ISSN: 0973-7510
E-ISSN: 2581-690X
Behavior of E. dissolvens under the exposure to weak direct current (DC) was investigated by using glucose as the sole carbon source. Continuous cultivation behavior of E. dissolvens under the exposure to weak direct current (DC) was investigated . Experimental results showed the electrolytic stimulation of the continuous culture, cell growth curve and glucose metabolic rate were significantly improve. After electrolytic stimulation for 10 hour, the specific dehydrogenase activity and glucose consumption were 2 and 1.8 fold as much as that determined in the absence of electric field. Owing to flowing of the culture medium, the cells by the DC application after the growth phase were better than the control.
Continuous cultivatation; Electrolytic stimulation; Enterobacter dissolvens; Dehydrogenase activity.
Electric field for cell growth and metabolism is the influence of the biological chemical industry, biological medicine and environmental biological engineering has the important significance of the research topic, so cause the attention of many researchers. Although it has been extensively reported that alternating current (AC) can be applied to alter the cell behaviors in terms of proliferation and activities,1,2 only a few studies had been presented regarding the implementation of direct current (DC) on biological systems. The application of DC current led to various consequences on different systems in the presence of electrode reaction. For instance, DC current was widely observed to be capable of enhancing the antibiotics’ efficacy against heterotrophic biofilms3,4,5, while it also promoted the yeast fermentation process6 and the bacterial denitrification process.7,8
using glucose as the sole substrate growth of E.d issolvens, have confirmed that application of electrolytic stimulation method can effectively strengthen the cell growth and activity9
But in batch experiments, with substrate consumption and cells step into stationary phase, Cells appear the phenomenon of accelerated decline, resulting in their own energy and biomass fell rapidly, the reason is by H2O2 and the accumulation of intermediate product Because the continuous culture conditions cells logarithmic phase can effectively extend, and maintain a relatively stable cell activity and growth rate. Therefore for electrolytic stimulation technology applied in the cells of the continuous culture process, is expected to overcome batch type training some drawbacks in and get a better effect of stimulation.
The objective of present study is to investigate the electrolytic stimulation process of Enterobacter dissolvens, in continuous cultivatation. Our aim is to apply DC for electrostimulation of micro-organisms in a laboratory-scale culture
Bacterium and chemicals
A gram-negative bacillus strain capable of utilizing phenanthrene as the sole carbon source was isolated from the petroleum-contaminated soil sampled in Tianjing, China. The strain was identified to be Enterobacter dissolvens. Phenanthrene and TTC as 99.0% analytical standards were both purchased from Alfa Aesar (Massachusetts, USA). Other reagents used are all in analytic grade.
Medium and pre-cultivation
The strain was pre-cultivated in shaking flask to multiply cell mass. The culture medium was prepared by mixing glucose with basal mineral medium containing, which finally contained (in g/l deionized water): glucose (6.7 g/L), K2HPO4 (0.8), KH2PO4 (0.2), NH4NO3 (0.8), MgSO4 (0.25), FeSO4•7H2O (0.09), and CaCl2 (0.032). Pre-cultivation was started by adding 0.1 ml of acclimatized inoculum into 150 ml of culture medium in shake flask (300 ml), and proceeded at 30°C (by shaker, 150 rpm) for approximately 10 hours when cell growth just entered the early log phase (0.2~0.3 in OD600 against fresh medium). The culture was then taken out and subjected to electrolytic stimulation experiments.
Electrolytic stimulation
The electrolytic stimulation experiments were conducted in two identical jacketed CSTR-type vessels (working volume: 100 ml) as shown in Peng She[9]. Two electrodes made of either platinum (Pt) wires (0.3 mm diameter) were installed on one vessel to implement AC current. The other vessel was then used for the control experiment without AC current. Before the electrolytic experiment, the two vessels were both autoclaved and then filled with 70 ml of pre-cultivated cell culture in each one. During the experiments, the broths were continuously cultivated on a multi-heads magnetic stirrer with the same mixing intensity. AC current was supplied by an electrophoresis apparatus (Liuyi Instrument, Beijing, China) and thermostatic water at 30°C continuously flowed through the jackets of both vessels. Culture samples were periodically taken out from each vessel, then immediately subjected to further analysis.
Analysis
Dehydrogenase activity
The dehydrogenase activity was measured by using 2,3,5-triphenyl tetrazolium chloride (TTC) as a terminal acceptor of protons and electrons from glucose being oxidized. After sampling 2 ml of the cell suspension was immediately mixed with 2 ml of glucose solution (0.1 M), 2 ml of Tris-HCl buffer (pH 8.4) and 2 ml of TTC solution (0.5%). After 3 hours incubation at 30°C, the mixture was extracted by 5 ml of toluene. The 2,3,5-triphenyl formazan (TPF) product in the organic phase was then spectrometrically measured at 490 nm against toluene. After divided by the corresponding cell density value (OD600), the OD490/ OD600 ratio was directly used as an index to the specific dehydrogenase activity.
