ISSN: 0973-7510
E-ISSN: 2581-690X
Soil microbe and soil enzyme activities are closely related to the fertilizer inputs. Our objective was to explore the dynamic change of soil microbe and soil enzyme activities in paddy field during early and late rice (Oryza sativa L.) main growth stages with different long-term fertilizer managements in the double cropping rice system. we analyzed the soil microbe and soil enzyme activities with mineral fertilizer alone (MF), rice residues and mineral fertilizer (RF), 30% organic matter and 70% mineral fertilizer (LOM), 60% organic matter and 40% mineral fertilizer (HOM), and without fertilizer (CK) basis on long-term fertilizer experiment. The results showed that the soil enzyme activities were increased by application of mineral fertilizer along with manure or rice residues at the main growth stages of early and late rice. The soil urease activities for different fertilizer managements was RF>HOM>LOM>MF>CK at the main growth stages of early and late rice. The soil catalase activities with HOM, LOM treatments was highest, and was significantly higher than that of RF, MF, CK treatments at the main growth stages of rice. The soil invertase and dehydrogenase activities with HOM treatment were highest, and were significantly higher than that of CK at the main growth stages of rice. And the soil cellobiohyrolase activities with RF treatment were highest, and were significantly higher than that of MF, CK treatments at the main growth stages of rice. Meanwhile, the results indicated that different fertilization managements were significantly affected some physiological function soil microbial quantity. The amount of nitrifying and denitrification bacteria in soil for different fertilizer managements was HOM>LOM>RF>MF>CK at the main growth stages of early and late rice. Meanwhile, the amount of ammonifiers and cellulose”decomposing bacteria in soil for different fertilizer managements was MF>RF>LOM>HOM>CK and LOM>RF>MF>HOM>CK at the main growth stages of rice, respectively. The amount of azotobacteria in soil with RF treatment was highest, and was significantly higher than that of MF, CK treatments at the main growth stages of rice. As a result, the soil microbe and soil enzyme activities were increased by applied of fertilizer practices, the effect of mineral fertilizer combined with manure or rice residues is better than that of only mineral fertilizer.
Rice; Paddy field; Long-term fertilizer management; Soil microbe; Soil enzyme activity.
Enzymes is an index of soil microbial activity and fertility for that it was respond to changes in soil management more quickly than other soil variables, and they play an important role in the cycling of nutrients in nature. Therefore, it was useful as early indicators of biological changes.1 It was involved in energy transfer, environmental quality and crop productivity.2,3 The number and activities of soil enzymes were influenced by soil microorganisms, plants and animals, and is closely related to environmental and ecological factors. Among the different enzymes in soils, the soil urease, catalase, invertase, dehydrogenase and cellobiohyrolase activities were important for the soil nutrients, for that they are play a key role in C, N, P cycling in ecological systems.
Soil microbial communities also play a critical role in the cycling of carbon and nutrients in terrestrial ecosystems, as well as regulating plant productivity and diversity.4 Therefore, there has a closely relationship between soil functions and its community structure, activity.5,6 Meanwhile, the communities and activities of soil bacterial were affected by the physicochemical properties of the soil, such as the pH,7 water content,8 mineral composition, fertilizer managements9 and planting systems10.
In recent years, many studies have indicated that the enzyme activities and microbial communities of soil are known to be affected by the field managements, including the vegetation types and crop rotations,11 soil tillage,12 fertilization regime13,14 and so on. Meanwhile, fertilization is an important factor that influences the soil enzyme activities and diversity and growth of microorganisms in agricultural soils.
However, relatively few studies have investigated how different fertilizer managements affect the soil enzyme activities and soil microbes in double cropping rice system paddy field in southern China. It is a traditional practice that application with organic manure used as the main nutrient source for rice production in China. In recently yeas, it was commonly accepted that application of mineral fertilizer along with rice residues practices in rice production systems enhance the soil quality.15 Therefore, the rice residues returning to the field practices is another important source of nutrients during rice production.16 However, in recent years, there has been a large increase in the use of mineral fertilizer and decrease in the use of organic fertilizer in rice production. Therefore, the soil quality was decreased in this fertilizer regime, such as decrease in soil organic matter (SOM) content and soil microbial community.17 Rather than applying with mineral fertilizer alone, the fertilizer regime of application mineral fertilizer with manure or rice straw is benefit for maintain both soil quality and high grain yields in double-cropping production systems.
The double-cropping production systems is the main crop rotation in southern China, and the fertilizer regime are very important for rice production and the paddy agroecosystems, however, limited information about the changes of soil enzyme activities and soil microbe under different long-term fertilization schemes in the double-cropping rice systems in southern China. Therefore, the objective of the present research was to study the soil enzyme activities and soil microbe at the early and late rice main growth stages as affected by long-term fertilization managements.
Sites and cropping system
The experiment was established in 1986. It was located in Ningxiang County (28°07 N, 112°18 E) of Hunan Province, China. Under a continental monsoon climate, the annual mean precipitation is 1553 mm and potential evapotranspiration of 1354 mm. The monthly mean temperature is 17.2°C. Soil texture in the plough layer (0–20 cm) was silt clay loam with 13.71% sand and 57.73% silt. At the beginning of the study, the surface soil characteristics (0–20 cm) were as follows: soil organic carbon (SOC) 29.4 g/kg, total N 2.0 g/kg, available N 144.1 mg/kg, total phosphorous (P) 0.59 g/kg, available P 12.87 mg/kg, total potassium (K) 20.6 g/kg, and available K 33.0 mg/kg. There were three crops in a year, barley (Hordeum vulgare L.), early rice and late rice (Oryza sativa L.). Barley was sown in the middle of November and harvested in early May of the following year. Early rice was then transplanted and harvested in the middle of July. The growing season of late rice from late July to the end of October.
