ISSN: 0973-7510
E-ISSN: 2581-690X
The discharge of textile effluents enriched with reactive azo dyes is of critical importance owing to inability of the dyes to degrade in waste water and their carcinogenic, mutagenic effects to various organisms. This study initiated based on the need to gaze into molecular mechanism of marine bacterial bioremediation process to develop strategies for the decolorization and detoxification of the synthetic azo dyes. The experimental work carried out to explore decolorization and degradation efficacy of laccase derived from marine actinobacteria, Streptomyces sviceus by choosing Congo red-21 as model azo dye. The extracellular production of laccase was confirmed with plate assay in medium supplemented with ABTS as substrate. Laccase was purified to homogeneity from 72hrs culture of Streptomyces sviceus by Fast performance liquid chromatography and the molecular size of laccase was noticed as 60 kDa. The purified laccase was immobilized with an efficiency of 82% by Calcium alginate method. The crude, purified and immobilized forms of the laccase enzyme was used to decolorize the Congo red-21. Crude laccase enzyme showed 69% of decolorization of Congo red-21 after 48h where as purified and immobilized laccase represented 78% and 92% of colour removal after 24 h respectively. Fourier-transform infrared spectroscopy, High Performance Liquid Chromatography and Gas chromatography–mass spectrometry were used to unravel the molecular mechanism of dye detoxification and also identify nontoxic products released from Congo Red-21 upon administration with immobilized laccase. Based on GC-MS data, it may deduce that immobilized laccase of Streptomyces sviceus cleaves the Congo red-21 dye followed by oxidative cleavage, desulfonation, deamination, demethylation process.
Marine bacteria, Reactive Azo dyes, Decolorization, Streptomyces sviceus, Immobilized Laccase
The Synthetic azo dyes are the major constituents of fabric colours and consequently accountable for substantial environmental pollution during dying process1. The azo dyes are one of the most popularly used textile dyes owing to their versatility and based on the chromophore structure they are categorized as acidic, reactive, basic, disperse dyes2,3. In the global textile market, the reactive dyes are chiefly used in the dying process owing to varied color spectrum and adherence features4. Dumping of azo dyes into the water streams and ponds is a great concern in textile effluent treatment due to their effect on the biological oxygen demand (10-30%), photosynthetic potential of under water plants by blocking the light penetration and toxicity to aquatic organisms due to the aromatic amines released during the cleavage of azo bonds5,6.
During the past fifteen years, bioremediation has been in practice as a novel strategy for the decolourization and the biotransformation of the azo dyes into non toxic products. Bioremediation at the present is being considered as a more eco-friendly alternative to the physical and chemical degradation methods of textile effluents while at the same time being cost effective. The efficacy of the bio degradation is dependent on the nature of the organisms available in the dwelling sites.
Literature replete with bioremediation of textile azo dye effluents using bacteria7, fungi8 yeast9 , Actinomycetes10 and Algae11. Research reports on the degradation of azo dyes by bacteria and fungi have revealed the contribution of either intracellular enzymes like azo reductases or extracellular oxidative enzymes like laccase, manganese peroxidases and tyrosinase12,13. Among the oxidative enzymes, laccase attracted the scientific community due to its physic-chemical properties and catalytic function in the bioremediation of textile effluents14. Laccase is a glycoprotein involved in the direct oxidation of phenolic compounds, indirect oxidation of non-phenolic compounds and coupling reactions15, Lacasses are extensively available in eukaryotes like fungi, plants, insects and mammals16 and also in bacteria17. By keeping in view the importance of laccase mediated degradation of azodyes, the research study is formulated to analyze the effect of immobilized laccases on degradation of reactive red azo dye.
The reactive azo dye Congo red – 21 was purchased from the local retail vendors situated in Tirupati with coordinates of 13.65°N 79.42°E, Andhra pradesh. India. Nutrient glucose broth medium inoculated with marine actinomycetes was taken for the analysis of dye degradation potential. Fine grade solvents and chemicals used for the experimental analysis. These were purchased form Sigma and SD – fine. Streptomyces sviceus strain KN3 isolated from the marine sediments, Bay of Bengal, Nellore, A.P with coordinates of 14.5131°N and 80.1791°E 18 was exploited for the production of extracellular laccase.
Screening for extracellular laccase
Qualitative screening approach was utilized to detect the extracellular production of laccase by using ABTS (2,2′-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid),and Guiacol as substrates. The ABTS plate was prepared by mixing 100ml nutrient glucose agar with 10ml of 0.2mM ABTS and 1ml of CuSo4.The appearance of green color halos around the bacterial colony after 48h incubation at 30°C, demonstrates the extracellular production of laccase from Streptomyces sviceus strain KN3. The production of laccase was further confirmed with guiacol plate assay. The assay was performed by supplementing 15ml nutrient glucose agar with 0.1% guiacol and the plates were inoculated with the isolate KN3 and incubated at 30°C for 48h. The laccase induced oxidation of guiacol was visualized with formation of reddish brown colour on the guiacol plates inoculated with S. sviceus KN319.
