ISSN: 0973-7510
E-ISSN: 2581-690X
The current study was focused on patients with Multiple sclerosis between the ages group of 20 – 60. The objective of this study is to decide the level of 25-hydroxyvitamin D3 status in patients with multiple sclerosis (MS) and its engagement, with disease activity. A total of 31 cases have participated from Baghdad Teaching Hospital in Baghdad, Iraq during the period between March and April 2018. The results shows that, only significant finding was that, supplements for Vitamin D significantly increased its mean level in patients with Multiple Sclerosis (P < 0.05).
Multiple sclerosis, Vitamin-D.
Multiple sclerosis is an inflammatory, demyelinating disease of the CNS and it’s characterized by myelin loss, inflammatory lesions, and varying degrees of axonal pathology1. Multiple sclerosis (MS) is a common neurological disease, and its etiology remains unknown1. However, despite the unknown etiology, there is an increasing evidence for the possible association between MS and vitamin D deficiency in recent years2. This proves correlates the role from Vitamin-D in the expansion of MS1. Vitamin D is a fat-soluble vitamin; its 2 principle shapes are ergocalciferol (vitamin D2), which is of plant source, and cholecalciferol (vitamin D3), which is of animal origin2. Vitamin D2 is considered less bioactive than vitamin D32. Total-body sun exposure easily supply the equivalent of 250 mg (10000 IU) vitamin D/d2. It is known that MS patients have an increased incidence of vitamin D deficiency 2. Vitamin D deficiency and increased disease activity have been related to established relapsing-remitting multiple sclerosis (RRMS).3 It is currently established that MS is a T helper type 1 (Th1) and Th17- driven immune-mediated disease4. This was illustrated by immune cell infiltration and accompanying inflammatory processes leading to damage of myelin4. Treatment of dendritic cells (DC) with 1, 25(OH)2 D3 renders these cells in a semi-mature state, as evidenced by low expression levels of costimulatory molecules, such as CD40, CD80, and CD86, increased IL-10 production, and impaired IL-12 secretion4. Consequently, 1, 25(OH)2D3- treated DC display a reduced capacity to activate T cells 4. Immune cells are hence can to synthesize and excrete Vitamin-D in together autocrine and paracrine methods, exhibiting that Vitamin-D has a serious role in the immune system, where it impacts antigen presentation, innate immunity, and T-cell activation, differentiation, and migration5. In vitro, the increment of Vitamin-D to antigen-presenting cells inhibited the roof expression from antigens by major histocompatibility complex (MHC) grade II and its costimulatory molecules, leading to reduced T-cell stimulatory capacity6. Moreover, 1, 25(OH)2D3 prohibit T cell and B cell reproduction and blocks B cell differentiation and immunoglobulin secretion2. All from these immunomodulatory wares of 1, 25(OH), 2D3 can be leading to the protection of objective tissue in autoimmune diseases2.
A total of 31 cases have participated from Baghdad Teaching Hospital in Baghdad, Iraq during the period between March and April 2018. Before conducting the study an ethical approval was taken from the hospital. The cases were taken from consultant ward. All subjects gave written consent after they were informed about the nature of the study. And then patients were given a questionnaire paper about the disease state. The patients who had been selected randomly had given their information regarding their age, sun exposure, diagnosis time, relapses, mood disturbances, previous treatment history and whether patients experienced previous vitamin D therapy, adherence to treatment, signs, and symptoms of the disease, issues with walking, fatigue, cognition, and memory, tremors and finally their quality of life score. From the patients documented information at the ward, an EDSS score had been taken with their brain MRI reports and medication history After taking the information from questionnaires and patients documented files, blood samples were taken from patients and then sent to the lab in which serum was separated from the blood by centrifuge and then were stored at -80°C.
Measurements
The 25-hydroxy vitamin D3 conce-ntration was measured from serum samples using enzyme-linked immunofluorescence (ELFA) assay on the Mini Vidas device.
