ISSN: 0973-7510
E-ISSN: 2581-690X
Acute diarrheal illness is a serious public health issue in developing and developed economies, with high morbidity and mortality rates. Children below five years are the most vulnerable, with one in every six dying from diarrhea. Diarrhea may be caused by various viruses, protozoa, and bacteria. Rotavirus, in particular, is often associated with diarrhea in children. Early detection of rotavirus infection reduces the need for medication and the spread of the virus and helps determine the optimal treatment strategies. In this study, we compared the efficacy of the rapid immunochromatography technique (ICT) with that of enzyme-linked immunosorbent assay (ELISA) to detect rotavirus antigens in stool samples. Overall, 361 stool samples were obtained from hospitalized children under five-years-old, with acute watery diarrhea symptoms and were tested by both rapid ICT and ELISA. Among these samples, 104 (28.81%) tested positive by ICT and 109 (30.19%) by ELISA. Furthermore, the specificity and sensitivity of ICT were assessed using ELISA, the gold standard test. ICT had a 95.41% sensitivity and a 100% specificity. Thus, during emergency testing, ICT may be utilized as an alternative test for quick diagnosis with high specificity and sensitivity in patients with rotavirus diarrhea.
Acute diarrhea, Rotavirus, Immunochromatography, ELISA, Sensitivity, Specificity
Acute diarrheal illness is a serious public health issue in developing and developed economies, with significant morbidity and mortality.1 Diarrhea is a common cause of pediatric hospitalization worldwide. Children under the age of five are the most vulnerable, with one in six children dying from diarrhea. In India, diarrhea affects roughly 3,34,000 children each year.2 Different viruses, protozoa, and bacteria, may be responsible for causing diarrhea. Rotavirus, in particular, is often associated with diarrhea in children.3 Group-A rotaviruses cause 30–60% of severe watery diarrhea cases in young children.4 Early detection of rotavirus infection reduces the requirement for medications, curbs the spread of the virus, and aids in determining the optimal therapy.5 Latex agglutination (LA), enzyme immunoassay (EIA), reverse transcriptase-polymerase chain reaction (RT-PCR), lateral flow immune-chromatography (ICT), and electron microscopy (EM) are some of the methods used to identify rotavirus in stool specimens. The EIA is a highly sensitive test that can be performed in a laboratory setup by trained laboratory personnel. A rapid test employing immunochromatography lateral flow or latex agglutination is a suitable alternative to EIA and has excellent sensitivity. The latex agglutination approach is widely applied for rapid testing. However, its reduced sensitivity compared to ELISA complicates the interpretation of the findings. Furthermore, the LA test results cannot be preserved. Therefore, ICT has become increasingly accessible; this test is cost-effective, reliable, quick, and simple.6
Hence, this study was undertaken to compare the two techniques, rapid ICT and ELISA, for detecting rotavirus antigens in fecal samples from children less than five years of age. Furthermore, we determined the prevalence of rotavirus-induced diarrhea in hospitalized children.
This cross-sectional study was performed in the Department of Microbiology at Krishna Institute of Medical Sciences & Research, Medical College, Karad, a tertiary care hospital (Maharashtra, India), between July 2016 and June 2020. Ethical approval was received from the institutional ethics committee before the study was performed. From the Pediatric Department, 361 fecal samples were collected from children under five years old and hospitalized with severe watery diarrhea.
Inclusion Criteria
- Those who provide written informed permission (parents/guardians of minors).
- Acute watery diarrhea with the occurrence of at least four loose bowel movements per day in hospitalized children under five years old.
Exclusion Criteria
- Those who refuse to provide written informed permission (parents/guardians of minors).
- Children who have received the rotavirus vaccine.
- Acute watery diarrhea with at least four bowel loose bowel movements per day in hospitalized children over five years of age.
Sample Collection and Transport
Fresh fecal specimens were collected in appropriately labeled, dry, sterile, wide-mouth, clean, leak-proof plastic containers within 24–48 h after patient admission. Upon collection, the samples were sent to the Microbiology Department for laboratory testing.
Sample Storage
Until they are processed, the stool samples are labeled and stored at -20°C.
Sample Processing
Rotavirus Detection by ICT
The Arco Biotech ICT Quick Rotavirus kit (Germany) was used to identify rotavirus antigens in the fecal samples. ICT test is a one-step immunochromatographic lateral flow test performed as directed by the manufacturer. Rotavirus positivity is confirmed by the presence of control and test bands. The test results were considered negative if only the control band was observed and invalid if the control band was not visible.
Rotavirus Detection by ELISA
The rotavirus antigen was detected in stool samples using a PREMIER® Rotaclone® Rotavirus kit from Meridian Bioscience®. Monoclonal antibodies against the product of the sixth viral gene (VP6) were used in a sandwich-type approach. The antigen is sandwiched between anti-rotavirus monoclonal and enzyme-linked antibodies.