Assay for Laccase enzyme
The quantitative analysis of the Laccase was carried out at 420 nm by calculating the oxidation of ABTS. S.sviceus KN3 indicated maximum production of laccase in nutrient glucose broth (pH 7.0) incubated at 30°C.The oxidation of ABTS was performed by adding the culture supernatant of S.sviceus (100µl) to the reaction mixture containing 0.1M Acetate buffer pH 4.5 and 10mm ABTS. The reaction mixture was incubated at a temperature of 25°C for 10 minutes and the oxidation was stopped with the 50% (w/v) Tri chloro Acetic acid. One unit of enzyme is defined as the amount of enzyme required to oxidize 10µl ABTS per minute. Triplicates were maintained for the experimental analysis. Lowry method was adopted to determine the protein concentration20.
Purification of laccase enzyme
The selected S.sviceus KN3 was grown under the optimum pH, temperature, Carbon and Nitrogen sources for 4 days. After incubation the culture filtrate was subjected to centrifugation for 30 mins at 11308xg. The supernatant acquired from the centrifugation was subjected to ammonium sulphate precipitation of 20 – 60 % saturation by addition of ammonium sulphate and centrifuged at 11308xg. for 30mins at 4°C. The pellet with protein activity was selected for further analysis while the supernatant was discarded. The pellet was dialyzed by dissolving in 10mM potassium phosphate buffer (pH 7.0) and dialyzed for 24hrs at 4°C. After dialysis the sample was centrifuged and the supernatant was selected for further purification by Fast performance liquid chromatography (FPLC) and protein content was determined by the Lowry method .20
Purification of Laccase by FPLC and molecular weight determination
The chromatographic purification steps were performed in a fast protein liquid chromatography (GE Healthcare, Uppsala, Sweden) with a flow rate of 1 ml/min, temperature of 20°C and volume fraction of 50µl and the elution was monitored by UV-detector at 280 nm. The dialyzed enzyme was filtered through a 0.45 μm membrane filter (Millipore, Billerica, MA, USA) and loaded onto a Sephadex G-75 column (GE Healthcare, Uppsala, Sweden) pre-equilibrated with 10 mol/L Tris–HCl (pH 7.0). The bound enzyme was eluted with the 0.3M KCl gradient and fractions were pooled and used for determining the specific activity if laccase with standard substrates (ABTS and guiacol). Sodium dodecyl sulphate Polyacrylamide gel electrophoresis (SDS – PAGE) with protein molecular weight markers was used in the determination of the molecular mass of the purified laccase21.
Immobilization of laccase Enzyme
Calcium alginate method was adopted for the immobilization of laccase enzyme. Sodium alginate solution (4%) was mixed with FPLC purified laccase enzyme solution (1 mg/mL) at a ratio of 1:2 (v/v) and stirred to facilitate complete mixing. The mixture was slowly dropped into CaCl2 solution (2% w/v) using a sterile syringe needle and stirred gently for the formation of immobilized enzyme beads. The Calcium alginate beads were washed with distilled water and refrigerated at a temperature of 4°C for the conduction of experimental work. The efficiency of immobilization was measured by with the help of the formula.
Percentage of immobilization = Initial activity of laccase – Activity of laccase in Washings
Degradation of Congo red dye by Immobilized laccase
Decolorization experiment was carried out in 25ml conical flasks with 10ml of potassium phosphate buffer(PH6.5) amended with 400 ppm reactive Congo red-21 and laccase enzyme (Crude/purified /immobilized enzyme) at different concentrations such as 25 mg/l and 50 mg/l and the samples were incubated at 30°C and 100 rpm for different time points. Control was maintained without the laccase enzyme. The speed of colour elimination of the Congo red-21 was measured at λ max 540 nm using UV-spectrophotometer (Schimadzu UV 1601) at a regular interval of 12h. The percentage decolorization of textile reactive Congo red -21dye was determined 18
Analysis of degradation Products of Congo red dye
The congo red-29 dye sample degraded by immobilized laccase was centrifuged at 11308xgfor 15 min and the supernatant containing the degraded products was subjected to liquid – liquid extraction using ethyl acetate as a solvent (1:1 ratio). The organic phase was collected and evaporated by using a rotary evaporator and the products were dried with anhydrous Na2So4. Further the extracted residue was analyzed by FTIR, HPLC, and GC – MS analysis.
Fourier transform infrared spectroscopy analysis (FTIR)
The profile of functional groups in the parent Congo red-21 and laccase degraded dye was assessed through Fourier transform infrared spectroscopy. The molecular signatures of functional groups were recorded with 16 scan speed in the mid-IR region of 400–4000 cm-1. The parent Congo red-21 dye sample and the sample extracted after treatment with laccase was mixed independently with 0.02 g of sample with KBr at a ratio of 1:20 and the spectra were measured using FT-IR Spectrophotometer (BRUKER Model ALPH).
