ISSN: 0973-7510
E-ISSN: 2581-690X
Health care workers (HCW) are a high-risk population for Hepatitis B infection. Hepatitis B vaccine which is 95% effective confers long-term protection and anti-HBs titre is a marker for protective immune response. Our objective was to assess the status of hepatitis B vaccination and to evaluate the anti-HBsAg titres among health care workers in a tertiary care hospital, Tamil Nadu. It was an observational study conducted among 610 Health care workers in a tertiary care teaching hospital from June to December 2018 after obtaining clearance from IEC. Workers were assessed for their HBV vaccination status and for their anti-HBsAg titre after getting informed consent. The antibody titres were measured using CLIA (chemiluminescent Immunoassay) supplied by Abbott diagnostics. The data was entered and analyzed using a Microsoft Excel sheet. In our study, 80.5% were fully vaccinated, 18.5% of them were defaulters which comprised the HCW with 2 doses and 1 dose of vaccine and 0.9% were not vaccinated. In the fully vaccinated group, 37% showed Anti HBs titres of 10-100 mIU/ml, 59.2% showed the titre of > 100 mIU/ml and 3.6% did not show the protective antibody titre (<10 mIU/ml ). The 3.6% who did not show protective antibody titre were given the booster dose of vaccine. Among those who received a booster dose, 61.1% responded with the titre of 10-100 mIU/ml and 22.2% responded with the titre of > 100 mIU/ ml and 16.6% did not respond even with the booster dose. In the defaulters 86.7% had titres < 10 mIU/ml , 9.7% had titres of 10 mIU/ml and 3.6% had titres > 10 mIU/ml. In the non vaccinated group all had titres < 10 mIU/ml. The present study emphasizes the importance of screening of anti-HBsAg titres to be made mandatory for all the health care workers along with HBV Vaccination.
Hepatitis B, Health care workers, HBsAg titre, Vaccination
Hepatitis B is a viral infection that attains greater significance because of its high risk of transmission. HBV infection occurs in a large proportion of the world’s population.1 It is a leading cause of Chronic hepatic diseases ultimately ending in hepatocellular carcinoma.2 Health care professionals are a high-risk group for Hepatitis B and are advised vaccination against hepatitis B.3 According to WHO, “5.9% of HCWs are each year exposed to blood-borne HBV infections corresponding to about 66,000 HBV infections in HCWs worldwide”.4 Data from various studies conducted in different parts of India revealed only <60% Health care community received Complete HBV vaccination. This is eventually who are at high risk of acquiring the infection.5,6
HBV infection can be prevented by timely vaccination at 0,1,6 Months and the efficacy of the vaccine is around 95%. Hepatitis B vaccine confers long-term protection, and anti-HBs titre is a marker for protective immune response.7 It is very essential to look for Anti-HBsAg titres since their levels decrease over time.8
Though guidelines suggest three doses of vaccine is effective in producing protective antibody levels, there has been always a debate regarding the need for an additional booster dose.9 The evaluation of immune status following vaccination is important in susceptible populations as there is always a group who remain as non-responders to the hepatitis B vaccine and continue to be at risk for acquiring the infection.10 Hepatitis B vaccine confers long-term protection against both clinical illness and its sequelae. An easily detectable anti-hepatitis B surface antigen (Anti-HBs) level is a marker for protection.11,12. So it is essential to screen the health care workers for hepatitis B vaccination and to check the titre of anti-HBs which serves as the marker of protective status.
Objectives
- To Check for Hepatitis B vaccination status among the high-risk group.
- To evaluate the anti-HBsAg titres in completely vaccinated and defaulters.
- To check the efficacy of a booster dose in the non-responders to the Primary vaccine schedule of HBV.
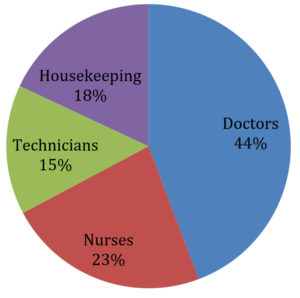
A total of 610 Health care workers working in a tertiary care teaching hospital which includes 270 doctors, 150 Nurses, 90 Technicians and 100 Housekeeping staff were assessed for their HBV vaccination status and for their anti-HBsAg titre. The antibody titres were measured using CLIA (chemiluminescent Immunoassay) supplied by Abbott diagnostics.