Study Population
Demographic information about the patients participated in the study are shown in (Table 1).
information about the patients participated in the study are shown in (table 1).
Table (1):
Baseline clinical and demographic characteristics of the study group.
No. |
Demographic characteristics |
comments |
|---|---|---|
1 |
Age |
A range between ( 20 – 60 ) years. |
2 |
sex |
12 Males, 17 Females. |
3 |
EDSS Score |
A range between ( 0 – 10) |
4 |
Quality of life score |
A range between ( 0 – 10 ) |
5 |
Brain MRI reports |
|
6 |
Previous and current therapy used |
Natalizumab, Fingolimod, and Interferon beta 1b. |
7 |
Living condition |
City or Rural culture. |
Statistical analysis
Data were first input into an excel file then transformed for analysis into a statistical package for social sciences file version 24 (SPSS v24). Continuous variables presented as means with their standard deviations and discrete variables presented as numbers with their percentages. T-test for two independent samples and Mann-Whitney test were used as appropriate to test the significance of the difference of means between two independent samples. Chi-square test for independence and Fisher’s exact test were used as appropriate to test the significance of the association between discrete variables. Pearson’s correlation test used to test the significance of the correlation between continuous variables. Level of significance was set at P value equals or less than 0.05.
Demographic Data and Patients Information
A total of 31 subjects were assessed and blood samples were drawn and sent for analysis. During the blood sample taking process, two samples’ serum were hemolyzed after blood gel tubes were separated by centrifuge, indicating an error had happened during blood sample taking process from those two patients and hence they are excluded from the study. Leaving a total number of 29 samples to be included. Demographic and clinical characteristics of patients are summarized in Table 2. The only significant finding was that supplements for Vitamin D significantly increased its mean level in patients with Multiple Sclerosis (P<0.05) as shown in Table 2. Otherwise other data and patient’s information were not significantly different.
Table (2):
Characteristics of sampled patients:
| Variables | Total | On vitamin D treatment | P
value |
|||||
|---|---|---|---|---|---|---|---|---|
| Yes | No | |||||||
| N=29 | 100% | N=7 | 100% | N=22 | 100% | |||
| Age (years) | Mean±SD | 33.4±9.9 | 33.6±8.2 | 33.4±10.6 | 0.963 | |||
| Min-Max | 20-60 | 25-48 | 20-60 | — | ||||
| Age Group | 0.841* | |||||||
| 20-29 | 13 | 44.8% | 3 | 42.9% | 10 | 45.5% | ||
| 30-39 | 10 | 34.5% | 3 | 42.9% | 7 | 31.8% | ||
| 40-49 | 4 | 13.8% | 1 | 14.3% | 3 | 13.6% | ||
| 50-60 | 2 | 6.9% | 0 | 0.0% | 2 | 9.1% | ||
| Disease Modifying Therapy Group | — | |||||||
| Fingolimod | 10 | 34.5% | 2 | 28.6% | 8 | 36.4% | ||
| Beta Interferon 1b | 13 | 44.8% | 4 | 57.1% | 9 | 40.9% | ||
| Natalizumab | 6 | 20.7% | 1 | 14.3% | 5 | 22.7% | ||