Experimental design
The experiment had five treatments: control (without fertilizer input, CK), mineral fertilizer (MF), rice residue and mineral fertilizer (RF), low manure rate and mineral fertilizer (LOM), and high manure rate and mineral fertilizer (HOM). The design ensured all fertilized treatments received the same amount of N, phosphorus pentoxide (P2O5), potassium oxide (K2O) (the amount of N, P2O5, K2O in mineral fertilizer plus that from rice residue or manure) during the early and late rice growing season, respectively. The mineral fertilizers included urea, ordinary superphosphate and potassium chloride. Details about the fertilizer managements are listed in Table 1. Before transplanting rice seedlings, air-dried rice residue was manually spread onto the soil surface and incorporated into the soil at a cultivation depth of 20 cm. For early and late cropped rice, 70% and 60%, respectively, of mineral N fertilizer was applied at seedling and the remaining N fertilizer was applied by top dressing (7–10 days after transplanting) during crop growth. All the P and K fertilizers were applied at seedling. There were three replications and each plot size was 66.7 m2 (10 × 6.67 m).
Table (1):
Nutrient supply from rice straw, manure and mineral fertilizer under different fertilizer treatments
| Treatment | Early rice | Late rice | Total | ||||||
|---|---|---|---|---|---|---|---|---|---|
| N | P2O5 | K2O | N | P2O5 | K2O | N | P2O5 | K2O | |
| CK | 0+0* | 0+0 | 0+0 | 0+0 | 0+0 | 0+0 | 0 | 0 | 0 |
| MF | 142.5+0 | 54.0+0 | 63.0+0 | 157.5+0 | 43.2+0 | 81.0+0 | 300.0 | 97.2 | 144.0 |
| RF | 124.4+18.1 | 50.4+3.6 | 38.3+24.7 | 133.0+24.5 | 37.8+5.4 | 48.2+32.8 | 300.0 | 97.2 | 144.0 |
| LOM | 96.0+46.5 | 33.0+21.0 | 33.6+29.4 | 110.2+47.3 | 21.8+21.4 | 51.1+29.9 | 300.0 | 97.2 | 144.0 |
| HOM | 49.6+92.9 | 12.0+42.0 | 4.2+58.8 | 63.0+94.5 | 0.5+42.7 | 21.2+59.8 | 300.0 | 97.2 | 144.0 |
MF: Mineral Fertilizer alone; RF: Rice residues and mineral fertilizer; LOM: 30% organic matter and 70% mineral fertilizer; HOM: 60% organic matter and 40% mineral fertilizer; CK: without fertilizer.
*Input from mineral fertilizer + input from rice residue or manure. The numbers are in kg/ha.
For the RF treatment, rice straw return rate (air dry) was 2780, 3600 kg/ha for early and late rice.
For the LOM treatment, manure application rate (decomposed) was 2625.0, 2670.0 kg/ha for early and late rice.
For the HOM treatment, manure application rate (decomposed) was 5250.0, 5340.0 kg/ha for early and late rice.
The N, P, and K content of air-dry early rice straw was 6.5 g/kg, 1.3 g/kg, and 8.9 g/kg, N, P, and K content of air-dry late rice straw was 6.8 g/kg, 1.5 g/kg, and 9.1 g/kg, respectively, and N, P, and K content of decomposed chicken manure was 17.7 g/kg, 8.0 g/kg, and 11.2 g/kg, respectively.
Soil sampling
Soil samples were collected from the plow layer (0–20 cm) at seedling stage, tillering stage, jointing stage, heading stage, and mature stage of the early and late rice growing season in 2016, respectively. Three soil samples were taken from each plot at the main growth stages of early and late rice. The soil samples were passed through a 2-mm sieve and kept moist in a refrigerator at 4°C until use.
Laboratory analyses
Enzyme activities
The urease and invertase activity was measured according to Kandeler et al.(2006),18 and the invertase activity results were expressed as mg/(g soil•h) glucose released. Urease activity is calculated by the conversion of NH4-N after 24 h and colorimetric assessment at 690 nm.
Catalase activity was measured using the method of Roberge(1978).19 Based on the H2O2 transformation efficiency, the residual H2O2 was determined by titration with potassium permanganate, and catalase activity was expressed as mL KMnO4 consumed per gram per hour. Dehydrogenase activity was measured using triphenyl tetrazolium chloride (TTC) colorimetric analysis.3 The enzyme substrate TTC is degraded into triphenyl formazan (TPF), which is quantified colorimetrically at 485 nm.
Soil cellobiohyrolase was measured by 3, 5-dinitrosalicylic acid regent. Briefly, 5 g of air-dried soil (<1 mm), 15 mL of 8% sucrose, and 5-mL phosphate buffer at pH 5.5 were added to a 50-mL conical flask with 0.2-mL toluene. After shocking, the suspensions were incubated for 24 h at 37°C. Following filtration, 1-mL filtrate and 3-mL 3,5-dinitrosalicylic acid were added to a 50-mL flask and heated for 5 min at 100°C. The solution was immediately cooled and diluted to 50 mL with distilled water. Absorbance was then measured at 508 nm using a spectrophotometer.20
Soil microbial populations
The number of culturable nitrifying and denitrification bacteria, ammonifiers bacteria and azotobacteria in a sample was determined by counting colony forming units (CFU). The number of culturable nitrifying and denitrification bacteria, ammonifiers bacteria and azotobacteria were measured using the method of Xu and Zheng (2006).21 The density of culturable cellulose-decomposing bacteria was determined as colony forming units (CFU) using a soil dilution plate-count technique.22
Statistical analysis
All data were expressed as mean ± standard error. The data were analyzed as a randomized complete block, using the PROC ANOVA procedure of SAS.23 Mean values were compared using the least significant difference (LSD) test, and a probability value of 0.05 was considered to indicate statistical significance.