Ethics
The study was conducted after obtaining clearance from the ethical committee.
Consent
Informed consent was obtained from the study group before enrolling in the study.
Inclusion criteria
All the health care workers irrespective of age, sex were included in the study
Exclusion criteria
People other than health care workers were excluded from the study.
The distribution of the study group based on the profession is given in Fig. 1.
491 HCW out of 610 (80.5%) had completed all the three doses of the vaccine as recommended.
The distribution of HCW with all three doses of vaccine is given in Table 1.
Table (1):
Study groups with three doses of the vaccine.
No |
Category |
Total (n= 491) |
Percentage (%) 80.5% |
|---|---|---|---|
1. |
Doctors |
258 |
95.5% |
2. |
Nurses |
122 |
87.1% |
3. |
Technicians |
65 |
72.2% |
4. |
House keeping staff |
46 |
46% |
The study groups with two doses of vaccine is given in Table 2.
Table (2):
Study group with two doses of the vaccine.
No |
Category |
Total (n= 87) |
Percentage (%) ( 14.2%) |
|---|---|---|---|
1. |
Doctors |
12 |
13.7% |
2. |
Nurses |
11 |
12.6% |
3. |
Technicians |
24 |
27.5% |
4. |
House keeping staff |
40 |
45.9% |
The distribution of the study group with one dose of vaccine is given in Table 3.
Table (3):
Study group with one doses of the vaccine.
No |
Category |
Total (n= 26) |
Percentage (%) (4.2%) |
|---|---|---|---|
1. |
Doctors |
0 |
0 % |
2. |
Nurses |
7 |
26.9% |
3. |
Technicians |
1 |
3.8% |
4. |
House keeping staff |
18 |
69.2% |
6 of the 610 HCW in the study gave history of no vaccination. All of the 6 were housekeeping staff.
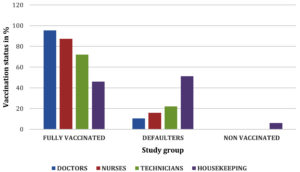
Based on the above data that was obtained the HCW were divided into three groups, the fully vaccinated group, the defaulters and the non-vaccinated group. The distribution of the study group based on vaccination status is shown in Fig. 2.
Of the 491 (80.5%) HCW who were fully vaccinated, 18 (3.6%) had titres < 10 mIU/mL, 182 (37%) had titres of 11-100 mIU/mL, 291 (59.2% ) had titres >100 mIU/mL. Out of the 113 defaulters (18.5%) (2 doses 88, one dose 25) 04 (3.5%) had titres of >10 mIU/mL, 11 (9.7%) had titre of 10 mIU/mL and 98 (86.7%) had titres <10mIU/mL. Booster dose were given for the 18 fully vaccinated HCW with titres <10 mIU/mL. 15/18 (83.3%) responded to the booster of which 11 (61.1%) had titres >10 mIU/mL, 4 (22.2%) had titres of >100 mIU/mL and 3 (16.6%) were non responders. Of the 6 (0.9%) non vaccinated HCW all had titre <10 mIU/mL.
Comparing the data obtained from all the three groups it was clear that compliance was higher with doctors followed by nurses and technicians. Compliance was least observed with housekeeping staff which is similar to the study done by Vishal Batral et al., Iftekhar et al. and Syed moayed alavian et al.13-14 Housekeeping staff are the major risk group for HBV because these are the population involved in handling bio-medical waste. Awareness programmes should be conducted highlighting the importance of HBV vaccination in Health care setup.
We found in the fully vaccinated group 59.2% had titres of >100 mIU/mL, 37% had titres of 10-100 mIU/mL. It was also found even in the fully vaccinated group 3.6% were non-responders with titres of <10 mIU/mL. In a study done by Alekhya et al.,15 it was found 6% were found non-responders in the completely vaccinated group of HCW. In the present study, a booster dose to the non-responders to the routine (0,1,6 Months) dose and 83.3% responded with protective titres of ≥10 mIU/mL. However, 16.6% were permanent non-responders.
