ISSN: 0973-7510
E-ISSN: 2581-690X
Flacourtia indica (Burm. F). Merr., a plant widely used in ethnomedicine, was investigated for its antibacterial properties and chemical composition using preparative thin-layer chromatography (TLC) bioautography, gas chromatography-mass spectrometry (GC-MS), and nuclear magnetic resonance (NMR) spectroscopy. Bioautography was performed on silica gel-coated TLC plates against Streptococcus pneumoniae, Vibrio cholerae, and Escherichia coli. The ethanolic extract exhibited a clear inhibition zone against E. coli, while the chloroform extract showed significant inhibition against S. pneumoniae. GC-MS analysis revealed 48 compounds in the ethanolic extract and 20 in the chloroform extract. Major compounds identified in the chloroform fraction included sulfurous acid hexyl nonyl ester, phenol derivatives, phthalic acid esters, siloxanes, heneicosane, and piperidinone derivatives. To further confirm the bioactive constituents, 1H NMR spectroscopy was performed on bands exhibiting antibacterial activity. The analysis indicated the presence of aliphatic acyclic, β-substituted aliphatic, and α-monosubstituted aliphatic compounds. These findings suggest that the chloroform extract of F. indica contains several aliphatic group chemicals with notable antibacterial activity. The study highlights the potential of isolating, purifying, and characterizing these compounds for pharmaceutical applications, reinforcing the ethnomedicinal relevance of F. indica as a source of bioactive molecules.
Streptococcus pneumoniae, Vibrio cholerae, TLC-Bioautography, Gas Chromatography-mass Spectrometry (GC-MS), Nuclear Magnetic Resonance (1H-NMR)
Since ancient times, the biological activity of plant extracts have been evaluated. Medicines made from plants have greatly enhanced human health and wellbeing. The demand for novel antimicrobials that can successfully combat drug-resistant microorganisms has skyrocketed.1 Secondary metabolites are mostly responsible for the antibacterial qualities of plant materials.1,2 The primary classes of plant antimicrobial compounds include phenols and polyphenols (flavonoids, quinones, tannins, coumarins, etc.), terpenoids, alkaloids, saponins, steroids, lectins, fatty acids, and polypeptides.1 The purpose of the current study was to investigate Flacourtia indica a medicinal plant for phytochemical and antibacterial properties.
Flacourtia indica, the governor’s plum or Indian plum, is a member of the Flacourtiaceae family. A bushy shrub with spiky branches, it has been recognized for its medicinal properties and has been reviewed as an endemic vascular plant limited to the Eastern Ghats of India, namely Odisha.3 It is utilized as indigenous medicine for a number of illnesses and has a lengthy folklore history. It has been discovered that the phytochemical-rich plant F. indica possesses a number of biological qualities, such as antibacterial, antifungal, antioxidant, and anticancer effects.4 Its antibacterial, antifungal, antioxidant, antimalarial, anti-diabetic, analgesic, anti-inflammatory, and antipyretic properties have all been validated in studies to demonstrate its effectiveness against human infections. The analgesic, anti-inflammatory, and antipyretic effects of Flacourtia indica’s ethanolic extract have also been investigated.5 Forty-one different secondary metabolites with antibacterial, antifungal, antioxidant, anti-cancer, and anti-proliferative qualities have been identified in Flacourtia indica root extract. According to Al Bashera et al.6 these extracts have also demonstrated anti-proliferative efficacy against a lung cancer cell line. Thirteen phenolic glycosides and fourteen additional compounds were discovered from the methanolic extract of Flacourtia indica leaves, twelve of which were composed of different oxidized forms of pyrocatechuic acid.7 For more research on the possible health advantages of F. indica, it is imperative to examine these bioactive components.
Crude ethanol and chloroform extracts of F. indica have shown strong antibacterial activity against a variety of bacterial strains, as demonstrated in previous investigations by our researchers. The antibacterial activity of the chromatographic separated antimicrobial potential of tested plant species has not been evaluated using TLC bioautographic techniques, despite the fact that they are commonly used for evaluating the antimicrobial potential of plant materials, according to a thorough review of the literature on this plant. In order to investigate the phytochemical contents of ethanol and chloroform extracts of F. indica which showed notable antibacterial action, the TLC bioautography technique was applied to a test bacterial strain, followed by GC-MS and 1H NMR spectroscopy.