Dynamics of soil urease activities
At the early rice main growth stages, the activities of soil urease were affected by the different long-term fertilizer managements. The RF treatment has the highest soil urease activities and the CK had the lowest activities (Fig. 1), and the activity increased in the following order: CK<MF<LOM<HOM<RF. At the late rice main growth stages, the highest activity was the treatment that the rice straw was used as residues, and the activity trend was as follows: CK<MF<LOM<HOM<RF.
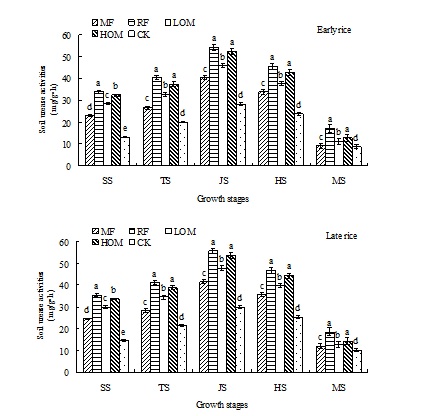
MF: mineral fertilizer alone; RF: rice residues and mineral fertilizer; LOM: 30% organic matter and 70% mineral fertilizer; HOM: 60% organic matter and 40% mineral fertilizer; CK: without fertilizer.
SS: seedling stage; TS: tillering stage; JS: jointing stage; HS: heading stage; MS: mature stage.
Bars represent standard deviation (S.D.) of three replicates. Different letters indicate significance at P < 0.05 among the fertilization treatments at the same rice growth stage, according to the least significant difference test.
Fig. 1. Effects of different long-term fertilizer treatments on soil urease activities during rice growth stages in paddy field
Dynamics of soil catalase activities
With the addition of the rice residue and manure, soil catalase activities in the HOM, LOM and RF treatments were higher than that of in CK (Fig. 2). In other words, the soil catalase activities were enhanced by application of mineral fertilizer along with manure or rice residues during the rice growth season. In the early and late rice seasons, soil catalase activities decreased as follows: HOM>LOM>RF>MF>CK. And there were no significant differences (P>0.05) in soil catalase activities under the HOM and LOM treatments at the early and late rice main growth stages. In the early and late rice seasons, the soil catalase activities of different treatments at different growth stages was in the range of 15.23–41.07 and 16.48–42.12 0.1 mol/L KMnO4 ML/(g•h), respectively. The highest activities were detected at the tillering stage.
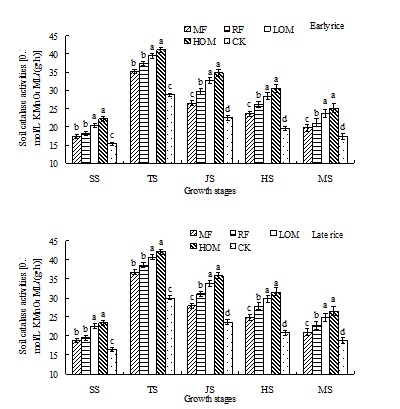
Fig. 2. Effects of different long-term fertilizer treatments on soil catalase activities during rice growth stages in paddy field
Dynamics of soil invertase activities
At the early and late rice main growth stages, soil invertase activities with different treatments was in the range of 4.34–5.39 mg/(g•h) and 4.56–5.52 mg/(g•h), respectively (Fig. 3). During the rice growth period, there was no significant difference in soil invertase activities among the HOM, LOM, RF and MF treatments, but the activity of this enzyme in HOM, LOM, RF and MF treatments was significantly (P<0.05) higher than that of in CK treatment at the different main growth stages of early and late rice.
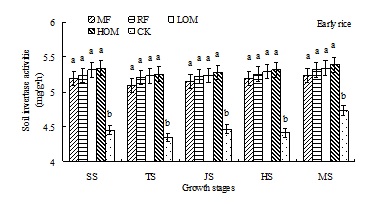
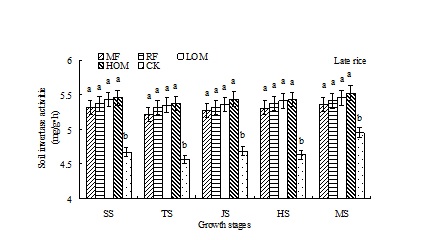
Fig. 3. Effects of different long-term fertilizer treatments on soil invertase activities during rice growth stages in paddy field
Dynamics of soil dehydrogenase activities
In the early and late rice growing seasons, soil dehydrogenase activities decreased as follows: HOM>LOM>RF>MF>CK. And there were no significant differences (P>0.05) in soil dehydrogenase activities under the HOM, LOM and RF treatments at the early and late rice main growth stages, but there were significant differences (P<0.05) among other treatments. And there were no significant differences (P>0.05) in soil dehydrogenase activities under the RF and MF treatments at the early and late rice main growth stages. In the early and late rice growing seasons, the soil dehydrogenase activities of different treatments at different growth stages was in the range of 25.46–72.64 µg/(g•h) and 26.21–73.66 µg/(g•h), respectively. The highest activities were detected at the jointing stage.
Dynamics of soil cellobiohyrolase activities
At the early rice main growth stages, the activities of soil cellobiohyrolase were affected by the different fertilizer managements. The RF treatment has the highest soil cellobiohyrolase activities and the CK had the lowest activities (Fig. 5), and the activity decreased in the following order: RF>HOM>LOM>MF>CK. At the late rice main growth stages, the highest activity was the RF treatment, and the activity trend was as follows: RF>HOM>LOM>MF>CK.
