ISSN: 0973-7510
E-ISSN: 2581-690X
The present study evaluates the effect of mothers lactation age and storage on the antibacterial potency of breast milk against common pathogens (E.coli, S.aureus and coagulase negative staphylococci) affecting newborns and toddlers. Breast milk expressed at four months and twelve months post-partum was subjected to antibacterial assay by agar well diffusion against the above bacterial isolates in comparison to cow milk. Also, breast milk stored at 4°C was evaluated for its antibacterial effect to study the effect of storage. Results indicated that human breast milk was far more superior in its anti-bacterial effect seen (in terms of zone diameter) than cow milk against all the five strains tested. Also, early post partum milk (four months post-partum) was significantly superior (in terms of zone size) than twelve months post partum milk. Storage studies showed that breast milk retained its activity upto 24 hours of storage at 4°C against the five test strains.The present study indicates that even late lactation milk has enough anti-bacterial properties to protect babies beyond one year and toddlers as well thus promoting the concept of continued breast feeding for one year and beyond. Further, study highlighted that lactating mother can pump the milk and refrigerate it for later consumption without compromising on the antibacterial potency of their milk for upto 24 hours.
Breast Milk, Neonatal Pathogens, coagulase negative Staphylococci
Breast milk is a complete nourishment and medicinal shield for the newborn. Its multiple benefits have been known since ages. They include strengthening baby’s immune system, anti-inflammatory activity, lowering risk of allergies, chronic diseases, mental ailments etc.1-3 Human milk contains cellular and hormonal factors that serve to protect infants against numerous bacteria, viruses, and fungi.4 Human breast milk exhibits antibacterial effect due to the presence of antimicrobial peptides (AMP) in it. It contains comparatively higher levels of AMP than those present in formula feed or cow milk.5,6 Antimicrobial peptides present in breast milk include lactoferrin, alpha defensins beta defensin, lysozyme, lacto-peroxidase. All these factors in breast milk significantly lower the incidence of number of bacterial infections in babies such as ear infections, respiratory illnesses, and bouts of diarrhea.7, 8 According to numerous reports, infants who breastfed appear to be less susceptible to certain infection than bottle-fed infants9. A number of health organizations — including the American Academy of Paediatrics (AAP), the American Medical Association (AMA), and the World Health Organization (WHO) — recommend breastfeeding as the best choice for babies.
The evaluation of antibacterial effect of breast milk has been investigated by past workers[10-12] which clearly shows its useful potential against bacterial infections. However, not much work has been focussed into studying the effect of mother’s lactation age and storage of expressed milk on the antibacterial effect against the common neonatal pathogens. This is essential to know as working mothers store their expressed milk for their babies for later consumption. The present study gives an insight into 1) Antibacterial effect of human breast milk against common neonatal pathogens such as E.coli, Staphylococcus aureus, Coagulase negative staphylococci (CoNS) 2) Effect of age of lactation on antibacterial efficacy and 3) Effect of storage of expressed milk on its antibacterial efficacy.
Isolation of CoNS and S. aureus from nasal and skin swabs
Skin swabs and nasal swabs from n=12 healthy volunteers were taken. Four swabs were pooled with three sets each for skin swab and nasal swab in nutrient broth tubes. These swabs were streaked on nutrient agar plates and incubated at 37°C for 24 hours. Colonies so obtained on nutrient agar plates were further identified on the basis of a) Microscopic analysis: Gram reaction and morphology b) Colony morphology c) Catalase reaction d) Coagulase test e) Mannitol fermentation and growth on selective medium i.e Mannitol salt agar (MSA).Strains isolated were later stored as slants at 4°C till further use.
E.coli strain used in the present study was an already characterized strain present in the laboratory. Its identification was again performed on the basis of Gram reaction, motility, IMViC, lactose fermentation, gas production and growth on Endo agar with metallic sheen.
Milk Sample
Human milk was donated by six lactating mothers (one included the corresponding author itself) with no clinical evidence of infection or inflammation and who had not taken any antibiotics previously by manual expression. Out of the six, three (N=3) healthy mothers were of lactation age of four months post-partum. Similarly, the rest three (N=3) healthy mothers were of lactation age of 12 months post-partum. Informed consent and oral consent was obtained from the mothers of infants for their participation in the study.
