ISSN: 0973-7510
E-ISSN: 2581-690X
Diabetes is a systemic inflammatory condition. The modulation of cytokine levels caused by Asymptomatic Bacteriuria (ASB) infection can lead to altered inflammatory status in various tissues and may affect their homeostasis and function. We aim to estimate the cytokine (IL-6, TNF-α and IL-10) levels in serum of diabetic individuals with and without ASB to study their clinical significance. The current study evaluates the role of cytokines in inflammation caused due to ASB in T2DM patients. Increase in IL-6 and TNF-α levels may indicate renal tissue damage caused due to inflammation by ASB. Whereas increased IL-10 levels is a possible indicator of persistent infection with bacteria which may lead to diabetic renal disease.
Diabetes Mellitus, Asymptomatic Bacteriuria, Cytokines, Inflammation
The constant increase in diabetic population in India has been a concern. Incidence of diabetes is expected to increase upto 79.4 million by 2030.1 Diabetes leads to several other complications such as cardiovascular, diabetic retinopathy, diabetic kidney disorders, etc. with the latter being more prevalent.2 The pathogenesis and the complications arising with it needs to be studied. Diabetics are more prone to infections as there is immune dysfunction linked with diabetes. Bacteriuria in individuals with diabetes mellitus is more frequent in people with poor glycaemic control.
Prevalence of Asymptomatic bacteriuria (ASB) is less studied as the pathological effect it may create leading to complications such as diabetic kidney diseases and more fatal conditions such as renal failure in long term diabetics with poor glycemic controls.3 It is important to study the effect of the infections caused by organisms causing ASB apart from identifying and treating them. Infection either symptomatic or asymptomatic is known to cause an inflammatory response in the system. In the current study, we have investigated the cytokine levels (IL-6, TNF-α and IL-10) in both non-ASB and ASB patients.
Diabetes mellitus is low grade systemic inflammatory condition, further increase in the cytokine levels,4,5 and frequent episodes of acute/chronic inflammation can lead to significant damage to tissue causing dis-regulation of homeostasis and loss of function. Inflammatory cytokines are also known to be associated with increased insulin resistance in diabetes which may further lead to disease progression.4 Cytokines being key mediators can attract white blood cells leading to amplification of inflammation in various tissues including the renal tissues causing renal injury.5 Previous studies reveal that cytokine levels are significantly high in chronic diabetic patients with renal dysfunction/ diabetic nephropathy compared to diabetics with controlled glycemic index.6 The current study revolves around the hypothesis that recurrent ASB if left undetected/untreated can lead to prolonged increase in the levels of inflammatory cytokines and renal dysfunction. Hence, it is highly essential to identify various causes and address them at the earliest to prevent further complication such as kidney disease/renal failure in diabetics.
A laboratory-based follow-up study conducted in Gayatri Vidya Parishad Institute of Health Care and Medical Technology, a teaching hospital located in Visakhapatnam, Andhra Pradesh, India.
Study was conducted in 258 type-2 DM patients aged between 30-60 years. Immunocompromised patients, individuals with symptomatic UTIs receiving antibiotics, obstructive uropathy, renal failure, indwelling catheters and pregnant women were excluded. Institutional Ethical Committee approval (GVPIHCMT/IEC/20201208/03) was obtained and samples were collected by receiving an informed consent to the patients inducted in the study. Serum samples were also obtained from the same patients after a gap of 6 months as part of the follow-up to estimate the prevalence of ASB and comparison of cytokine levels.
Testing for ASB
Urine samples were collected from all the individuals included in the current study following standard procedure. Collected urine samples were inoculated on MacConkey agar, nutrient agar and blood agar and incubated at 37°C for 18-24 hours. Standard microbiological procedures were followed to identify the organism if any growth was observed.
HbA1c estimation
HbA1c was quantified in whole blood using Fluorescent Immuno Chromatographic method (Finecare F2071720C AD). The estimations were done following the manufacturer’s instructions.