In the current investigation, a few unique bioactive components were discovered from the aerial portions of F. indica. Keeping in view the plant extract’s separation in TLC-IB and demonstration of antibacterial activity, 1H NMR spectroscopy was used to get exact structural information on the constituent chemicals.
Plant material and extract preparation
Plant materials were collected, identified, and prepared in accordance with procedure.8 Dr. P.C. Panda, the lead scientist at the Regional Plant Resource Centre (RPRC), Bhubaneswar, identified and validated fresh Flacourtia indica samples that were obtained from the Regional Plant Resource Centre. The plant’s fresh stem and leaves were rinsed with tap water and then distilled water, let to air dry for 15 days in the shade, then cut into small pieces and processed into a coarse powder using an electric grinder before being placed in airtight bottles for further analysis. Ethanol and chloroform were used as solvents for a 72 hour Soxhlet extraction of the coarse powder at 30 °C. Following filtering, a rotary vacuum evaporator (BUCHI TYPE, CEMC/RS/01) was used to concentrate the liquid extract, which was then kept in a sterile bottle at 4 °C for further examination.
Bacterial strains
The Microbial Type Culture Collection (MTCC), Institute of Microbial Technology (IMTECH), Chandigarh, provided the microbial cultures used in the experiment. The study employed Gram-positive bacteria, Streptococcus pneumoniae (MTCC 655), Gram-negative bacteria, Vibrio cholerae (MTCC 3900) and Escherichia coli (MTCC 1687).
TLC-Bioautography of the chromatograms
The process of bioautography is used to isolate the physiologically active ingredient in plant extract. TLC-IB, or thin-layer chromatography-agar overlay, is an experiment used to perform separation and visualization on a TLC barrier. The eluent system ethyl acetate:benzene (1:5) is employed in this study to thin-layer chromatograph ethanolic and chloroform extracts on pre-coated commercial aluminium TLC plates (Merck, silica gel 60 F254). It was decided on a suitable solvent system before the chromatographic separation. Following chromatographic separation, the resulting TLC plates were allowed to thoroughly dry at room temperature for two hours in order to guarantee that all of the solvents had been removed. The bioautography screening of the chromatographically separated fractions of the F. indica leaf was performed using conventional techniques. The produced chromatogram is immersed in a molten 1% nutritional agar bacterial solution. For the investigation, bioautography was done using a culture of Streptococcus pneumoniae, Vibrio cholerae, and Escherichia coli, which had previously demonstrated high sensitivity to the ethanolic and chloroform extracts of the aerial sections of the plant F. indica. After solidification, the plates were incubated for 24 hours at room temperature (37 °C) before staining with FCR. TLC-IB is widely used to discover physiologically active substances that may be further spectroscopically analyzed to learn more about their structure.
Gas chromatography and mass spectrometry
The chloroform and ethanolic extract of F. indica was subjected to a GC-MS analysis utilizing an Instrument MassHunter GC/MS Acquisition. This gadget operates at 230 °C and uses mass selective detectors and a detector (MS source). The auxiliary temperature (interphase) was 270 °C, while the injector temperature was 240 °C. Using a single quadrupole of version 6.00.34, the quadrupole temperature in this technique was 150 °C. Helium gas was used as the carrier gas with a flow rate of 1.5 ml/min. The split ratio was 100:1, where the mass spectral scan range was 40 to 550 amu, and the ionization voltage used was 70 eV (International). HP-5MS capillary column was used (30 mm length, 0.25 mm in diameter, 0.025 µm in film thickness at temperatures of 50-325 °C/350 °C). The spectra of the unknown component and the known components, which included the compound’s name, chemical formula, molecular weight, and structure, were compared using the NIST data library (NISTVer.17.LMS data library). The temperature programming used for the samples had an initial temperature of 80 °C and a hold period of two minutes. At a rate of 4.5 C/min, the temperature was raised to 180 °C, held for 5 minutes, and then increased to 230 °C, for a total run length of 61.5 minutes. The proportional percentage amount of each component is calculated by comparing the average peak area of each component to the total area.