Evaluation of anti-bacterial efficacy of human breast milk using agar well diffusion assay
The antibacterial efficacy of breast milk was evaluated using the agar well diffusion assay as per the method of CLSI.[13] Sample was collected by first cleaning the nipple area with clean wet tissue paper followed by disinfection using 70% alcohol. Three samples (n=3) from three different mothers (4 month post-partum; referred as BM-4) were collected aseptically by self expression in sterile vials and labeled. The samples were stored at 4°C till further use.
Overnight cultures of E.coli, S.aureus S1 and S.aureus S2 and CoNS-S3, CoNS-S4 were used. Optical density (O.D) of the cultures were adjusted to 0.1 at 600 nm that corresponds to 108 CFU/ml. The cultures were spread plated followed by boring of wells. Wells were marked as RM (raw milk), BM (breast milk) and normal saline (0.9% NaCl) respectively. 0.1 ml of raw milk, breast milk and saline water was added to the wells respectively. Each sample was tested in duplicate along with cow milk and normal saline. All the plates were incubated in upright position at 37°C for 24 hours. Next day, zone of inhibition were measured.
Effect of lactation period on anti-bacterial efficacy of human breast milk
The effect of different lactation age on the antibacterial efficacy of breast milk was evaluated using samples collected from mothers of three different lactation ages. The breast milk form 4 month post-partum (BM-4) and breast milk post-1 year referred as BM-12 were used. Three samples each of BM-4 and BM-12 were used in the study. The antibacterial effect of all the samples was determined by agar well diffusion assay as explained above.
Effect of storage on anti-bacterial efficacy of human breast milk
Expressed milk sample (BM-4) was collected in four sterile vials respectively. Breast milk of one vial was subjected to agar well diffusion assay within one-two hour of collection. Rest of the vials were stored at 4°C for 24 h, 48 h and 72 h respectively and labelled accordingly. At 24 h, 48 h and 72 h, the stored samples were again subjected to agar well diffusion assay respectively by the same process as described above. Zone of inhibitions were measured on next day and compared with the zones obtained by using samples of 48 h and 72 h. Similarly, cow milk samples were also processed.
Statistical Analysis
Statistical analysis of the data was performed using Microsoft Excel 2007. Most of the data are expressed as the mean ± standard deviation (SD) of more than two experimental values for every variable. Student’s t test was used to compare different variables. P values of <0.05 were considered significant.
A total of two strains of S.aureus and two strains of CoNS were isolated (Table-I). All the strains were gram positive cocci in bunches and catalase positive. S.aureus strains gave a golden yellow colonies on nutrient agar. They were coagulase positive and showed mannitol fermentation. The colonies seen on selective media i.e Mannitol salt agar (MSA) exhibited change in colour to yellow confirming positive mannitol fermentation. The other two strains exhibited no change in color with non-mannitol fermentation on MSA plates.
Table (1):
Strains isolated and their characteristic features.
| Source | Gram Reaction | Colony Morphology | Catalase & Coagulase Reaction |
Mannitol Fermentation | |
|---|---|---|---|---|---|
| S.aureus (S-1) | Spoiled food sample | Gram Positive Cocci in bunches; Golden Yellow colonies on Nutrient agar | (+);(+) | (+) | |
| S.aureus –(S-2) | Nasal | Gram Positive Cocci in bunches; Golden Yellow colonies on Nutrient agar | (+) ;(+) | (+) | |
| CoNS (S-3) | Skin | Gram Positive Cocci in bunches; White colonies on Nutrient agar | (+);(-) | (-) | |
| CoNS(S-4) | Nasal | Gram Positive Cocci in bunches; White colonies on Nutrient agar | (+) ;(-) | (-) | |
Evaluation of anti-bacterial effect of breast milk against five test strains
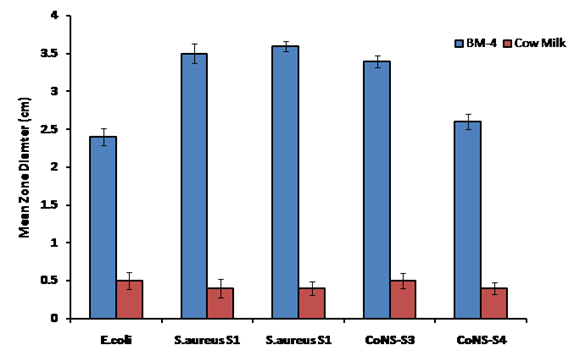
Human breast milk (4 month post-partum) was evaluated for its anti-bacterial effects using agar well diffusion assay against two strains of S.aureus, two strains of CoNS and one E.coli strain. The results are depicted in Fig.1. Human breast milk was far more superior in its anti-bacterial effect seen (in terms of zone diameter) than cow milk against all the five strains tested. Breast milk showed the largest zone diameter for S.aureus (3.5 cm) followed and CoNS and E.coli that gave an average zone diameters of 3.0 cm and 2.4 cm respectively. Cow milk showed negligible activity against all the test strains.