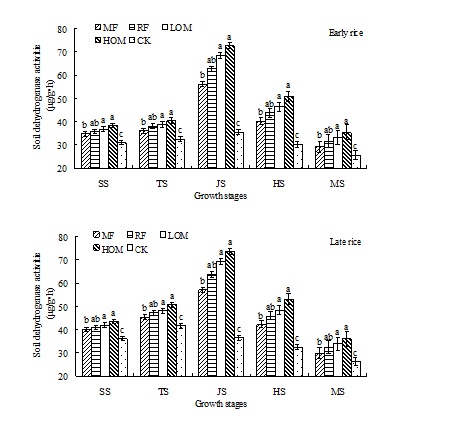
Fig. 4. Effects of different long-term fertilizer treatments on soil dehydrogenase activities during rice growth stages in paddy field
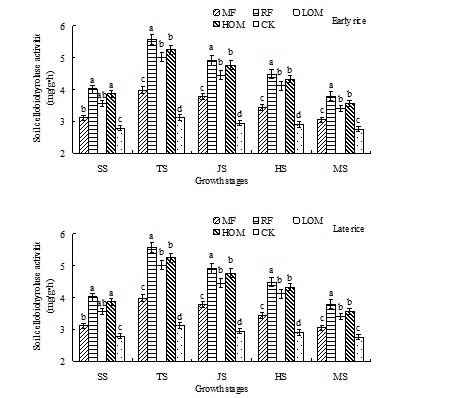
Fig. 5. Effects of different long-term fertilizer treatments on soil cellobiohyrolase activities during rice growth stages in paddy field
Dynamics of soil nitrifying and denitrification bacteria
At the early and late rice main growth stages, the numbers of soil nitrifying and denitrification bacteria decreased as follows: HOM>LOM>RF>MF>CK. There were significant differences (P<0.05) in numbers of soil nitrifying and denitrification bacteria under the HOM, LOM and RF, MF, CK treatments at the early and late rice main growth stages. But there were no significant differences (P>0.05) in numbers of soil denitrification bacteria under the HOM and LOM treatments at the early and late rice main growth stages (Table 2).
Table (2):
Effects of long−term fertilization treatments on the amount of nitrifying bacteria and denitrification bacteria in soil at early and late rice main growth stages
| Items | Treatment | Early rice | Late rice</th | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| SS | TS | JS | HS | MS | SS | TS | JS | HS | MS | ||
| Nitrifying bacteria
(×105 cfu/g) |
MF | 10.24±0.69d | 159.05±1.88d | 69.81±1.97d | 31.59±1.15d | 11.51±0.12d | 12.38±0.82d | 153.48±5.61d | 68.45±3.91d | 34.66±2.21d | 13.37±1.46d |
| RF | 12.18±0.40c | 240.39±3.31c | 120.15±1.97c | 42.93±1.62c | 25.37±1.12c | 14.55±0.57c | 231.62±4.88c | 117.73±4.03c | 43.82±1.65c | 26.42±1.15c | |
| LOM | 17.84±0.33b | 282.54±4.22b | 177.30±2.17b | 56.19±1.21b | 38.74±0.67b | 19.92±0.42b | 273.28±4.68b | 174.38±3.39b | 57.37±1.26b | 39.89±1.76b | |
| HOM | 24.59±0.27a | 307.06±4.88a | 206.82±1.98a | 75.34±0.85a | 53.15±0.22a | 28.75±0.35a | 298.47±2.43a | 204.74±2.97a | 76.62±1.01a | 50.58±1.38a | |
| CK | 8.17±0.26d | 38.41±1.11e | 28.17±0.81e | 20.52±0.59e | 8.13±0.11e | 10.31±0.29e | 29.78±1.85e | 25.81±0.74e | 17.54±0.51e | 8.92±0.25e | |
| Denitrification
bacteria (×105 cfu/g) |
MF | 22.93±2.38c | 39.63±3.79c | 30.99±3.05c | 27.83±2.16c | 22.22±1.74d | 25.47±2.45c | 42.76±3.59c | 33.46±3.12c | 29.63±2.20c | 24.37±1.80d |
| RF | 41.73±2.28b | 76.31±2.93b | 64.99±2.48b | 37.09±2.03b | 30.35±1.53c | 44.24±2.35b | 79.48±3.05b | 67.43±2.55b | 38.39±2.07b | 32.26±1.58c | |
| LOM | 79.14±1.20a | 101.62±2.20a | 85.94±1.87a | 70.51±1.07a | 53.12±0.87b | 81.73±1.27a | 105.73±2.29a | 98.45±1.94a | 71.73±1.11a | 54.94±0.93b | |
| HOM | 82.57±0.66a | 111.53±1.14a | 95.85±0.89a | 74.93±0.81a | 60.46±0.64a | 85.15±0.73a | 114.66±1.23a | 108.35±0.96a | 76.36±1.85a | 62.36±1.71a | |
| CK | 12.31±0.35d | 38.42±1.11d | 22.78±0.65d | 17.25±0.49d | 10.63±0.31e | 14.87±0.42d | 41.58±1.21d | 25.28±0.72d | 18.58±0.53d | 12.47±0.36e | |
MF:mineral fertilizer alone;RF:rice residues and mineral fertilizer;LOM:30% organic matter and 70% mineral fertilizer;HOM:60% organic matter and 40% mineral fertilizer;CK:without fertilizer.
SS:seedling stage; TS:tillering stage; JS:jointing stage; HS:heading stage; MS:mature stage.