High performance liquid column chromatography (HPLC)
The parent and degraded samples were extracted and dissolved methanol, filtered and purification was carried out on silica C18 (250 X 4.6mm) column in an isocratic mode with methanol and water (1:1) as mobile phase and detected at 316nm using Shimadzu LC solution. The purified products were eluted with a flow rate of 0.75 ml/min.
Gas chromatography and Mass spectrometry (GC-MS)
GC-MS analysis of parent Congo red-21 dye and the enzyme mediated products was carried out using GCMS analyzer equipped with integrated gas chromatogram. The injector temperature was maintained at 300°C with an oven conditions at 100°C kept constant for 2 min then increased up to 250°C with 10oC/min and raised up to 300°C with 30°C/min rate. NIST library was accessed to detect and identify the chemical compounds based on the mass spectra.
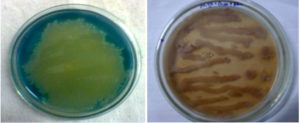
Qualitative screening approach was utilized to detect the extracellular production of laccase by using ABTS and Guiacol as standard substrates. The appearance of green color halos around the bacterial colony after 48h incubation, demonstrates, the extracellular production of laccase from S.sviceus KN3 (Fig. 1a). The production of laccase was further confirmed with guiacol plate assay. The enzymatic reaction of laccase with guiacol was visualized with the formation of reddish brown colour on the guiacol plates inoculated with S.sviceus KN3(Fig. 1b). Seven different species of Pseudomonas were tested for the production of laccase by using standard substrates like syringaldazine or guiacol22,23. The laccase activity was reported in Streptomyces lavendulae, Streptomyces psammoticus, Streptomyces ipomoeae, and Streptomyces sviceus 24. The supernatant of B. subtilis demonstrated the oxidation ABTS and guaiacol due to the extracelluar production of laccase25,26 reported the decolourisation activity of laccase against azo dyes and the plate assay has been used to confirm the secretion of laccase in to the extracellular fluid when grown in the nutrient glucose medium.
Fig. 1. Extra cellular production of Laccase by Streptomyces syiceus (a) ABTS as substrate (b) Guiacol as substrate
Production of Laccase from Streptomyces sviceus strain KN3
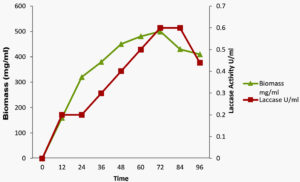
The extracellular production of laccase by Streptomyces sviceus strain KN3 was assessed at different time points. As shown in the Figure.2 the maximum enzyme yield and biomass was noticed at 72h and considerable production of laccase and biomass was retained up to 84h of incubation. The declined trend was observed at later period and the data showed a linear correlation between laccase production and biomass. Among the textile degrading enzymes, Laccase is considered to be potential bio-remediating enzyme in the treatment of textile dyestuff and textile industry effluents27. The decolorization of azo dyes with laccase enzymes depends on the source of laccase and number azo bonds in the dye28-31.
Fig. 2. Effect of time on production of laccase by Streptomyces syiceus strain KN3 purification and characterization of Laccase enzyme
Purification and characterization of Laccase
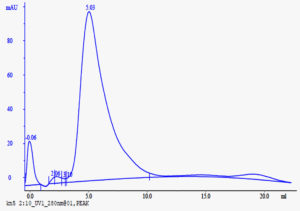
All the three steps of purification were carried out at 4°C. At the initial step, the culture supernatant of S.sviceus containing laccase with an initial activity (860U) was concentrated by ammonium sulphate precipitation. The optimum ammonium sulphate fractionation (80% W/V saturation) showed 2.02 U/mg specific activity compared to crude enzyme. The protein pellet obtained after 80% saturation with ammonium sulphate was dialyzed with 10mM potassium phosphate buffer pH 7.0 (Table 1). Further, the dialyzed sample was subjected to purification by Fast protein liquid chromatography (FPLC). The fractions were collected at a flow rate of 1ml/min on gradient mode using 0.3M KCl as elution buffer. As shown in FPLC chromatogram (Fig. 3) a single peak was resolved at 5.63 min. The fraction collected at 5.63 demonstrated positive reaction with the formation of green and reddish brown complexes with ABTS and guiacol respectively (Fig. 4).
Table (1):
Purification profile of laccase from Streptomyces sviceusstrain KN3.
