ISSN: 0973-7510
E-ISSN: 2581-690X
Five plant species namely, Phyllanthus acidus, Piper aduncum, Pandanus amaryllifolius, Macaranga peltata and Acacia mangium were analysed for their effective in-vitro bioactivity. The chloroform and aqueous extracted of the selected plants were subjected to TLC bioautography for antioxidant activity later all the extracted were subjected for DPPH assay where the chloroform extracts were found to express maximum antioxidant property. Amongst all the plants, Macaranga peltata accounted to 95% DPPH scavenging activity. The antimicrobial studies of the plant extracts were performed via agar well diffusion method, MIC determination, Biofilm inhibition assay in microtitre plate against clinical isolates like Staphylococcus aureus (MRSA) and Pseudomonas aeruginosa. It was found that Piper aduncum (chloroform and aqueous extract) and Macaranga peltata (only aqueous) expressed antibacterial activity, of which only chloroform extract of Piper aduncum could show negative influence against the biofilm development of P.aeruginosa.
DPPH scavenging, antibacterial activity, Staphylococcus aureus and Pseudomonas aeruginosa.
The ideology of utilising plants to treat diseases and to preserve human health dates back to ancient civilization which took its form into different system of medicinal practices namely Siddha, Unani and Ayurveda which are in practise till date. It is estimated that around 2,50,000 to 5,00,000 different types of plant source exist on earth but however, less than 11% are utilised as food or medicinal source by humans and other animals1,2. In recent years, the advancements in technology and techniques had led to better understanding of plants components which helped in recognizing various plant species for their bioactivity3-5. Plants are believed to produce bioactive compounds in order to protect themselves from microbial invasion. The presence of phytochemicals especially secondary metabolites such as alkaloids, tannins, saponins, flavonoids, sterols, terpenoids are deemed to be the main supporting factor of the antimicrobial activity of the plant against invading pathogens6-13. Though the ancient system of medicine gives knowledge about treatment of various diseases using medicinal plants, only the in-vitro and in-vivo analyses of these herbal plants could possibly explain and describe the effect of bioactive compounds against the causing microbes. However, the microbial revolution has led to the rise of new resistant strains against the existing drug. Apparently, the formulation or discovery of a new effective drug against the pathogens is very essential. The broad range and diversity of phytomolecules with potent bioactivity remains as the promising source for drug synthesis and new formulation14. To discuss a few bioactive compounds, Tannins are found to possess antiviral, antineoplastic and antibacterial activity15,16. Flavonoids are widely known for their antimicrobial activity, allergies, inflammation, prevent platelet aggregation17. Plant phenolics, vitamin C, vitamin E and carotenoids are known for their antioxidant activity15. Glycoside are reported to increase myocardial contraction18. Alkaloids are referred for range of pharmacological activities such as antimalarial, antineoplastic, antibacterial and anti-hyperglycaemic activities19,20. Other alkaloids like cocaine, nicotine and caffeine are known as nervous system stimulants and psychotropic agents.
Likewise, numerous new plants species were surveyed as crude extracts to report their in-vitro bioactivities. Analgesic, Anti-inflammatory, Antiviral, Anti-hepatotoxic and Anti-pyretic are the medicinal activities of Phyllanthus spp.21,22. Fruits of P. acidus and P. emblica are proven as rich source of vitamin C hence they are often used to improve general body wellness as well as preventive measures against diabetes and to relieve coughing symptoms21. A wide variety of medicinal plants belonging to Piperaceae, Moraceae, Euphorbiaceae, Clusiaceae, Berberidaceae, Asteraceae, etc., were identified for its anti-HIV activity, which also serves as the base molecule to derive effective modern drugs against HIV23. P. granatum and C. cyminum was seen effective in inhibiting growth of pathogenic bacteria such as S. aureus24. Even plant derived silver nanoparticles too showed antibacterial activity25,26.
This study deals with five plant species namely Phyllanthus acidus, Piper aduncum, Pandanus amaryllifolius, Macaranga peltata and Acacia mangium. The phytochemical separation was carried out by TLC bioautography for their crude extracts and their antioxidant property was analysed via TLC-DPPH bioautography and DPPH assay27-31. These plant extracts are subjected for in-vitro studies against multidrug resistant strains of Staphylococcus aureus and Pseudomonas aeruginosa to evaluate its antimicrobial and biofilm inhibition property.