| Female sex | 17 | 58.6% | 5 | 71.4% | 12 | 54.5% | 0.430 | |
| Disease duration (months) | Mean±SD | 49.4±29.7 | 45.4±25.8 | 50.7±31.3 | 0.689 | |||
| Min-Max | 6-120 | 6-72 | 6-120 | |||||
| No. of disease exacerbations last year | Mean±SD | 2.9±5.3 | 1.1±1.2 | 3.4±6.0 | 0.616 | |||
| Min-Max | 0.0-20.0 | 0.0-3.0 | 0.0-20.0 | |||||
| Regular sun exposure | 13 | 44.8% | 2 | 28.6% | 11 | 50.0% | 0.321 | |
| Exacerbation during treatment | 10 | 34.5% | 2 | 28.6% | 8 | 36.4% | 0.706 | |
| Depression/Mood Disorder | 14 | 48.3% | 2 | 28.6% | 12 | 54.5% | 0.231 | |
| Preexisting seizure | 2 | 6.9% | 1 | 14.3% | 1 | 4.5% | 0.431 | |
| Variables | Total | On vitamin D treatment | P
value |
|||||
| Yes | No | |||||||
| N=29 | 100% | N=7 | 100% | N=22 | 100% | |||
| Compliance with treatment | 26 | 89.7% | 6 | 85.7% | 20 | 90.9% | 1.000 | |
| Disability is worsening during treatment | 8 | 27.6% | 2 | 28.6% | 6 | 27.3% | 0. 947 | |
| Walking issues | 15 | 51.7% | 4 | 57.1% | 11 | 50.0% | 0.742 | |
| Use of walking assistance device | 4 | 13.8% | 0 | 0.0% | 4 | 18.2% | 0.546 | |
| Fatigue | 21 | 72.4% | 5 | 71.4% | 16 | 72.7% | 0.947 | |
| Bladder infection | 5 | 17.2% | 0 | 0.0% | 5 | 22.7% | 0.166 | |
| Constipation | 10 | 34.5% | 2 | 28.6% | 8 | 36.4% | 0.706 | |
| Memory issue | 13 | 44.8% | 1 | 14.3% | 12 | 54.5% | 0.62 | |
| Complaints of pain | 13 | 44.8% | 4 | 57.1% | 9 | 40.9% | 0.452 | |
| Tremor | 7 | 24.1% | 2 | 28.6% | 5 | 22.7% | 1.000 | |
| Quality of life (score) | Mean±SD | 6.0±2.5 | 7.0±1.7 | 5.7±2.7 | 0.187 | |||
| Min-Max | 0-10 | 5-10 | 0-10 | |||||
| EDSS (score) | Mean±SD | 1.7±1.0 | 1.3±0.3 | 1.9±1.1 | 0.068 | |||
| Min-Max | 0.0-4.5 | 1.0-1.5 | 0.0-4.5 | |||||
| EDSS category | 0.354* | |||||||
| < 1.5 | 7 | 24.1% | 3 | 42.9% | 4 | 18.2% | ||
| 1.5-2.49 | 16 | 55.2% | 4 | 57.1% | 12 | 54.5% | ||
| 2.5-3.49 | 4 | 13.8% | 0 | 0.0% | 4 | 18.2% | ||
| 3.5-4.5 | 2 | 6.9% | 0 | 0.0% | 2 | 9.1% | ||
| 25-Hydroxyvitamin D3 level(ng/ml) | Mean±SD | 19.7±12.6 | 32.7±17.6 | 15.5±7.1 | 0.011 | |||
| Min-Max | 6.0-65.1 | 18.7-65.1 | 6.0-26.9 | |||||
*Could be invalid due to expected cell count is less than one; EDSS: Expanded Disability Status Scale; SD: Standard Deviation; Min: Minimal Value; Max; Maximal Value; N= Number of samples
As shown in table 2 above, 17 patients (58.6%) of the study group were women. Five of them (71.4%) were on vitamin D therapy group, and 12 of them (54.5%) were not taking any vitamin D therapy. Mean age of patients at inclusion was 33.4 years (±9.9), of which 13 patients (44.8%) were between 20 to 29 years old; 10 patients (34.5%) were between 30 to 39 years old; 4 patients (13.8%) were between 40 to 49 years and 2 of them (6.9%) where between 50 to 60 years old. Mean EDSS score was 1.7(±1.0) of which 1.3(±0.3) was from vitamin D therapy group and 1.9(±1.1) of the non-therapy group. And mean disease duration was 49.4 (±29.7) months in which the means of vitamin D and non-vitamin D therapy groups were 45.4(±25.8) months and 50.7(±31.3) months, respectively.