Effect of lactation period on the anti-bacterial properties of human breast milk
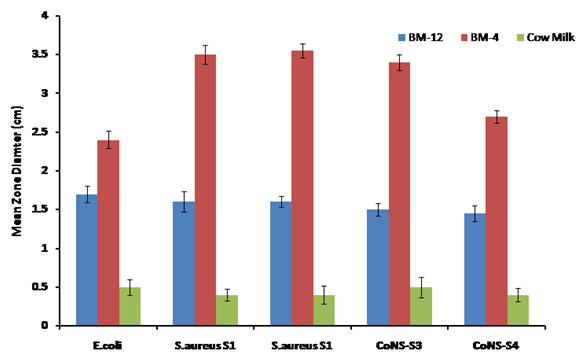
Results showed significant anti-bacterial activity (measured as mean zone of inhibition) against test strains i.e (isolated from spoiled food sample).BM-4 showed higher inhibition against all the strains in the agar well diffusion assay (as seen Fig.2).Cow milk showed negligible zones. Equal volumes of normal saline (0.85%) NaCl) was used as negative control showing no zone of inhibition.
Fig. 2. Comparison of antibacterial activity of BM-12, BM-4 and Cow milk using agar well diffu-sion assay against all five test strains
As can be seen in the graph below, BM-4 breast milk showed a mean zone diameter of 3.5 cm against S.aureus strains and >3 cm against CoNS strains. There was more than two fold increase in the zone diameters with BM-4 as compared to BM-12 against the strains tested. Also, a significant increase from 1.6 cm (BM-12) to 2.5 cm (BM-4) was obtained against E.coli tested.
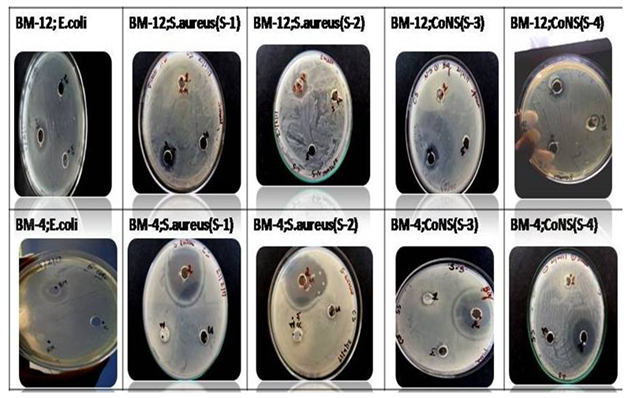
Fig. 3. Pictoral representation of anti-bacterial effect of two types of breast milk i.e BM-12 (late lactation) and BM-4 (early lactation) against five test strains
Fig.3 clearly indicates the significant increase in the zone diameters (in cm) of BM-4 as compared to BM-12 against all the four strains tested. Although there was less activity with BM-12 milk , but still it indicates that even late lactation milk has enough anti-bacterial properties to protect babies beyond 1 year and toddlers as well.
Effect of storage on anti-bacterial activity of human breast milk
In an attempt to study the effect of storage of breast milk on its anti-bacterial properties, breast milk was sampled and distributed into four aliquots and stored at 4°C.The first was processed immediately within 2 h of sampling, the second was processed after 24 h, 48 h and 72 h respectively.