Data Analysis
Statistical Package for Social Sciences, 21 version, was employed to analyze the data. The chi-square test was used to compare categorical variables, and a p-value of 0.05 was assumed to be statistically significant.
The study included 361 hospitalized children ≤ five years old who had symptoms of acute watery diarrhea. All 361 stool samples from children were diagnosed using ICT and ELISA for rotavirus antigen detection. According to test results, 104 (28.81%) children tested positive and 257 (71.19%) negative by ICT, whereas 109 (30.19%) tested positive and 252 (69.80%) negative by ELISA.
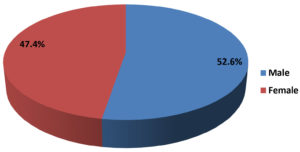
Among 361 children, 190 (52.6%) were male, and 171 (47.4%) were female. The male-to-female ratio was 1.1:1, as shown in Figure 1.
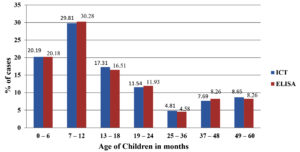
The most rotavirus-positive samples were observed in children within the age range of 7–12 months, including 31 children by ICT (29.81%) and 33 (30.28%) by ELISA, followed by children between 0–6 months [21 children (20.19%) by ICT, and 22 (20.18%) by ELISA] as shown in Table 2 and Figure 2. Table 1 shows that the association between age and positive cases detected by ICT and ELISA was not statistically significant (p-value = 0.999).
Table (1):
Age-wise distribution of ICT and ELISA positive rotavirus watery diarrhea cases.
| Age group (in months) | ICT | ELISA | p-value | ||
|---|---|---|---|---|---|
| + ve | – ve | + ve | – ve | ||
| 0 – 6 | 21 (20.19%) | 43 (16.73%) | 22 (20.18%) | 42(16.67%) | 0.999* |
| 7 – 12 | 31(29.81%) | 51 (19.84%) | 33(30.28%) | 49(19.44%) | |
| 13 – 18 | 18(17.31%) | 37 (14.40%) | 18(16.51%) | 37(14.68%) | |
| 19 – 24 | 12(11.54%) | 37 (14.40%) | 13(11.93%) | 36(14.29%) | |
| 25 – 36 | 05(04.81%) | 23 (08.95%) | 05(04.58%) | 23(09.13%) | |
| 37 – 48 | 08(07.69%) | 37 (14.40%) | 09(08.26%) | 36(14.29%) | |
| 49 – 60 | 09(08.65%) | 29 (11.28%) | 09(08.26%) | 29(11.50%) | |
| Total | 104 (100%) | 257 (100%) | 109 (100%) | 252 (100%) | |
The value of Chi-square test (X2) = 0.067, degree of freedom (df) = 5, P-value = 0.999, *Not significant
Table 2 reveals that 104 (28.81%) patients tested positive for the rotavirus antigen by both ELISA and ICT, whereas five (1.36%) tested positive by ELISA but negative by ICT. We did not observe any ELISA-negative cases among those that tested positive by ICT. ELISA and ICT confirmed that 252 (69.81%) cases were negative for the rotavirus antigen.
Table (2):
Comparison of rapid ELISA and ICT test to detect rotavirus antigen from the stool sample.
| ICT | ELISA | Total | |
|---|---|---|---|
| Positive | Negative | ||
| Positive | 104 (28.81%) | 00 (00%) | 104 (28.81%) |
| Negative | 05 (01.36%) | 252 (69.81%) | 257 (71.19%) |
| Total | 109 (30.19%) | 252 (69.81%) | 361 (100%) |
The diagnostic effectiveness of the rapid ICT test, compared to ELISA (the gold standard test), had 95.41% sensitivity and 100% specificity. ICT registered positive and negative predictive values of 100 and 98.05%, respectively. Thus, the accuracy of the ICT test is 98.61%.
Rotavirus is the most prevalent cause of dehydration in children with severe diarrhea. Developing nations, such as India, have high mortality rates. Estimating the true burden of gastroenteritis caused by rotavirus is easier with detecting this infection in children with severe diarrhea. Laboratory diagnosis can be improved by a cost-effective and sensitive approach to detecting rotavirus.7
Clinically differentiating between diarrhea caused by rotavirus and other illnesses is impossible. Therefore, a reliable diagnostic test is required to confirm rotavirus as the causal agent of gastroenteritis. Children infected with rotavirus do not require antibiotic treatment unless they have other coexisting bacterial illnesses. The indiscriminate use of antibiotics leads to antibiotic resistance in bacteria, as well as increased treatment expenses and adverse side effects. Currently, rotavirus disease is confirmed by detecting viral antigens in stool samples. Commercially-accessible rapid diagnostics include immunochromatographic testing, latex agglutination, and enzyme-based immunoassays. Rapid tests are straightforward to perform, cost-effective, and require no expertise; however, the sensitivity of ELISA is higher than that of the rapid test. ELISA is the gold standard with high sensitivity compared to other rapid testing strategies.8
We thus compared the efficiency of the rapid fast immunochromatography technique with that of ELISA in identifying rotavirus in fecal samples from children under the age of five. All 361 stool samples from hospitalized children ≤ five years old with acute diarrhea were analyzed for rotavirus antigen detection using a rapid test during our investigation (lateral flow immunochromatography) and ELISA.
