ISSN: 0973-7510
E-ISSN: 2581-690X
In the treatment of influenza, Neuraminidase inhibitors (NAIs) (Oseltamivir and Zanamivir) play a major role. The emergence of variants of influenza A (H1N1) pdm09 virus resistant to Oseltamivir is a matter of great concern as it limits its usage. Therefore, vigilant monitoring for Oseltamivir-resistant viruses has been recommended by the World Health Organization (WHO). Our study aimed to screen the influenza A (H1N1) pdm09 virus for NAI drug resistance during the outbreak of 2015-16 in North-Western India. A total of 640 H1N1pdm09 virus-positive samples were screened for drug resistance to Oseltamivir by WHO allelic discrimination real-time RT-PCR protocol. The allelic discrimination PCR protocol can detect the presence of single nucleotide polymorphisms (SNPs), the H275Y mutation is detected by this method which causes resistance to Oseltamivir. Sanger sequencing of partial fragment of NA gene (fragment IV), of 90 samples were performed to confirm the presence of NA-H275Y mutation. Neuraminidase susceptibility of 20 randomly selected isolates to Oseltamivir was tested using NA inhibition chemiluminiscence based assay. Among 640 H1N1pdm09 positive samples tested, H275Y mutation was detected in one sample (0.15%) by PCR and confirmed by Sanger sequencing also. All the 20 isolates tested for NAI susceptibility by NA star assay were found to be sensitive to Oseltamivir. WHO allelic discrimination PCR is an easy, rapid and sensitive method for high-throughput detection of resistance to Oseltamivir. Systematic regular drug resistance surveillance of Influenza A is essential to monitor the emergence and spread of drug-resistant strains.
Influenza A (H1N1) pdm09 virus, (NAIs) Neuraminidase inhibitors, Oseltamivir, real-time polymerase chain reaction
The first case of Influenza A (H1N1) pdm09 was reported in Mexico in April 2009 which quickly spread to various countries to cause the pandemic1. Neuraminidase inhibitors (NAIs) (Oseltamivir and Zanamivir) play an important role in treating influenza infection and are recommended by the Center for Disease Control and Prevention (CDC) for high-risk patients or hospitalized patients. NAIs act by interfering with release of newly assembled viruses from the infected cells2. Oseltamivir has a large hydrophobic side chain that requires a conformational rearrangement in the viral NA that is essential to accommodate the drug. Any mutations that affect this rearrangement may reduce the binding affinity of the drug leading to viral resistance to Oseltamivir3-5.
In seasonal Influenza A (H3N2) virus Oseltamivir resistance emerged during 2007–2008 which became widespread by 2008–2009 season6. The first case of Oseltamivir resistant H1N1pdm09 was reported in June 20097 and up to October 2011, over 600 Oseltamivir-resistant H1N1pdm09 virus cases were reported in 32 countries8. Emergence of H1N1pdm09 virus resistant to Oseltamivir is a matter of great concern as it can limit the use of the drug which is the mainstay of treatment of cases infected with H1N1pdm09.
A point mutation found on the neuraminidase gene, H275Y, is known to cause Oseltamivir resistance. Many methods are available to test for drug resistance to oseltamivir. The allelic discrimination PCR protocol can detect presence of single nucleotide polymorphisms (SNPs) and the H275Y mutation is identified by this method to detect Oseltamivir. In sanger sequencing protocol given by WHO fragment IV of NA gene can confirm the presence of NA-H275Y mutation. Phenotypically Neuraminidase susceptibility can be tested using NA inhibition chemiluminiscence based assay, NA star.
Oseltamivir-resistant H1N1pdm09, with the H275Y substitution were reported from two clusters of immunocompromised and hospitalized patients at United Kingdom (UK)9 and North Carolina, US10. Therefore, vigilant monitoring for Oseltamivir-resistant viruses has been recommended by the World Health Organization (WHO). Only sporadic reports of Oseltamivir resistance testing are available from India and data from North Western India is lacking. In the present study, we tested H1N1pdm09 positive samples from North Western India for Oseltamivir resistance by allelic discrimination real time PCR to detect for the presence of H275Y mutation. Sanger sequencing and phenotypic assay for drug resistance were done in representative samples.