NMR
NMR spectra were obtained using a Bruker Avance III 400 spectrometer fitted with a 5 mm multinuclear inverse probehead and running at a frequency of 400.13 MHz for protons. An NMR-400 MHz was employed to record the NMR spectra of the ethanol and chloroform extracts of F. indica. Chemical shifts are represented as δ values. Using CDCl3 (deuterated chloroform) as a baseline, the result graph was compared to the reference chart to identify any possible functional groups in the plant.9
TLC bioautography assay
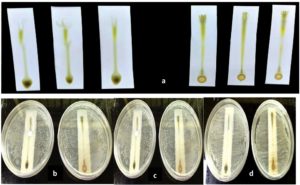
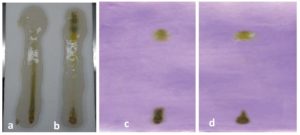
The TLC fractionation of the chloroform and ethanolic extracts of F. indica revealed the presence of four distinct bands (Figure 1a). The findings revealed high antibacterial characteristics of a fraction. Both the chloroform and ethanolic extracts showed considerable antibacterial action at the point of spotting against Gram-positive bacteria Streptococcus pneumoniae (MTCC 655) and Gram-negative bacteria Vibrio cholerae (MTCC 3900) and Escherichia coli (MTCC 1687) (Figure 1b-d). After separation, only one of the four bands remained physiologically active (Figure 1b-d). On TLC plates, light-colored dots against a purple background indicated the presence of antibacterial compounds at ZOI (Figure 2). The development of a zone of inhibition surrounding the band containing the chemical or substances with antibacterial activity served as proof of this activity.
Figure 1. Bioautography Screening: (a) Developed chromatograms of chloroform and ethanolic extract of F. indica on TLC silica gel 60 F254 plates, Bioautogram screening of chromatograms (TLC-IB) against: (b) Streptococcus pneumoniae, (c) Escherichia coli (d) Vibrio cholerae. Eluent system ethyl acetate: Benzene (1:5)
Figure 2. Bioautograms: (a) Developed bioautogram of chloroform extract with 1% molten agar S. pneumoniae solution, (b) Developed bioautogram of ethanolic extract with 1% molten agar E. coli solution, bioautogram of (c) chloroform and (d) ethanolic extract of F. indica after staining with FCR/derivatization with FCR
GC-MS analysis
The major compounds with their retention time (RT), molecular formula, and chemical structure from the GC-MS analysis of the chloroform and ethanolic extracts of F. indica aerial parts are shown in Table 1. The current study demonstrated that a total of 20 biomolecules were analyzed from the F. indica leaf extract using chloroform as a solvent, while 48 biomolecules were analyzed from the ethanolic extract.
Table (1):
Major Compounds identified in the chloroform and ethanol extract of F. indica using GC-MS
RT |
Phytocomponents |
Molecular formula |
Chemical structure |
Chemical Nature |
Biological activity |
|---|---|---|---|---|---|
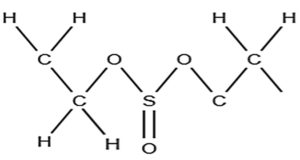
5.749 |
Sulfurous acid, hexyl nonyl ester |
C15H32O3S |
Aliphatic |
Antioxidants10, 11 |
|
8.000 |
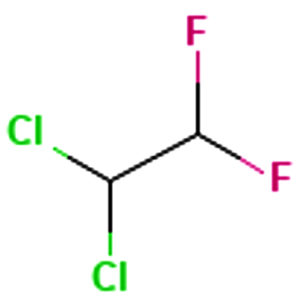
Ethane, 1,1-dichloro-2,2difluoro |
C2H2Cl2F2 |
Aliphatic |
Antibacterial12 |
|
9.104 |