Rotavirus infection rates fluctuate across countries, and across states in India.9-11 Rotavirus is the primary cause of severe, life-threatening gastroenteritis in small children, accounting for 20–50% of gastroenteritis-related hospitalizations across the globe.12 With ELISA, we observed a 30.19% prevalence of rotavirus-induced diarrhea in children under the age of five. The findings of this study were corroborated by reports from different parts of India, including Mumbai, Maharashtra 28%,13 Vijayapur, Karnataka 31%,14 Agartala, Tripura 36% .15
In our study, the highest prevalence of rotavirus infection was observed in male children (52.6%) compared to female children (47.4%), with a male-to-female ratio of 1.1:1. The male preponderance observed in the present study is comparable to that reported by Majumder et al. (1.59:1),15 Habib et al. (1.62:1),16 and Mansabdar et al. (1.17:1).17 The reason for this male predominance was unclear. However, parents may prioritize male children over females when seeking healthcare. Further, this finding could be explained by the XX chromosome, which confers higher resistance to infection in women.18
In the present study, the majority of the rotavirus infections in children occurred in the 7-12 months age group (30.28%), which is similar to data published by Sharma et al., Dhiman et al., Yadav et al., Mansabdar et al., who reported an estimate of 33.33%,19 38.1%,18 21.7%,20 and 55.17%, respectively.17 The 7–12 month age group had the highest incidence, followed by 0–6 months. This could be due to partial or exclusive breastfeeding in the first six months, as well as acquired immunity from the mother, which could prevent infection in the first few months. The incidence of rotavirus infection dropped dramatically after 18 months of age due to the development of antibodies in response to earlier exposure, causing subsequent infections to be less severe.
The rapid test is easy to perform and saves time, cost, and labor; Although it does not require expertise, the results must be corroborated with ELISA, a sensitive method for rotavirus detection. In this study, rapid ICT suggested a rotavirus prevalence rate of 28.81%; however, five cases that were positive by ELISA were negative by ICT. The present study showed that the rapid ICG test sensitivity was 95.41%, and specificity was 100%. Dennehy et al. reported 94% sensitivity in rapid ICG, whereas the specificity was 100%.21 Ibrahim et al. observed that the rapid ICG test had a specificity and sensitivity of 100 % and 90%, respectively, compared to ELISA22; Yadav et al. reported a sensitivity of 90.9% and specificity of 94.5%,20 and Habib et al. observed 90.70% sensitivity and 93.88% specificity.16 Our findings were consistent with the previous reports.
In summary, rotavirus is the primary cause of acute diarrhea in newborns and children under five years old. Late detection of rotavirus may result in increased morbidity and mortality. Most hospitals do not regularly identify rotavirus owing to the high expense of diagnostics. The clinical range of symptoms and signs is comparable to those of other gastroenteritis cases. To reduce excessive antibiotic usage and the development of drug resistance, fecal samples from all children with acute gastroenteritis should be subjected to rotavirus detection assays. Due to restricted availability and increased costs, only a few laboratories in India use ELISA to diagnose rotavirus infection. In this study, a rapid ICG test was compared with the conventional ELISA. We report that the rapid ICG test has 95.41% sensitivity and 100% specificity. Consequently, the rapid ICG test is comparable to ELISA and has several advantages. The ICG test is quicker, cost-effective, facilitates single-sample assessment, convenient, straightforward, accessible, and requires no extra equipment. Finally, the rapid ICG test does not require expertise. It can be performed for a single specimen collected directly from hospitalized children by the clinician, thereby enabling medical personnel to make prompt management decisions.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct, and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This study was approved by the Institutional Ethics Committee, Krishna Institute of Medical Sciences, Karad, India with reference number KIMSDU/IEC/01/2016
- Pang X, Hodinka RL. Gastroenteritis Viruses. In Jorgensen JH, Pfaller MA (eds.). Manual of Clinical Microbiology. 11th Ed. Washington, D.C.: ASM Press; 2015:2:1617-1632.
Crossref - Morris SK, Awasthi S, Khera Aet al. Rotavirus mortality in India: estimates based on a nationally representative survey of diarrhoeal deaths. Bull World Health Organ. 2012;90(10):720-727.
