ISSN: 0973-7510
E-ISSN: 2581-690X
Trigonella foenum-graecum L. is a widely used herb in traditional medicine. The aim of this study was to evaluate the genetic polymorphisms in fenugreek plants following the treatment of fenugreek seeds with different combinations of biotic and abiotic agents using the random amplified polymorphic DNA (RAPD)-PCR technique. We assessed the effects of two strains of the fungus Trichoderma harzianum (Th-1 and Th-2), methyl jasmonate (MeJA), and Aloe vera gel (AVG) on growth parameters of fenugreek plants. Combinations of Th-1, MeJA, AVG significantly increased fenugreek root length, shoot length, shoot fresh weight, number of true leaves, and chlorophyll content. The Th-2 isolate, on the other hand, markedly slowed plant development (except for root length which was not affected significantly). In contrast, the combination with MeJA had no considerable effect on all growth measures, whereas the combination with VAG resulted in a substantial drop in shoot height and chlorophyll content when compared to other growth parameters that were unaffected. The present study has shown that the PCR amplification of DNA, using five primers for RAPD analysis, produced 62 DNA fragments that could be scored in all genotypes. The total number of polymorphic bands was 26, and the average percentage of polymorphism was 54.21%. The RAPD-PCR results showed that the treatment of fenugreek seeds with Th-1 alone or in combination with MeJA and AVG induced polymorphisms in fenugreek leaves.
Genetic polymorphisms, Trigonella foenum-graecum, Trichoderma harzianum, methyl jasmonate, Aloe vera, RAPD-PCR
Fenugreek Trigonella foenum-graecum L. (family: Fabaceae) is an annual herb that has been used as an essential spice and traditional medicine since ancient times1. It has therapeutic properties such as anti-diabetic, anti-carcinogenic, hypocholesterolemic, antioxidant, and immunological activity2.
Fenugreek is native to southeastern Europe and western Asia, but is now grown all over the world, including South Africa, northern Africa, Austria, Australia, Europe, India, and the United States3. Fenugreek seeds provide a naturally rich source of vitamins, such as thiamine, and biogenic elements, such as phosphorous, sulfur, iron, silicon, and sodium3. The chemical constituents of fenugreek include fibers, saponins, flavonoids and alkaloids, namely, trigonelline and choline4. Additionally, the steroidal sapogenin diosgenin is a secondary metabolite produced by fenugreek and other plants that is widely studied due to its bioactive properties and potential applications in the medical field5. The nutritional benefits and curative applications of fenugreek as an effective therapeutic agent against inflammation and diseases, such as cancer and diabetes, have been reported6. Various scientists from all around the world have identified it as an essential medicinal plant. It has been extensively recognized to be efficacious in the prevention of a variety of ailments, including cancer, hypercholesterolemia, diabetes, and inflammation7,8.
Fungi of the genus Trichoderma have an enormous capacity to produce secondary metabolites7; thus, playing an important role in ecosystem health9. The efficacy of Trichoderma spp. have been identified as effective plant-beneficial microbial inoculants, these fungi act as bio-stimulants that promote plant growth10. Plant associations with Trichoderma strains have been shown to enhance plant growth by improving root development, water-holding capacity11. and nutrient uptake12. Also, the Fenugreek growth factors and Trigonelline biosynthesis can be affected by Trichoderma strains13. Moreover, it was recently reported that fenugreek inoculation with Trichoderma treatment increased trigonelline accumulation, which acts as a strong inducing factor for secondary metabolite production14.
MeJA may be considered a promising elicitor for diosgenin production in fenugreek plants15. Simultaneously, Jasmonic Acid (JA) has synergistic and antagonistic effects with abscisic acid (ABA), ethylene (ET), salicylic acid (SA), and other plant hormones in the process of resisting environmental stress16. Aloe leaf extract (ALE) has been used to improve the vegetative growth of Abelmoschuses culentus, Oenothera biennis and Majorana hortensis17. El Sherif18 suggested that ALE is an efficient alternative source to improve the growth of Populus trees grown under in vitro conditions. Many biologically active chemicals can be found in aloe vera, more scientific research into this medicinal plant would be beneficial, as would promoting its widespread use19. Mirihagalla and Fernando20 reported that aloe vera could be used in developing natural root inducing substance and resolving and reducing the risk of chemical toxicity in plants due to plant growth regulators, since, aloe vera leaves produce more rooting hormones after separated from the plant.
Fenugreek seed quantity and quality can be improved via cultivation, irrigation, and harvest management practices14. However, the potential for genetic advancement and successful conservation and management of germplasm resources in Trigonella sp. is limited due to a lack of knowledge on genetic variation and intra-specific relatedness21. Random amplified polymorphic DNA (RAPD) is a PCR-based DNA amplification technique that is widely used in the field of applied plant breeding. RAPD is a simple and efficient tool that does not require full sequence information21. Karp et al22, and can be used for genetic diversity analyses23. Furthermore, RAPD techniques are advantageous because they require only a small quantity of DNA and can uncover a large number of polymorphisms24. The relative genetic distances across fenugreek types did not always correspond to the geographic distance of their growth. And the genotypes that fall into distinct groupings show that there is genetic variation among the members of different clusters. Because the members of distinct clusters are genetically diverse, they can be crossed for future crop improvement25. Other studies, reported that the SRAP technique is effective in detecting genetic diversity and population structure of Iranian fenugreek varieties26.
The aim of the present study was to conduct random amplified polymorphic DNA (RAPD)-PCR to investigate the extent of genetic polymorphisms and to correlate genetic relatedness in fenugreek plants following seed treatments with various combinations of T. harzianum, MeJA, and AVG.