RT-PCR and sample collection
Throat or nasal swab samples of suspected cases with influenza-like illness (ILI) were collected in Viral Transport Medium (VTM) and transported maintaining cold-chain. The study was conducted at the Advanced Research Laboratory, SMS Medical College, Jaipur, Rajasthan, India. The RNA was isolated by the QIAamp Viral RNA Mini Kit (Qiagen, Germany). The RNA of each sample was tested using primers and probes sets of InfA, Universal Swine (swFluA), Swine H1 (swH1) and RNase P according to CDC protocol 10 on Step One Plus real time PCR instrument (Applied Biosystems,USA).
Detection of H275Y mutation by allelic discrimination real time PCR
Total 640 H1N1pdm09 virus positive samples (n=640) were selected and screened for the presence of H275Y mutation using primers and probes as per WHO protocol12 (Table 1). This method detects a single nucleotide mutation H275Y mutation in the NA gene from C to T which is known to be associated with Oseltamivir resistance in H1N1 influenza virus by allelic discrimination real time PCR that uses a pair of primers with two TaqMan MGB probes each targeting a different allele i.e., wild type and mutant virus that differ by a single base substitution or single nucleotide polymorphism (SNP).
Table (1):
Primers and probe sequences for allelic discrimination PCR11.
Names |
Sequence (5’-3’) |
Position |
|---|---|---|
H1N1 NA F690-719 (Forward primer) |
ATGTGCATGTGTAAATGGTTCTTGCTTTAC |
690-719 |
H1N1 NA R847-872 (Reverse primer) |
ACACATGTGATTTCACTAGAATCAGG |
847-872 |
Probe 1 FAM-274Ya-swH1N1-F823-835 (Specific for Mutant allele) |
(FAM)TACTATGAGGAAT(MGB) |
823-835 |
Probe 2 VIC-H274a- swH1N1-F823-835 (Specific for wild type allele) |
(VIC)CACTATGAGGAAT(MGB) |
823-835 |
Detection of H275Y mutation by Sanger sequencing
Sanger sequencing of a fragment IV of NA gene of representative 90 samples was done as per WHO sequencing protocol to detect the presence of H275Y mutation. For Sanger sequencing, reverse transcriptase PCR (RT-PCR) was performed to amplify partial regions of the NA gene using Super Script III one-step RT-PCR kit with platinum Taq (Invitrogen) and specific primers targeting the fragment from nucleotides 726 to 1346 of NA gene (fragment IV) as described earlier13. RT-PCR products were used for cycle sequence PCR reaction which was set up using Big Dye Terminator Kit (Applied Biosystems) and gene specific primers13 (Table 2). The reaction products were purified by Big Dye X Terminator Purification Kit (Applied Biosystems) and run on an ABI 3500 Dx Genetic analyser (Applied Biosystems). Sequences were analysed using the Mega Version 5.2 sequence analysis software and were submitted to BLAST (Basic Local Alignment Search Tool) program of NCBI (National Centre for Biotechnology Information)14.
Table (2):
Sequence of Primers used for one step RT PCR12.
Name |
Primer Sequence (5’-3’) |
|---|---|
NA gene fragment IV Forward Primer |
726 TGT AAA ACG ACG GCC AGT AAT GGR CAR GCC TCR TAC AA |
NA gene fragment IV Reverse Primer |
1346 CAG GAA ACA GCT ATG ACC GCT GCT YCC RCT AGT CCA GAT |
Virus Culture
Madin-Darby Canine Kidney (MDCK) cells, acquired from the National Center for Cell Sciences (NCCS) stored in Minimal Essential Medium (MEM, Himedia, Mumbai, India) were enriched with 10% fetal bovine serum (FBS) (HiMedia, Mumbai, India). Twenty H1N1pdm09 positive samples were selected based on Ct values <35 and volume of samples available. These clinical samples were inoculated onto confluent MDCK cells in serum free medium containing 2 μg/ml of Tosyl Phenylalanyl Chloromethyl Ketone (TPCK) trypsin for virus isolation. The samples were passaged until sufficient titer was reached. Tissue culture fluid was harvested after observing the infected cells for cytopathic effects. Virus stocks were aliquoted and stored at -80°C until use.
NA inhibition Assay
Neuraminidase susceptibility testing was done for 20 representative isolates to Oseltamivir by NA-star Influenza neuraminidase inhibitor resistance detection Kit (Applied Biosystems, USA), which is a inhibition chemiluminescence based assay. The 50% inhibitory concentration (IC50) of Oseltamivir was determined using the NA Star by regression analysis15.
