the shorter time of colonization and lower percentage of contamination is the sorghum. In the case of treatments in which 50% combinations were used, better results were obtained in those whose combination included sorghum.
On the other hand, the production cycles had a variation of up to eight days between treatments, obtaining a more cut cycle when the sorghum + rice formulation was used.
The treatment with the largest diameter in the carpophores was with the sorghum formulation followed by the sorghum + rice formulation. However, in the other treatments, commercially acceptable diameters were obtained.
The data provided by the bromatological analyzes and those reported by other authors suggest that fungi are a good source of protein and fiber as well as being rich in minerals and low in fat content, this composition makes them a food of high nutritional value and medicinal. Currently, humanity faces serious food problems, one of the main ones being protein deficiency, especially in rural areas where the consumption of mushrooms is a good alternative in terms of food safety.
ACKNOWLEDGMENTS
The authors thank the VIEP Benemerita Universidad Autonoma de Puebla (BUAP) and CONACyT for the support in this research project.
- Albores-Pérez, B., Álvarez-Gutiérrez, P.E. Análisis de la cadena de valor de producción de setas (Pleurotus spp.) en cuatro municipios de Chiapas. Acta Universitaria. 2015; 25(6): 51-58.
- Alemu, F. Cultivation of Pleurotus ostreatus mushrooms on Coffee Arabica and Ficus sycomorus leaves in Dilla University, Ethiopia. Journal of Yeast and Fungal Research. 2013; 4(7): 103-108.
- AOAC. Official methods of analysis. (18th ed.), Association of Analyticla Chanemist, Washington, DC, USA. 2006.
- Bandopadhyay, S. Effect of supplementing rice straw with water hyacinth on the yield and nutritional qualities of oyster mushrooms (Pleurotus spp). Micología Aplicada International. 2013; 25(2):15-21.
- Bautista, N., Bautista-García, N. I., Acosta, U.L. Evaluación del cultivo de Pleurotus ostreatus sobre paja de trigo bajo condiciones rústicas en Galeana, Zacatepec, Estado de Morelos. Resúmenes, Primera Reunión Nacional sobre el Cultivo de Pleurotus en México. San Cristóbal de las Casas, Chiapas. 2005a; p. 13.
- Bermúdez, R., García, N., Gross, P., Serrano, M. Cultivation of Pleurotus on agricultural substrates in Cuba. Micología Aplicada International. 2001; 13(1): 25-29.
- Chairez-Aquino, J., Enríquez del Valle, L.J., Ruíz, G.V., Campos, R., Martínez-García. Uso del bagazo de Agave spp y hojas de maíz para cultivar el hongo Pleurotus ostreatus. Revista Mexicana de Agroecosistemas. 2015: 2(1): 23-28.
- Enciclopedia de los Municipios en Puebla. 2006.
- Fernández, A., Barros, L., Martins, A., Herbert, P., Ferreira, I. Nutritional characterisation of Pleurotus ostreatus (Jacq. ex Fr.) P. Kumm. produced using paper scraps as substrate. Food Chemistry. 2015; 396-400.
- Gaitán, H.R., Salmones D., Pérez-Merlo, R., Mata, G. Manual práctico del cultivo de setas Aislamiento, siembra y producción. Xalapa, Veracruz. Instituto de Ecología A.C. 2006.
- García, H.T. 2016. Transferencia de tecnología. Revista Facultad de Ingeniería. 2016; 12: 9-15.
- Garzón, G.J., Cuervo, A.J. Producción de Pleurotus ostreatus sobre residuos sólidos lignocelulósicos de diferente procedencia. Ciencias Biomédicas. 2008; 6(10): 126-140.
- Girmay, Z., Gorems, W., Birhanu, G., Zewdie, S. Growth and yield performance of Pleurotus ostreatus (Jack. Fr.) Kumm (Oyster mushroom) on different substrates. AMB Express. 2016; 6(8): 2-7.
- Hung, P., Nhi, N. 2012. Nutritional composition and antioxidant capacity of several edible mushrooms grown in the Southern Vietnam. International Food Research Journal. 2012; 19(2): 611-615.
- Jiménez, R.M., Pérez, M. J. Almaraz, S. J., Torres, A.M. Hongos silvestres con potencial nutricional, medicinal y biotecnológico comercializados en Valles Centrales, Oaxaca. Revista mexicana de ciencias agrícolas. 2013; 4(2): 199-213.
- Koehler, L.H. Differentiation of carbohydrates by antrone reaction, rate and color intensity. Analytical Chemistry. 1952; 24(10): 1576-1591.