3-Ethyl-3-methylnonadecane |
C22H46 |
Aliphatic |
Antimicrobial13 |
|
9.614 |
Phenol, 2,5-bis(1,1dimethylethyl) |
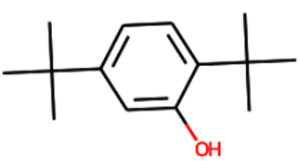
C14H22O |
Aromatic |
Antioxidant10,11 |
|
11.177 |
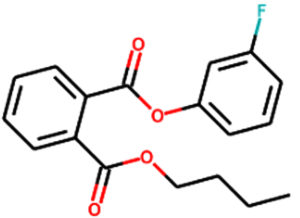
Phthalic acid, 3-fluorophenyl heptadecyl ester |
C31H43FO4 |
Aromatic |
Antibacterial, antimicrobial, antiinflammatory, antitumor12, antimicrobial, anti-inflammatory, anti-tumor14 |
|
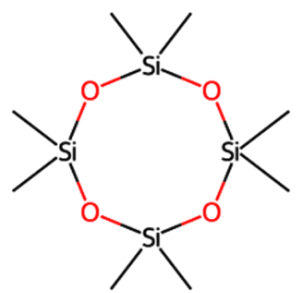
12.112 |
Cyclooctasiloxane, hexadecamethyl |
C16H48O8Si8 |
Aromatic |
Antioxidant and antimicrobial10,11 |
|
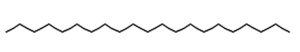
13.131 |
Heneicosane |
C21H44 |
Aliphatic |
Antimicrobial12,15,16 |
|
14.287 |
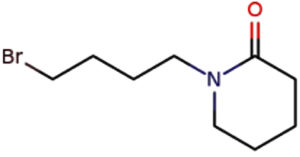
2-Piperidinone, N-[4-bromo-nbutyl] |
C9H16BrNO |
Aromatic |
Antimicrobial activity17,18
. |
|
18.391 |
Cyclotetrasiloxane, octamethyl- |
C8H24O4Si4 |
Aromatic |
Antioxidant13 |
NMR
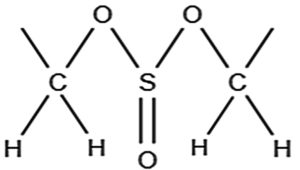
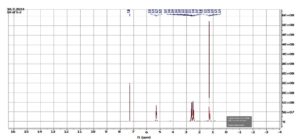
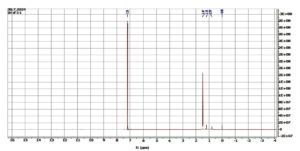
A 1H NMR study of the chloroform extract of F. indica revealed various peaks between δ 0-2, δ 2-3 and δ 5-6 (Figure 3), whereas the ethanolic extract showed signals only in the δ 0-2 ppm region (Figure 4). According to the reference charts by Silverstein et al.,19 the peaks observed in the chloroform extract correspond to acyclic aliphatic protons, β-substituted aliphatic protons, and α-monosubstituted aliphatic systems (Table 2). A strong, broad signal at δ 1.2-1.5 ppm indicates the presence of methyl (-CH3) and methylene (-CH2) groups in long aliphatic chains. Minor peaks between δ 2.0-3.0 ppm can be attributed to methylene groups adjacent to carbonyl functionalities (-CH‚ -CO-) or protons near ester groups, while protons on carbons adjacent to electronegative atoms (Oxygen, nitrogen or halogens) may also resonate in this region. Additional signals at δ 3.5-3.75 ppm suggest protons in close proximity to oxygen atoms, consistent with alcohol, ether, or ester moieties. A small but distinct peak at δ 5.3-5.4 ppm represents vinylic or deshielded protons adjacent to strong electron-withdrawing groups. The absence of downfield aromatic signals (6-8 ppm), aside from the residual CDCl3 signal at δ~7.26 ppm, indicates a lack of aromatic ring structures. There may be alcohol groups (-OH), and the peaks around 3.5-4.0 ppm could correspond to protons in proximity to hydroxyl groups. A small but clear peak around 5.3-5.4 ppm represent protons on carbon adjacent to strong electron-withdrawing groups. A small residual peak at δ~7.26 ppm was given by CDCl3. Whereas, in ethanolic extract, δ 1.0-2.2 ppm indicates the presence of methyl (-CH3) and methylene (-CH2) groups, revealing the presence of aliphatic alicyclic compounds only (Figure 4). Muharni et al.20 revealed chemical constituents from the stem bark of Flacourtia rukam, which align with the FTIR-identified functional groups observed in the crude ethanolic and chloroform extracts in the present study.