Collection of Plants
The selected plants namely Phyllanthus acidus, Piper aduncum, Pandanus amaryllifolius, Macaranga peltata and Acacia mangium were collected from Kuala Lumpur and Puchong, Malaysia. Fresh and healthy leaves were collected and cleansed in running tap water and left to shade dry for 1 – 2 weeks.
Preparation of Plant Extracts
The dried plant leaves were ground into fine powder by an electric blender. The plant extracts were prepared by maceration using solvents (chloroform and water) in the ratio 1:10 (g of powder: mL of solvent)32. 50g of each plant powder were mixed with 500ml of respective solvents (chloroform and water) separately and placed under magnetic agitation for 30 min. The plant-solvent mixtures were allowed to react for a week and later vacuum filtered, followed by centrifugation at 8000 rpm for 5 min. The supernatant was lyophilized to obtain the solid residue of the crude extracts which was weighed and dissolved in respective solvents prior every analysis.
Phytochemical Screening
The plant extracts were screened for secondary metabolites such as Tannins, Phlobatannins, Saponins, Flavonoids, Terpenoids and Glycosides following Edeoga et al 33.
TLC Bioautography
TLC Bioautography were performed for separation of individual plant component in the extract using silica plates (Merck, F245)34. Each plant extract was loaded onto silica plates and the chloroform extracts were run having chloroform: benzene: acetonitrile: ethanol (3:2:0.5:0.5) and water extracts were run with the mobile phase of water: butyl alcohol: acetonitrile: ethanol: ethyl acetate (1:1:1:0.5:0.5) respectively. Thus, developed TLC plates were visualized under visible light, UV and iodine vapor. Rf (retention factor) were calculated from the below formula for each band developed under the different conditions.
Rf = Distance travelled by sample/Distance travelled by Solvent
TLC DPPH Bio-autography for Antioxidant Activity
The plant extracts were screened for antioxidant property by a preliminary method of DPPH (1, 1- diphenyl-2-picrylhydrazyl) spray technique. The developed plates were dried and sprayed with DPPH (0.004 % w/v in 95% methanol) and observed for development of yellow spot27.
Antioxidant DPPH assay
The free radical (DPPH) scavenging activity of the plant extracts were evaluated in terms of % by following Annegowda et al35 and Selvarani et al36. Ascorbic acid was used as the standard solution. Different concentrations of each plant extract such as 50, 100, 150, 200, 250 µg/ml were made up to 1 mL with methanol and added with 1 mL of DPPH solution (0.004% w/v in 95% methanol). The reaction mixture was incubated for 30 min at room temperature under dark. After incubation, the absorbance was read at 517nm against a reagent blank. The radical scavenging activity is calculated from the formula,
% free radical scavenged = [(Abcontrol – Absample) / Abcontrol] x 100
Antimicrobial Activity
Agar well diffusion method
Clinical isolated Staphylococcus aureus and Pseudomonas aeruginosa was used for antibacterial activity studies. The antibacterial activity of the plant extracts against the isolated cultures was checked by agar well diffusion method on Muller Hinton Agar (MHA) plates37. Stock solution of the plant extracts was prepared using DMSO as solvent for chloroform extracts and sterile water for aqueous extracts. Having their respective solvents as the negative control, the plant extracts were loaded in different concentrations such as 5 mg/ml, 10 mg/ml, 15 mg/ml and 20 mg/ml and positive control was maintained. Then, the plates were incubated at 37°C for 24 h.
Minimum Inhibition Concentration
The plant extract that showed positive results for antibacterial activity are proceeded to determine the minimal concentration of inhibition. From the zone of clearance (if any) formed in antibacterial activity, the respective concentration was serially diluted and added to culture laden microtitre plate and the MIC was identified according to Mgbeahuruike et al38.
Biofilm Inhibition Assay
To a sterile 96-well tissue culture plate, the plant extracts were pipetted in varied concentrations such as 4mg/mL, 8mg/mL, 12mg/mL and 16mg/mL in triplets and total volume of each well was made up to 100µL with Muller Hinton broth. Biofilm inhibition assay was performed as described earlier39.