Different letters indicate significance at P < 0.05 among the fertilization treatments at the same rice growth stage, according to the least significant difference test.
Dynamics of soil ammonifiers bacteria
At the early and late rice main growth stages, the numbers of soil ammonifiers bacteria decreased as follows: MF>RF>LOM>HOM>CK. The MF treatment has the highest numbers of soil ammonifiers bacteria and the CK had the lowest numbers of soil ammonifiers bacteria (Table 3). There were significant differences (P<0.05) in numbers of soil ammonifiers bacteria under the RF, MF and HOM, LOM, CK treatments at the early and late rice main growth stages.
Dynamics of soil cellulose-decomposing bacteria
At the early and late rice main growth stages, the numbers of soil cellulose-decomposing bacteria were affected by the different fertilizer managements. The LOM treatment has the highest numbers of soil cellulose-decomposing bacteria and the CK had the lowest numbers of soil cellulose-decomposing bacteria (Table 3), and the numbers of soil cellulose-decomposing bacteria decreased in the following order: LOM>RF>MF>HOM>CK. The numbers of soil cellulose-decomposing bacteria in LOM treatment was significantly (P<0.05) higher than that of in MF, HOM, and CK treatments at the different main growth stages of early and late rice.
Table (3):
Effects of long−term fertilization treatments on the amount of ammonifiers bacteria, azotobacteria and cellulose-decomposing bacteria in soil at early and late rice main growth stages.
| Items | Treatment | Early rice | Late rice | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| SS | TS | JS | HS | MS | SS | TS | JS | HS | MS | ||
| Ammonifiers bacteria (×106 cfu/g) | MF | 65.53±1.89a | 71.26±2.05a | 56.93±1.64a | 45.83±1.32a | 47.96±1.38a | 66.78±1.92a | 72.17±2.08a | 57.26±1.65a | 46.62±1.34a | 48.12±1.38a |
| RF | 40.36±1.16b | 42.26±1.21b | 40.26±1.16b | 39.17±1.13b | 29.83±0.86b | 41.58±1.21b | 43.54±1.25b | 41.05±1.18b | 35.04±1.01b | 37.95±1.09b | |
| LOM | 35.68±1.03c | 37.84±1.09c | 29.96±0.86c | 28.73±0.82c | 20.04±0.57c | 36.74±1.06c | 38.63±1.11c | 32.33±0.93c | 27.23±0.78c | 28.53±0.82c | |
| HOM | 12.99±0.37d | 28.86±0.83d | 15.56±0.44d | 10.88±0.31d | 13.32±0.38d | 14.31±0.41d | 29.75±0.85d | 15.94±0.46d | 12.69±0.33d | 13.25±0.38d | |
| CK | 11.25±0.32d | 19.48±0.73e | 11.53±0.33e | 8.85±0.25d | 9.93±0.28e | 12.29±0.35d | 13.36±0.38e | 11.82±0.34e | 9.67±0.27e | 10.14±0.29e | |
| Cellulose-decomposing bacteria (×103 cfu/g) | MF | 34.25±1.44c | 39.09±1.70c | 33.06±1.39c | 30.88±1.17b | 32.07±1.23b | 36.56±1.59c | 41.24±1.76c | 36.49±1.51c | 31.88±1.08b | 32.06±1.12b |
| RF | 46.11±1.33b | 51.70±1.49b | 41.58±1.20b | 39.11±1.12a | 40.55±1.17a | 48.63±1.40b | 54.41±1.57b | 45.97±1.32b | 35.11±1.01a | 36.25±1.04a | |
| LOM | 50.08±0.98a | 58.97±1.12a | 48.27±0.95a | 40.63±0.89a | 42.95±0.92a | 55.12±1.05a | 61.28±1.19a | 52.37±1.05a | 37.63±0.92a | 38.67±0.92a | |
| HOM | 29.37±0.84d | 33.28±0.96d | 28.06±0.81d | 23.14±0.66c | 25.58±0.73c | 28.68±0.82d | 32.78±0.94d | 31.06±0.89d | 28.14±0.81c | 30.74±0.89b | |
| CK | 17.42±0.51e | 21.11±0.61e | 12.74±0.36e | 10.17±0.29d | 11.06±0.31d | 16.95±0.48e | 19.46±0.56e | 15.48±0.44e | 12.12±0.34d | 13.35±0.39c | |
| Azotobacteria (×106 cfu/g) | MF | 18.54±0.72d | 25.78±1.25d | 20.06±1.13d | 17.38±0.78c | 10.93±0.33b | 19.97±0.76d | 27.62±1.31d | 22.54±1.17d | 18.42±0.82c | 12.45±0.41b |
| RF | 50.26±0.53a | 85.17±0.74a | 66.36±1.98a | 34.15±0.50a | 23.52±0.67a | 51.62±0.57a | 86.02±0.79a | 67.86±0.65a | 34.95±0.53a | 24.68±0.35a | |
| LOM | 31.75±1.45b | 55.48±2.45b | 43.71±1.91b | 32.42±0.98a | 20.85±0.31a | 33.15±1.49b | 57.35±2.48b | 46.25±1.95b | 33.26±1.01a | 21.76±0.71a | |
| HOM | 25.23±0.91c | 43.63±1.60c | 39.35±1.26c | 27.31±0.93b | 11.65±0.60b | 26.56±0.95c | 45.47±1.65c | 40.85±1.33c | 28.55±0.65b | 14.26±0.62b | |
| CK | 14.36±0.41e | 20.13±0.58e | 16.26±0.46e | 10.22±0.29d | 8.05±0.23c | 15.72±0.45e | 20.95±0.60e | 17.86±0.51e | 10.56±0.30d | 8.67±0.33c | |
Dynamics of soil azotobacteria
In the early and late rice growing seasons, the numbers of soil azotobacteria decreased as follows: RF>LOM>HOM>MF>CK. There were significant differences (P<0.05) in the numbers of soil azotobacteria under the RF, LOM and HOM, MF, CK treatments at the early and late rice main growth stages. In the early and late rice growing seasons, the numbers of soil azotobacteria of different treatments at different growth stages was in the range of 8.05–85.17 ×106 cfu/g and 8.67–86.02 ×106 cfu/g, respectively.