- Maftoun, P., Helmi, J., Mohammad, S., Roslinda, M., Nor, Z., Hesham, A. The Edible Mushroom Pleurotus spp., I. Biodiversity and Nutritional Values. International Journal of Biotechnology for Wellness Industries. 2015; 4(2): 67-83.
- Mamiro, D., Royse, D. The influence of spawn type and strain on yield, size and mushrooms solids contents of Agaricus bisporus produced on non-composted and spent mushroom compost. Bioresource Technology. 2008; 99(8): 3205-3212.
- Martínez, G.M., Sihuanca, D., Macías, L.A., Pérez, M., Martínez, C.D., López, O. Characterization and production of Shiitake (Lentinula edodes) in Mexico using supplemented sawdust. African Journal of Biotechnology. 2012; 11(46): 10582-10588.
- Mattila, P., Salo-Väänänen, P., Könkö, K., Aro, H., Jalava, T. Basic composition and amino acid contents of mushroom cultivated in Finland. Journal of Agricultural and Food Chemistry. 2002; 50(22): 6419-6422.
- Miles, P.G., Chang, S.T. Mushroom biology: Concise basics and current developments. Singapore: World Scientific. 1997.
- Mora, V., Martínez, C.D. Investigaciones básicas, aplicadas y socioeconómicas sobre el cultivo de setas (Pleurotus) en México. Capítulo 1.1, 17 pp. In: El Cultivo de Setas Pleurotus spp. en México. J.E. Sánchez, D. Martínez-Carrera, G. Mata & H. Leal (eds). ECOSUR-CONACYT, México, D.F. 2007; 230 pp.
- Ohaco, E., De Michelis, A,; Valiente, L. Antioxidant capacity of frozen Pleurotus ostreatus during convective drying. Micología Aplicada International. 2016; 1-10.
- Pathmashini, L., Arulnandhy, V., Wilson, W. Cultivation of oyster mushroom (Pleurotus ostreatus) on sawdust. Ceylon Journal of Science. 2008; 37(2): 177-182
- Ríos, M., Hoyos, J.L., Mosquera, S.A. Evaluación de los parámetros productivos de la semilla de Pleurotus ostreatus propagada en diferentes medios de cultivo. Biotecnología en el Sector Agropecuario y Agroindustrial. 2010; 8(2): 86-94.
- Romero, A.O. M. Sc Thesis: Technological development to control green mold (Trichoderma spp.) during the commercial cultivation of edible fungi (Pleurotus and Lentinula) in México. Posgraduate College. Campus Puebla. 2007; 136 p.
- Romero, A.O., Damian-Huato M. A., Parraguirre-Lezama J. F. C., García-Espinoza A., Hernández Treviño I., Macías López A., Juárez H.J. Preparation of inoculum of Pleurotus ostreatus in laminar flow hood rustic. Scientific Research and Essays. 2010a; 5(24), 3945-3949.
- Romero-Arenas, O., Huerta, L., Damián H., Macías, L.A., Tapia, A., Parraguirre L.C., Juárez, H. Evaluación de la capacidad productiva de Pleurotus ostreatus con el uso de hoja de plátano (Musa paradisiaca L., cv. Roatan) deshidratada, en relación con otros sustratos Agrícolas. Agronomía Costarricense. 2010b; 34(1):53-63.
- Royse, D. Cultivation of oyster mushroom. Penn State’s College of Agricultural Sciences: Agricultural Research and Cooperative Extension. 2003.
- Sales, C.C., Medina, A.L., Texeira, M.M., Nogueira, M.C. Physiochemical analysis and centesimal composition of Pleurotus ostreatus mushroom grown in residues from the Amazon. Food Science and Technology. 2011; 31(2): 456-461.
- Salmones, D., Gaitan-Hernández, R., Pérez, R., Guzmán, G. Estudios sobre el género Pleurotus VIII. Interacción entre el crecimiento micelial y productividad. Revista Iberoamericana de Micología. 1997; 14:173-176.
- Suárez A., C., Holguín H.M. Evaluación de medios de cultivo sintéticos y cereales para la producción de semilla de setas comestibles. Revista Colombiana de Ciencias Hortícolas. 2012; 5(1): 130-140.
- Van Soest, P.J. Robertson, J.B., Lewis, B.A. Methods of dietary fibers, and nonstarch polysacarides in relations to animal nutrition. Symposium: Carbohidrate methodology, metabolism and nutricional implications in dairy cattle. Journal of Dairy Science. 1991; 74:3583-3597.
- Yang, J.H., Lin, H.C. Mau, J.L. Non-volatile taste components of several commercial mushrooms. Food Chemistry. 2001; 72(4): 465-467.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