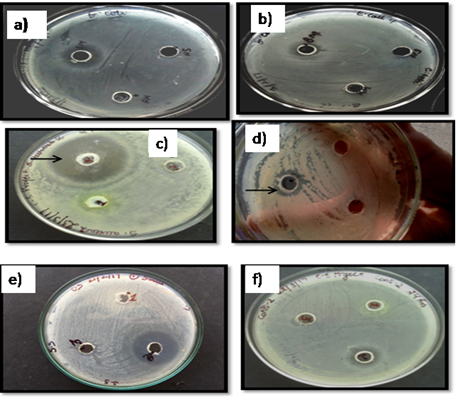
Fig. 4. A) E.coli zone immediate plating B) E.coli zone 24 h post sam-pling C) S.aureus zone immediate plating D) S.aureus zone 24 h post sampling E) CoNS immediate plating and F) CoNS zone 24 h post sampling
The pictoral representation of zone diameters is shown in Fig. 4, indicates the zone diameters (cm) obtained after immediate sampling and 24 h sampling. No zones were detected at 48 h and 72 h post-sampling. This indicates that breast milk retained its activity upto 24 h of storage at 4°C.There was an approximate 50% decrease in activity in terms of zone diameters after 24 h storage of breast milk against the three test strains.
Table (2):
Results of storage effect of breast milk (in terms of % activity retained).
1 h post sampling |
24 h post sampling |
% activity retained after 24 h |
|
|---|---|---|---|
E.coli |
2.4 cm |
1.3 cm |
54.20% |
S.aureus-S1 |
3.8 cm |
1.4 cm |
38.80% |
S.aureus-S2 |
3.5 cm |
1.3 cm |
37.1% |
CoNS-S3 |
3.2 cm |
1.4 cm |
43.7% |
CoNS-S4 |
3.0 cm |
1.4 cm |
46.6% |
As seen in Table-2, 54% activity was retained even after 24 hours of storage against E.coli.Similarly, 38% and 46% activity was still present in the stored breast milk against S.aureus and CoNS strains resepctively as seen after 24 h plating.
The present study was aimed at evaluating the anti-bacterial properties of human breast milk against three bacterial species that are commonly associated with infections of newborns and infants. These included E.coli which is a leading cause of gastroenteritis, abdominal cramps ,and urinary tract infections, neonatal sepsis among newborns and infants.14-16 It has also been lately associated with sudden infant death syndrome (SIDS).17 In addition, S.aureus and coagulase negative Staphylococci (CoNS) both of which are commonly isolated from cases of skin infections. They are also the leading cause of conjunctivitis/ocular infections, pneumonia, bloodstream sepsis and mortality among neonates especially preterm and very low birth weight infants.18-20 These are gaining more importance in the clinical world due to the increasing prevalence of antibiotic resistant strains common in neonatal and paediatric wards. Therefore, we isolated these species from their natural habitat i.e. skin and nasal area of healthy volunteers. Two strains of CoNS and two strains of S.aureus were isolated (skin and nasal samples) and characterized.
The antibacterial effect of sampled breast milk was assessed on the basis of agar well diffusion assay. Since cow milk is considered the easy to digest milk choice for infants (in cases where breastfeeding is not possible or it is not done); therefore freshly drawn cow milk was collected and used as control. Results clearly depicted that breast milk (4 months post partum) was highly superior (in terms of zone diameter) against all the test strains . Cow milk was not able to show any clearance zone against bacteria. Breast milk displayed biggest zone against S. aureus and CoNS followed by E. coli. Antibacterial effect of human breast milk is attributable to many components present in the milk. Antimicrobial peptides present in breast milk include lactoferrin, alpha defensins, beta defensin, lysozyme, lacto-peroxidase. In addition, immunological factors such as complement, immunoglobulins also contribute to bacterial phagocytosis and killing action. Avery and Gordon,21 studied the opsonic components of breast milk responsible for phagocytosis of surface-adherent Staphylococcus aureus by human polymorphonuclear leukocytes. Results showed that there was significantly greater phagocytosis of bacteria pre-opsonized with 100% breast milk than of unopsonized bacteria. Hancock et al.22 also delineated the possible role of xanthine-oxidase (abundant in human milk) as a potential antibacterial components involved in generation of nitric oxide that kills bacteria. Recently, workers have also reported the antibacterial and anti-biofilm role of human milk Oligosaccharides(HMOs) against Gram positive pathogens.23 Study has showed that the concentration at which beta-defensin showed antimicrobial activity against enteropathogens and nosocomial strains is the same as human milk’s, highlighting its physiological function.24 All these factors contribute towards greater antibacterial activity of breast milk essential for infants protection.