Soil enzyme activities and fertilizer managements
Soil enzymes are an index of soil microbial activity and fertility, and they are playing an important role in C, N, P cycling in ecological systems. Zhao et al. (2009)24 results showed that the soil invertase activity were increased by combined application of straw and manure. In our study, compared with MF and CK treatments, the soil urease and catalase activities were increased by application of mineral fertilizer along with manure or rice residues at the early and late rice main growth stages. The reason maybe application of mineral fertilizer and organic (manure or rice residues) provides energy material for soil microbial activity, conducive to the straw mineralization and nutrient release, soil microbial metabolic activity and soil enzyme activity were increased.25 Secondly, with the increase of temperature and rice growth, the plant root exudates were promoted, which also stimulated the microbial activity and enhanced the metabolic activity of microorganisms.26 In the present study, the RF treatment has the highest soil urease activities at the early and late rice main growth stages. This may be application of rice straw provides energy and nutrition source for microorganisms, so that the soil urease activity were promoted. In the low and high rate of manure combine with mineral fertilizer treatments, the soil urease activities increased with the increase of organic application, that is, the soil urease activities in HOM treatment were higher than that of the LOM treatment, this may be application of high rate of manure and mineral fertilizer is benefit to soil C/N ratio and organic decomposition.27
In our study, compared with the RF, MF and CK treatments, the soil catalase, invertase and dehydrogenase activities were increased by application of manure along with mineral fertilizer (LOM and HOM treatments) at the early and late rice main growth stages. The reason maybe application of manure were promote the growth and physiological metabolism of the underground of rice plant, increased the root exudates, and promoted the activity of soil enzyme.28 Meanwhile, application of the manure provides carbon and nitrogen source for soil enzyme activity. Secondly, soil physical and chemical properties were improved under long-term application of organic practices, which also promoted the improvement of enzyme activity.29
Soil invertase activities play a critical role in the cycling of carbon in terrestrial ecosystems, it is an indictor of the accumulation and decomposition characteristic of soil organic carbon (SOC). And it was closely related to mineralization intensity of soil organic matter.28 In the present study, soil invertase activities with application of mineral fertilizer along with manure or rice residues were higher than that of the merely mineral fertilizer inputs and without fertilizer input treatments, which suggested that application with organic manure (chicken manure or rice residues) practices had increased of soil organic matter content in a long-term experiment, and provide substrate for the soil invertase activities. Thus, the soil invertase activities were greatly increased by application of mineral fertilizer along with manure or rice residues practices.
Soil catalase is an important oxidation–reduction enzyme, which is involved in the synthesis of humic compounds and protects the cells from damage caused by hydrogen peroxide.30 SOC content was the most important factor influencing soil catalase activities. During the rice growth period, the soil catalase activities were increased by application of mineral fertilizer along with manure or rice residues compared with application of mineral fertilizer practices. That is, the soil catalase activities were inhibited under application of mineral fertilizer condition, compared with the combined application of organic and inorganic fertilizers treatments.
Soil dehydrogenase activity can be used as the index of microbial redox system, and its activity is closely related to soil fertility. Soil dehydrogenase is typically present in all intact and viable microbial cells.31 We found that the soil dehydrogenase activities were higher in RF, LOM and HOM treatments than that of in MF and CK treatments. This might because the soil physical and chemical properties and soil micro environment were improvement by combined application of manure or rice straw with miner fertilizer, which provides a good environment and carbon sources for growth and reproduction of soil dehydrogenase.32 Meanwhile, the application of organic fertilizer were contained some microorganisms, which provide source for activities of the soil dehydrogenase.
Soil microbial community and fertilizer managements
Fertilizer managements are important practices for soil microbial community, soil quality and sustainable development for agriculture production.33 Soil microbial community was control by different fertilizer managements, and it was closely related to the amount of application with manure. In the present study, the results indicated that different fertilization managements were significantly affected some physiological function soil microbial quantity, including the soil nitrifying and denitrification, ammonifiers, cellulose-decomposing and azotobacteria bacteria. Compared with the RF and CK treatments, the number of some physiological function soil microbial in the paddy field were increased by application of mineral fertilizer along with manure or rice residues at the early and late rice main growth stages. The reason may be application of mineral fertilizer and organic (manure or rice residues) increase crop growth and root biomass.26 Therefore the number of some physiological function soil microbial is raised by inputs of organic and straw residues.