Cell density and glucose concentration
Cell density in culture solution was determined by the optical density at 600 nm, and also checked by colony counting on agar plate in case of necessity. Glucose concentration was analyzed by dinitrosalicylic colorimetric (DNS) method.[10] Electrolysis test in fresh medium confirmed that glucose is stable in the presence of electrode reactions (data omitted).
Redox potential and pH
The redox potential and pH of culture solution were off-line measured by a combined platinum-redox electrode (Pt4865-50-S7, Mettler Toledo, Switzerland) and a pH sensor (Z71201AP40, Applikon, the Netherlands), respectively.
Cell growth and glucose uptake in electrolytic continuous cultivation process
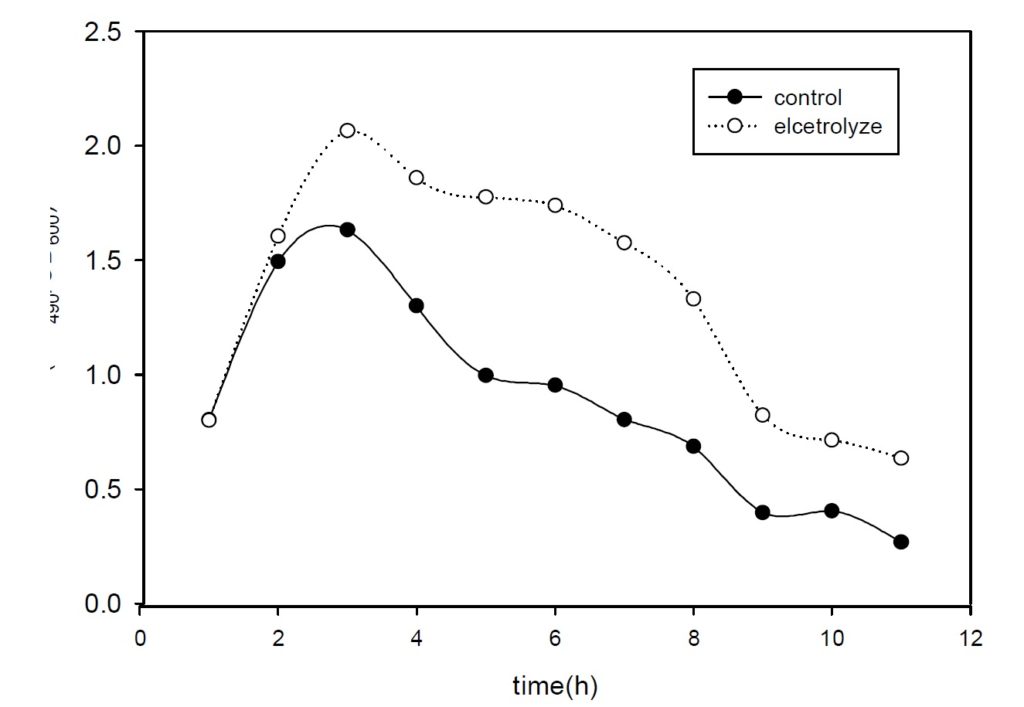
Based on she[9] by batch culture electrolytic stimulation of cell culture process research, The suitable current intensity is 10 ma in DC. So 10mA current were applied In the experimental process Monitoring cell dehydrogenase live (DHA), glucose degradation rate and growth curve three changes Because the dehydrogenase activity (DHA) was often used to represent the microbial activity. In order to characterize the effects of water electrolysis on cell growth and glucose uptake, electrolytic stimulation experiments with DC current at various intensity were conducted. Changes in cell DHA, glucose concentration and cell growth were measured in both electrolyzed culture and the control culture as a function of time. The results as shown in figure 1 to figure 3.
