ISSN: 0973-7510
E-ISSN: 2581-690X
The resistance to MLSB antibiotics, i.e. Macrolide-Lincosamide-Streptogramin B (MLSB), is an increasing problem among Methicillin-resistant Staphylococci. The resistance to macrolides can be by efflux mechanism or via inducible or constitutive resistance. Unfortunately, routine clindamycin susceptibility testing fails to detect the inducible resistance, which commonly results in treatment failure and necessitates incorporating a simple D-test to detect such resistance. A retrospective observational study was performed on S. aureus isolates from patients. The strains were subjected to antibiotic susceptibility testing followed by detection of mecA gene by a polymerase chain reaction and, the ‘D-test’ was performed to know the inducible resistance to clindamycin. A total of 235 isolates were identified as S. aureus. Antibiotic susceptibility test indicated 190 MRSA and 45 are sensitive to MLSB (MS). Inducible clindamycin resistance was found among 48 (20.4%) isolates and constitutive resistance in 104 (44.2%). MRSA strains had higher inducible and constitutive resistance than MSSA strains (22.1%, 51.6% and 13.3%, 13.3%, respectively). Clindamycin is a commonly used antibiotic in patients with MRSA infections to spare higher-end anti-MRSA antibiotics like linezolid and vancomycin. To detect inducible clindamycin to avoid treatment failures; the study showed the importance of incorporating the D-test in routine testing.
Antibiotic, Clindamycin, MRSA, Macrolide, Methicillin-resistant, Staphylococcus, rRNA methylase
Skin and soft tissue infections, if not appropriately treated, may lead to disfiguring, amputation, sepsis and extended hospital stays which results in a significant increase in mortality and morbidity rates across the world. A variety of micro-organisms can be associated with skin and soft tissue infections, Staphylococcus aureus being the commonest isolate.1,2 b-lactam group of antibiotics is the most commonly used antibiotic for the treatment of Staphylococcus aureus infections.3,4 However, Methicillin-resistant S. aureus (MRSA) poses severe therapeutic challenges. During the past decades, a steep rise has been observed in methicillin-resistant S. aureus isolates.5 MRSA strains are often multidrug-resistant, leaving few therapeutic options. The macrolide-lincosamide-streptogramin B (MLSB) antibiotics are commonly used to treat MRSA infections, and among them, the use of clindamycin as an alternative therapy has risen in India and other parts of the world.1 The advantages of choosing clindamycin are—availablility of both parenteral and oral formulations, high bio-availability, soft tissue permeability, inhibits toxin production, and is relatively cheap.6 However, its increased use has resulted in widespread resistance against clindamycin.
The resistance to clindamycin depends upon the expression of rRNA methylase (RM) by S. aureus, either produced constitutively or induced by an inducing agent. Erythromycin is known to be an effective inducer of the enzyme. S. aureus isolates with constitutively expressing RM (strains termed cMLSB) resist clindamycin and erythromycin while the strains with inducibler RNA methylase enzyme (iMLSB) show resistance only to erythromycin but are sensitive to clindamycin.7,8 However, in vivo clindamycin can be ineffective in such cases. Interestingly, the difference between iMLSB and cMLSB can be detected by D-test.
Three different phenotypes can be exhibited by strains for MLSB antibiotics 9 as shown in Table 1.
Table (1):
Phenotypes of strains towards MLSB antibiotics.
Phenotype |
Erythromycin Sensitivity |
Clindamycin Sensitivity |
Remarks |
|---|---|---|---|
MLSB sensitive (MS) |
Resistant (zone size ≤13mm) |
Sensitive ( ≥21mm zone) |
Circular zone of inhibition |
InducibleMLSB |
Resistant (zone size ≤13mm) |
Sensitive ( ≥21mm zone) |
D-shaped zone of inhibition with flattening towards erythromycin. |
Constitutive MLSB |
Resistant (zone size ≤13mm) |
Resistant ( ≤14mm zone) |
Nozone of inhibition |
In the present study, the antibiotic sensitivity testing of the isolates was done along with D-test to figure out the rate of inducible resistance to clindamycin in our hospital and to formulate treatment options for iMLSB isolates.
Sample collection
The current study is a retrospective observational study done for one year (January, 2019 – December, 2019). Pus samples from patients from all age groups with skin and soft tissue infections received in the Department of Microbiology, M.M. Institute of Medical Science and Research, were processed according to standard laboratory guidelines.
Inclusion criteria
Patients with skin and soft tissue infection of any age or sex.