Meanwhile, the communities and activities of soil physiological function bacterial (nitrifying and denitrification, ammonifiers, cellulose-decomposing and azotobacteria bacteria) were affected by the soil physicochemical properties,7,8 fertilizer regimes,9,34 type of crops and planting systems,10 and the fertilizer managements is an important factor for affect the communities of soil bacterial. In the present study, the numbers of soil bacterial were increased by application of mineral fertilizer along with manure or rice residues at the early and late rice main growth stages. The reason may be animal manure contains large amounts of soluble, readily available, organic substrates for microorganisms growth by application of mineral fertilizer and organic (manure or rice residues). Secondly, both manure and rice residues act as a substrate for microbes, and this will result in an increase in the numbers of soil bacterial. Meanwhile, with the increase of temperature during the rice growth period, the plant physiological activity and root exudates were increased, which also contribute to increase of the numbers of soil bacterial. And there have straw cellulose and hemicellulose contained in the application of straw, which provide the carbon and energy source for growth and reproduction of the microbial, therefore, the microorganisms were promoted during the rice growth period. Similarly, in a long-term experiment, some studies results showed that the soil microbial were greater in application of manure (farmyard manure, straw) along with NPK fertilizer than that of the merely NPK fertilizer inputs in field conditions.35
The numbers of soil nitrifying and denitrification bacteria are related with soil redox potential (Eh), soil organic matter and pH. The numbers of soil nitrifying bacteria were higher in application of manure (manure or rice residues) along with mineral fertilizer than that of the without fertilizer input treatment. The reason maybe it did not inhibit nitrification by application of manure. Meanwhile, the numbers of soil denitrification bacteria were increased by application of mineral fertilizer along with manure or rice residues at the early and late rice main growth stages, which suggested that application with organic manure (chicken manure) practices had decreased of soil Eh and pH in a long-term experiment. This results was coincides with application of organic manure boosted bacterial growth and increased the number of bacterial in agroecosystems.36
The results showed that the activities of soil enzyme were affected by the different long-term fertilizer managements at the rice main growth stages. Compared with the MF and CK treatments, the soil enzyme activities were increased by application of mineral fertilizer along with manure or rice residues at the early and late rice main growth stages. That is, application of manure or rice residues can improve the soil urease, catalase, invertase, dehydrogenase and cellobiohyrolase activities at the rice main growth stages. Compared with CK treatment, the soil urease activities with RF treatment were increased by 20.92, 20.45, 25.95, 21.86, 8.44 mg/(g•h) and 20.90, 19.63, 25.75, 21.76, 8.36 mg/(g•h) at the early and late rice main growth stages, respectively. And the soil catalase activities with HOM treatment were increased by 6.65, 12.31, 12.34, 10.97, 7.66 mol/L KMnO4 ML/(g•h) and 6.94, 12.11, 12.12, 10.67, 7.76 mol/L KMnO4 ML/(g•h) at the early and late rice main growth stages, respectively. But there was no significant difference in soil invertase activities among the HOM, LOM, RF and MF treatments. The soil dehydrogenase activities with HOM treatment were increased by 7.26, 7.95, 37.19, 20.49, 9.75 µg/(g•h) and 7.40, 8.92, 37.27, 20.53, 9.80 µg/(g•h) at the early and late rice main growth stages, respectively. And the soil cellobiohyrolase activities with RF treatment were increased by 1.23, 2.45, 1.96, 1.58, 1.03 mg/(g•h) and 1.24, 2.44, 2.06, 1.60, 1.02 mg/(g•h) at the early and late rice main growth stages, respectively.
The number of some physiological function soil microbial in the paddy field also increased by application of mineral fertilizer along with manure or rice residues at the rice main growth stages. At the early and late rice main growth stages, the numbers of soil nitrifying and denitrification bacteria higher in HOM treatment, and the numbers of soil ammonifiers bacteria higher in MF treatment, the numbers of soil cellulose-decomposing bacteria higher in LOM treatment, the numbers of soil azotobacteria bacteria higher in RF treatment, respectively. In conclusion, application of mineral fertilizer along with manure or rice residues is an effective way of enhancing soil enzyme activities and soil microbial community.
ACKNOWLEDGMENTS
This study was supported by the National Natural Science Foundation of China (No. 31571591), and the Public Research Funds Projects of Agriculture, Ministry of Agriculture of the P.R. China (No. 201503123).
- Benitez, E., Melgar, R., Sainz, H., Gomez, M., Nogales, R. Enzyme activities in the rhizosphere of pepper (Capsicumannuum L.) grown with olive cake mulches. Soil. Biol. Biochem. 2000; 32: 1829–1835.
- Dick, R.P. Soil Enzyme Activities as Indicators of Soil Quality. In: Doran JW, Coleman DC, Bezdicek DF, Stewart BA (Eds.). Defining soil quality for a sustainable environment. American Society of Agronomy, Madison, WI, 1994; 107–124.
- Tabataba,i M.A. Soil Enzymes. In: Page AL, Miller RH, Keeney DR. (Eds.). Methods of soil analysis. American Society of Agronomy, Madison, 1994; 775–833.
- Zul, D., Denzel, S., Kotz, A., Overmann, J. Effects of plant biomass plant diversity, and water content on bacterial communities in soil lysimeters: implications for the determinants of bacterial diversity. Appl. Environ. Microbiol. 2007; 73: 6916–6929.
- Coleman, D.C., Whitman, W.B. Linking species richness, biodiversity and ecosystem function in soil systems. Pedobiologia 2005; 49: 479–497.
- Dunbar, J., Barns, S.M., Ticknor, L.O., Kuske, C.R. Empirical and theoretical bacterial diversity in four Arizona soils. Appl. Environ. Microbiol. 2002; 68: 3035– 3045.
- Lauber, C.L., Hamady, M., Knight, R., Fierer, N. Pyrosequencing-based assessment of soil pH as a predictor of soil bacterial community structure at the continental scale. Appl. Environ. Microbiol. 2009;75: 5111–5120.
- Drenovsky, R.E., Vo, D., Graham, K.J., Scow, K.M. Soil water content and organic carbon availability are major determinants of soil microbial community composition. Microb. Ecol. 2004; 48: 424–430.
- Carson, J.K., Campbell, L., Rooney, D., Clipson,N., Gleeson, D.B. Minerals in soil select distinct bacterial communities in their microhabitats. FEMS Microbiol. Ecol. 2009; 6: 381–388.