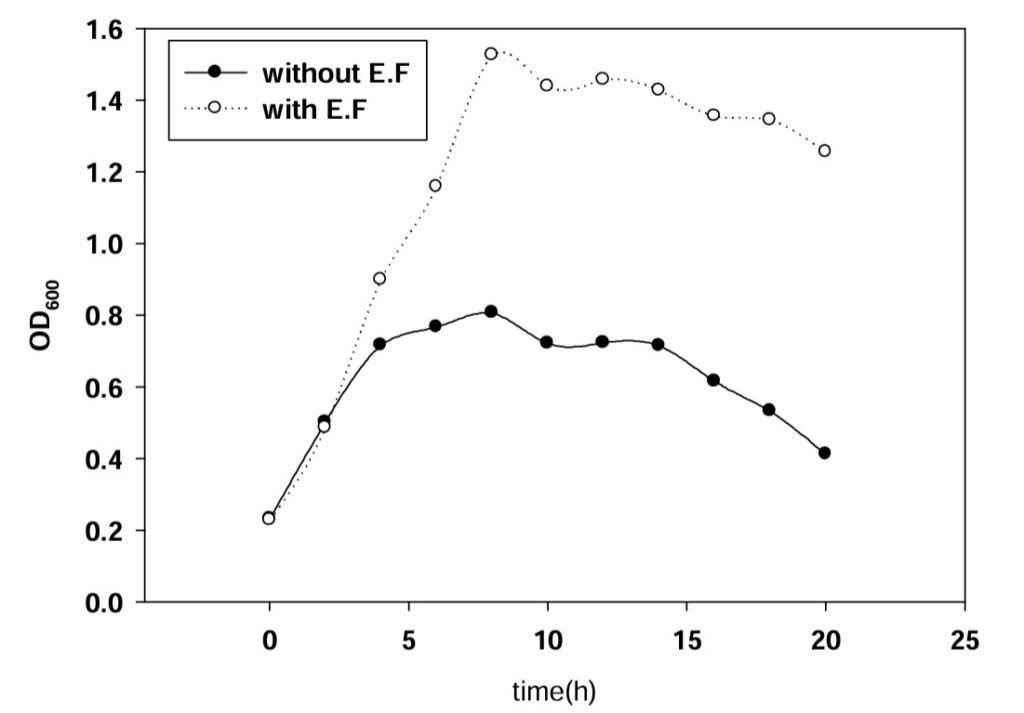 Fig. 1. Cell growth under 10 mA DC current
Fig. 1. Cell growth under 10 mA DC current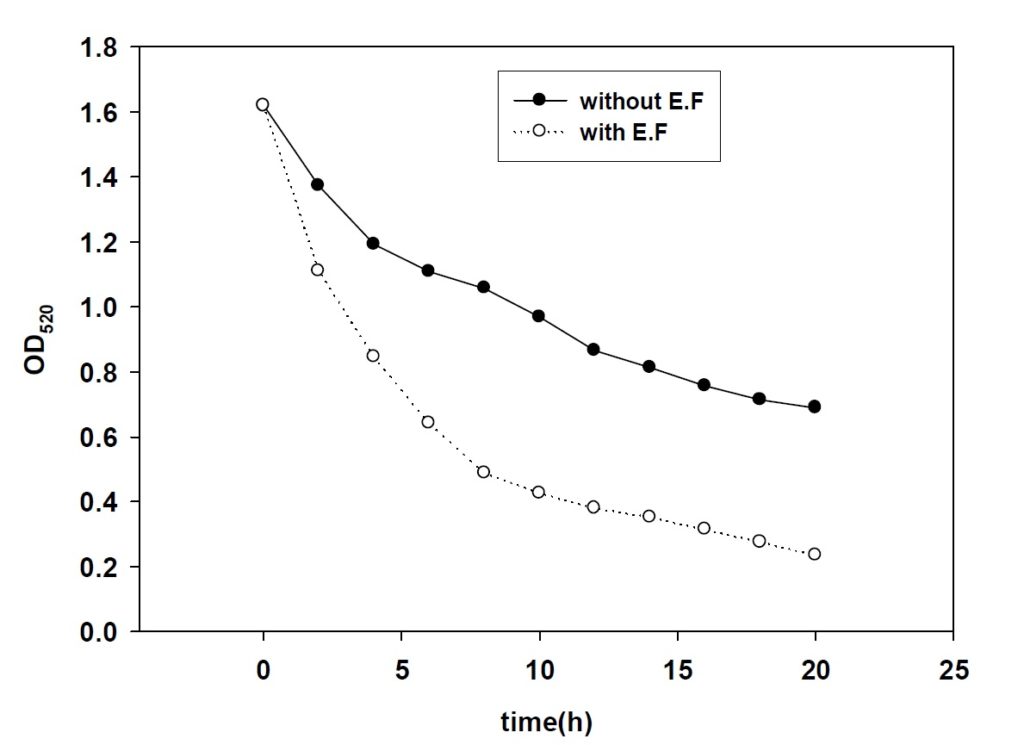 Fig. 2. Cell glucose uptake under 10 mA DC current
Fig. 2. Cell glucose uptake under 10 mA DC currentAs shown in figure 1 and figure 3, notable increases in cell DHA and glucose uptake were obtained in electrolyzed culture. After 8 hours exposure, the specific DHA represented by TPH(OD490)/Cell density (OD600) had reached 2 times in electrolyzed culture over the control value, and 3 times in terms of the glucose uptake. Besides the relevancy between glucose utilization and cell DHA, present results clearly indicated glucose metabolism was enhanced in the presence of electrolytic reaction. Compared with batch growth, continuous culture growth in good condition And the Cells is in the stationary phase
Chang of cell solution properties in electrostimulation
The influence from electrolysis reaction by AC current was also indicated by the changes in solution properties. During all the AC electrostimulation experiments presented above, there is visible changes in solution pH Changes in the physicochemical properties (pH and redox potential) of culture solutions were simultaneously recorded and shown in figure 4. The culture pH continuously ascend during the cultivation process, indicating the capability of producing organic alkali by the strain. Furthermore, no apparent distinction in pH was observed on both cultures within the first 12 hours. Although a 1 difference in pH appeared at the end of the experiment, this change was obviously not enough to fully account for the greatly accelerated ascend of cell DHA shown in figure 3. On the other hand, the implementation of AC current also led to an immediate decrease in the redox potential (Eh) of electrolyzed culture, which is always below -200 mV and much lower than the control value. This indicates the presence of water electrolysis maintained a strong reductive environment during the electrolytic cultivation process, which is probably the reason for enhanced cell DHA and glucose utilization.