The mean for the number of disease exacerbations during the last year prior of this study with a range between 0 to 20 exacerbations like walking difficulties, blurred vision, muscle spasms, peripheral neuropathies and hearing problem was 2.9 (±5.3), 1.1(±1.2) mean score was from therapy with vitamin D group while mean from non-therapy group was 3.4(±6.0). Further more, the quality of life score mean for the study was 6.0 (±2.5), with mean values between the therapy and the non- therapy groups were 7.0 (±1.7) and 5.7 (±2.7), respectively. The mean 25-hydroxy vitamin D3 level was 19.7 (±12.6) ng/ml in the study group, from which the mean level of vitamin D therapy group was 32.7 (±17.6) ng/ml while for the non-therapy group was 15.5 (±7.1) ng/ml, as shown in Fig. 1.
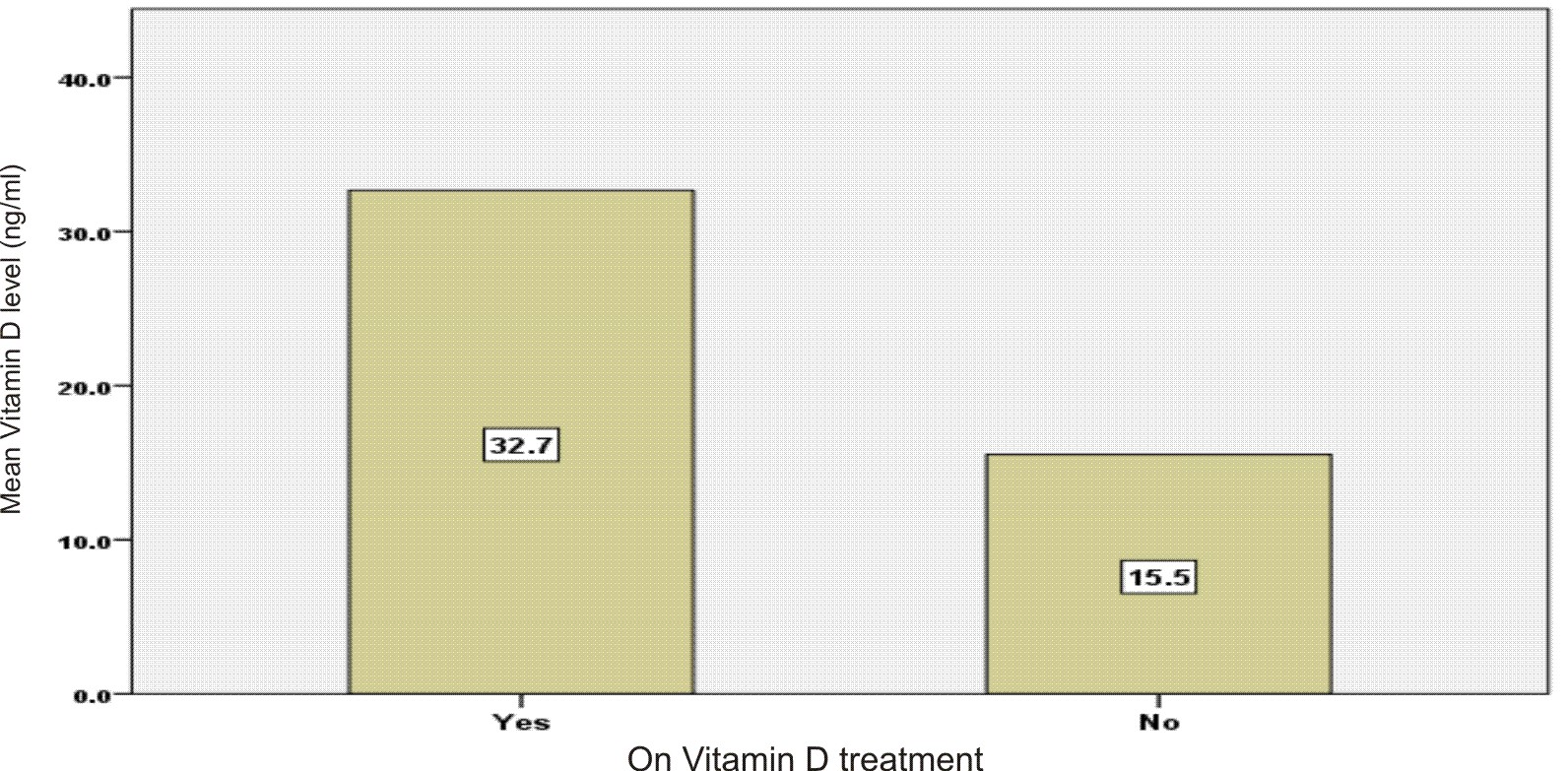
Fig. 1. Mean 25-hydroxyvitamin D3 level in patients with multiple sclerosis according to presence or absence of vitamin D treatment (supplements).