Purification steps |
Volume (ml) |
Total Laccase activity (U) |
Total protein (mg) |
Specific activity (U/mg) |
Purification Fold |
Yield (%) |
|---|---|---|---|---|---|---|
Crude |
1000 |
860 |
450 |
1.60 |
1 |
100 |
Ammonium sulphate |
25 |
700 |
320 |
2.02 |
1.10 |
82 |
Dialysed |
15 |
610 |
192 |
3.00 |
1.70 |
70 |
FPLC |
5 |
300 |
30 |
10.20 |
5.82 |
36 |
Fig. 4. Reaction of Purified laccase with substrates A) FPLC purified lacase B) Fractio with guiacol C) Fraction with ABTS
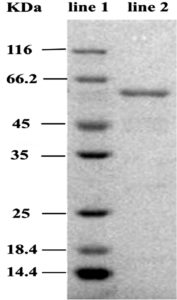
The purity as well as molecular weight of laccase enzyme was detected by SDS-PAGE. Laccase showed the monomeric structure as visualized by single band with a molecular weight of about 60kDa (Fig. 5). The molecular weight of the laccase enzyme varies depends on the source of the organism32 and the molecular mass of laccase was found to be between 50-130 kDa33. Few bacterial species with laccase activity have been reported in the degradation of azo dyes34,4 However, the laccases isolated from Streptomyces are found to be effective for the degradation of reactive azo dyes. And the Laccase enzyme purified from Streptomyces cyaneus CECT demonstrated the degradation of azo dyes 36.
Fig. 5. Molecular weight of laccase derived from Streptomyces syiceus strain KN3 line 1: Molecular weight marker Line 2: FPLC purified enzyme
Decolorization of Congo Red-29 by Laccases
Laccase has been widely applied in the decolouisation of textile dyes compared with azo reductases due to the generation of nontoxic phenolic compounds by free radical mechanism37,38 .To evaluate the decolorization efficiency of laccase, the Congo red -21 dye was treated with different concentrations (25mg/l and 50 mg/l) of Crude/purified /immobilized enzyme for different time periods. As shown in the Table 2 the decolorization of congored-21 was commenced within 24 h and the rate of decolorization accelerated with increase in time. Crude enzyme showed 69% of decolorization after 48h where as purified and immobilized laccase represented 78% and 92% after 24 h respectively. The data demonstrates the efficacy of immobilized laccase in the decolourisation and degradation of synthetic azo dyes. Podoscypha elegans demonstrated maximum degradation of 70% of Congo red and Rose Bengal after 72 days of incubation39 where as bacterial consortium of P. rettgeri strain HSL1 and Pseudomonas sp. SUK1 represented the degradation of 22% of 100 mg/L of azo dyes within 48 hours40. Aspergillus niger exhibited 70% decolourisation of Congo red after 6 days of incubation due to the presence of Lignin peroxidise and Manganese peroxidise and the laccase is totally absent in the culture filtrate of A.niger 41.
Table (2):
Congo Red-21 Decolorization by Laccase.
| Dye | Enzyme | Conc. (mg/l) | decolorization (%) | |||
|---|---|---|---|---|---|---|
| 12 h | 24 h | 36 h | 48 h | |||
| Congo red-21 | Crude | 25 | 12±0.20 | 25±0.10 | 35±0.30 | 52±0.12 |
| 50 | 22±0.20 | 44±0.20 | 53.3±0.10 | 69±0.20 | ||
| Purified | 25 | 43±0.21 | 62±0.30 | 71±0.12 | 80±0.04 | |
| 50 | 58±0.02 | 78±0.05 | 84±0.03 | 84±0.12 | ||
| Immobilized | 25 | 68±0.10 | 82±0.11 | 92±0.24 | 96±0.02 | |
| 50 | 78±0.03 | 92±0.12 | 96±0.11 | 96±0.20 | ||
Immobilized Laccases were considered as potential enzymes to be potential candidates in the bioremediation of synthetic dyes as well as textile effluents42. Cross-linked aggregates of Laccases enzyme derived from Cerrena sp. demonstrated 90% decolorization of Remazol Brilliant Blue Reactive dye43. Similarly, immobilized laccase showed better elimination of colour from Procion Green H4G, Brilliant Blue G, and Crystal Violet on comparison with free enzyme17,45.
Congo red dye degradation by immobilized laccase
Dyes are decolorized either through adsorption or degradation. The dyes are adsorbed only on the surface of the bacterial cells where as degradation of dyes by bacterial enzymes, results in the formation new products. Different analytical methods were adopted to detect feasible degradation pathway and the metabolites generated from Congo red-21after treatment with immobilised laccase derived from Streptomyces sviceus KN3.