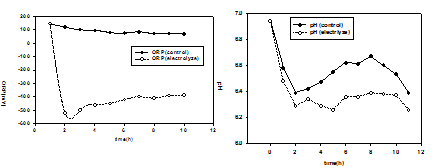 Fig. 4. Time course profiles of the cell solution properties(ORP) and (PH)
Fig. 4. Time course profiles of the cell solution properties(ORP) and (PH) The electrolytic stimulation process of strain E. dissolvens was investigated in present research. Compared with the batch culture, using continuous culture way, on the one hand, culture medium with bacteria liquid flow on the harmful intermediate product has the dilution effect, on the other hand the substrate of the continuous supply helps make the cell vigor has always been maintained at a high level. Through the cell electrolytic stimulation continuous cultivation model show that the continuous training mode can be compared with the batch culture higher positive electrolytic stimulation effect, and bacteria concentration and the cell vitality level also in more long training period maintain relative stability.
Through the electrolytic stimulation to cells continuous culture research seldom reported. This experiment through the electrolytic stimulation in the process of hydrolysis reaction control reaction system in ORP potential, thus make the cell culture of each parameter has improved markedly, this shows that direct current field stimulation microbial reaction has very good application value and development potential. On the microorganisms of industrial production has a certain significance.
Project supported by China west normal university doctor Starting Foundation(No:15E023)
- E.M. Goodman, B. Greenebaum, M.T. Marron, Effects of electromagnetic fields on molecules and cells, International Review of Cytology – a Survey of Cell Biology 158 (1995) 279 – 338.
- H. Berg, Problems of weak electromagnetic field effects in cell biology, Bioelectrochemistry and Bioenergetics 48(1999) 355 – 360.
- S.A. Blenkinsopp, A.E. Khoury, J.W. Costerton, Electrical Enhancement of Biocide Efficacy against Pseudomonas aeruginosa Biofilms, Applied and Environmental Microbiology, 58(11) 1992, 3770 – 3773.
- N. Wellman, S.M. Fortun, B.R. McLeod, Bacterial Biofilms and the Bioelectric Effect, Antimicrobial Agents and Chemotherapy, 1996; 40(9); 2012–2014.
- P.S. Stewart, W. Wattanakaroon, L. Goodrum, S.M. Fortun, B.R. McLeod, Electrolytic Generation of Oxygen Partially Explains Electrical Enhancement of Tobramycin Efficacy against Pseudomonas aeruginosa Biofilm, Antimicrobial Agents and Chemotherapy, 1999; 43(2): 292-296.
- K. Nakanishi, H. Tokuda, T. Soga, T. Yoshinaga, Takeda M. Effect of Electric Current on Growth and Alcohol Production by Yeast Cells, Journal of Fermentation and Bioengineering, 1998; 85(2); 250 – 253.
- A.M. Hayes, J.R.V. Flora, J. Khan, Electrolytic Stimulation of Denitrification in Sand Columns, Water Research, 1998; 32(9); 2830 – 2834.
- 8. Beschkov, S. Velizarov, S.N. Agathos, V. Lukova, Bacterial Denitrification of Waste Water Stimulated by Constant Electric Field, Biochemical Engineering Journal, 2004; 17(2); 141–145.
- Peng She,Bo Song,Xin-Hui Xing etl. Electrolytic stimulation of bacteria Enterobacter dissolvens by a direct current.Biochemical Engineering Journal.2006; 28: 23-29.
© The Author(s) 2017. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.