Phytochemical Screening for Secondary Metabolites
The results obtained for qualitative phytochemical screening are tabulated as Table 1. According to Edeoga et al33 the presence of phytochemicals were represented via colour changes upon reaction with reagents. The chloroform plant extracts did not respond for presence of secondary metabolites but the aqueous extracts have projected the presence of several phytochemicals. The aqueous extracts of Phyllanthus acidus showed the presence of flavonoid and glycosides, Piper aduncum showed the presence of tannins and flavonoid while the methanol extract of Piper aduncum was reported to have other phytocompounds like alkaloids; triterpenoids, sterols, tannins, saponins and coumarins40. Tannin, terpenoids and glycosides were present in Pandanus amaryllifolius, tannin and flavonoid were present in Macaranga peltata and Acacia mangium was identified with tannin, flavonoid, terpenoid and glycosides. Jain et al.41 has identified the presence of flavonoids, glycosides, phenolic compounds, proteins, amino acids, carbohydrates and saponins from the aqueous extract of Phyllanthus acidus. Al-Rifai et al42 has also seen with similar outcome where secondary metabolite compounds such as flavonoids are more favourably isolated from polar extract.
Table (1):
Phytochemical screening from various qualitative biochemical tests.
| Phytochemicals | PLANT EXTRACTS | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| PA-C | PSPP-C | MP-C | AM-C | PA-W | PSPP-W | PDA-W | MP-W | AM-W | |
| Tannin | – | – | – | – | – | + | + | + | + |
| Phlobatannins | – | – | – | – | – | – | – | – | – |
| Flavonoids | – | – | – | – | + | + | – | + | + |
| Terpenoids | – | – | – | – | – | – | + | – | + |
| Glycosides | – | – | – | – | + | – | + | – | + |
(-) indicates the absence, and (+) indicates the presence. C- chloroform extract, W – aqueous extract. PA- Phyllanthus acidus, PSPP- Piper aduncum, PDA- Pandanus amaryllifolius, MP- Macaranga peltate, AM- Acacia mangium.
TLC and TLC- Bioautography
The chloroform extracts of P. acidus, P. aduncum, P. amaryllifolius, M. peltata and A. mangium were run with chloroform, benzene, acetonitrile and ethanol in the ratio 3:2:0.5:0.5 respectively and their respective water extracts were run with water, butyl alcohol, acetonitrile, ethanol and ethyl acetate in the ratio 1:1:1:0.5:0.5. And the plates were exposed to UV and iodine vapours to visualize the other possible components. On comparing the TLC results in Fig. 1 & 2, the chloroform extracts resolved better to water extracts with maximum number of bands. The Rf of each band was calculated and tabulated in Table 2 and Table 3. DPPH was sprayed on to the TLC developed plates for chloroform and water extracts in order to identify the antioxidant property. The scavenging activity of DPPH as yellow spots was observed over the resolved bands as seen in Fig. 1 (a-e iv) and Fig. 2 (a-e iv). This confirmed the presence of potent antioxidant phytochemicals. TLC Bioautography method is rather an easy way to screen plant extracts for its antioxidant activity, where Wang et al44 was able to identify three antioxidant compounds with the incorporation of this technique alongside with UV, MS and NMR spectra.
Table (2):
Rf value of compounds separated from chloroform plant extracts.
P.acidus |
P.aduncum |
P. amaryllifolius |
M. peltate |
A. mangium |
|---|---|---|---|---|
0.11 |
0.62 |
0.08 |
0.73 |
0.46 |
0.33 |
0.64 |
0.55 |
0.89 |
0.55 |
0.62 |
0.73 |
0.60 |
0.95 |
0.62 |
0.68 |
0.77 |
0.67 |
0.97 |
0.66 |
0.77 |
0.86 |
0.73 |
0.71 |
|
0.86 |
0.93 |
0.88 |
0.80 |
|
0.93 |
0.97 |
0.93 |
0.88 |
|
0.97 |
0.93 |
|||
0.97 |
Table (3):
Rf value of compounds separated from aqueous plant extracts.
P.acidus |
P.aduncum |
P. amaryllifolius |
M. peltate |
A. mangium |
|---|---|---|---|---|
0.13 |
0.33 |
0.39 |
0.90 |
0.23 |
0.19 |
0.78 |
0.51 |
0.97 |
0.27 |
0.47 |
0.90 |
0.90 |
0.48 |
|
0.88 |
0.92 |
0.70 |
||
0.94 |
0.87 |
Fig. 1. TLC and TLC DPPH Bio-autography results of chloroform extracts of a) Phyllanthus acidus, b) Piper aduncum, c) Pandanus amaryllifolius, d) Macaranga peltata, e) Acacia mangium. i) Visible light ii) UV iii) Iodine vapour and iv) DPPH Sprayed.
Fig. 2. TLC and TLC DPPH Bio-autography results of aqueous extracts of a) Phyllanthus acidus, b) Piper aduncum, c) Pandanus amaryllifolius, d) Macaranga peltata, e) Acacia mangium. i) Visible light ii) UV iii) Iodine vapour and iv) DPPH Sprayed.