Fenugreek seed preparation
T. foenum-graecum L. seeds were purchased from the local market in Al-Hilla City, Babylon Province, Iraq. We selected homogeneous one-year-old fenugreek seeds with no cracks or other visible deformations. Seeds were suspended in distilled water to assess viability; seeds that settled to the bottom were selected and surface sterilized using 2% NaOCl for 5 min. Seeds were rinsed several times with sterile distilled water and then dried on sterile filter paper (filter papers were sterilized by enveloping them in foil and placed in an autoclave basket at 121°C for 15 minutes) under a laminar flow hood for 30 min (Allegrucci et al)27.
Trichoderma harzianum source and culture
Two T. harzianum isolates, Th-1 and Th-2 (distinguished by phenotypic characteristics), were obtained from Dr. Jawad K. Abood Aljanabi, Proffesor of Advanced Mycology, College of Science, the University of Babylon in September 2020.
To prepare T. harzianum cultures, autoclaved Potato Dextrose Agar medium (PDA)-manufacture by Titan Biotech Limited/ India, was poured into sterilized test tubes and allowed to solidify at an angle. Loopful inoculums of T. harzianum isolates were transferred to PDA slants and incubated at 25°C for 7 d. Pure cultures were maintained at 4°C until use28.
To prepare spore suspensions, T. harzianum isolates were cultured on Petri plates containing sterilized PDA, incubated in the dark at 26°C for 7 days, and then placed under continuous light to promote sporulation29. Fungal spores were obtained by adding 10 ml of sterilized distilled water containing 0.1% Tween 80 (v/v) to each Petri dish to decrease the surface tension and promote the release of fungal spores (conidia). The colony surfaces were scraped gently with a sterile lancet30 to prepare the suspensions, and the mixture was filtered through a three-layer muslin cloth. The filtrate was collected, and the spore suspension concentration was adjusted to 1.0 × 108 conidia per milliliter using a hemocytometer under a light microscope31,32.
Soil preparation and planting
Peat moss and sand were mixed 1:1, wet with distilled water, and packed in appropriate plastic bags for autoclaving. Approximately 50 g of soil was transferred into each plastic container (3.5 cm diameter at the bottom and 5.5 cm diameter at the top; 5.5 cm depth).
Seed treatment
A preliminary experiment was conducted with and without 2% Arabic Gum to ensure the gum did not affect seed viability using three replicates. No differences in percentage germination of fenugreek seeds were recorded. Ten treatments were prepared using two isolates of T. harzianum (Th-1 and Th-2) at concentration shown in 2.2, while 100 µL/L (0.01%) was used from methyl jasmonate (95% MeJA; Sigma-Aldrich and 100% from fresh- natural Aloe vera gel (AVG) was prepared. Fenugreek seeds were dipped separately (25 seeds each) for two hours in each of the follows treatments:
Control, distilled water;
Th-1;
Th-2;
20 µM MeJA;
Th-1 + 10 µM MeJA;
Th-2 + 10 µM MeJA;
AVG (100% concentration);
AVG (50%) + 10 µM MeJA;
Th-1 + AVG;
Th-2 + AVG.
In all experimental formulations, 2% Arabic Gum was used to ensure the adhesion of T. harzianum conidia to fenugreek seeds. All seeds were drained and dried in a laminar airflow hood for two hours33,34.
Five fenugreek seeds were sown per mini pots in a growth chamber at 25±1°C temperature and at a relative humidity of 70%. After two weeks, germination took place within three days of sowing, and the standard germination rate was 98%.
Experimental treatment and design
Germinated seeds were transferred after two weeks to plastic pots (12 cm diameter at the top and 7 cm diameter at the bottom; 12 cm depth) filled with 200 g of autoclaved soil, as five seedlings per pot, in a greenhouse with 17-hour light periods and a light intensity of 200 mol quanta m-2 s-1, and day/night temperatures of 25°C/18°C. In four replicates, the pot experiment was set up in a completely randomized designs following35,36 with modification. A total of 40 pots, each containing four plants, were used in the experiment. Seedlings were watered with an equal amount of tap water when required
Growth parameters
Fenugreek plants were harvested soon after flowering at 45 days old. Plants were removed from the pots, and the shoots and roots were separated and washed with distilled water three times, dried, and weighed. The number of leaves, chlorophyll content (using a self-calibrating chlorophyll meter- model SPAD 502), shoot length, and fresh weight of the shoots were all measured. The length of the roots and shoots was measured from the soil line to the top of the shoot37 and then weighed using a sensitive balance.
Genotypic identification
Sampling and DNA extraction
Two plants were used for DNA extraction to ascertain the changes in genetic diversity in fenugreek using RAPD markers. DNA from fenugreek plants was obtained and purified using an extraction and purification kit (Wizbio, South Korea).
Primers
Five arbitrary or “random” primers were obtained from Bioneer (Integrated DNA Technologies, USA). Fenugreek plant DNA was tested for single primers for RAPD-PCR amplification (Table 1).
Table (1):
Primer sequences used in RAPD-PCR amplification.