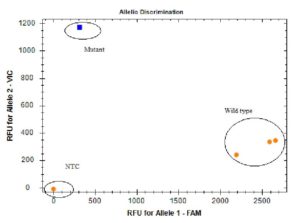
In our study H275Y mutation associated with Oseltamivir drug resistance was detected in only 1/640 (0.15%; confidence interval: 0.000-0.004) H1N1pdm09 positive samples by allelic discrimination real time RT PCR (Fig. 1). On sequencing of a partial fragment of NA gene, only 1/90 (1.1%) sample was found to have the mutation H275Y causing drug resistance, which was detected by RT-PCR assay also, rest of the samples were found to have wild type sequences only. The partial NA gene sequences were submitted to NCBI (accession numbers: MN210329 to MN210341 and MN252131 to MN252207). Fig. 2 shows the dendrogram of the partial sequences of NA gene of both wild type and mutant samples aligned with reference to California strain (NCBI Accession. No NC_026434.1). All the representative 20 (100%) isolates tested for neuraminidase susceptibility to Oseltamivir by NA Star were found to be sensitive. NAI susceptibility of the sample which was found to be resistant by Sanger sequencing could not be tested as the sample quantity was insufficient to perform the assay. The results of the present study are summarized in Table 3.
Table (3):
Results of tests performed for drug resistance in Influenza A (H1N1) pdm 09 samples.
S. No. |
Test Done |
No. of Samples tested (N) |
Results |
|---|---|---|---|
1 |
WHO allelic discrimination real time RT PCR |
640 |
1/ 640 (0.15%) Positive for H275Y mutation, |
2 |
Sanger sequencing |
90 |
One, Positive for H275Y mutation by allelic discrimination real time PCR confirmed by sequencing also |
3 |
NA star kit (Chemiluminiscence based assay) |
20 |
All sensitive (The sample found to be resistant by Sanger sequencing and real time PCR could not be tested using chemiluminescence based assay as the sample quantity was insufficient to perform the assay). |
Fig. 1. Real Time RT PCR Allelic discrimination plot for detection of substitution at amino acid 275 in Neuraminidase
Fig. 2. Sequence dendrogram of the partial sequences of NA gene of wild type and mutant samples aligned with reference to California strain (NCBI Accession. No NC_026434.1).
A study from Scotland reported 0.55% (10/1802) Oseltamivir resistance in H1N1pdm09 strains15.Whereas study from South Korea reported 16% (11/67) drug-resistance in H1N1pdm09 virus during May 2009–January 2010 and virus isolates from all the patients had the H275Y mutation in the neuraminidase gene17. Till now there have been very few studies on drug resistance surveillance in H1N1pdm09 from India15,18-20. In a study from Eastern India, H275 mutation was not detected (0%) in any of the 25 H1N1pdm09 isolates analyzed by sequencing method17. Study from Pune reported 0.07% Oseltamivir resistance in 1524 H1N1pdm09 positive samples screened for the presence of H275Y mutation by allelic rRT-PCR18. In study from Mumbai, all the 47 isolates of H1N1pdm09 virus tested for NAI susceptibility were found to be susceptible to Oseltamivir using NA inhibition chemiluminescence based assay20. In a study from Brazil, 261 virus isolates were tested for mutations by Sanger sequencing of the NA gene and H275Y mutation was detected in only one sample21.
Few studies have reported drug resistance by using enzyme inhibition assays21,23. In a study by Gubareva et al., 0.68% (23/3335) H1N1pdm09 virus isolates were found to be resistant by chemiluminescent NI assay22. Whereas study from China reported higher resistance rate of 2.5% in 2013 and 2.3 % in 2014 to Oseltamivir tested by NAI susceptibility method23.
Several methods have been used in various studies worldwide for surveillance of antiviral resistance in influenza viruses, but allelic discrimination real time PCR has been reported to be most rapid and high throughput method that has the benefits of lower cost and generally straightforward interpretation. Major advantage is that most clinical molecular diagnostic laboratories have the capabilities required for its use, it’s easy to perform, and most of the steps can be automated and has shorter turnaround time. Another major advantage with RTPCR is that they have increased sensitivity over that of sequencing assays. Sanger sequencing has a sensitivity of ca. 20%, and the initial screening for rare resistant species in patient samples requires pure growth of virus21. Sanger sequencing is cumbersome, time consuming, expensive, requires technical skill. Also, the equipment is not available in most laboratories, but it has the advantage that it can identify mutations in whole NA gene. The neuraminidase inhibition assay is also time consuming, needs to culture the virus and requires specialized infrastructure but can give clear answer as to the susceptibility of the virus. We carried out all three methods to confirm findings of real time PCR in representative samples.