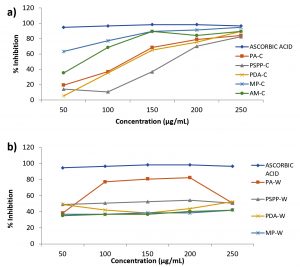
DPPH assay
Having ascorbic acid as the standard sample, the % of DPPH scavenged by the plant extracts were calculated and compared. On relating with the aqueous extracts in Fig. 3b, the chloroform extracts were found to show better radical scavenging activity where the maximum inhibition was expressed by Macaranga peltata (Fig. 3a). With increase in concentration of plant extracts, the percentage of DPPH scavenged was found to increase, reaching a maximum inhibition of 80 % – 95 % at 250µg/mL by all the chloroform extracts. The IC 50 of chloroform extracts were as follows, Acacia mangium at 75µg/mL, Phyllanthus acidus at 120µg/mL, Pandanus amaryllifolius at 125µg/mL and Piper aduncum at 170µg/mL. The aqueous extracts showed a constant inhibition of 35% to 50% with increase in concentration and the maximum scavenging activity was expressed by Phyllanthus acidus (80 %). 50% of DPPH inhibition was noticed in Phyllanthus acidus, Piper aduncum, Pandanus amaryllifolius whereas Macaranga peltata and Acacia mangium showed a maximum of 35% and hence the IC 50 of Phyllanthus acidus, Piper aduncum, Pandanus amaryllifolius were 70µg/mL, 50µg/mL and 50µg/mL respectively.
Fig. 3. Antioxidant activity by DPPH assay a) Chloroform extracts and b) Aqueous extracts
PA- Phyllanthus acidus, PSPP- Piper aduncum, PDA- Pandanus amaryllifolius, MP- Macaranga peltata, AM- Acacia mangium.
Antibacterial Activity by agar well diffusion method
The zone of clearance was observed (Table 4 & 5) for the aqueous extracts of Piper aduncum and Macaranga peltata against Staphylococcus aureus (MRSA) and by the chloroform extract of Piper aduncum against MDR – Pseudomonas aeruginosa. No zone of clearance was noticed for other plant extracts. This confirms the antibacterial activity of Piper aduncum (chloroform & aqueous extract) against both the isolated cultures and only the aqueous extract of Macaranga peltata to have antibacterial activity against Staphylococcus aureus. A similar antimicrobial activity of P. aduncum have been reported against its inhibitory actions against S. aureus45. Likewise, the methanolic extract of P. aduncum was reported to exhibit antimicrobial action against Staphylococcus aureus, Pseudomonas aeruginosa, Salmonella typhimurium, Shigella sonnei, Klebsiella pneumoniae40. M. peltata are reported to aid in wound healing as its antimicrobial effect studied against various wound pathogen46. The results are supported by the believed potential of Piper spp. against pathogenic Gram-positive and Gram-negative bacteria such as Staphylococcus aureus, Bacillus cereus, Bacillus subtilis, Streptococcus pyogenes, Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa and Acinetobacter baumanii that have been investigated and reported from published researches43.
Table (4):
Zone of Inhibition by plant extracts against Staphylococcus aureus.
| S. aureus | Zone of Inhibition (mm) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Plant Extract | PA-C | PA-W | PSPP-C | PSPP-W | PDA-C | PDA-W | MP-C | MP-W | AM-C | AM-W |
| Methicillin (5µg) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Control | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 5mg/ml | 0 | 0 | 0 | 12 | 0 | 0 | 0 | 6 | 0 | 0 |
| 10mg/ml | 0 | 0 | 0 | 15 | 0 | 0 | 0 | 7 | 0 | 0 |
| 15mg/ml | 0 | 0 | 0 | 17 | 0 | 0 | 0 | 8 | 0 | 0 |
| 20mg/ml | 0 | 0 | 0 | 20 | 0 | 0 | 0 | 10 | 0 | 0 |
C- chloroform extract, W – aqueous extract. PA- Phyllanthus acidus, PSPP- Piper aduncum, PDA- Pandanus amaryllifolius, MP- Macaranga peltate, AM- Acacia mangium
Table (5):
Zone of Inhibition by plant extracts against Pseudomonas aeruginosa.