Exclusion criteria
S. aureus isolates for infection other than skin and soft tissue infection.
Isolation and characterization of S. aureus isolates
The samples were streaked for culture on MacConkey agar and blood agar media and were incubated at 37°C for 24-48 h. After the Gram staining and biochemical tests as per standard laboratory protocols10, the S. aureus isolates were collected and examined for antibiotic susceptibility. The Kirby Bauer‘s disc diffusion antibiotic sensitivity testing (AST) was done, and interpretations were made as per the Clinical and Laboratory Standards Institute (CLSI). The antibiotics tested include penicillin (10 units), co-trimoxazole (1.25/23.75μg), ciprofloxacin (5μg), gentamicin (10μg), erythromycin (15μg), clindamycin (2μg), azithromycin (15μg) and linezolid (30μg) (HiMedia,Mumbai, India). Vancomycin sensitivity was determined by broth dilution test to determine MIC. Cefoxitin (30µg) disc was used to detect methicillin resistance and the isolates that yielded a zone diameter of <22 mm around the cefoxitin disc were considered as MRSA.11 The control strain used was S. aureus (ATCC 25923).
Detection of mecA gene by PCR for molecular confirmation of MRSA12
DNA of the cefoxitin resistant strains was extracted using QIA amp DNA mini kit (QIAGEN). Isolated DNA was amplified using the primers as described by Geha et al.
Forward primer sequence – 5’-GTA GAA ATG ACT GAA CGT CCG ATAA – 3’
Reverse primer sequence- 5’-CCA ATT CCA CAT TGT TTC GGT CTA A –3’
D-test
For determining the phenotype of strains against MLSB group of antibiotics, the clindamycin (2µg) disc and erythromycin (15µg) disc were placed at a distance of 15mm (edge to edge) on a Mueller Hinton agar inoculated 0.5 McFarland bacterial suspension of the test organism and the strains were allowed to grow for 24 h at 37°C.12
Statistical Analysis
Performed by using SPSS, version 18.0 (SPSS Inc., Chicago, IL).
In our study, a total of 235 S. aureus isolates were obtained from patients with infections of the skin and soft tissue. Out of 235 S. aureus strains screened 80.5% (190/235) strains were cefoxitin resistant. The DNA from these strains was isolated, and amplicons were detected for mecA gene. We found that all the cefoxitin-resistant strains harbor mecA gene, showing that among all the collected samples, 80.5% were MRSA (190/235) strains and rest were MSSA. Of the 190 MRSA strains, 69.5% (132/190) were from hospitalized patients.
After that, the antibiotic sensitivity testing for MRSA strains was performed, and phenotype was examined and recorded (Table 2). Resistance to various classes of antibiotics was higher among the MRSA isolates than among methicillin-sensitive S. aureus (MS).
Table (2):
Antibiotic sensitivity pattern of the S.aureus isolates.
| Isolates | Sensitivity Pattern | Anti Microbial Agent / N | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Cip | E | CL | TE | Gen | Ln | Vn | COT | Rf | ||
| MRSA (n=190) | S | 11 | 44 | 86 | 143 | 135 | 183 | 190 | 91 | 164 |
| R | 179 | 146 | 104 | 47 | 55 | 7 | 0 | 99 | 26 | |
| MSSA (n=45) | S | 15 | 30 | 37 | 37 | 7 | 44 | 45 | 29 | 45 |
| R | 30 | 15 | 7 | 7 | 38 | 1 | 0 | 16 | 0 | |
| Total (n=235) | S | 26 | 74 | 123 | 180 | 142 | 227 | 235 | 120 | 209 |
| R | 209 | 161 | 111 | 54 | 93 | 8 | 0 | 115 | 26 | |
S=Sensitive, R=Resistant, Cip= Ciprofloxacin, E= Erythromycin, CL= Clindamycin, TE= Tetracycline,Gen= Gentamycin, Ln= Linezolid, Vn= Vancomycin, COT= Co-trimoxazole,Rf= Rifampicin
Table (3):
Susceptibility pattern ofclindamycin and erythromycin among the isolates.