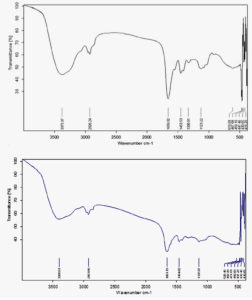
The molecular signatures indicating the functional groups of Congo red-21 parent dye were contrasted with the functional of detoxified Congo red-21 dye. The FTIR spectrum of Congo red-21 control demonstrates molecular signatures at 3250,3100,2370,2080, 1620, 1598,970 and 765 cm-1 (Fig.6a). The molecular signatures from 3250 to 2980 indicates the functional groups such as OH stretch, C-H stretching of aromatic compounds, C≡N stretching , C≡C stretching of and 765 cm-1 demonstrated the aromatic nature of the congo red dye. Specifically, the control dye showed signatures at 1620cm-1 and 1598cm-1due to the presence of azo linkage and N=N stretching respectively. The FTIR spectrum of extracted metabolites after decolorization by Immobilized laccase demonstrated the absence of IR signatures at 1620cm-1 and 1598cm-1indicates the cleavage of azo linkage (Fig. 6b). The signature at 1096 cm-1, 1371 cm-1 indicates S-O stretching and S=O stretching indicated the formation of sulfonates after the degradation by laccases.
Fig. 6. FTIR spectrum of Congo red-21 (a) control dye (b) Congo red-21 dye after decolorization by immobilized laccase
These changes in the IR signatures are clear evidence for the degradation of Congo red-21 dye by immobilized laccase. Our spectral data is in agreement with decolorization of textile effluent by Enterobacter asburiae46. FTIR spectrum provides the information about the type and strength of interactions that occurs with in azo-dyes containing different functional groups after degradation by bacteria. The FTIR technique has been used previously by some workers for analyzing azo-dye degraded products47 .
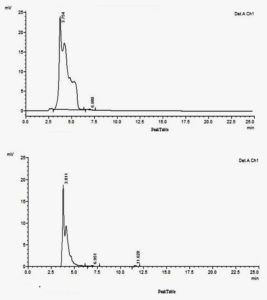
HPLC study of Congo red-21 dye and laccase degraded dye was carried out and retention profiles were recorded. Both the samples demonstrated elution peaks at varied retention times. The Congo red -21 dye showed a major peak at 3.754 (Fig.7a) where as dye metabolites of degradation by Streptomyces sviceus KN3 exhibited two major peaks at 3.81 and 4.6 and two minor peaks at 6.95and 11.6 (Fig.7b). Two peaks indicated in degraded sample compared with single peak in parent Congo red-21dye clearly indicates the degradation of Congo red-21 dye. Our results were supported with the demonstration of new peaks in the decolouzised sample of CR by DTS26 due to the production of different intermediate metabolites.
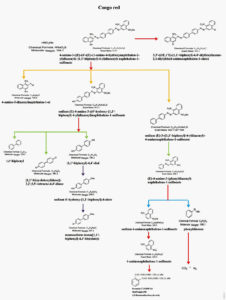
Mass spectrometry is an advanced analytical tool used for the detection of intermediates generated during laccase mediated degradation of reactive synthetic dyes. The proposed enzymatic cleavage pathway involved in the degradation of dyes can be deduced based on the GC-MS data which provides the information about the structure and molecular weight of the degraded products on comparison with NIST data library. GC-MS analysis was performed to characterize the products formed during Laccase mediated degradation of Congo red-21. The Gas chromatogram of Congo red-21 control dye exhibited minor peaks at 1.1, 15.8,16.8 and 19.5 min. The gas chromatogram of Congo red-21 dye degraded by Streptomyces sviceus strain KN3 showed two major peaks at 25.33 and 26.00min and minor peaks at 16.78,18.75and 19.15min. The structure and molecular weight of identified degraded compounds were validated based on fragmentation pattern and m/z values. Based on GC-MS data, it may be deduced that laccase cleaves the Congo red-21 dye molecule through asymmetric cleavage, followed by oxidative cleavage, desulfonation, deamination, demethylation process.
The degradation of Congo red-21 dye was initiated with asymmetric cleavage which results in the formation of sodium (E)-4-amino-3-((4’-hydroxy-[1,1’-biphenyl]-4-yl)diazenyl)naphthalene-1-sulfonate(molecularweight441.1),4-amino-3-diazenylnaphthalen-1-01molecularweight186.2), Sodium (E)-3-(1,1, ‘-biphenyl)-4-yldiazenyl)-4-aminonaphalene-1-sulfonate (molecular weight 464). Further, sodium (E)-4-amino-3-((4’-hydroxy-[1,1’-biphenyl]-4-yl)diazenyl)naphthalene-1-sulfonate was transformed to 1,1’-biphenyl(molecular weight 154.2), 1,1-bi(cyclohexyliden)1-2,2’,5,5’- tetraene-4,4’-dione (molecular weight 184.2), -[1,1’-biphenyl]-4,4’-diol(molecular weight 186.2)by desulfonation and deamination and finally results in the formation of monosodium mono (1,1’-biphenyl)-4,4’-bis(olate) (molecular weight 207.2) (Table.3). As shown in the pathway (Fig.8), Sodium(E)-3-(1,1,’-biphenyl)-4-yldiazenyl)-4-aminonaphalene-1-sulfonate was converted into 4-amino-3-phenyldiazenyl naphthalene -1- sulfonated(molecular weight 326.1) which further results in the formation of sodium 4-aminonaphthalene-1-sulfonate(molecular weight 245), and phenyldiazene. The data indicated the accumulation of 1,2 benzendicarboxylicacid (molecular weight 390) as the end product as evidenced from the major peaks of GC-MS chromatograph.