- Tang, H.M., Xiao, X.P., Tang, W.G., Lin, Y.C., Wang, K., Yang, G.L. Effects of winter cover crops residue returning on soil enzyme activities and soil microbial community in double-cropping rice fields. PLoS ONE 2014; 9: e100443.
- Hamido, S.A., Kpomblekou, A.K. Cover crop and tillage effects on soil enzyme activities following tomato. Soil Till. Res. 2009; 105: 269–274.
- Ekenler, M., Tabatabai, M.A. Tillage and residue management effects on b–glucosaminidase activity in soils. Soil. Biol. Biochem. 2003; 35: 871–874.
- Carmine, C., Magda, C., Maria, D.R.P., Patrizia, R., Pacifico, R. Effects of municipal solid waste compost amendments on soil enzyme activities and bacterial genetic diversity. Soil. Biol. Biochem. 2004; 36: 1595–1605.
- Tejada, M., Garcia, C., Gonzalez, J.L., Hernandez, M.T. Use of organic amendment as a strategy for saline soil remediation: Influence on the physical, chemical and biological properties of soil. Soil. Biol. Biochem. 2006; 38: 1413–1421.
- Lal, R. Soil carbon sequestration in China through agricultural intensification, and restoration of degraded and desertified ecosystems. Land Degrad Dev 2002; 13: 469–478.
- Li, Q.K. Paddy soil of China. Science Press, Beijing, China. 1992.
- Xu, M.G. The evolvement of soil fertility in China. China Agricultural Science and Technology press, Beijing, China. 2006.
- Kandeler, E., Mosier, A.R., Morgan, J.A., Milchunas, D.G., King, J.Y., Sabine, R., Tscherko, D. Response of soil microbial biomass and enzyme activities to the transient elevation of carbon dioxide in a semi-arid grassland. Soil. Biol. Biochem. 2006; 38: 2448–2460.
- Roberge, M.R. Methodology of enzymes determination and extraction, in Soil Enzymes (Ed: Burns RG.), Academic Press, New York, 1978; 341–373.
- Guan, S.Y. Soil Enzyme and its Approach. Beijing: Agriculture Press. 1986.
- Xu, G.H., Zheng, H.Y. 2006. Handbook of soil microbiological analysis methods. China Agricultural press, Beijing, China.
- Ulrich, A., Wirth, S. Phylogenetic diversity and population densities of culturable cellulolytic soil bacteria across an agricultural encatchment. Microb. Ecol. 1999; 37: 238–247.
- SAS Institute. SAS Version 9.1.2 2002–2003. SAS Institute Inc., Cary, NC. 2003.
- Zhao, Y.C., Wang, P., Li, J.L., Chen, Y.R., Ying, X.Z., Liu, S.Y. The effects of two organic manures on soil properties and crop yields on a temperate calcareous soil under a wheat-maize cropping system. Eur. J. Agron. 2009; 31: 36–42.
- Liu, E.K., Yan, C.R., Mei, X.R., He, W.Q., Bing, S.H. Long-term effect of chemical fertilizer, straw and manure on soil chemical and biological properties in northwest China. Geioderma 2010; 158: 173–180.
- Kundu, D.K., Ladha, J.K. Efficient management of soil and biologically-fixed N2 in intensively cultivated rice fields. Soil. Biol. Biochem. 1995; 27: 431–439.
- Anthony, M.W., Graeme, J.B., Lefroy, R.D.B. Managing legume leys, residues and fertilizers to enhance the sustainability of wheat yields and nutrient balance 2. Soil physical fertility and carbon. Soil Till. Res. 2000; 54: 77–89.
- Zhang, B.C., Zhou, X.B., Zhang, Y.M. Responses of microbial activities and soil physical-chemical properties to the successional process of biological soil crusts in the Gurbantunggut Desert, Xinjiang. J. Arid Environ. 2015; 7: 101–109.
- Katsalirou, E., Deng, S.P., Nofziger, D.L., Gerakis, A. Long-term management effects on organic C and N pools and activities of C–transforming enzymes in prairie soils. Eur. J. Soil. Biol. 2010; 46: 335–341.
- Guwy, A.J., Martin, S.R., Hawkes, F.R., Hawkes, D.L. Catalase activity measurements in suspended aerobic biomass and soil samples. Enzyme Microb. Technol. 1999; 25: 669–676.
- Singh, D.K., Kumar, S. Nitrate reductase, arginine deaminase, urease and dehydrogenase activities in natural soil (ridges with forest) and in cotton soil after cetamiprid treatments. Chemosphere 2008; 71: 412–418.
- Powlson, D.S., Brookes, P.C., Christensen, B.T. Measurement of soil microbial biomass provides an early indication of changes in total organic matter due to straw incorporation. Soil. Biol. Biochem. 1987; 19: 159–164.
- Plaza, C., Hernandez, D., Garcia-Gil, J.C., Polo, A. Microbial activity in pig slurry-amended soils under semiarid conditions. Soil. Biol. Biochem. 2004; 36: 1577–1585.
- Kautz, T., Wirth, S., Ellmer, F. Microbial activity in a sandy arable soil is governed by the fertilization regime. Eur. J. Soil. Biol. 2004; 40: 87–94.
- Hopkins, D.W., Shiel, R.S. Size and activity of soil microbial communities in long-term experimental grassland plots treated with manure and inorganic fertilizers. Biol. Fertil. Soils 1996; 22: 66–70.
- Marschner, P., Kandeler, E., Marschner, B. Structure and function of the soil microbial community in a long-term fertilizer experiment. Soil. Biol. Biochem. 2003; 35: 453–461.
© The Author(s) 2017. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