Primer name |
Sequence |
|---|---|
OPW-2 |
5`ACCCCGCCAA3` |
OPA-18 |
5’AGGTGACCGT3’ |
OPA-5 |
5’AGGGGTCTTG3’ |
OPF-5 |
5’CCGAATTCCC3’ |
OPX-8 |
5’CAGGGGTGGA3’’’ |
RAPD-PCR amplification
Reaction mixtures with a final volume of 30 μl were prepared with 5 μl of a single primer, 12.5 µl of Green Master Mix, 3 μl of genomic DNA, and also the reaction volume was rounded to 30 µl with the addition of 8.5 µl of nuclease-free water. The reaction mixture was dispensed in a thermo-cycler (Eppendorf) programmed for 3 min at 94°C, followed by 42 cycles of 1 min at 94°C, 1 min at 42°C, and 3 min at 72°C; then a final extension of 7 min at 72°C. Amplification products were electrophoresed in 2% agarose gels and visualized via staining with ethidium bromide. Standard molecular markers were conjointly enclosed in every electrophoresis run. Images of the ultraviolet trans-illuminated gels were captured (Gel documentation system, UV source\ Cleaver Scientific – UK)
Phylogenetic analysis
Locations of scorable RAPD bands were remodeled to a matrix of binary characters (“1” for the existence of a band at a certain location; “0” for the absence of a band at the location). Cluster analysis was conducted based on Jaccard’s similarity coefficient matrices, calculated from RAPD data to create a dendrogram of fenugreek genotypes. A phylogenetic tree was generated from the similarity matrices using the un-weighted pair-group method arithmetic (UPGMA) average cluster analysis (Program \ past3).
Statistical analysis
Data were subjected to mono-factorial or bi-factorial analysis of variance (ANOVA) depending upon the experimental design. Treatment means were compared using a least significant difference range test at P < 0.05 with standard error.
Fenugreek growth
Fenugreek plant growth varied among treatments (Table 2). The results are expressed as the mean of four replicates revealed that treatment with Th-1, MeJA, AVG, and their combinations enhanced the growth capacity of fenugreek plants. According to our findings, the Th-1 and Th-2 isolates reacted differently to the growth of the fenugreek plant.
Table (2):
Effects of fenugreek (Trigonella foenum-graecum) seed treatment with Trichoderma harzianum isolates (Th-1 and Th-2) in combination with methyl jasmonate (MeJA) and/or Aloe vera gel (AVG) on root length, shoot height, shoot fresh weight, number of true leaves, and chlorophyll content. Data are means ± standard errors of four measurements.
Treatments |
Root length |
Shoot height |
FW of of shoots |
No. of true leaves |
Chlorophyll content |
|---|---|---|---|---|---|
Control |
9.12 ± 0.59 |
23.30 ± 1.09 |
2.10 ± 0.27 |
9.20 ± 0.47 |
49.07 ± 0.59 |
-1 |
14.87 ± 0.65 |
32.00 ± 0.70 |
3.50 ± 0.29 |
12.00 ± 0.40 |
56.12 ± 0.62 |
Th-2 |
8.35 ± 0.69 |
19.57 ± 0.33 |
1.10 ± 0.29 |
6.50 ± 0.86 |
40.94 ± 0.33 |
MeJA |
15.55 ± 0.68 |
29.77 ± 1.08 |
3.07 ± 0.21 |
20.25 ± 0.47 |
53.80 ± 0.51 |
Th-1+MeJA |
16.75 ± 0.75 |
30.12 ± 0.31 |
4.30 ± 0.45 |
20.50 ± 0.64 |
56.15 ± 0.55 |
Th-2+MeJA |
10.0 ± 0.40 |
25.05 ± 0.55 |
1.60 ± 0.13 |
8.75 ± 0.25 |
48.77 ± 0.45 |
AVG |
14.95 ± 0.68 |
31.92 ± 0.07 |
7.90 ± 0.49 |
23.50 ± 0.64 |
59.25 ± 0.61 |
Th-1+AVG |
14.00 ± 1.35 |
33.75 ± 0.47 |
6.52 ± 0.57 |
28.50 ± 0.75 |
61.45 ± 0.86 |
Th-2+AVG |
10.67 ± 0.41 |
20.25 ± 0.47 |
2.11 ± 0.12 |
8.25 ± 0.47 |
45.32 ± 1.07 |
+ MeJA |
16.0 ± 0.40 |
32.32 ± 0.87 |
5.12 ± 0.09 |
29.50 ± 0.64 |
62.27 ± 0.75 |
LSD0.05 |
1.75 |
2.52 |
0.81 |
1.38 |
1.8 |
LSD, least significant difference (P < 0.05)
Root length
Root length was significantly (P < 0.001) higher in plants treated with Th-1 (14.87 cm), MeJA (15.55 cm), AVG (14.95 cm), and combined treatments of Th-1 + MeJA (16.75 cm), Th-1 + AVG (14.00 cm) and AVG + MeJA (16.0 cm) than in the control (9.12 cm), Th-2 (8.35 cm), Th-2 + MeJA (10 cm), and Th-2 + AVG (10.67 cm) treatments. Th-2 alone or in combination with other agents did not stimulate root growth.
Shoot height
The shoot height was significantly (P < 0.001) taller when seeds were treated with Th-1 alone or in combination with MeJA and AVG. The tallest plant height was recorded in Th-1 + AVG (33.75), followed by AVG + MeJA (32.32 cm), Th-1 (32.00 cm), AVG (31.92 cm), Th-1 + MeJA (30.12 cm), and MeJA (29.77 cm), which were all significantly taller than the control. The minimum height was observed in Th-2 (19.57cm) and in Th-2 + AVG (20.25 cm), which both produced shorter plants than the control (23.30 cm).