Association between plasma 25-hydroxy vitamin D3 concentrations, demographic and studied variable characteristics in the MS Study
There were no significant correlations between studied variables and 25-hydroxy vitamin D3 level in MS patients (P > 0.05), as shown in table 3.
Table (3):
Correlations of the studied variable with the level of 25-hydroxyvitamin D3:
Variables |
Pearson Correlation |
P value |
|---|---|---|
Age (years) |
-0.052 |
0.789 |
Duration of Disease (months) |
0.023 |
0.904 |
Number of exacerbations last year |
-0.219 |
0.254 |
Quality of life score |
0.262 |
0.169 |
EDSS score |
-0.242 |
0.206 |
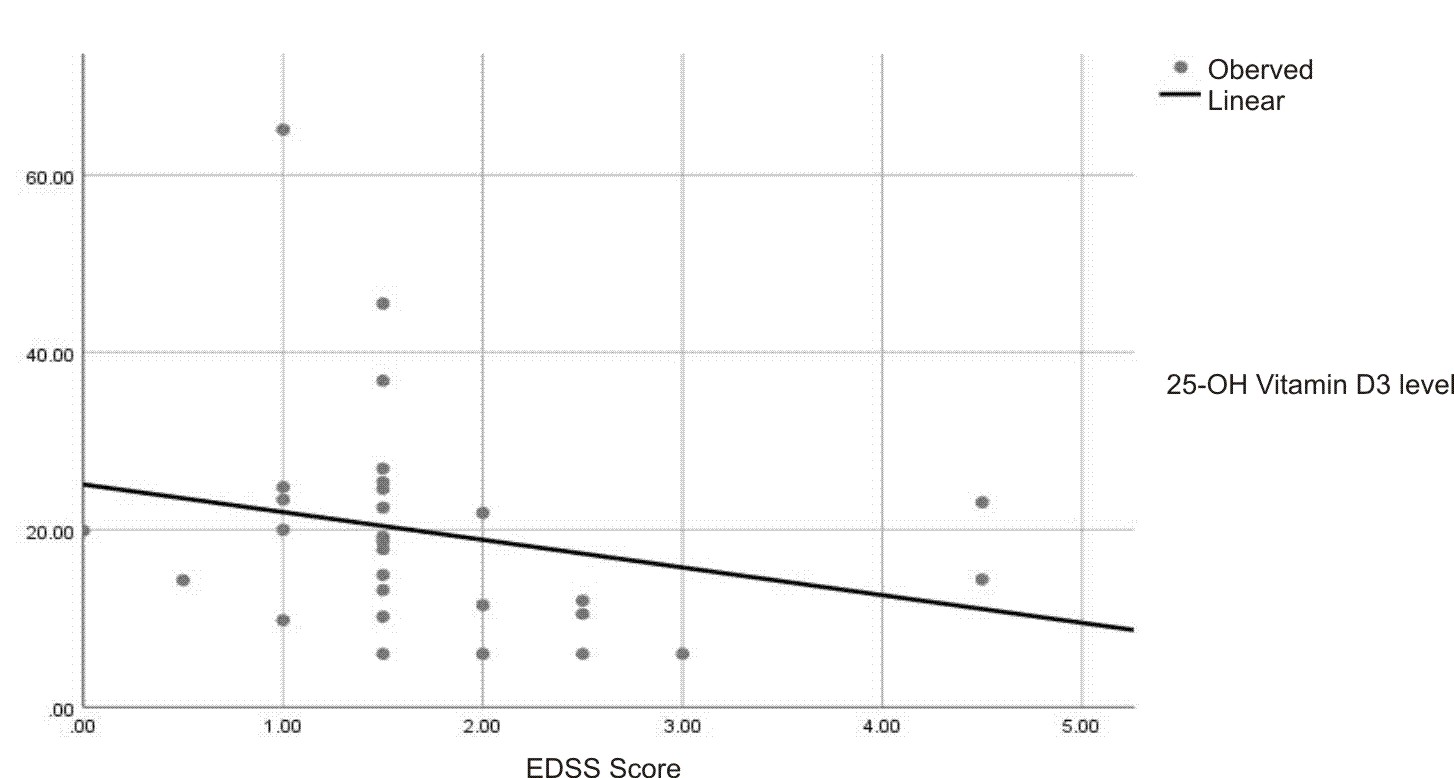
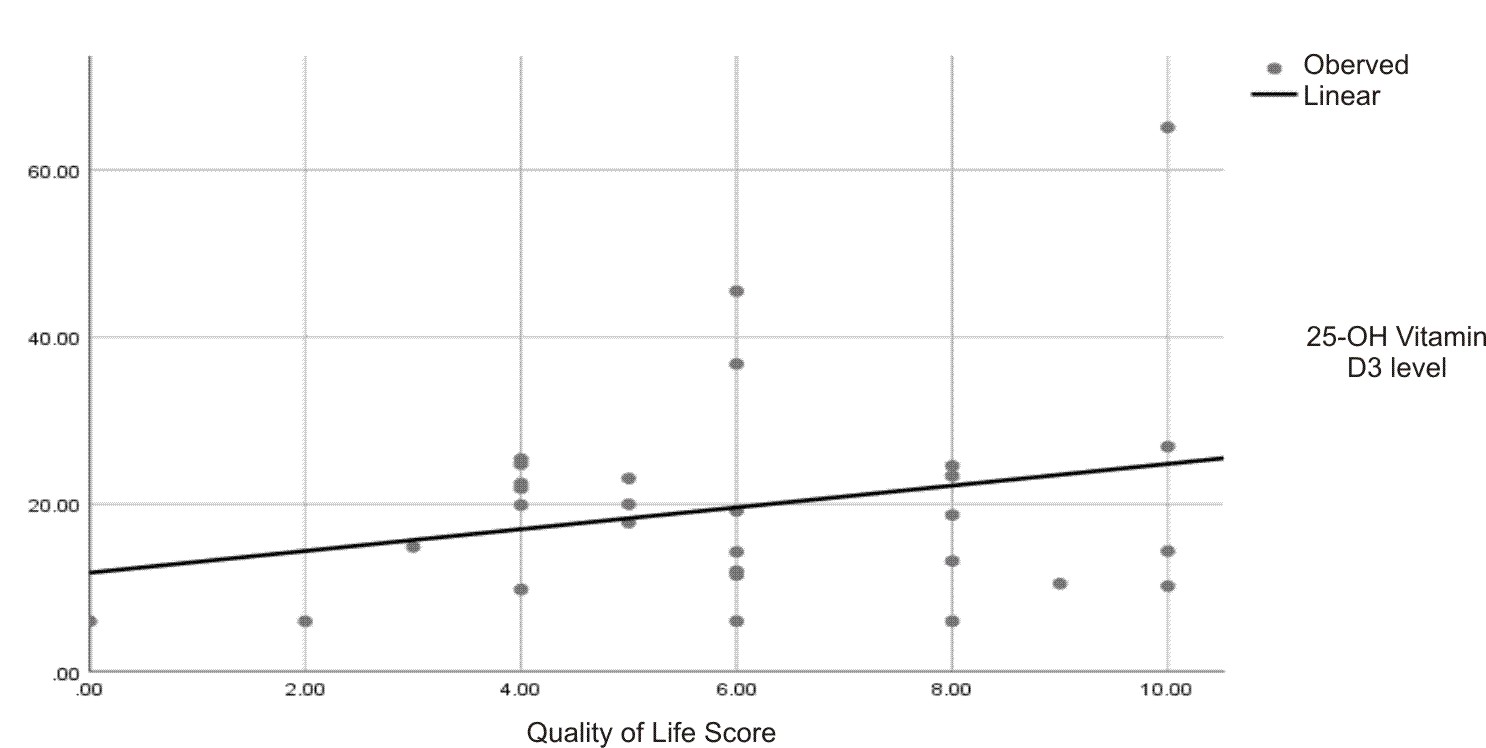
Despite the non-significant correlation results between 25-hydroxy vitamin D3 level and both EDSS score and fineness from life score, there is some enhancement in these scores as the level of 25-hydroxy vitamin D3 was higher and vice versa, as seen in figures 2 and 3.