Table (3):
GC-MS profile of Congo red-21 degradation products after laccase action.
S.no |
R.T |
Molecular weight |
Compound Name |
|---|---|---|---|
1 |
18.750 |
186.2 |
1,1′-biphenyl]-4,4′-diol |
2 |
19.158 |
184.2 |
[1,1′-bi(cyclohexylidene)]-2,2′,5,5′-tetraene-4,4′-dione |
3 |
31.050 |
441.1 |
sodium (E)-4-amino-3-((4′-hydroxy-[1,1′-biphenyl]-4-yl)diazenyl)naphthalene-1-sulfonate |
4 |
16.783 |
154.2 |
1,1′-biphenyl |
5 |
29.117 |
207.2 |
monosodium mono([1,1′-biphenyl]-4,4′-bis(olate)) |
6 |
31.717 |
326.1 |
(E)-4-amino-3-(phenyldiazenyl) naphthalene-1-sulfonate |
7 |
24.042 |
245.0 |
sodium 4-aminonaphthalene-1-sulfonate |
8 |
25.33 |
390 |
1,2-Benzenedicarboxylic acid |
9 |
26.00 |
390 |
1,2-Benzenedicarboxylic acid |
Degradation of azo dye is a complex process and the degradation of azo dyes by laccases has been initiated with asymmetric cleavage of the azo bond48. After cleavage, the products are subjected to oxidative cleavage desulfonation, deamination, demethylation and dehydroxylation, depending on the azo dye structure48,14. Research reports demonstrated the degradation through non- specific free-radical mechanism without the cleavage of azo bonds50,34. The Mass spectral data of Congo red -21 degraded by laccase is in agreement with literature reported the asymmetric cleavage of the azo bond52. Our results revealed the possible mechanism for the cleavage of congo red-21 dye which involves asymmetric cleavage followed by oxidative cleavage, desulfonation, deamination and demethylation which results in the production of benzene dicarboxylicacid and CO2. The degradation of Congo red -21 by Trametes pubescence exhibited the formation of naphthalene amine53. As per the literature, the degradation of azo dyes is a complicated process and involves asymmetry. In earlier study, we reported the nontoxic nature of degraded products based on their insignificant effect on choromosomal pattern and the germination potential higher plants54. The current data confirms the bioremediation potential of immobilized laccase of Streptomyces sviceus strain KN3.
From the present study, it can be concluded that Congo red dye was effectively degraded by laccase of S.sviceus. High decolourisation efficiency was noticed with purified laccase (78%) and immobilised laccase (92%) after 24 h. FT-IR and GC-MS analysis proved the significance of immobilised laccases in the mineralization and detoxification of the reactive azo dyes, particularly Congo red-21 dye. Thus, degradation by immobilised laccase derived from marine actinomycetes promotes a promising approach in the treatment of textile effluents.
ACKNOWLEDGMENTS
We would like to express our thanks to CURIE-Laboratory at SPMVV, Tirupati for providing assistance for HPLC and FPLC .
CONFLICT OF INTEREST
The authors declares that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
ETHICS STATEMENT
This article does not contain any studies with human participants or animals performed by any of the authors.
AVAILABILITY OF DATA
All datasets analyzed during this study are included in the manuscript.
- Langhals H. Color Chemistry. Synthesis, Properties and Applications of Organic Dyes and Pigments. 3rd revised edition. By Heinrich Zollinger Angewandte Chemie International Edition. 2004;43(40):5291-5292.
- Lorimer JP, Mason TJ, Plattes M, Phull SS, Walton DJ. Degradation of dye effluent. Pure Appl Chem. 2001;73(12):1957-1968.
Crossref - Raffi F, Hall JD, Cernigila CE. Mutagenicity of azo dyes used in foods, drugs and cosmetics before and after reduction by Clostridium species from the human intestinal tract. Food Chem Toxicol. 1997;35(9):897 – 901.
Crossref - Singh G, Capalash N, Goel R, Sharma P. A pH-stable laccase from alkali-tolerant γ-Proteobacterium JB: purification, characterization and indigo carminedegradation. Enzyme Microb Technol. 2007;41(6-7):794-799.
Crossref - Pinheiro HM, Touraud E, Thomas O. Aromatic Amines from Azo Dye Reduction: Status Review with Emphasis on Direct UV Spectrophotometric Detection in Textile Industry Waste waters. Dyes Pigm. 2004;61(2):121-139.
Crossref - Aksu Z. Application of biosorption for the removal of organic pollutants: a review. Process Biochem. 2005;40(3-4):997-1026.
Crossref - Srivastava AK, Tripathi SN, Dey S, Kanawade VP, Tiwari S. Inferring aerosol types over the Indo-Gangetic basin from ground based Sunphotometer measurements. Atmos Res. 2012;109-110:64-75.