Shoot fresh weight
Compared with the control, fenugreek shoot fresh weights were significantly (P < 0.001) increased by seed treatment with Th-1, MeJA, AVG, and their combinations. AVG produced the highest shoot weight (7.90 g), followed by Th-1 + AVG (6.52 g), AVG + MeJA (5.12 g), Th-1 + MeJA (4.30 g), Th-1 (3.50 g), and MeJA (3.07 g), which were also significantly (P < 0.001) higher than those of Th-2 and its combinations. The lowest shoot fresh weight was recorded in Th-2 + MeJA (1.60 g), Th-2 (1.10 g), and the control (2.10), which were not significantly different from one another.
Number of true leaves
Significant variation was observed in the number of true leaves among treatments. The AVG + MeJA treatment produced the maximum number of leaves (29.50) followed by Th-1 + AVG (28.50), AVG (23.50), Th-1 + MeJA (20.50), MeJA (20.25), and Th-1 (12), which were significantly higher than Th-2 (6.50), Th-2 + MeJA (8.75), Th-2 + AVG (8.25), and the control (9.20 cm).
Chlorophyll percentage
Significant differences in chlorophyll percentage were observed among treatments. The maximum percentage of leaf chlorophyll was recorded in AVG + MeJA (62.27%), followed by Th-1 + AVG (61.45%), AVG (59.25%), Th-1 + MeJA (56.15%), Th-1 (56.12%). The lowest chlorophyll percentage was observed in Th-2 (40.94%) and Th-2+AVG (45.32%), which were both significantly lower than the control (49.07%). Th-2 + MeJA (48.77%) was not significantly different from the control but was significantly lower than Th-1, MeJA, AVG, and their combinations.
RAPD analysis of fenugreek
In the RAPD analysis, five RAPD primers were screened, and amplified bands were selected for further study. The size of the PCR products ranged from 100 bp to 1500 bp; the average number of polymorphic bands for each primer was 5.1 (Table 3).
Table (3):
The number of amplified products, the number of polymorphic bands, and the percentage of polymorphism obtained using five primers for random amplified polymorphic DNA (RAPD) analysis of fenugreek (Trigonella foenum-graecum) leaves.
Primer Name |
Total no. of band |
No. of polymorphic bands |
% polymorphism |
|---|---|---|---|
OPW-2 |
10 |
5 |
50 |
OPA-18 |
9 |
4 |
44.4 |
OPA-5 |
11 |
6 |
54.54 |
OPF-5 |
9 |
5 |
55.55 |
OPX-8 |
9 |
6 |
66.66 |
Total |
48 |
26 |
54.21 |
PCR amplification of DNA using the five primers for RAPD analysis produced 62 DNA fragments that could be scored in all genotypes. The total number of polymorphic bands was 26, and the average percentage of polymorphism was 54.21%. Cluster analyses were conducted using RAPD data and the similarity matrices were used to generate a dendrogram using the UPGMA method (Table 2). RAPD profiles detect alterations in genomic DNA with the use of arbitrarily primed PCR reactions, with the potential to detect treatment-induced DNA effects. However, RAPD is a qualitative method through which the nature and amount of DNA can only be speculated. The results revealed substantial effect of Th-1 of MeJA and AVG treatments and their combination on RAPD profiling in fenugreek (Fig. 1-6).
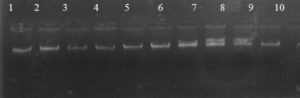
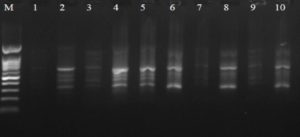
Fig. 1. The electrophoresis pattern of genomic DNA extracted from fenugreek (Trigonella foenum-graecum) leaf samples: 1, control; 2, Th-1; 3, Th-2; 4, AVG; 5, MeJA; 6, Th-1 + AVG; 7 Th-1 + MeJA; 8, Th-2 + AVG; 9, Th-2 + MeJA; 10, MeJA + AVG.
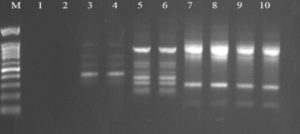
Fig. 2. Agarose gel electrophoresis of OPW-2 amplified product. The electrophoresis pattern of genomic DNA extracted from fenugreek (Trigonella foenum-graecum) leaf samples: 1, control; 2, Th-1; 3, Th-2; 4, AVG; 5, MeJA; 6, Th-1 + AVG; 7 Th-1 + MeJA; 8, Th-2 + AVG; 9, Th-2 + MeJA; 10, MeJA + AVG; M, DNA size marker. Electrophoresis conditions: 1.5% agarose concentration, 75 V, 20 mA for 120 min, stained with ethidium bromide.
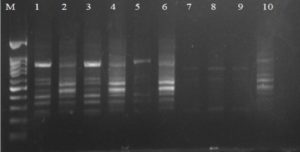
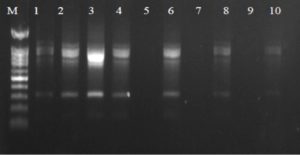
Fig. 3. Agarose gel electrophoresis of OPA-18 amplified product. The electrophoresis pattern of genomic DNA extracted from fenugreek (Trigonella foenum-graecum) leaf samples: 1, control; 2, Th-1; 3, Th-2; 4, AVG; 5, MeJA; 6, Th-1 + AVG; 7 Th-1 + MeJA; 8, Th-2 + AVG; 9, Th-2 + MeJA; 10, MeJA + AVG; M, DNA size marker. Electrophoresis conditions: 1% agarose concentration, 75 V, 20 mA for 120 min, stained with ethidium bromide.