Susceptibility pattern (Phenotype) |
Number(Percentage) MRSA (N= 190) |
Number (Percentage) MSSA strains (N=45) |
Number (Percentage) All S.aureus (N=235) |
|---|---|---|---|
E= S, CL= S |
44 (23.1%) |
30 (66.7%) |
74 (31.5%) |
E= R, CL= R (cMLSB) |
98 (51.6%) |
6 (13.3%) |
104 (44.2%) |
E= R, CL= S (D test +ve) =iMLSB |
42 (22.1%) |
6 (13.3%) |
48 (20.4%) |
E= R, CL= S (D test negative) = MS |
6 (3.1%) |
3 (6.7%) |
9 (3.8%) |
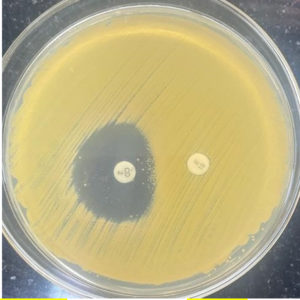
After that, we examined MRSA and MSSA strains to exhibit resistance towards erythromycin and clindamycin by performing D-test by putting the discs carrying antibiotics. As summarized in Table 3, we found that among MRSA strains (n=190), 23% were sensitive to both erythromycin and clindamycin, and ~51% showed resistance to both. Importantly, we found that 22% showed positive D-test in MRSA strains (Fig 1), thus belonging to iMLSB, indicating inducible expression of RM enzyme. On the other hand, among MSSA strains, 13% showed positive D-test, 66% showed sensitivity towards both antibiotics, and 13% showed resistance. Thus, in totality, our study showed that 44% of all the strains showed the presence of constitutive RM enzyme while 20% of strains showed that RM is inducible.
MRSA isolation is increasing from infections of the skin and soft tissue. The increasing resistance to the available therapeutic options for the treatment of MRSA is worrisome. Our study detected a high prevalence of MRSA (80.5%) comparable to studies performed in Ethiopia1,13 and Kenya.14 However, the results of our study are higher from the studies from India3,5 and other parts of the world.15-17 The difference in prevalence from different regions could be due to the variation in the study population, antibiotics used, sample size, and infection control policies. As, mostly the treatment of skin and soft tissue infections is on an empirical basis; the increase of antimicrobial resistance; the update on the susceptibility pattern of the isolates can help in the selection of empirical therapy. Clindamycin is one of the commonly used drug for Staphylococcal infections, particularly skin and soft tissue infections.4 The differentiation of inducible MLSB (iMLSB phenotype) isolates from isolates with MS phenotype is a critical as use of clindamycin in iMLSB phenotype results in treatment failures. In our study, iMLSB phenotype (Positive for D-test) was 22.1% and 13.3% in MRSA and MS isolates, respectively. The prevalence of MS phenotype (D test- Negative) was 3.1% and 6.7% among MRSA and MSSA isolates, respectively. Similar results were observed from a study from in India where MS phenotype was found among 8% isolates.18 However, Patel et al.19 and Lall M et al.20 reported higher rates 50% and 37.5%, respectively, of iMLSB phenotype among MRSA strains.
Limitation and suggestion
The study is performed on patients from Northern Part of India, so the data need to be further verified by different research groups to know prevalence of iMLSB at National level. We would be further working on charchterization of genes responsible for the inducibilty of RM gene.
The incidence of MLSB resistance varies with geographical area, with patterns of infections and drug use. Therefore, constant surveillance should be done for MLSB resistance in S. aureus using D-test on erythromycin-resistant isolates in every health care setting to avoid therapeutic failures. Routine testing of all Staphylococcal isolates for iMLSB is the recommendation by CLSI to minimize the misuse of the drug. The clinicians should also be aware of in vitro inducible clindamycin resistance. Vancomycin and linezolid are the drugs to be considered in such cases for therapy.
ACKNOWLEDGMENTS
We would like to thank Mamharishi Maharishi (Deemed to be University) for providing facility.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
ETHICS STATEMENT
The study was approved by the institutional Ethics Committee, Maharishi Markandeshwar Institute of Medical Sciences & Research, Mullana, Ambala, India (Project No: IEC- 87E).
AVAILABILITY OF DATA
All datasets generated or analyzed during this study are included in the manuscript
- Mama M, Aklilu A, Misgna K, Tadesse M, Alemayehu E. Methicillin- and Inducible Clindamycin-Resistant Staphylococcus aureus among Patients with Wound Infection Attending Arba Minch Hospital, South Ethiopia. Int J Microbiol. 2019;2965490.
Crossref - Navidinia M, Goudarzi H, Pouriran R, Azimi H, Goudarzi M. Molecular Analysis and Integron Carriage of Mupirocin-Resistant Staphylococcus aureus Strains Isolated from Burn Wound Infections, Tehran, Iran. Archives of clinical and Infectious diseases. 2018;14 (1); e84764.