Further, the effect of lactation period on the antibacterial activity against the test strains was performed. The concentrations of total and specific components of human breast milk vary between mothers, based on infant’s gestational age, and over the course of lactation.[25,26] Baricelli et al.24 reported higher anti-microbial peptide (AMP) and beta-defensin levels in colostrum and a progressive decline with the lactation age. Castellote et al.27 reported that the concentration of immune factors in colostrum samples is richer than both transitional and mature milk. Our results also support this as it clearly indicate that BM-4 exhibit greater zone of clearance for all five test strains as compared to BM-12.
Certain lactating mother who are either working or find it difficult to breastfeed often pump the milk and refrigerate it for later consumption whenever the babies required . Hence, experiment was conducted to study the effect of storage on antibacterial properties of breast milk .It was found that breast milk retained its antibacterial properties on 24 h storage but had negligible activity on 48 h of storage. Our results are in line with the finding of Marrtinez-Costa and co-workers,28 who reported that human milk possessed bactericidal activity that remained stable during the first 48 hours of refrigerated storage, but thereafter showed a significant decline beyond 72 hours. Another study showed that expressed breast milk displayed little change in the immunological components even after 72 h of storage at 4°C.29 Hanna et al.30 who studied the effect of storage on the antioxidant effect of breast milk also clearly stated that in order to preserve the antioxidant activity of human milk, storage time should be limited to 48 hours. Takci et al.31 also reported that breast milk stored at 4°C for 24 h in Pyrex containers did not show significant decline in antibacterial potential emphasizing on the choice of storage containers used for storage.
The present study has emphasised on the antibacterial efficacy of breast milk. It clearly indicates that breast milk is significantly superior over cow milk. Further, age of lactation affects the antibacterial potency although breast milk 12 months post-partum also retained significant activity. Also, refrigerated storage upto 24 h did not decrease its antibacterial effect. Future studies focusing on effect and mechanism of human milk on different clinical strains especially resistant bacterial strains need to be looked into.
Compliance with ethical standards
Conflict of Interest Disclosure: The authors have no competing interests to declare. Ethics of Human and Animal Experimentation: No experimentation on humanas or animals performed Informed Consent: Informed and verbal consent was obtained from participating mothers (which includes the corresponding author as one of the milk donor).
ACKNOWLEDGMENTS
The assistance provided by Department of Food Science and MCM DAV College for Women, Chandigarh is gratefully acknowledged.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
- Oddy WH. The impact of breast milk on infant and child health [online].Breastfeeding Review, 2002; 10(3):5-18.
- Lessen R, Kavanagh K. Position of the academy of nutrition and dietetics: Promoting and supporting breastfeeding. J Acad Nutr Diet, 2015; 115:444–449.
- Martin CR, Ling PR, Blackburn GL. Review of Infant Feeding: Key Features of Breast Milk and Infant Formula. Nutrients, 2016; 8(5):279.
- Oddy WH. Infant feeding and obesity risk in the child [online].Breastfeeding Review, 2012; 20(2):7-12.
- Lonnerdal B. Bio-active proteins in breast milk. J Paediatr Child Health, 2013; 49:1–7.
- Hakansson AP. Protective effects of human milk antimicrobial peptides against bacterial infection. J. Pediatr, 2015; 91(1): 04-05.
- Lopez-Alarcon M , Villalpando S , Fajardo A .Breast-feeding lowers the frequency and duration of acute respiratory infection and diarrhea in infants under six months of age. J Nutr, 1997; 127(3):436-443.
- Tromp I, Kiefte-de Jong J, Raat H, Jaddoe V. Breastfeeding and the risk of respiratory tract infections after infancy: The Generation R Study. PLoS ONE, 2017; 12(2): e0172763.
- Stuebe A. The Risks of not Breastfeeding for Mothers and Infants. Rev Obstet Gynecol, 2009; 2(4): 222–231.
- Silvestre D, Lopez MC, March L, Plaza A. Bactericidal activity of human milk: stability during storage. Br J Biomed Sci, 2006; 63:59–62.