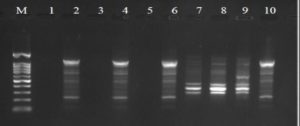
Fig. 4. Agarose gel electrophoresis of OPA-5 amplified product. The electrophoresis pattern of genomic DNA extracted from fenugreek (Trigonella foenum-graecum) leaf samples: 1, control; 2, Th-1; 3, Th-2; 4, AVG; 5, MeJA; 6, Th-1 + AVG; 7 Th-1 + MeJA; 8, Th-2 + AVG; 9, Th-2 + MeJA; 10, MeJA + AVG; M, DNA size marker.
Fig. 5. Agarose gel electrophoresis of OPF-5 amplified product. The electrophoresis pattern of genomic DNA extracted from fenugreek (Trigonella foenum-graecum) leaf samples: 1, control; 2, Th-1; 3, Th-2; 4, AVG; 5, MeJA; 6, Th-1 + AVG; 7 Th-1 + MeJA; 8, Th-2 + AVG; 9, Th-2 + MeJA; 10, MeJA + AVG; M, DNA size marker. Electrophoresis conditions: 1% agarose concentration, 75 V, 20 mA for 120 min, stained with ethidium bromide.
Fig. 6. Agarose gel electrophoresis of OPX-8 amplified product. The electrophoresis pattern of genomic DNA extracted from fenugreek (Trigonella foenum-graecum) leaf samples: 1, control; 2, Th-1; 3, Th-2; 4, AVG; 5, MeJA; 6, Th-1 + AVG; 7 Th-1 + MeJA; 8, Th-2 + AVG; 9, Th-2 + MeJA; 10, MeJA + AVG; M, DNA size marker. Electrophoresis conditions: 1% agarose concentration, 75 V, 20 mA for 120 min, stained with ethidium bromide.
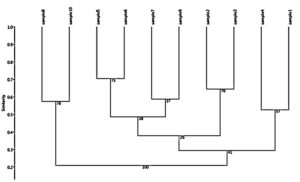
Fig. 7. Dendrogram analysis showing the phylogenetic diversity of fenugreek (Trigonella foenum-graecum) leaf samples: 1, control; 2, Th-1; 3, Th-2; 4, AVG; 5, MeJA; 6, Th-1 + AVG; 7 Th-1 + MeJA; 8, Th-2 + AVG; 9, Th-2 + MeJA; 10, MeJA + AVG; characterized by random amplified polymorphic DNA (RAPD) markers (OPX-8, OPA-18, OPA-5, OPW-2, OPF2).
UPGMA analysis for the dendrogram made based on the RAPD data generated by primers (OPX-8, OPW-2, OPA-18, OPA-5, OPF-2) were performed and shown in Fig. (7). Analysis showed that the 10 Trigonella foenum-graecum L were grouped into three clustered and five classes. The first class comprised the sample 1 & sample 4 a second class included sample 3 & sample 2, and the third class include sample 9 and sample 7 and the last class comprised of sample 6 & sample 5, second cluster comprised of one class include sample 10 & sample 8.
Trichoderma is one of the most economically important microorganisms in the agro-ecosystem industry, influencing soil health and crop productivity. The anti-pathogenic, plant-growth promoting, and bioremediating effects of Trichoderma increase crop yield in terms of both biological and economic output38.
The present experiment demonstrated the substantial effects of T. harzianum on the growth of fenugreek plants. However, the effects of T. harzianum were dependent on the isolate applied, since differential reaction patterns were observed between the two T. harzianum isolates toward the growth parameters of the studied plant. The Th-1 strain, alone and in combination with MeJA and AVG, produced an increase in fenugreek root length, shoot height, shoot fresh weight, number of true leaves, and chlorophyll content, reflecting growth improvement consistent with previous reports39,40. Several mechanisms for plant growth stimulation via Trichoderma have been described. When root endophytic fungi in the genus Trichoderma colonize the roots of crop plants, upregulation of genes and pigments that enhance the photosynthesis is observed41. It was reported42 that the release of soluble compounds by Trichoderma may exert beneficial effects on microbial groups cohabiting the rhizosphere, compounding the positive impact on plants. Moreover, Trichoderma produces auxins that can stimulate plant growth and root development43. Trichoderma can also improve plant uptake of copper, phosphorus, iron, cobalt, arsenic, cadmium, zinc, boron, aluminum, manganese, and sodium44 by increasing the bioavailability of these nutrients45. It is most likely that Trichoderma stimulates growth by influencing the balance of hormones such as IAA, gibberellic acid and ethylene46.
Conversely, the Th-2 strain negatively impacted fenugreek growth, the Authors have no explanation for this phenomenon which never previously demonstrated. Our findings demonstrate that combination treatments with Th-1 and MeJA or AVG produced significantly higher fenugreek growth than Th-1 alone. Li et al47, reported increases in plant height and biomass as a result of MeJa application in Helianthus annuus (sunflower), Solanum lycopersicum (tomato), and Glycine max (soybean). Another study demonstrated that MeJA has an impact on gene expression and stomatal opening48. The present study revealed a substantial effect of AVG on the growth of fenugreek whether used alone or in combination with MeJA or Th-1 which possibly due to its content of β-polysaccharides, amino acids, vitamins, glycoproteins, soluble sugars, anthraquinones, and enzymes49,50. El Sherif16 concluded that the leaf extract of Aloe Vera is an efficient alternative source to improve the growth of Populus clones. T. harzianum plays an important role in inducing the expression of defense genes in infected plants51. They used biotic and abiotic agents to suppress Fusarium oxysporum, which infects tomato plants.