The study fundamental is to measure serum 25-hydroxy vitamin D3 concentration in Multiple Sclerosis patients and to evaluate the association between serum 25-hydroxy vitamin D3 concentration and disease activity in Iraqi patients with MS. The only statistically significant finding is that patients taking vitamin D supplements significantly improved their serum 25-hydroxy vitamin D3 level above their normal lower level limit (>30 ng/ml) with a mean of 32.7 (±17.6) ng/ml. While for the majority of the patients who didn’t have taken any form of vitamin D therapy, their serum 25-hydroxy vitamin D3 level was obviously far below their normal lower level limit with a mean 15.5 (±7.1) ng/ml. This was confirmed by many studies like the Shaygannejad et al. study in 2010 in Iran in which lower serum 25-hydroxy vitamin D3 level was found in MS patients in comparison to the normal population, despite sufficient sun exposure in Isfahan region7. Also the Portuguese study by Bettencourt et al. in 2017 which showed significantly lower serum 25-hydroxy vitamin D3 from that of healthy controls8. Female patient’s ratio was found higher in the study group than male patients. A study by Smolders et al. in 2007 also revealed higher levels in female patients with MS than in male patients. This is perhaps caused by lower percentage chance for sun exposure for females (with insufficient ultraviolet exposure) than males or suggesting that vitamin D requirements may differ between the sexes, as well as by underlying disease state and reflect as one of the environmental factors involved in MS pathogenesis of the sex difference9 . The other compared variable with vitamin D is the Expanded Disability Status Scale (EDSS), from the results of the study it’s noticeably shown that patients with higher serum 25-hydroxy vitamin D3 level whom they were on previous vitamin D therapy have a lower EDSS score than that of those on lower serum 25-hydroxy vitamin D3 level despite the little difference between the mean number of the 2 groups and the non- significant correlation between 25-hydroxy vitamin D3 level and mean EDSS score owing to the fact that limitation of this study population was too small to permit a definitive assessment of the clinical outcome measures. Methodologically, it is not unable to assess the causality agent amidst the two objects; MS vitality and vitamin D3 deficiency, except if a longitudinal study could be performed with serial mensuration of vitamin D3 in MS patients. As the study is planned as cross-sectional research, so it is not logically possible to pretend whether MS cases were prone to vitamin D3 insufficiency or the patients with low serum level of this vitamin are vulnerable to forward to a severe form of MS. In another hand, it is not able to interpret a correlation or a relationship among MS and depress concentrations from vitamin D3 with such cross-sectional study because of limitations, including its cross-sectional designing, small sample size and shortened study period. Additionally, there are only a few studies to compare vitamin D3 level between MS patients with regard to malady severity.