- Martinez-Costa C, Silvestre MD, Lopez MC, Plaza A.Effects of refrigeration on the bactericidal activity of human milk: a preliminary study. J Pediatr Gastr Nutr, 2007; 45:275-7.
- Silvestre PK, Carvalhaes MABL, Venancio SI, Tonete VLP. Breastfeeding knowledge and practice of health professionals in public health care services. Rev Latino-am Enfermagem, 2009; 17(6): 953-960.
- CLSI, Performance Standards for Antimicrobial Disk Susceptibility Tests, Approved Standard, 7th ed., CLSI document M02-A11. Clinical and Laboratory Standards Institute, 950 West Valley Road, Suite 2500, Wayne, Pennsylvania 19087, USA, 2012.
- Polin RA, St Geme JW, III. Neonatal sepsis. Adv Pediatr Infect Dis, 1992; 7:25–61.
- Camacho-Gonzalez A, Spearman PW, Stoll BJ. Neonatal infectious diseases: evaluation of neonatal sepsis. Pediatr Clin North Am, 2013; 60:367–389.
- Simonsen KA, Anderson-Berry AL, Delair SF, Davies HD. Early-Onset Neonatal Sepsis. Clin Microbiol Rev.2014; 27(1): 21–47.
- Bettelheim KA, Goldwater PN. Escherichia coliand Sudden Infant Death Syndrome. Front Immunol, 2015; 6:343. [18] Venkatesh MP, Placencia F, Weisman LE. Coagulase-negative staphylococcal infections in the neonate and child: an update. Semin Pediatr Infect Dis, 2006; 17(3):120–127.
- Rogers KL, Fey PD, Rupp ME. Coagulase-negative Staphylococcal infections. Infect Dis Clin North Am, 2009; 3(1):73–98.
- Marchant EA, Boyce GK, Sadarangani M, Lavoie PM. Neonatal Sepsis due to Coagulase-Negative Staphylococci. Clin Dev Immunol, 2013; 586076.
- Avery VM, Gordon DL. Antibacterial properties of breast milk: requirements for surface phagocytosis and chemiluminescence. Eur J Clin Microbiol Infect Dis, 1991; 10(12):1034-9.
- Hancock JT, Salisbury V, Ovejero Boglione MC, Cherry R. Antimicrobial proteins of milk: dependence of presence of xanthine oxidase and nitrite. Antimicrob Agents Chemother, 2002; 46:3308-3310.
- Ackerman DL, Doster RS, Weitkamp JH, Aronoff DM. Human Milk Oligosaccharides Exhibit Antimicrobial and Antibiofilm Properties against Group B Streptococcus.ACS Infect Dis. 2017; 3(8):595-605.
- Baricelli J, Rocafull MA, Vazquez D, Bastidas B. â-defensin-2 in breast milk displays a broad antimicrobial activity against pathogenic bacteria. J Pediatr, 2015; 91:36–43.
- Armogida SA, Yannaras NM, Melton AL, Srivastava MD. Identification and quantification of innate immune system mediators in human breast milk. Allergy Asthma Proc, 2004; 25(5):297-304.
- Ballard O, Morrow AL. Human milk composition: nutrients and bioactive factors. Pediatr Clin North Am, 2013; 60(1):49-74.
- Castellote C, Casillas R, Ramírez-Santana C, Perez-Cano FJ. Premature delivery influences the immunological composition of colostrum and transitional and mature human milk. J Nutr, 2011; 141:1181-1187.
- Martínez-Costa C, Silvestre MD, Lopez MC, Plaza A. Effects of refrigeration on the bactericidal activity of human milk: a preliminary study. J Pediatr Gastroenterol Nutr, 2007; 45(2):275-7.
- Lawrence RA. Storage of human milk and the influence of procedures on immunological components of human milk. Acta Paediatr Suppl, 1998; 88(430):14-8.
- Hanna N, Ahmed K, Anwar M, Petrova A. Effect of storage on breast milk antioxidant activity. Arch Dis Child Fetal Neonatal, 2004; Ed 89:F518-F520.
- Takci S, Gulmez D, Yigit S, Dogan O, Hascelik G. Container type and bactericidal activity of human milk during refrigerated storage. J Hum Lact, 2013; 29(3):406-11.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.