The observed DNA profile modifications, such as changes in band intensity and loss of bands, may be due to the changes in oligonucleotide priming sites due to genomic rearrangements, DNA damage in the primer binding sites, or the presence of DNA photoproducts, which can block or reduce the polymerization of DNA in the PCR reaction52.
RAPD markers have been used to determine genetic variation and relationships among many plants53. RAPD molecular markers are useful tools for assessing genetic variability because they are abundant, very reproducible, highly polymorphic, highly informative, and simple54,55. In this study, the genetic diversity among some Iraqi populations of fenugreek was investigated using RAPD markers.
The five selected primers are sensitive enough to evaluate the diversity and intergenic or intragenic phylogenetic relationship among fenugreek plants. These markers can help in understanding the population structure, variation in genetic diversity within the species related to the geographic range, mode of reproduction, mating system, seed dispersal, and fecundity56. The genetic variability that detected in the present studies may be due to these prevalent background factors as the genotype of fenugreek. Dangi et al57, reported 70–72% of polymorphisms using RAPD and ISSR markers to analyze fenugreek collected from different countries. Other researchers58, observed an average of 76.78% polymorphism using RAPD in the 30 accessions collected from a western part of India.
Finally, the investigation has demonstrated that cluster analysis could be profitably used in unraveling the genetic variation within the accessions and the two molecular markers, RAPD could be used as an effective tool to evaluate genetic diversity and assess the genetic relationship in fenugreek.
The positive impact of combinations of T. harzianum, VAG, and MeJA on fenugreek plant growth was not previously reported, according to the researcher’s knowledge. Treatments with strain Th-1 produced higher plant shoot and root growth and increased biomass and chlorophyll production. However, Th-2 negatively impacted plant growth. Some combinations show potential as fertilizer alternatives to stimulate fenugreek growth and production; however, our results demonstrate the importance of T. harzianum strain selection. Moreover, our findings show that RAPD can detect treatment-induced differences in fenugreek.
The current study also revealed that some primers failed to give different bands to the treatments under study, which could be due to a lack of suitable conditions for the operation of the polymerase chain reaction, such as temperature or nucleic acid quantity, GC ratio to primers, or the number of cycles used.
ACKNOWLEDGMENTS
The authors greatly thank the Deanship of the College of Science and the Head of the Department of Biological Sciences for providing the necessary facilities for the implementation of this work. The authors sincerely thank Dr Zahraa Ossam for her generous technical assistance.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
ETHICS STATEMENT
This article does not contain any studies with human participants or animals performed by any of the authors.
AVAILABILITY OF DATA
All datasets generated or analyzed during this study are included in the manuscript.
- Aasim M, Baloch FS,Nadeem M A, Bakhsh A, Sameeullah M, Day S. Fenugreek (Trigonella foenum-graecum L.): An underutilized edible plant of modern world. In Global perspectives on underutilized crops. Springer, Cham. 2018;381-408.
Crossref - Wani SA, Kumar P. Fenugreek: A review on its nutraceutical properties and utilization in various food products. Journal of the Saudi Society of Agricultural Sciences. 2018;17(2):97-106.
Crossref - Sundaram S, Purwar S. Assessment of genetic diversity among fenugreek (Trigonella foenum-graecum L.), using RAPD molecular markers. Journal of Medicinal Plants Research, 2011;5(9):1543-1548.
- Bienkowski T, Zuk-Golaszewska K, Kaliniewicz J, Golaszewski J. Content of biogenic elements and fatty acid composition of fenugreek seeds cultivated under different conditions. Chilean Journal of Agricultural Research. 2017;77(2):134-141.
Crossref - Shailajan S, Sayed N, Menon S, Singh A, Mhatre M. A validated RP-HPLC method for quantitation of trigonelline from herbal formulations containing Trigonella foenum-graecum (L.) seeds. Pharmaceutical Methods. 2011;2(3):157-160.
Crossref - Jesus M, Martins AP, Gallardo E, Silvestre S. Diosgenin: recent highlights on pharmacology and analytical methodology. J Anal Methods Chem. 2016;2016:4156293.
Crossref - Goyal S, Gupta N, Chatterjee S. Investigating therapeutic potential of Trigonella foenum-graecum L. as our defense mechanism against several human diseases. J Toxicol. 2016;2016:1250387.
Crossref - Syed QA, Rashid Z, Ahmad MH, et al. Nutritional and therapeutic properties of fenugreek (Trigonella foenum-graecum): a review. International Journal of Food Properties. 2020;23(1):1777-1791.
Crossref - Zeilinger S, Gruber S, Bansal R, Mukherjee PK. Secondary metabolism in Trichoderma-chemistry meets genomics. Fungal Biology Reviews. 2016;30(2):74-90.
Crossref - Rajput AQ, Khanzada MA, Shahzad S. Effect of different organic substrates and carbon and nitrogen sources on growth and shelf life of Trichoderma harzianum. J Agr Sci Tech. 2014;16:731-745.
- Lombardi N, Caira S, Troise AD, et al. Trichoderma applications on strawberry plants modulate the physiological processes positively affecting fruit production and quality. Front Microbiol. 2020;11:1364.
Crossref - Harman GE, Howell CR, Viterbo A, Chet I, Lorito M. Trichoderma species-opportunistic, a virulent plant symbiont. Nat Rev Microbiol. 2004;2:43-56.
Crossref - Yildirim E, Taylor AG, Spittler TD. Ameliorative effects of biological treatments on growth of squash plants under salt stress. Sci Hortic (Amst.). 2006;111(1):1-6.