Table (2):
Potential functional groups and their structures present in the chloroform and ethanolic extract of F. indica as analyzed by 1H NMR
Peak in PPM |
Possible type of group of compounds |
Possible structure |
|---|---|---|
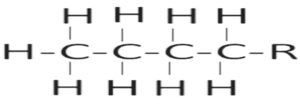
0-2 |
Aliphatic alicyclic compounds |
|
2-3 |
β-Substituted aliphatic compounds |
|
5-6 |
α-Mono substituted Aliphatic compounds |
‘R’ = Elongation of chain; ‘X’ = Attachment of any functional group
Figure 3. NMR Spectroscopy of chloroform extract of F. indica. 1H NMR (400 MHz, CDCl3): δ 1.27 (d, J = 6.4 Hz, 7H), 2.47 (dd, J = 6 Hz, 2H), 2.61 (dd, J = 7.6 Hz, 2H), 5.25 (q, J = 6.4 Hz, 2H)
Figure 4. NMR Spectroscopy of ethanolic extract of F. indica. 1H NMR (400 MHz, CDCl3): δ 1.47 (s, 3H)
The spectrum of 1H NMR indicated a doublet at δ 1.27 ((d, J = 6.4 Hz, 7H), a doublet doublet at δ 2.47 ((dd, J = 6 Hz, 2H), another doublet doublet at δ 2.61 ((dd, J = 7.6 Hz, 2H), and a quartet at d δ.25 (q, J = 6.4 Hz, 2H) in the chloroform extract whereas the ethanolic extract indicated a singlet at δ 1.47 (s,3H).
The present study successfully employed TLC-bioautography, GC-MS, and 1H NMR spectroscopy to identify and characterize antibacterial constituents in chloroform and ethanolic extracts of Flacourtia indica. The bioautography assay revealed clear zones of inhibition against Streptococcus pneumoniae and Escherichia coli, confirming the presence of bioactive compounds with antimicrobial potential. Notably, the chloroform extract demonstrated stronger activity against S. pneumoniae, while the ethanolic extract was more effective against E. coli, suggesting differential solubility and specificity of active constituents.
GC-MS analysis identified 20 compounds in the chloroform extract and 48 in the ethanolic extract. Key constituents included sulfurous acid hexyl nonyl ester, heneicosane, 2-piperidinone derivatives, siloxanes, and phenolic esters. These compounds have been previously reported for their antimicrobial, antioxidant, and anti-inflammatory properties.21,22 The presence of fatty acid methyl esters and alkaloid derivatives further supports the therapeutic potential of F. indica23,24 as recorded during this investigation.
1H NMR profiling revealed peaks corresponding to aliphatic chains, β-substituted, and α-monosubstituted aliphatic compounds. These structural features are consistent with hydrophobic molecules capable of disrupting bacterial membranes, a mechanism supported by previous studies on plant-derived antimicrobials.25,26
The use of TLC-bioautography proved effective for rapid screening and localization of antibacterial activity within complex plant matrices. This method has gained traction in phytochemical research due to its ability to preserve compound integrity while enabling direct visualization of bioactivity.27,28 When integrated with GC-MS and NMR, it offers a robust analytical framework for natural product discovery.
Recent investigations have emphasized the broad pharmacological spectrum of F. indica, including its antioxidant,29 anti-diabetic,24 hepatoprotective,30 and wound-healing properties.23 These findings align with the current study’s identification of multifunctional phytoconstituents. Moreover, trace metal analysis by Koperuncholan and Kulandaivel31 revealed safe levels of essential elements, supporting the plant’s suitability for therapeutic use.
The observed antibacterial activity against both Gram-positive and Gram-negative strains is consistent with earlier reports on F. indica’s efficacy against Staphylococcus aureus, Pseudomonas aeruginosa, and Vibrio cholerae.32 The presence of flavonoids, tannins, and terpenoids confirmed through FTIR and HPTLC fingerprinting further validates the plant’s antimicrobial profile.33
This study underscores the antibacterial potential of chloroform and ethanolic extracts of Flacourtia indica, through TLC-bioautography enabled direct visualization of active zones, while GC-MS and 1H NMR profiling revealed a diverse array of bioactive compounds, including phenolic esters, aliphatic derivatives and siloxanes.
It supports the ethnomedicinal relevance of F. indica and highlights its promise as a source of plant-based antibacterial agents. Future research should focus on isolating individual constituents, validating their pharmacological effects, and exploring compound-target interactions through metabolomics and molecular docking approaches.
ACKNOWLEDGMENTS
The authors would like to acknowledge Kalinga Institute of Social Sciences for their constant support in carrying out this work.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analysed during this study are included in the manuscript.