| P. aeruginosa | Zone of Inhibition (mm) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Plant Extract | PA-C | PA-W | PPSP-C | PPSP-W | PDA-C | PDA-W | MP-C | MP-W | AM-C | AM-W |
| Ciprofloxacin (5µg) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Control | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 5mg/ml | 0 | 0 | 15 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 10mg/ml | 0 | 0 | 17 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 15mg/ml | 0 | 0 | 19 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 20mg/ml | 0 | 0 | 22 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
C- chloroform extract, W – aqueous extract. PA- Phyllanthus acidus, PSPP- Piper aduncum, PDA- Pandanus amaryllifolius, MP- Macaranga peltate, AM- Acacia mangium
Minimal Inhibition Concentration (MIC)
From the antibacterial results of Piper aduncum and Macaranga peltata, the minimum inhibition concentration (MIC) of the plant extracts was determined. It was found that the aqueous extract of Piper aduncum and Macaranga peltata inhibited the bacterial growth of Staphylococcus aureus (MRSA) at 1.25 mg/mL and 0.1563 mg/mL respectively. And a minimal concentration of about 1.25 mg/mL was required by chloroform extract of Piper aduncum to inhibit the growth of MDR – Pseudomonas aeruginosa (Table 6).
Table (6):
MIC for plant extracts with Antibacterial activity.
| s.no | S.aureus MIC (mg/ml) | Pseudomonas aeruginosa MIC (mg/ml) | |
|---|---|---|---|
| PSPP-W | MP-W | PSPP-C | |
| 1 | 1.25mg/ml | 0.1563mg/ml | 1.25mg/ml |
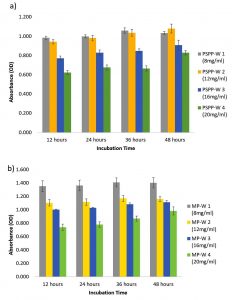
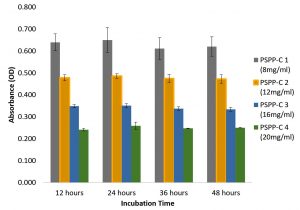
Fig. 4. Antibiofilm activity of aqueous extracts of a) Piper aduncum and b) Macaranga peltata against Staphylococcus aureus.
Antibiofilm Activity
On 48 h of observation, it was found that the aqueous extracts of Piper aduncum and Macaranga peltata did not seem to affect the biofilm formation of Staphylococcus aureus (Fig. 4a and b). The bacteria maintained its growth and prevailed the whole incubation period. However, the population of treated culture was minimal at the highest concentration (20 mg/mL of plant extract) when compared with the control. While treating Pseudomonas aeruginosa with chloroform extract of Piper aduncum, the biofilm formation decreased gradually with increase in concentration and the density of culture remained the same as in 12h with no further growth till the lasting incubation period (48 h) (Fig. 5).
It can be concluded that the chloroform and aqueous extracts of the selected plants, i.e. Phyllanthus acidus, Piper aduncum, Pandanus amaryllifolius, Macaranga peltata and Acacia mangium were found to possess antioxidant property which was confirmed by TLC-DPPH bioautography and DPPH assay. All the plant extracts were examined against the isolated cultures, Staphylococcus aureus and Pseudomonas aeruginosa, where, the aqueous extract of Macaranga peltata, Piper aduncum and chloroform extract of Piper aduncum showed antibacterial activity and their respective minimum inhibition concentration was determined. And only the chloroform extract of Piper aduncum was found to have impact on biofilm formation of P.aeruginosa.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All the authors have made direct contribution on idea creation, research work and editing of the manuscript.
FUNDING
None.
ETHICS STATEMENT
This article does not contain any studies with human participants or animals performed by any of the authors.
AVAILABILITY OF DATA
All datasets generated or analyzed during this study are included in the manuscript.
- Silva, N., Junior, F.A. Biological properties of medicinal plants: a review of their antimicrobial activity. J. Venom Anim. Toxins., 2010; 16(3): 402-413.
Crossref - Cowan, M. Plant Products as Antimicrobial Agents. Clin. Microbiol. Rev., 1999; 12(4): 564-582.
Crossref - Sharifi-Rad, J., Miri, A., Hoseini-Alfatemi, S.M., Sharifi-Rad, M., Setzer, W.N., Hadjiakhoondi, A. Chemical Composition and Biological Activity of Pulicaria vulgaris Essential Oil from Iran. Nat Prod Commun., 2014; 9(11): 1633-1636.