Estimation of Cytokines
The serum obtained from patients were stored in -80°C and were used for estimation of IL-6; Interlukin-6 (BioLegend, USA, 430501), IL-10; Interlukin-10 (BioLegend, USA, 430601) and TNF-α; Tumor Necrosis Factor-α (BioLegend, USA, 430201) as per manufacturer instructions.
Statistical analysis
Samples were analysed in duplicates. Values expressed as mean±Standard Error Mean (SEM). Data was analysed and graphical representations were plotted using MS Excel. Statistical significance was calculated using t-test.
Testing for ASB
A total of 258 T2DM patients included in the study, 124 were female and 134 were male. Growth was observed in 93 (i.e. 36% of total) specimens of which, females were the majority (61.3%).
Klebsiella was identified as predominant isolate (44.09%) succeeded by Escherichia coli (27.96%) Staphylococcus aureus (22.58%) and Enterococci species (5.38%) [Table 1].
Table (1):
Pathogens among ASB positive and non-ASB patients with respect to gender. E. coli – Escherichia coli, S. aureus – Staphylococcus aureus
Gender |
Sterile |
Enterococci |
Klebsiella |
E. coli |
S. aureus |
Total |
|---|---|---|---|---|---|---|
M |
98 |
5 |
10 |
5 |
16 |
134 |
F |
67 |
– |
31 |
21 |
5 |
124 |
Total |
165 |
5 |
41 |
26 |
21 |
258 |
As part of studying the persistence pattern of ASB, urine samples were obtained from 93 previously positive ASB diabetic patients and subjected to culture. Of the 93 patients 24 (25.81%) developed recurrent ASB. In recurrent ASB Klebsiella predominantly contributed 50%, followed by Escherichia coli (33.33%), Staphylococcus aureus (12.50%) and Enterococci species (4.17%).
HbA1c estimation
22.22% of diabetic patients with ≤ 6.5 HbA1c levels developed ASB. 43.45% specimens with ≥6.5 HbA1c levels yielded bacterial growth. Patients with HbA1c >6.5% exhibited high prevalence of ASB [Table 2].
Table (2):
HbA1c levels in ASB positive and non-ASB patients. ASB- Asymptomatic Bacteriuria
| HbA1c | Sterile | ASB | Total | ||
|---|---|---|---|---|---|
| Number | % | Number | % | ||
| ≤ 6.5 | 70 | 77.08 | 20 | 22.22 | 90 |
| > 6.5 | 95 | 56.55 | 73 | 43.45 | 168 |
| Total | 165 | 63.95 | 93 | 36.05 | 258 |
Estimation of Cytokines
Cytokines levels were measured in the serum of the patients in both non-ASB and ASB groups.
IL-6 estimation
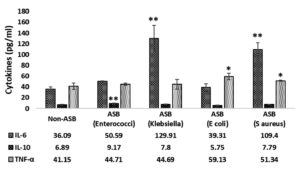
IL-6 levels increased significantly (p<0.005) in patients infected with Klebsiella (129.91±24.00 pg/ml) and S. aureus (109.40± 12.52 pg/ml) when compared to patients not having ASB (36.09±5.74 pg/ml). It is also to be noted that patients infected with Enterococci and E. coli did not show any significant difference in IL-6 levels compared to non-ASB group (Figure 1).
IL-10 estimation
IL-10 levels were significantly (p<0.005) increased in patients infected with Enterococci (9.17±0.05 pg/ml) when compared to patients not having ASB (6.90±0.82 pg/ml). Other ASB groups did not show any significant difference in IL-10 levels compared to non-ASB group (Figure 1).
TNF-α estimation
TNF-α levels increased significantly (p<0.05) in patients infected with E. coli (59.13±6.26 pg/ml) and S. aureus (51.34±1.04 pg/ml) when compared to patients not having ASB (41.15±5.71 pg/ml). Other ASB positive groups did not show any significant difference in TNF-α levels compared to non-ASB group (Figure 1).