Crossref - Kumar PGN, Bhat SK. Fungal Degradation of Azo dye- Red 3BN and Optimization of Physico-Chemical Parameters. ISCA J Biol Sci. 2012;1(2):17-24.
- Waghmode TR, Kurade MB, Khandare RV, Govindwar SP. A sequential aerobic/microaerophilic decolorization of sulfonated mono azo dye Golden yellow HER by microbial consortium GG-BL. Int Biodeterior. Biodegradation. 2011;65(7):1024-1034.
Crossref - Parvez RS, UMD Palempalle. Decolorization and detoxification of Reactive azo dyes by Saccharothrix aerocolonigenes TE5. Appl Environ Microbiol. 2015;3(2):58-62.
Crossref - Dilek Y, Thy P, Hacker B, Grundvig S. Structure and petrology of Tauride ophiolites and mafic dike intrusions (Turkey): implications for the Neotethyan Ocean. Geological Society of America Bulletin. 1999; 111:1192-1216
Crossref - Joshi SM, Inamdar SA, Telke AA, Tamboli DP, Govindwar SP. Exploring the potential of natural bacterial consortium to degrade mixture of dyes and textile effluent. Int Biodeterior Biodegradation. 2010;64(7):622-628.
Crossref - Kurade MB, Waghmode TR, Govindwar SP. Preferential biodegradation of structurally dissimilar dyes from a mixture by Brevibacillus laterosporus. J Hazard Mater. 2011;192(3):1746-1755.
Crossref - Legerska B, Chmelova D, Ondrejovic M. Degradation of synthetic dyes by laccases – A mini-review. Nova Biotechnol Chim. 2016;15(1):90-106.
Crossref - Agrawal K, Chaturvedi V, Verma P. Fungal laccase discovered but yet undiscovered. Bioresour Bioprocess. 2018;5:4.
Crossref - Janusz G, Pawlik A, Swiderska-Burek U, et al. Laccase properties, physiological functions, and evolution. Int J Mol Sci. 2020;21(3):966.
Crossref - Deska M, Konczak B. Immobilized fungal laccase as “green catalyst” for the decolorization process – State of the art. Process Biochem. 2019;84:112-123.
Crossref - B Chakravarthi, Vijayasree J, Swathi V, Sudhira DS, UMD Palempalle. Screening and Exploration of azo dye decolorizing Actinomycetes from Marine Sediments. Int J Eng Res. 2015;6(2):27-30.
- Muthukumarasamy NP, Jackson B, Raj AJ, Sevanan M. Production of Extracellular Laccase from Bacillus subtilis MTCC 2414 Using Agroresidues as a Potential Substrate. Biochem Res Int. 2015;2015:765190.
Crossref - Lowry OH, Rosebrough NJ, Farr AL, Randall RJ. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951;193(1): 265-275.
Crossref - Laemmli U. Cleavage of structural protein during the assembly of the head of bacteriophage T4. Nature. 1970;277:680-685.
Crossref - Sakurai T, Kataoka K. Basic and applied features of multicopper oxidases, CueO, bilirubin oxidase, and laccase. Chem Rec. 2007;7:220-229.
Crossref - Mandic M, Djokic L, Nikolaivits E, et al. Identification and Characterization of New Laccase Biocatalysts from Pseudomonas Species Suitable for Degradation of Synthetic Textile Dyes. Catalysts. 2019;9:629.
Crossref - Fernandes TAR, da Silveira WB, Passos FML, Zucchi TD, Laccases from Actinobacteria-what we have and what to expect. Advances in Microbiology. 2014;4(6):285-296.
Crossref - Sheikhi F, Ardakani MR, Enayatizamir N, Rodriguez-Couto S. The Determination of Assay for Laccase of Bacillus subtilis WPI with Two Classes of Chemical Compounds as Substrates. Indian J Microbiol. 2012; 52(4): 701-707.
Crossref - Telke AA, Ghodake GS, Kalyani DC, Dhanave RS, Govindwar SP. Biochemical characteristics of a textile dye degrading extracellular laccase from a Bacillus sp. ADR. Bioresour Technol. 2011;102:1752-1756.
Crossref - Faraco V, Pezzella C, Miele A, Giardina P, Sannia G. Bio-remediation of colored industrial waste waters by the white-rot fungi Phanerochaete chrysosporium and Pleurotus ostreatus and their enzymes. Biodegradation. 2009;20:209-220.
Crossref - Abadulla E, Tzanov T, Costa S, Robra KH, Cavaco-Paulo A, Gübitz GM. Decolorization and detoxification of textile dyes with a laccase from Trametes hirsuta. Appl Environ Microbiol. 2000;66:3357-3362.
Crossref - Claus H, Faber G, Konig, H. Redox-mediated decolorization of synthetic dyes by fungal laccases. Appl Microbiol Biotechnol. 2002;59(6):672-678.