ETHICS STATEMENT
This article does not contain any studies on human participants or animals performed by any of the authors.
- Barbieri R, Coppo E, Marchese A, et al. Phytochemicals for human disease: an update on plant-derived compounds antibacterial activity. Microbiol Res. 2017;196:44-68.
Crossref - Savoia D. Plant-derived antimicrobial compounds: alternatives to antibiotics. Future Microbiol. 2012;7:979-990.
Crossref - Panda PC, Kar SK, Das PK, Mallick SN, Kamila PK. A reassessment of the status of endemic vascular plants of Eastern Ghats of India with special reference to Odisha. Front Plant Sci. 2019:143-161.
- Velmurugan G, Anand SP. GC-MS analysis of bioactive compounds on ethanolic leaf extract of Phyllodium pulchellum L. Desv. Int J Pharmacogn Phytochem Res. 2017;9(1):114-118.
Crossref - Al Foyjul IM, Masuma R, Rahman AMR, et al. Evaluation of analgesic, anti-inflammatory and antipyretic properties of the Flacourtia indica extract in laboratory animals. J Phytomol Pharmacol. 2022;1(2):66-74.
Crossref - AI Bashera M, Parvin MS, Islam MB, Rana GM, Rony SR, Islam ME. Exploring the antioxidant and antiproliferative properties of Flacourtia indica extracts on lung cancer cells: a comprehensive analysis utilizing GC-MS, molecular docking, and PASS analysis. Appl Food Res. 2025;5(2):101275.
Crossref - Nguyen PD, Sayagh C, Massiot G, Lavaud C. Phenyl glycosides from the leaves of Flacourtia indica (Burm. f.) Merr (Salicaceae). Phytochem. 2021;190:112891.
Crossref - Ginovyan M, Ayvazyan A, Nikoyan A, Tumanyan L, Trchounian A. Phytochemical screening and detection of antibacterial components from crude extracts of some Armenian herbs using TLC-bioautography technique. Curr Microbiol. 2020;77(7):1223-1232.
Crossref - Patra JK, Gouda S, Sahoo SK, Thatoi HN. Chromatography separation, 1H NMR analysis and bioautography screening of methanol extract of Excoecaria agallocha L. from Bhitarkanika, Orissa, India. Asian Pac J Trop Biomed. 2012;2(1):50-56.
Crossref - Sardar N, Bibi Y, Arshad M, Ahmad A, Almoallim HS, Ansari MJ. Identification of natural antioxidants using GC-MS analysis from Moringa oleifera with meat preservative potential. Emir J Food Agric. 2024;36:1-10.
Crossref - Kabuka R, Mudenda S, Kampamba M, et al. Phytochemical analysis of leaf, stem bark, and root extracts of Cassia abbreviata grown in Zambia. Pharm Pharmacol Int J. 2022;13(5):119-128.
Crossref - Thejashree AB, Naika R. Identification of bioactive compounds in acetone leaf and stem-bark extracts of Psychotria dalzellii hook.f. by GC-MS analysis and evaluation of in vitro antibacterial properties. Asian J Biol Life Sci. 2023;12(3):499-509.
Crossref - Eramma N, Patil SJ. Exploration of the biomolecules in roots of Flacourtia indica (Burm f.) Merr. methanol extract by chromatography approach. Lett Appl NanoBioSci. 2023;12(4):166-177.
Crossref - Perera HDSM, Samarasekera JKRR, Handunnetti SM, et al. In vitro pro-inflammatory enzyme inhibition and antioxidant potential of selected Sri Lankan medicinal plants. BMC Complement Altern Med. 2018;18(1):2-15.
Crossref - Ganesan MP, Prabhu K, Rao MRK, Janaki CS, Kalaivannan J, Kavimani M. The GC-MS analysis of ethyl acetate extract of one herbal plant Jatropha curcus. Nat. Volatiles & Essent. Oils. 2021;8(4):6347-6454. https://www.nveo.org/index.php/journal/article.
- Vanitha V, Vijayakumar S, Nilavukkarasi M, Punitha VN, Vidhya E, Praseetha PK. Heneicosane—a novel microbicidal bioactive alkane identified from Plumbago zeylanica L. Ind Crop Prod. 2020;154:112748.