Crossref - Mishra, A.P., Saklani, S., Salehi, B., Parcha, V., Sharifi-Rad, M., Milella, L., Iriti, M., Sharifi-Rad, J., Srivastava, M. Satyrium nepalense, a high altitude medicinal orchid of Indian Himalayan region: chemical profile and biological activities of tuber extracts. Mol. Cell. Biol., 2018; 64(8): 35-43.
Crossref - Sharifi-Rad, J., Ozleyen, A., Tumer, T.B., Adetunji, C.O., El Omari, N., Balahbib, A., Taheri, Y., Bouyahya, A., Martorell, M., Martins, N., Cho, W.C. Natural products and synthetic analogs as a source of antitumor drugs. Biomolecules, 2019; 9(11): 679.
Crossref - Ali, M., U. Zage, A., Ahmed, I., D. Fagwalawa, L. In Vitro Antibacterial Activity and Phytochemical Screening of Garcinia kola Extracts Against Methicillin Resistant Staphylococcus aureus (MRSA). Journal of Pharmacy and Pharmaceutics, 2018: 5(1): 13-18.
Crossref - Sharifi Rad, M., Roberts, T.H., Matthews, K.R., Bezerra, C.F., Morais Braga, M.F.B., Coutinho, H.D.M., Sharopov, F., Salehi, B., ZubaidaYousaf, Sharifi Rad, M., Contreras, M.D.M., Varoni, E.M., Verma, D.R., Iriti, M., Sharifi Rad, J. Ethnobotany of the genus Taraxacum—Phytochemicals and antimicrobial activity. Phytother Res., 2018; 32(11): 2131-2145.
Crossref - Sharifi-Rad, J., Soufi, L., Ayatollahi, S.A., Iriti, M., Sharifi-Rad, M., Varoni, E.M., Shahri, F., Esposito, S., Kuhestani, K., Sharifi-Rad, M. Anti-bacterial effect of essential oil from Xanthium strumarium against shiga toxin-producing Escherichia coli. Cell Mol. Biol., 2016; 62(9): 69-74.
- Sharifi-Rad, M., Iriti, M., Sharifi-Rad, M., Gibbons, S., Sharifi-Rad, J. Anti-methicillin-resistant Staphylococcus aureus (MRSA) activity of Rubiaceae, Fabaceae and Poaceae plants: A search for new sources of useful alternative antibacterials against MRSA infections. Cell Mol. Biol., 2016; 62(9): 39-45.
- Setzer, M.S., Sharifi-Rad, J., Setzer, W.N. The search for herbal antibiotics: An in-silico investigation of antibacterial phytochemicals. Antibiotics, 2016; 5(3): 30.
Crossref - Sharifi-Rad, J., Mnayer, D., Roointan, A., Shahri, F., Ayatollahi, S.A., Sharifi-Rad, M., Molaee, N., Sharifi-Rad, M. Antibacterial activities of essential oils from Iranian medicinal plants on extended-spectrum β-lactamase-producing Escherichia coli. Cell Mol Biol., 2016; 62(9): 75-82.
- Sahraie-Rad, M., Izadyari, A., Rakizadeh, S., Sharifi-Rad, J. Preparation of strong antidandruff shampoo using medicinal plant extracts: A clinical trial and chronic dandruff treatment. Jundishapur J. Nat. Pharm. Prod., 2015; 10(4): e21517.
Crossref - Stojanovic-Radic, Z., Pejcic, M., Stojanovic, N., Sharifi-Rad, J., Stankovic, N. Potential of Ocimum basilicum L. and Salvia officinalis L. essential oils against biofilms of P. aeruginosa clinical isolates. Cell Mol. Biol., 2016; 62(9): 27-32.
- Newman, D., Cragg, G. Natural Products as Sources of New Drugs from 1981 to 2014. J. Nat. Prod., 2016; 79(3): 629-661.
Crossref - Haslam, E. Natural Polyphenols (Vegetable Tannins) as Drugs: Possible Modes of Action. J. Nat. Prod., 1996; 59(2): 205-215.
Crossref - Kakiuchi, N., Hattori, M., Nisizawa, M., Yamagushi, T., Okuda, T., Namba, T. Studies on dental caries prevention by traditional medicines. VIII. Inhibitory effect of various tannins on glucan synthesis by glucosyltransferase from Streptococcus mutans. Chem. Pharm. Bull., 1986; 34(2): 720-725.
Crossref - Dillard, C., German, J. Phytochemicals: nutraceuticals and human health. J. Sci. Food Agric., 2000; 80(12): 1744-1756.