The only drug resistant sample in present study was from a patient who was a medical student, 28-year-old female, having a history of contact with a hospitalized four-year-old child who was reported positive for H1N1pdm09. After two days of exposure, she developed symptoms of ILI and was further reported positive H1N1pdm09. She was otherwise healthy and immuno-competent. She received Oseltamivir for treatment of H1N1 pdm09 infection after diagnosis, was successfully treated and is alive and healthy. There was no history of Oseltamivir prophylaxis and vaccination in the medical student. The sample of the four-year-old boy from whom she was presumed to have acquired the infection was also tested for H275Y mutation, but it was found to be sensitive to NAI.
Detection of mutation in our study indicates the emergence and circulation of low-level populations of Oseltamivir-resistant virus in North Western Indian population which has been reported for the first time but this could not be attributed to selection pressure owing to drug therapy because the patient’s sample was collected for testing prior to initiation of treatment. This is similar to that reported by Potdar et al., who reported first case of Oseltamivir drug resistance from India tested by real time PCR18.Although the clinical significance of low-level populations of Oseltamivir-resistant virus is indeterminate, early detection of Oseltamivir-resistant virus can facilitate the use of alternative antiviral drugs, which may help in improving patient’s treatment outcome.
A major limitation of our study was that other mutations conferring drug resistance were not analyzed as only partial fragment of NA gene (fragment IV as per WHO protocol) was sequenced which covered only 726 to 1346 nucleotide positions and resistant case couldn’t be confirmed by NA star.
To conclude the present study reports the first case of emergence and circulation of low-level populations of Oseltamivir-resistant H1N1pdm09 virus in North Western Indian population this highlights the need for continuous antiviral drug resistance surveillance for influenza. Such studies are essential to indicate efficacy of the prescribed neuraminidase inhibitors by clinicians and have public health importance. For rapid detection of drug resistance easy and sensitive screening methods like WHO allelic discrimination PCR are preferable over other methods like Sanger sequencing and chemiluminescence based assays as they require specialized facilities of sequencing and virus isolation, technical expertise, are labor intensive, time consuming and results cannot be available in clinically useful time period.
ACKNOWLEDGMENTS
We would like to express our heartfelt thanks to Dr. Varsha Potdar and Dr. Mandeep Chadha from NIV Pune for providing training.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
BM conceived the research. KT, PS, FD, JT and AC performed the experiments. KT wrote the manuscript. BM, WD and PS edited the manuscript. All authors read and approved the manuscript for publication.
FUNDING
This work was supported by the ICMR (Indian Council of Medical Research) (project number VIR/7/2011-ECD-I) 23.03.2011.
ETHICS STATEMENT
The study was approved by the ethics committee of SMS Hospital Jaipur Rajasthan No. MC/EC/2011/227.dated March 3, 2011.
AVAILABILITY OF DATA
The sequences analysed during the study are available in the NCBI database. NA gene sequences (accession numbers: MN210329 to MN210341 and MN252131 to MN252207).
- Suppiah J, Othman KA, Thayan R, et al. Screening of H275Y mutation in influenza a (H1N1) pdm09 isolated in Malaysia by rapid real-time PCR assays. Innovative Journal Of Medical And Health Science. 2013;3(5):226-229.
- Moscona A. Neuraminidase inhibitors for influenza. N Engl J Med. 2005;353(13):1363-1373.
Crossref - Samson M, Abed Y,Desrochers FM, et al. Characterization of Drug-Resistant Influenza Virus A(H1N1) and A(H3N2) Variants Selected In Vitro with Laninamivir. Antimicrob Agents Chemother. 2014;58 (9):5220-5228.
Crossref - Collins PJ, Haire LF, Lin YP, et al. Crystal structures of Oseltamivir-resistant influenza virus neuraminidase mutants. Nature. 2008;453(7199):1258-1261.
Crossref - Malaisree M, Rungrotmongkol T, Decha P, Intharathep P, Aruksakunwong O, Hannongbua S. Understanding of known drug-target interactions in the catalytic pocket of neuraminidase subtype N1. Proteins. 2008;71(4):1908-1918.