Crossref - AI-Salman HNK. Antimicrobial activity of the compound 2-piperidinone, N-[4-bromo-n-butyl]-extracted from pomegranate peels. Asian J Pharm. 2019;13(1).
Crossref - Loganathan S, Selvam K. Phytochemical and antibacterial activity of hexane and ethyl acetate extract of Knoxia sumatrensis (Retz.) DC. Asian J Pharm Clin Res. 2021;14(7):35-39.
Crossref - Silverstein RM, Webster FX, Kiemle DJ. Spectrometric Identification of Organic Compounds. 7th ed. Hoboken, NJ: John Wiley & Sons; 2005
- Muharni E, Yohandini H, Julinar, Yasrina, Miranti. Chemical constituents from stem bark of Flacourtia rukam Zoll. & Mor. and their antioxidant activities. Sains Malaysiana. 2019;48(9):1899-1906.
Crossref - Makuttan S, Fernandes J, Maste MM, Dsouza NG. GC-MS characterization and in vitro anti-inflammatory activity of fruit extracts of Flacourtia indica. Res J Pharm Technol. 2025;18(5):2081-2089.
Crossref - Sasi S, Anjum N, Tripathi Y C. Ethnomedicinal, phytochemical and pharmacological aspects of Flacourtia jangomas: a review. Int J Pharm Pharm Sci. 2018; 10(3):9-15.
Crossref - Ahmad F, Taj MB, Ramzan M, Raheel A, Shabbir S, Imran M, Iqbal HMN. Flacourtia indica based biogenic nanoparticles: development, characterization, and bioactivity against wound associated pathogens. Mat Res Express. 2020;7(1):1-13.
Crossref - Ogboui A, Evenamede KS, Kpegba K, Simalou O, Agbonon A. Phytochemical study and antioxidant, antibacterial, and antidiabetic activities of Flacourtia indica leaves extracts from the Togolese flora. Int J Pharm Pharm Sci. 2023;15(8):50-56.
Crossref - Beulah GG, Soris PT, Mohan VR. GC-MS determination of bioactive compounds of Dendrophthoe falcata (LF) Ettingsh: An epiphytic plant. Int J Health Sci Res. 2018; 8(11):261-269
- Panda L, Duarte-Sierra A. Recent advancements in enhancing antimicrobial activity of plant-derived polyphenols by biochemical means. Horticulturae. 2022;8(5):1-19.
Crossref - Choma IM, Jesionek W. TLC-direct bioautography as a high throughput method for detection of antimicrobials in plants. Chromatography. 2015; 2(2):225-238.
Crossref - Zang Y, Cheng Z, Wu T. TLC bioautography for screening of bioactive natural products: an updated review. Curr Anal Chem. 2020; 16(5):545-556.
Crossref - Mallawaarachchi MALN, Madhujith T, Suriyagoda LDB, Pushpakumara DKNG. Antioxidant efficacy of selected underutilized fruit species grown in Sri Lanka. Trop Agric Res. 2021;32(1):68-80.
Crossref - Idoko A, Emmanuel UEG. Study on fresh leaf aqueous extract of Flacourtia indica for hepatoprotective, anti-anemic and hypoglycemic abilities in CCl4 induced hepatotoxicity in albino wistar rats. Univ J Pharma Res. 2019;4:17-23.
Crossref - Koperuncholan M, Kulandaivel S. Assessment of phytochemical constituents, trace metals, and antimicrobial efficacy of Flacourtia indica from Southern India. Int J Bot Stud. 2022;7(2):569-573.
- Mal B, Mondal S, Kuila D, Mridha M, Biswas G. Food value analysis, phytochemical profiling, LC/MS analysis, antioxidant and antimicrobial activity of methanolic extract of Flacourtia indica (Burm.f.) Merr. fruits of lateritic West Bengal. Prospect Pharm Sci. 2025;23(2):84-95.
Crossref - Hokonya N, Mahamadi C, Mukaratirwa-Muchanyereyi N, Gutu T, Zvinowanda C. Green synthesis of P “ ZrO2CeO2ZnO nanoparticles using leaf extracts of Flacourtia indica and their application for the photocatalytic degradation of a model toxic dye, Congo red. Heliyon. 2022;8(8):e10277.
Crossref
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.