Crossref - Braunwald, E., Bloodwell, R., Goldberg, L., Morrow, A. Studies on digitalis. IV. Observations in man on the effects of digitalis preparations on the contractility of the non-failing heart and on total vascular resistance. J. Clin. Invest., 1961; 40(1): 52-59.
Crossref - Qiu, S., Sun, H., Zhang, A., Xu, H., Yan, G., Han, Y., Wang, X. Natural alkaloids: basic aspects, biological roles, and future perspectives. Chin J Nat Medicines., 2014; 12(6): 401-406.
Crossref - Cushnie, T., Cushnie, B., Lamb, A. Alkaloids: An overview of their antibacterial, antibiotic-enhancing and antivirulence activities. Int. J. Antimicrob. Agents., 2014; 44(5): 377-386.
Crossref - Andrianto, D., Widianti, W., Bintang, M. Antioxidant and Cytotoxic Activity of Phyllanthus acidus Fruit Extracts. IOP Conference Series: Earth and Environmental Science, 2017; 58: 12-22.
Crossref - Unander, D., Webster, G., Blumberg, B. Usage and bioassays in Phyllanthus (Euphorbiaceae). IV. Clustering of antiviral uses and other effects. J. Ethnopharmacol., 1995; 45(1): 1-18.
Crossref - Salehi, B., Kumar, N.V.A., Sener, B., Sharifi-Rad, M., Kilic, M., Mahady, G.B., Vlaisavljevic, S., Iriti, M., Kobarfard, F., Setzer, W.N., Ayatollahi, S.A., Ata, A., Sharifi-Rad, J. Medicinal Plants Used in the Treatment of Human Immunodeficiency Virus. Int. J. Mol. Sci. 2018; 19(5): 1459
Crossref - Mostafa, A., Al-Askar, A., Almaary, K., Dawoud, T., Sholkamy, E., Bakri, M. Antimicrobial activity of some plant extracts against bacterial strains causing food poisoning diseases. Saudi J. Biol. Sci., 2018; 25(2): 361-366.
Crossref - Samrot, A.V., Shobana, N., Jenna, R. Antibacterial and antioxidant activity of different staged ripened fruit of Capsicum annuum and its green synthesized silver nanoparticles. Bionanoscience, 2018; 8: 632–646.
Crossref - Raji, P., Samrot, A.V., Keerthana, D., Karishma, S. Antibacterial Activity of Alkaloids, Flavonoids, Saponins and Tannins Mediated Green Synthesised Silver Nanoparticles Against Pseudomonas aeruginosa and Bacillus subtilis. J. Clus. Sci., 2019; 30 (4): 881 – 895.
Crossref - Samrot, A.V., Sahiti, K., Raji, P., Rohan, B.D., Kumar, D.M., Sharma, K.V. TLC bio-autography guided identification of antioxidant and antibacterial activity of Acacia senegal. Der. Pharmacia Lettre, 2016; 8(9): 41-47.
- Samrot, A.V., Rohan, B.D., Kumar, D.M., Sahiti, K., Raji, P., Samanvitha, S.K. Detection of antioxidant and antibacterial activity of Mangifera indica using tlc bio-autography. Int. J. Pharm. Sci. Res., 2016; 7(11): 4467-4472.
- Sahiti, K., Raji, P., Rohan, B.D., Kumar, D.M., Samrot, A.V. Invitro bioactivity screening of Desmostachya bipinnata. Research J. Pharm. and Tech., 2016; 9(4).
Crossref - Samrot, A.V., Suvedhaa, B., Raghu, R., Sarah, S. Bioactivity of some pigmented and non-pigmented vegetables and fruits. Research J. Pharm. and Tech., 2017; 10(12): 4152-4154.
Crossref - Raji, P., Samrot, A.V., Rohan, B.D., Kumar, D.M., Gandham, R.G., Sharma, V.K., Keerthana, D. Extraction, characterization and invitro bioactivity evaluation of alkaloids, flavonoids, saponins and tannins of Cassia alata, Thespesia populnea, Euphorbia hirta and Wrightia tinctoria. Rasayan J. Chem., 2019; 12(1): 123 – 137.
Crossref - Mekonnen, A., Atlabachew, M., Kassie, B. Investigation of antioxidant and antimicrobial activities of Euclea schimperi leaf extracts. Chem. Biol. Technol. Agric., 2018; 5: 16.