Crossref - Goudarzi M, Navidinia M, Beiranvand E, Goudarzi H. Phenotypic and Molecular Characterization of Methicillin-Resistant Staphylococcus aureus Clones Carrying the Panton-Valentine Leukocidin Genes Disseminating in Iranian Hospitals. Microb Drug Resist. 2018;24(10):1543-1551.
Crossref - Arora S, Devi P, Arora U, Devi B. Prevalence of Methicillin-resistant Staphylococcus Aureus (MRSA) in a Tertiary Care Hospital in Northern India. J Lab Physicians. 2010;2(2):78-81.
Crossref - World Health Organization. Global priority list of antibiotic-resistant bacteria to guide research, discovery, and development of new antibiotics. Geneva: 2017. https://www.who.int/medicines/publications/WHO-PPL-Short_Summary_25Feb-ET_NM_WHO.pdf
- Baker CJ, Frenck RW. Change in management of skin/soft tissue infections needed. Am Acad Ped News. 2004;25:105-117. https://www.aappublications.org/content/25/3/105.2. Accessed June 5, 2019.
- Gupta V, Datta P, Rani H, Chander J. Inducible clindamycin resistance in Staphylococcus aureus: a study from North India. J Postgrad Med. 2009;55(3):176-179.
Crossref - Ahmed MO, Alghazali MH, Abuzweda AR, Amri SG. Detection of inducible clindamycin resistance (MLSBi) among methicillin-resistant Staphylococcus aureus (MRSA) from Libya. Libyan J Med. 2010; 5:1,4636.
Crossref - Lata S , Bala R,Jindal N, Gupta N. In Vitro Study of Constitutive and Inducible Clindamycin Resistance in Staphylococcus aureus with Reference to Methicillin Resistant Staphylococcus aureus: Experience From Tertiary Care Hospital in Punjab. Ind J Pub Health Res Dev. 2020;11(2):314-317.
Crossref - Stephen DA, William MJ, Elmer WK, Paul C. Schreckenberger Washington C. Koneman’s Color Atlas and Textbook of Diagnostic Microbiology: 6th Edition, Washington C. 2015:8580000248357.
- Performance standards for Antimicrobial Susceptibility Testing. M100, 27th ed. Clinical and Laboratory Standards Institute.2017:143
- Geha DJ, Uhl JR, Gustaferro CA, Persing DH. Multiplex PCR for identification of methicillin-resistant staphylococci in the clinical laboratory. J Clin Microbiol. 1994;32(7):1768-1772.
Crossref - Godebo G, Kibru G, Tassew H. Multidrug-resistant bacterial isolates in infected wounds at Jimma University Specialized Hospital, Ethiopia. Ann Clin Microbiol Antimicrob. 2013;12:17.
Crossref - Maina EK, Kiiyukia C, Wamae CN, Waiyaki PG, Kariuki S. Characterization of methicillin-resistant Staphylococcus aureus from skin and soft tissue infections in patients in Nairobi, Kenya. Int J Infect Dis. 2013;17(2):115-119.
Crossref - Tsige Y, Tadesse S, G/Eyesus T, et al. Prevalence of Methicillin-Resistant Staphylococcus aureus and Associated Risk Factors among Patients with Wound Infection at Referral Hospital, Northeast Ethiopia. J Pathog. 2020;2020:3168325.
Crossref - Almeida GC, dos Santos MM, Lima NG, Cidral TA, Melo MC, Lima KC. Prevalence and factors associated with wound colonization by Staphylococcus spp. and Staphylococcus aureus in hospitalized patients in inland northeastern Brazil: a cross-sectional study. BMC Infect Dis. 2014;14:328.
Crossref - Ghebremedhin B, Olugbosi MO, Raji AM, et al. Emergence of a community-associated methicillin-resistant Staphylococcus aureus strain with a unique resistance profile in Southwest Nigeria. J Clin Microbiol. 2009;47(9):2975-2980.
Crossref - Prabhu K, Rao S, Rao V. Inducible Clindamycin Resistance in Staphylococcus aureus Isolated from Clinical Samples. J Lab Physicians. 2011;3(1):25-27.
Crossref - Patel M, Waites KB, Moser SA, Cloud GA, Hoesley CJ. Prevalence of inducible clindamycin resistance among community- and hospital-associated Staphylococcus aureus isolates. J Clin Microbiol. 2006;44(7):2481-2484.
Crossref - Lall M, Sahni AK. Prevalence of inducible clindamycin resistance in Staphylococcus aureus isolated from clinical samples. Med J Armed Forces India. 2014;70(1):43-47.
Crossref
© The Author(s) 2021. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.