Crossref - Antiviral Drug Resistance among Influenza Viruses http://www.cdc.gov/flu/professionals/antivirals/antiviral-drug-resistance.htm accessed on May 10, 2018.
- Hurt AC, Deng YM, Ernest J, et al.Oseltamivir-resistant influenza viruses circulating during the first year of the influenza A (H1N1)2009 pandemic in the Asia-Pacific region, March 2009 to March 2010. Euro Surveill. 2011;16(3):19770.
Crossref - Dixit R, Khandaker G, Hay P, et al. A randomized study of standard versus double dose oseltamivir for treating influenza in the community. Antiviral Therapy. 2015;20:689-698.
Crossref - Rapid risk assessment http://ecdc.europa.eu/en/publications/Publications/110906_TER_Rapid_Risk_Assessmet. accessed on March 5, 2018.
- World Health Organization. Pandemic (H1N1) 2009: briefing note 18, Geneva, Switzerland: World Health Organization. 2009. http://www.who.int/csr/disease/swineflu/notes/briefing_20091202/en/index.html. Accessed on May 16, 2018.
- CDC Protocol of Real Time RTPCR for Influenza A (H1N1). http://www.who.int/csr/resources/publications/swine flu/CDCRealtimeRTPCR_SwineH1Assay -2009_20090430.pdf. Accessed on Jan 10, 2015.
- WHO, Real-time RT-PCR Allelic Discrimination Analysis for Detection of the Substitution at Amino Acid 275 in the Neuraminidase (NA) of A(H1N1) pdm09 Influenza Viruses, Influenza Virus Research Center National Institute of Infectious Diseases Tokyo, Japan, 1-6. www.Origin.who.int/influenza/gisrs/antiviral/allelic discrimination-analysis-na275.pdf. Accessed on Jan 10, 2015.
- Sequencing primers and protocol. www.who.int/csr/resources/publications/swineflu/GenomePrimers_20090512.pdf. Accessed on June, 2015.
- Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ. Basic local alignment search tool. J Mol Biol. 1990;215(3):403-410.
Crossref - Gohil D, Kothari S, Shinde P, et al. Oseltamivir Resistant Influenza A (H1N1) Virus Infection in Mumbai, India, J Antivir Antiretrovir. 2015;7(4):108-114.
Crossref - Harvala H, Gunson R, Simmonds P, et al. The emergence of Oseltamivir-resistant pandemic influenza A(H1N1) 2009 virus amongst hospitalized immunocompromised patients in Scotland, November- December, 2009. Euro Surveill. 2010;15(14):19536.
Crossref - Shin SY, Kang C, Gwack J, et al. Drug-resistant pandemic (H1N1) 2009, South Korea. Emerg Infect Dis. 2011;17(4):702-704.
Crossref - Mukherjee A, Nayak MK, Dutta S, Panda S, Satpathi BR, Sarkar MC. Genetic Characterization of Circulating 2015 A(H1N1)pdm09 Influenza Viruses from Eastern India. PLoS ONE. 2016;11(12):e0168464.
Crossref - Potdar VA, Padbidri VV, Chadha MS. Oseltamivir-resistant influenza A (H1N1) pdm09 virus: first reported case from India. WHO South-East Asia J Public Health. 2013;2(3):181-183.
Crossref - Gohil D, Kothari S, Shinde P, et al. Genetic characterization of Oseltamivir-resistant seasonal influenza A (H1N1) virus circulating during 2009 pandemic influenza in Mumbai. IJAR. 2015;3(3):252-261.
- Marx C, Gregianini TS, Kieling FML, et al. Oseltamivir-resistant influenza A(H1N1) pdm09 virus in southern Brazil. Mem Inst Oswaldo Cruz. 2013;108(3):392-394.
Crossref - Gubareva LV, Trujillo AA, Okomo MA, et al. Comprehensive assessment of 2009 pandemic influenza A (H1N1) virus drug susceptibility in vitro, Antiviral Therapy. 2010;15(2):1151-1159.
Crossref - Liu S, Jiao X, Wang S, et al. Susceptibility of influenza A(H1N1)/pdm2009, seasonal A(H3N2) and B viruses to Oseltamivir in Guangdong, China between 2009 and 2014. Sci Rep. 2017;7:8488.
Crossref
© The Author(s) 2021. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.