The mean number of the disease symptoms exacerbations during the last year before the study was lower in the higher 25-hydroxy vitamin D3 level group than that of the lower 25-hydroxy vitamin D3 group (1.1±1.2 vs 3.4±6.0) respectively. And this was confirmed by some studies like the study by Runiaet al. in 2012 which referenced that higher vitamin D levels are related with decreased exacerbation danger in relapsing-remitting MS suggests a beneficial impact of vitamin D on disease course in MS. However, the likelihood of reverse causality can’t be ruled out completely. Randomized intervention studies are therefore needed to investigate the effect from vitamin D supplementation in MS10. Nonetheless, despite these promising results from the later study, the result of this pilot study showed no significant correlation between 25(OH)D3 and disease exacerbations. The same criteria apply for the mean Quality of life score between the 2 groups that it also yields no significant correlation even with the fact the mean was better with the high 25(OH)D3 group.
Vitamin D is an immune modulator; however, its clinical effect in MS remains unclear. This study demonstrated that MS patients have lower plasma 25-hydroxy vitamin D3 conce-ntrations. Plasma 25(OH)D concentrations in patients taking vitamin D therapy were significantly higher than those who were not taking any vitamin D supplements. Although this was the only significant value and other
variables like EDSS and disease exacerbations were not significantly correlated in this pilot study, these observations could provide support for further research in this direction to confirm the potential benefit of vitamin D supplements for early treatment of MS patients with longitudinal follow up studies.
None
The authors declare that they have no conflict of interest in the publication.
- Eva Koטovska., Fiona Gaughran., Amir Krivoy., and Ute-Christiane Meier. Vitamin-D Deficiency as a Potential Environmental Risk Factor in Multiple Sclerosis, Schiz-ophrenia, and Autism. J. Front Psychiatry., 2017; 8: 47.
- Fatimah M. Alharbi. Update in vitamin D and multiple sclerosis, Review Article from Neurosciences Journal, 2015; 20(4): 329-335.
- Dalia L. Rotstein, Brian C. Healy, and Muhammad T. Malik. Effect of vitamin D on MS activity by disease-modifying therapy class, Neurol Neuroimmunol Neuro-inflamm, 2015; 2(6): e167.
- Wai-Ping Lee, Barbara Willekens, and Patrick Cras.Immunomodulatory Effects of 1, 25-dihydroxy vitamin D3 on Dendritic Cells Promote Induction of T Cell Hypo-responsiveness to Myelin-Derived Antigens. Journal of Immunology Research, 2016.
- Chun RF, Liu PT, Modlin RL, Adams JS, Hewison M. Impact of vitamin D on immune function: lessons learned from the genome-wide analysis. Front Physio., 2014; 5:151.
- Bartels LE, Hvas CL, Agnholt J, Dahlerup JF, Agger R. Human dendritic cell antigen presentation and chemotaxisare inhibited by intrinsic 25-hydroxy vitamin D activation. Int Immunopharmacol., 2010; 10(8):922-8.
- Shaygannejad V, Golabchi KH, Haghighi S, Dehghan H, Moshayedi A. A comparative study of 25 (OH) vitamin D serum levels in patients with multiple sclerosis and control group in Isfahan, Iran. Int. J. Prev. Med., 2010; 1:195–201.
- Bettencourt A1, Boleixa D., Reguengo H., Samץes R., Santos E., Oliveira JC., Silva B., Costa PP. and da Silva AM. Serum 25-hydroxy vitamin D levels in multiple sclerosis patients from the north of Portugal. J. Steroid Biochem. Mol. Biol., 2017; 22: S0960-0760(17)30264-9.
- Barnes MS, Bonham MP, Robson PJ, Strain JJ, Lowe-Strong AS, Easton-Evans J, et al. Assessment of 25-hydroxy vitamin D and 1, 25-dihydroxy vitamin D3 concentrations in male and female multiple sclerosis patients and control volunteers. Mult Scler., 2007; 13:670–2.
- Runia TF, Hop WC, de Rijke YB, Buljevac D, Hintzen RQ. Lower serum vitamin D levels are associated with a higher relapse risk in multiple sclerosis. American Academy of Neurology, 2012 17; 79(3):261-6.
© The Author(s) 2019. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.