Crossref - Edeoga, H., Okwu, D., Mbaebie, B. Phytochemical constituents of some Nigerian medicinal plants. Afr. J. Biotechnol., 2005; 4(7): 685-688.
Crossref - Samrot, A.V., Raji, P., Selvarani, A.J., Nishanthini, P. Antibacterial activity of some edible fruits and its green synthesized silver nanoparticles against uropathogen – Pseudomonas aeruginosa SU 18, Biocatalysis and Agricultural Biotechnology, 2018; 16: 253 – 270.
Crossref - Annegowda, H.V., Tan, P.Y., Mordi, M.N., Ramanathan, S., Hamdan, M.R., Sulaiman, M.H., Mansor, S.M. TLC–Bioautography-Guided Isolation, HPTLC and GC–MS-Assisted Analysis of Bioactives of Piper betle Leaf Extract Obtained from Various Extraction Techniques: In vitro Evaluation of Phenolic Content, Antioxidant and Antimicrobial Activities. Food Anal. Methods., 2012; 6(3): 715–726.
Crossref - Selvarani, A., Nishanthini, P., Raji, P., Samanvitha, S.K., Paulraj, P., Iyappan, P., Chandramohan, M., Samrot, A.V. Antioxidant and Quorum Quenching Activity against Pseudomonas aeruginosa SU-18 of some Edible Fruit Juices. J Pure Appl Microbio., 2019; 13(3): 1863-1876.
Crossref - Muthukrishnan, S., Kumar, T.S., Gangaprasad, A., Maggi, F., Rao, M.V. Phytochemical analysis, antioxidant and antimicrobial activity of wild and in vitroderived plants of Ceropegia thwaitesii Hook – An endemic species from Western Ghats, India. Journal, Genetic Engineering & Biotechnology, 2018; 16(2): 621–630.
Crossref - Mgbeahuruike, E.E., Fyhrquist, P., Vuorela, H., Julkunen-Tiitto, R., Holm, Y. Alkaloid-Rich Crude Extracts, Fractions and Piperamide Alkaloids of Piper guineense Possess Promising Antibacterial Effects. Antibiotics (Basel)., 2018; 7(4): 98.
Crossref - Samrot, A.V, Raji, P., Selvarani, A.J, Sruthi, D.P., Angalene, L.A.J., Ponnaiah, P., Iyappan, P. A Handbook on Phytochemical extraction, screening and its in-vitro bioactivity assays. SARAS Publisher, India, ISBN: 978-93-86519-60-3.
- Bouzada, M., Fabri, R., Nogueira, M., Konno, T., Duarte, G., Scio, E. Antibacterial, cytotoxic and phytochemical screening of some traditional medicinal plants in Brazil. Pharmaceutical Biology, 2009; 47(1): 44-52.
Crossref - Jain, N.K., Singhai A.K. Protective effects of Phyllanthus acidus (L.) Skeels leaf extracts on acetaminophen and thioacetamide induced hepatic injuries in Wistar rats. Asian Pac. J. Trop. Med., 2011; 4(6): 470-474.
Crossref - Al-Rifai, A., Aqel, A., Al-Warhi, T., Wabaidur, S., Al-Othman, Z., Badjah-Hadj-Ahmed, A. Antibacterial, Antioxidant Activity of Ethanolic Plant Extracts of Some Convolvulus Species and Their DART-ToF-MS Profiling. Evid Based Complement. Alternat. Med., 2017; 5694305.
Crossref - da Silva, J., da Trindade, R., Alves, N., Figueiredo, P., Maia, J., Setzer, W. Essential Oils from Neotropical Piper Species and Their Biological Activities. Int. J. Mol. Sci., 2017; 18(12): 2571.
Crossref - Wang, J., Yue, Y., Tang, F., Sun, J. TLC Screening for Antioxidant Activity of Extracts from Fifteen Bamboo Species and Identification of Antioxidant Flavone Glycosides from Leaves of Bambusa. textilis McClure. Molecules, 2012; 17(10): 12297-12311.
Crossref - Monzote, L., Scull, R., Cos, P., Setzer, W. Essential Oil from Piper aduncum: Chemical Analysis, Antimicrobial Assessment, and Literature Review. Medicines, 2017; 4(3): 49.
Crossref - Nehete, M., Nipanikar, S., Kanjilal, A., Kanjilal, S., Tatke, P. Comparative efficacy of two polyherbal creams with framycetin sulfate on diabetic wound model in rats. J. Ayurveda Integr. Med., 2016; 7: 83-87.
Crossref
© The Author(s) 2019. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.