Cytokine levels in recurrent ASB
Cytokines levels were measured in the serum of the 93 diabetic patients who were ASB positive to study the influence of cytokine levels in recurrent ASB in diabetics after 6 months post-treatment.
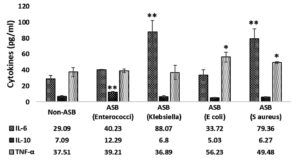
Of the 93 patients who were previously positive for ASB 69 patients (74.19%) were found to have responded positively for treatment and did not develop any culture positivity. In these patients it was observed that cytokine levels reverted back to levels (Figure 2) as found in non-ASB diabetic patients (IL-6 : 29.09 pg/ml, IL-10 : 7.09 pg/ml, TNF-α : 37.51 pg/ml).
Patients with Klebsiella infection had following cytokine levels: IL-6: 88.07 pg/ml, IL-10: 6.8 pg/ml and TNF-α: 36.89 pg/ml (Figure 2). E. coli infected patient serum had IL-6: 33.72 pg/ml, IL-10: 5.03 pg/ml and TNF-α: 56.23 pg/ml (Figure 2). Patients infected by S. aureus showed 79.36 pg/ml of IL-6, 6.27 pg/ml of IL-10 and 49.48 pg/ml of TNF-α (Figure 2). Enterococcal infection resulted in the following levels of IL-6: 40.23 pg/ml, IL-10: 12.29 pg/ml and TNF-α: 39.21 pg/ml (Figure 2).
The high rate of incidence of ASB (36%) is in concordance with previous literature.7 43.45% patients with poor glycaemic control (HbA1c >6.5%) were positive for ASB. This shows an increase in incidence of ASB with increased glycaemic levels compared to patients with controlled glycaemic levels (≤6.5%). Occurrence of ASB in women (45.96%) is higher than in men (26.86%) with poor glycaemic control. This can be attributed to variations in invading microbial species and respective host responses.8,9
Majority of patients in the current study were positive to Klebsiella, which is the second common cause for ASB in diabetic patients.10 This was followed by E. coli and S. aureus was the third, with Enterococci being the least infective. High number of patients getting affected with Klebsiella is an interesting aspect to be given importance in the current study. The study of their antibiotic susceptibility or resistance patterns will be of major interest in the treatment of such conditions.
T2DM onset and progression is associated with low levels of chronic inflammation. Previous studies in diabetics have revealed high plasma levels of pro and anti-inflammatory cytokines such as IL-6 and IL-10.5 The current study also revealed different pathogens causing ASB bring about an increase in various cytokines. The levels of cytokines were significantly high in ASB diabetic patients when compared to non-ASB diabetic patients (Figure 1). This shows that ASB can cause significant increase in cytokine production. S. aureus infection resulted in elevated serum IL-6 and TNF-α levels whereas patients infected with Klebsiella exhibited significantly increased serum IL-6 levels alone. In contrast, ASB patients infected with Enterococci showed significant increase in IL-10 levels. This indicates altered inflammatory status in patients with ASB which may further lead to significant change in the functioning of immune system (Figure 1).
Increased serum IL-6 levels observed prominently in ASB samples infected with S. aureus and Klebsiella (Figure 1) may play a role in the pathogenesis. Serum IL-6 can act in a paracrine manner recruiting lymphocytes and monocytes, inducing their proliferation which further leads to significant increase in production of inflammatory mediators and affect various renal tissues playing a significant role in development and progression of renal dysfunction.11 Raised levels of IL-6 have also revealed a positive correlation with urine albumin excretion. This indicates that IL-6 can serve as an early indicator of diabetic nephropathy.12,13 IL-6 apart from its role in the immune system functioning, has been known to have a role in metabolic functioning. IL-6 is known to regulate glucose homeostasis. It has been found that IL-6 is necessary for maintenance of glucose metabolism by inducing insulin clearance.14-16
When there is a bacterial infection, monocytes, macrophages and T-cells produce TNF-α leading to inflammation.11 Significant increase in serum TNF-α levels were observed in ASB diabetic patients infected with E. coli and S. aureus when compared with non-ASB diabetic patients (Figure 1). TNF-α exerts direct cytotoxic effect on renal cells inducing direct renal injury, apoptosis, and necrotic cell death.11 TNF-α induces generation of Reactive Oxygen Species (ROS) in local tissue causing alteration in barrier function of the capillary wall thus leading to increased albumin permeability. It has also been proved that patients with diabetic nephropathy exhibit increased urinary levels of TNF-α.17 This may serve as one of the red flags for diagnosis of secondary complications in diabetic patients.