It is clearly evident that routine screening of antibody titre is to be done among vaccinated HCW to identify the non-responders and implement policies that can suggest for providing additional booster doses if required and also take necessary precautionary measures to ensure their safety in the workplace.
Also in contrast to the belief that the partially vaccinated group does not develop protective titres, it was found in our study that 13.2 % has titres ≥10 mIU/mL.
The present study emphasizes the importance of screening of anti-HBsAg titres to be made mandatory for all the health care workers along with HBV Vaccination. It also implies the need for health education to the HCW on HBV to ensure compliance with the vaccine.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work and approved it for publication.
FUNDING
None.
ETHICS STATEMENT
The study was approved by the Institutional Ethics Committee, Bhaarath Medical College and Hospital, Chennai, Tamil Nadu, India. (BIEC-45-20).
AVAILABILITY OF DATA
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
INFORMED CONSENT
Written informed consent was obtained from the participants before enrolling in the study.
- Kao JH, Chen DS. Global control of hepatitis B virus infection. Lancet Infect Dis. 2002;2(7):395-403.
Crossref - Hollinger FB, Liang TJ, et al.: Hepatitis B virus. In Fields Virology. 4th edition. Edited by: Knipe DM, Howley PM, Griffin DE, Lamb RA, Martin MA, Roizman B. Lippincott-Raven Publishers, Philadelphia, PA; 2001:2971-3036.
- Mast EE, Margolis HS, Fiore AE, et al. A Comprehensive immunization strategy to eliminate transmission of hepatitis B virus infection in the United States: Recommendations of the advisory committee on Immunization Practices (ACIP) part 1: Immunization of infants, children and adolescents. MMWR Recomm Rep 2005;54:1-31
- Pruss-Ustun A, Rapiti E, Hutin Y. Estimation of the global burden of disease attributable to contaminated sharps injuries among health-care workers. Am J Ind Med. 2005;48(6):482-490.
Crossref - Pati NT, Sethi A, Agrawal K, et al. Low level of awareness, vaccine coverage, and the need for booster among health care workers in tertiary care hospitals in India. J Gastroenterol Hepatol. 2008;23(11):1710-1715.
Crossref - Duseja A, Arora L, Masih B, et al. Hepatitis B and C Virus-prevalence and prevention in health care workers. Trop Gastroenterol. 2002;23(3):125-126. PMID: 12693154
- Ciorlia LAS, Zanetla DMT. Hepatitis B in health care workers: Prevalence, vaccination and relation to occupational factors. Braz J Infect Dis. 2005;9(5):384-389.
Crossref - Sjogren MH. Prevention of hepatitis B in non responders to initial hepatitis B virus vaccination. Am J Med 2005;118(Suppl 10A):34-39.
Crossref - Hepatitis B virus: A Comprehensive Strategy for Eliminating Transmission in the United States through Universal Childhood Vaccination. Recommendations of the Immunization Practices Advisory Committee (ACIP). MMWR Recomm Rep. 1991;40:1- 25.
- Chaudhari CN, Bhagat MR, Shah T, Misra RN. Antibody to hepatitis B surface antigen in vaccinated health care workers. Med J Armed Forces India. 2008;64(4):329-332.
Crossref - Mast E, Mahoney F, Kane, M, Margolis H. Hepatitis B vaccine. In: Plotkin SA, Orenstein WA (Eds). Vaccines (4th ed). Philadelphia: Saunders, 2004, 299-337.
- Hutin Y, Hauri A, Chiarello L, et al. Best infection control practices for intradermal, subcutaneous, and intramuscular needle injections. Bulletin of the World Health Organization. 2003;81(7):491-500. PMID: 12973641
- Alavian S-M, Izadi M, Zare A-A, et al. Survey of the level of anti-HBs antibody titer in vaccinated Iranian general dentists. Spec Care Dentist. 2008;28(6):265-270.
Crossref - Batra V, Goswami A, Dadhich S, Kothari D, Bhargava N. Hepatitis B immunization in healthcare workers. Ann Gastroenterol. 2015;28(2):276-280. PMID: 25830669
- Jayalakshmi L, Alekhya P, Sasikala G. AntiHBs Titers in Health Care Persons of Clinical Laboratory. IOSR Journal of Dental and Medical Sciences. 2017;16(10):54-57.
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.