Crossref - Nyanhongo GS, Gomes J, Gübitz GM, Zvauya R, Read J, Steiner W. Decolorization of textile dyes by laccases from a newly isolated strain of Trametes modesta. Water Research. 2002;36(6):1449-1456.
Crossref - Couto SR, Toca-Herrera JL. Laccase production at reactor scale by filamentous fungi. Biotechnol Adv. 2007;25(6):558-569.
Crossref - Baldrian P. Fungal laccases occurrence and properties. FEMS Microb Rev. 2006;30(2):215-242.
Crossref - Kanagaraj J, Senthilvelan T, Panda RC. Degradation of azo-dyes by laccase: biological method to reduce pollution load in dye wastewater. Clean Technol Environ. 2015;17(6):1443-1456.
Crossref - Pereira L, Coelho AV, Viegas CA, Correia Dos Santos MM, Robalo MP, Martins LO. Enzymatic biotransformation of the azo dye Sudan Orange G with bacterial CotA-laccase. J Biotechnol. 2009;139(1): 68-77.
Crossref - Gottlieb A, Shaw C, Smith A, Wheatley A, Forsythe S. The toxicity of textile reactive azo dyes after hydrolysis and decolourisation. J Biotechnol. 2003; 101(1):49-56.
Crossref - Iark D, Buzzo AJ, Garcia JAA, et al. Enzymatic degradation and detoxification of azo dye Congo red by a new laccase from Oudemansiella canarii. Bioresour Technol. 2019;289:121655.
Crossref - Ramírez-Montoya LA, Hernández-Montoya V, Montes-Morán MA, Jáuregui-Rincón J, Cervantes FJ. Decolorization of dyes with different molecular properties using free and immobilized laccases from Trametes versicolor. J Mol Liq. 2015;212:30-37.
Crossref - Pramanik S, Chaudhuri S. Laccase Activity and Azo Dye Decolorization Potential of Podoscypha elegans. Mycobiology. 2018;46(1):79-83.
Crossref - Lade H, Govinwar S, Paul D. Mineralization and detoxification of the carcinogenic azo dye congo red and real textile effluent by a polyurethane foam immobilized microbial consortium in an upflow column bioreactor. Int J Environ Res Public Health. 2015;12(6): 6894-6918.
Crossref - Asses N, Ayed L, Hkiri N, Hamdi M. Congo Red Decolorization and Detoxification by Aspergillus niger: Removal Mechanisms and Dye Degradation Pathway. BioMed Res Int. 2018;2018:3049686.
Crossref - Bilal M, Rasheed T, Nabeel F, Iqbal HM, N, Zhao Y. Hazardous contaminants in the environment and their laccase-assisted degradation – A review. J. Environ. Manag. 2019;234:253-264.
Crossref - Arica MY, Salih B, Celikbicak O, Bayramoglu G. Immobilization of laccase on the fibrous polymer-grafted film and study of textile dye degradation by MALDI-ToF-MS. Chem Eng Res Des. 2017;128: 107-119.
Crossref - Singh AL, Chaudhary S, Kayastha AM, Yadav A. Decolorization and degradation of textile effluent with the help of Enterobacter asburiae. Indian J Biotechnol. 2015;14: 101-106
- Amin AW. Cytotoxicity testing of sewage water treatment using Allium cepa chromosome aberration assay. Pakistan Journal of Biological Sciences. 2002;5: 184-188.
Crossref - Yang J, Yang X, Lin Y, NG TB, Lin J, Ye X. Laccase-catalyzed decolorization of malachite green: performance optimization and degradation mechanism. Plos One. 2015;10(5):e0127714.
Crossref - Zbeng F, Cui BK, Wu XJ, Megn G, Liu HX, Si J. Immobilization of laccase onto chitosan beads to enhance its capacity to degrade synthetic dyes. Int Biodeterior Biodegradation. 2016;110(1): 69-78.
Crossref - Chen H. Recent advances in azo dye degrading enzyme research. Curr Protein Pept Sci. 2006;7(2):101-111.
Crossref - Adnan LA, Sathishkumar P, Yusoff ARM, Hadibarata T. Metabolites characterisation of laccase mediated Reactive Black 5 biodegradation by fast growing ascomycete fungus Trichoderma atroviride F03. Int Biodeterior Biodegradation. 2015;104:274-282.
Crossref - Si J, Peng F, Pui B. Purification, biochemical characterization and dye decolorization capacity of an alkali-resistant and metal-tolerant laccase from Trametes pubescens. Bioresour Technol. 2013;128:49-57.
Crossref - Chakravarthi B, Mathakala V, Palempalli UMD. Bioremediation of Textile Azo Dyes by Marine Streptomyces. Recent Trends in Waste Water Treatment and Water Resource Management. Springer Nature Singapore. 2020;129-142.
Crossref
© The Author(s) 2021. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.