Crossref - Hosseini Z, Hassanloo T, Kowsari M, Majidian M. Trigonelline as an anti-diabetic metabolite increased in inoculated fenugreek by Trichoderma. Advanced Research in Microbial Metabolites & Technology. 2019;2(1):129-139.
- Chaudhary S, Chikara SK, Sharma MC, et al. Elicitation of diosgenin production in Trigonella foenum-graecum (fenugreek) seedlings by methyl jasmonate. Int J Mol Sci. 2015;16(12):29889-29899.
Crossref - Wang J, Song L, Gong X, Xu, J, Li M. Functions of jasmonic acid in plant regulation and response to abiotic stress. Int J Mol Sci. 2020;21(4):1446.
Crossref - Padmaja CK, Kowsalya B, Seethalakshmi C. Efficacy of Aloe vera leaf powder as bio stimulant in enhancing the growth and yield of Lady’s Finger (Abelmoschus esculentus L.). Research on Crops. 2007;8:395-397.
- El Sherif F. Aloe vera leaf extract as a potential growth enhancer for Populus trees grown under in vitro conditions. American Journal of Plant Biology. 2017;(2)4:101-105.
Crossref - Christaki EV, Florou-Paneri PC. Aloe vera: a plant for many uses. J Food Agric Environ. 2010;8(2):245-249.
- Mirihagalla MKPN, Fernando KMC. Effect of Gel for Inducing Rooting of Stem Cuttings Aloe vera and Air layering of Plants. Journal of Dry Zone Agriculture. 2020;6(1):13-21.
- Raja V, Wani UM, Kudesia R, John R. Estimation of genetic variability in Trigonella cultivars by RAPD analysis. Int J Biotechnol Res. 2015;3:021-7.
- Karp, A., S. Kresovich, K.V. Bhat, W.G. Ayad and T. Hodgkin. Molecular tools in plant genetic resources conservation: a guide to the technologies. IPGRI Technical Bulletin No. 2. 1997. International Plant Genetic Resources Institute, Rome, Italy .
- Li L, Xiaoying, Z, Klocke E. Variation in some Lycopersicon esculentum and Capsicum annuum cultivars revealed by RAPD and AFLP markers. Guangxi Sci. 2004;11:249-257.
- Carelli BP, Gerald LTS, Grazziotin FG, Echeverrigaray S. Genetic diversity among Brazilian cultivars and landraces of tomato Lycopersicon esculentum Mill. revealed by RAPD markers. Genet Resour Crop Evol. 2006;53:395-400.
Crossref - Mamatha NC, Tehlan SK, Srikanth M, Ravikumar T, Yashveer S, Kumar M. Assessment of Genetic Diversity among Fenugreek (Trigonella foenum-graecum L.) Genotypes using ISSR Markers. Int J Curr Microbiol App Sci. 2017;6(6):2565-2572.
Crossref - Amiriyan M, Shojaeiyan A, Yadollahi A, Maleki M, Bahari Z. Genetic diversity analysis and population structure of some Iranian Fenugreek (Trigonella foenum-graecum L.) landraces using SRAP Markers. Molecular Biology Research Communications. 2019;8(4):181-190.
- Allegrucci N, Velazquez MS, Russo ML, Perez ME, Scorsetti AC. Endophytic colonisation of tomato by the entomopathogenic fungus Beauveria bassiana: the use of different inoculation techniques and their effects on the tomato leafminer Tuta absoluta (Lepidoptera: Gelechiidae). Journal of Plant Protection Research. 2017;57(4):205-211.
Crossref - Obaid AJ, Al-Janabi JK, Taj-Aldin WR. Bioactivities of anethole, astragalin and cryptochlorogenic acid extracted from anise oil and Moringao oleifera on the keratinase gene expression of Trichophyton rubrum. J Pure Appl Microbiol. 2020;14(1):615-626.
Crossref - Mohsen LY, Kadhim HJ, Al- Janabi JKA, Yassiry ZAN. Alternative culture media for growth and sporulation of Trichoderma harzianum. Pak J Biotechnol. 2017;14(4):587-593.
- Jansson HB, Jeyaprakash A, Zuckerman BM. Control of root-knot nematodes on tomato by the endoparasitic fungus Meriaconiospora. J Nematol. 1985;17:327-329.
- Sen A, Batra A. Evaluation of Antimicrobial Activity of Different Solvent Extracts of Medicinal Plant: Melia Azedarach L. Int J Curr Pharm Res. 2012;4(2):67-73.
- Obaid AJ, Al-Janabi JKA, Taj-Aldin WR. Chemical Composition and Bioactivity Characteristics of Pimpinella Anisum Essential Oil against Trichophyton rubrum. Journal of Global Pharma Technology. 2017;8(9):44-56.
- Anis M, Zaki MJ, Abbasi MW. Effect of seed coating with Trichoderma species on colonization of Macrophomina phaseolina and the growth of sunflower under field conditions. Int J Biol Biotechnol. 2013;10:207-212.
- Ruttanaruangboworn A, Chanprasert W, Tobunluepop P, Onwimol D. Effect of seed priming with different concentrations of potassium nitrate on the pattern of seed imbibition and germination of rice (Oryza sativa L.). Journal of Integrative Agriculture. 2017:16(3):605-613.
Crossref - Zhang S, Xu B, Gan Y. Seed treatment with Trichoderma longibrachiatum T6 promotes wheat seedling growth under NaCl stress through activating the enzymatic and non-enzymatic antioxidant defense systems. Int J Mol Sci. 2019;20(15):3729.
Crossref - Sellal Z, Touhami AO, Chliyeh M, et al. Effect of seeds treatment with Trichoderma harzianum on argan plants growth. Plant Cell Biotechnology and Molecular Biology. 2020;21:69-77.