Crossref - Salim H, Karyana IPG, Sanjaya-Putra IGN, Budiarsa S, Soenarto Y. Risk factors of rotavirus diarrhea in hospitalized children in Sanglah Hospital, Denpasar: a prospective cohort study. BMC Gastroenterol 2014;14(1):54.
Crossref - Blacklow NR, Greenberg HB. Review article: Viral gastroenteritis. N Engl J Med. 1991;325 (4):252-264.
Crossref - Christensen ML, Howard C. Viruses Causing Gastroenteritis. Manual of Clinical Microbiology, 5th Ed. American Society for Microbiology, Washington D.C. 2006:950-958.
- Lee SY, Hong JH, Lee SN, Lee M. Comparisons of latex agglutination, immunochromatography and enzyme immunoassay methods for the detection of rotavirus antigen. Korean J Lab Med.2007;27(6):437-441.
Crossref - Manjula G. Comparison of Immunochromatography with RT-PCR for Detection of Rotavirus in Fecal sample. Medical Science. 2013;2(12):479- 481.
Crossref - Pang XL, Lee B, Boroumand N, Leblanc B, Preiksaitis JK, Charlotte CYI. Increased detection of rotavirus using a real-time reverse transcription-polymerase chain reaction (RT-PCR) assay in stool specimens from children with diarrhea. J Med Virol. 2004;72(3):496-501.
Crossref - Ramani S, Kang G. Burden of disease & molecular epidemiology of group A rotavirus infections in India. Indian J Med Res. 2007;125(5):619-632. PMCID: PMC2474761
- De Villiers FPR, Sawyerr TN, De Villiers GK. The incidence and clinical presentation of infantile rotavirus diarrhea in Sierra Leone. S Afr Med J. 2009;99(4):249-252. PMID: 19588778
- Kahn G, Fitzwater S, Tate J, et al. Epidemiology and prospects for prevention of rotavirus disease in India. Indian Pediatrs. 2012;49(6):467-474.
Crossref - Glass RI, Parashar UD, Bresee JS, et al. Rotavirus Vaccines: Current Prospects and Future Challenges. Lancet. 2006;368(9532):323-332.
Crossref - Mehta N, Baveja S. Comparative study of enzyme-linked immunosorbent assay and immunochromatography for rotavirus detection in children below five years with acute diarrhea. Med Pulse International Journal of Microbiology. 2017;4(3):23-26.
- Patil SGV, Lakhkar BB, Kalyanshettr SS. Diarrhoea in north Karnataka: Rotavirus versus non-rotavirus.Sri Lanka Journal of Child Health. 2018;47(3):204-209.
Crossref - Majumder N, Barbhuiya NI, Majumder T, Datta SS. Prevalence of Rotaviral Infection among Children admitted with Acute Diarrhoea in a Tertiary Care Hospital of Tripura. Int J Sci Res. 2018;7(3):29-31.
- Habib FB, Rahman MM, Sinha SP, et al. A Comparative Study of Rotaviral Antigen Detection by ELISA and ICT in Children below Five Years with Acute Diarrhoea in A Tertiary Care Hospital. Bangladesh Med J. 2020;49(1):14-18.
Crossref - Mansabdar P, Sambrani PN. Mahesh kumar S. A study of rotavirus infection in acute diarrhoea in children less than 5 years of age. Indian J Microbiol Res. 2021;8(4):321-326.
Crossref - Dhiman S, Devi B, Sing K, Devi P. Comparison of Enzyme-Linked Immunosorbent Assay and Immunochromatography for Rotavirus Detection in Children Below Five Years with Acute Gastroenteritis. J Clin Diagn Res. 2015;9(9):6-9.
Crossref - Sharma A. Comparison of a Rapid Immunochromatography Test with Elisa to Detect Rotavirus. J Med Sci Clin Res. 2017;05(07):24334-24340.
Crossref - Yadav M, Agrawal P, Sharma M, Abrol P and Broor S. Comparison of Enzyme-Linked Immunosorbent Assay and Immunochromatography for Rotavirus Detection in Children below Two Years with Acute Gastroenteritis. Indian Journal of Health Sciences and Care. 2019;6(1):24-27.
Crossref - Dennehy PH, Hartin M, Nelson SM, Reising SF. Evaluation of the Immuno-Card-STAT! Rotavirus Assay for Detection of Group A Rotavirus in Fecal Specimens. J Clin Microbiol.1999;37(6):1977-1979.
Crossref - Ibrahim SB, Bialy AAE, Mohammed MS, Sheikh AO, Elhewala A, Bahgat S. Detection of Rotavirus in children with acute gastroenteritis in Zagazig University Hospitals in Egypt. Electron Physician. 2015;7(5):1227-1233.
Crossref
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.