- McGovern RJ, Datnoff LE, Tripp L. Effect of mixed infection and irrigation method on colonization of tomato roots by Trichoderma harzianuma and Glomus intraradix. Proc Fla State Hort Soc. 1992;105:361-363.
- Thapa S, Rai N, Limbu AK, Joshi A. Impact of Trichoderma sp. in Agriculture: A Mini-Review. Journal of Biology and Today’s World. 2020;9(7):1-5.
- Dubey SC, Suresha M, Singha B. Evaluation of Trichoderma species against Fusarium oxysporum f. sp. ciceris for integrated management of chickpea wilt. Bioogical Control. 2007;40(1):118-127.
Crossref - Islam MM, Shahid SB, Akter A, Hossain MS, Bhuiyan MSU. Effect of indigenous Trichoderma strains on growth of tomato seedlings. Int J Expt Agric. 2020;10(2):1-6.
- Harman GE, Doni, F, Khadka RB, Uphoff N. Endophytic strains of Trichoderma increase plants’ photosynthetic capability. J Appl Microbiol. 2019;130(2):529-546.
Crossref - Herrera-Jimenez E, Alarcon A, Larsen J, Ferrera-Cerrato R, Cruz-Izquierdo S, Ferrera-Rodriguez MR. Comparative effects of two indole-producing Trichoderma strains and two exogenous phytohormones on the growth of Zea mays L., with or without tryptophan. Journal of Soil Science and Plant Nutrition. 2018;18(1):188-201.
Crossref - Contreras-Cornejo H A, Macias-Rodriguez L, Cortes-Penagos C, Lopez-Bucio J. Trichoderma virens, a plant beneficial fungus, enhances biomass production and promotes lateral root growth through an auxin-dependent mechanism in Arabidopsis. Plant Physiology. 2009;149(3):1579-1592.
Crossref - Yedidia I, Benhamou N, Chet I. Induction of defense responses in cucumber plants (Cucumis sativus L.) by the biocontrol agent Trichoderma harzianum. Appl Environ Microbiol. 1999;65:1061-1070.
Crossref - Altomare C, Norvell WA, Bjorkman T, Harman GE. Solubilization of phosphates and micronutrients by the plant-growth-promoting and biocontrol fungus Trichoderma harzianum Rifai 1295-22. Appl Environ Microbiol. 1999;65:2926-2933.
Crossref - Stewart A, Hill R. Applications of Trichoderma in plant growth promotion. Biotechnology and Biology of Trichoderma. 2014;415-428.
Crossref - Li C, Wang P, Menzies NW, Lombi, E, Kopittke PM. Effects of methyl jasmonate on plant growth and leaf properties. Journal of Plant Nutrition and Soil Science. 2018;181(3):409-418.
Crossref - Kriegs B, Jansen, M, Hahn K, et al. Cyclic monoterpene mediated modulations of Arabidopsis thaliana phenotype. Plant Signal Behav. 2010;5:832-838.
Crossref - Chang X L, Chen BY, Feng YM. Water-soluble polysaccharides isolated from skin juice, gel juice and flower of Aloe vera Miller. J Taiwan Inst Chem E. 2011;42(2):197-203.
Crossref - Lucini L, Pellizzoni, M, Pellegrino R, Molinari GP, Colla G. Phytochemical constituents and in vitro radical scavenging activity of different Aloe species. Food Chem. 2015;170:501-507.
Crossref - Alnuaimy AA, AL-Janab JK, ALobaidi LA, Marjan AF. The expression of resistance genes in tomato induced by abiotic and biotic factors against Fusarium oxysporum f. sp. Lycopersici. Int J Chem Tech Res. 2017;10(6):838-850.
- Nelson JR, Lawrence CW, Hinkle DC. Thymine-thymine dimmer bypass by yeast DNA-polymerase-zeta. Science. 1996:272(5268):1646-1649.
Crossref - Pour-Aboughadareh A, Mohmoudi M, Ahmadi J, Moghaddam M, Mehrabi AA, Alavikia SS. Agro-morphological and molecular variability in Triticum boeoticum accessions from Zagros Mountains, Iran. Genet Resour Crop Evol. 2017;64:545-556.
Crossref - Guo DL, Zhang JY, Liu CH. Genetic diversity in some grape varieties revealed by SCoT analyses. Mol Biol Rep. 2012;39(5):5307-5313.
Crossref - Hamidi H, Talebi R, Keshavarzi F. Comparative efficiency of functional gene based markers, Start Codeon Targeted Polymorphism (SCoT) and Conserved DNA derived Polymorphism (CDDP) with ISSR markers for diagnostic fingerprinting in wheat (Triticuma estivum L.). Cereal Res Commun. 2014;42(4):558-567.
Crossref - Pasquet RS. Allozyme diversity of cultivated cowpea, Vigna unguiculata (L.) walp. Theor App Genet. 2002;101:211-219.
Crossref - Dangi RS, Lagu MD, Choudhary LB, Ranjekar PK, Gupta VS. Assessment of genetic diversity in Trigonella foenum-graecum and Trigonella caerulea using ISSR and RAPD markers. BMC Plant Biology. 2004:4:13.
Crossref - Tomar RS, Parakhia MV, Rathod VM, Thakkar GR, Golakiya BA. A comparative analysis of RAPD and ISSR markers for studying genetic diversity in Trigonella foenum-graceum genotypes. Res J Biotech. 2014;9:89-95.
© The Author(s) 2021. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.