Figure 1. Cytokine estimations (IL-6, IL-10 and TNF-α) in serum of diabetic patients with or without ASB by ELISA method. Values expressed as mean±SEM. ** p<0.005, * p<0.05 compared to Non-ASB group. ASB- Asymptomatic Bacteriuria, IL-6-Interleukin-6, IL-10 – Interleukin-10, TNF-α- Tumor Necrosis Factor alpha
IL-10 is anti-inflammatory in nature. Previous literature suggests that Enterococci and Enterobacteriaceae family is capable of producing IL-10.18 IL-10 modifies immune response such as to develop adaptability towards the bacteria causing the infection by over-riding the host inflammatory response to the infection. This creates an immune-suppressive environment where the micro-organism can thrive. Thus, elevation in IL-10 levels (Figure 1) may contribute to host tolerance towards a particular species and promote colonisation that may lead to recurrent/persistent bacterial infections with multiple drug resistance.19 Since we have performed this study with very limited number of patients, it needs further investigation in such patients with persistent UTI.
Further prevalence of recurrent ASB in diabetic patients was studied by following-up the patients who were ASB positive and urine and serum samples were obtained 6 months post-treatment. 74.19% patients with ASB responded well to antibiotic treatment therapy and turned sterile. Klebsiella was the most prominent causative organism for recurrent ASB followed by E. coli. The rate of infection of E. coli significantly increased from 27.96% to 33.33% in recurrent ASB samples. Prevalence of S. aureus infection significantly decreased from 22.58% to 12.50% in recurrent ASB samples. Rate of infection by Enterococci remained unaltered.
Cytokine levels in recurrent ASB samples were still significantly high compared to non-ASB samples. IL-6 levels in recurrent ASB samples were significantly low when compared to samples prior to treatment. IL-10 and TNF-α levels did not show any significant change in samples prior to treatment and 6 months post treatment (Figure 1 and Figure 2). It is well established that levels of inflammatory cytokines such as IL-6 and TNF-α are significantly increased in T2 diabetes mellitus indicating low grade inflammation.11 The current study reveals that in case of an acute infection like ASB the levels of serum cytokines are still higher than that of the non ASB diabetic patients. ASB patients who responded well to antibiotic therapy had their serum cytokine levels comparable to that of non-ASB diabetic patients 6 months post therapy, whereas patients with recurrent infection still had significantly high levels of cytokines (Figure 2). This stands as a proof that ASB can cause significant increase in cytokine levels of patients with T2diabetes mellitus.
Figure 2. Cytokine estimations (IL-6, IL-10 and TNF-α) in serum of diabetic patients with recurrent ASB six months post treatment by ELISA method. Values expressed as mean±SEM. ** p<0.005, * p<0.05 compared to non-ASB group. ASB- Asymptomatic Bacteriuria, IL-6-Interleukin-6, IL-10- Interleukin-10- Interlukin-10, TNF-α- Tumor Necrosis Factor alpha
Prevalence of ASB in T2DM patients is a major concern, as such prolonged condition can affect kidney functioning and can lead to dire condition such as renal failure. The current study will be of great significance since we have evaluated the cytokine levels in patients with ASB. This may help us estimate the renal functioning in patients with T2DM. Cytokines either inflammatory or anti-inflammatory can serve as early markers for indicating the disease progression. Thus, the current study evaluates the role of cytokines in inflammation caused due to ASB in T2DM patients. Increase in IL-6 and TNF-α levels may indicate renal tissue damage caused due to inflammation by ASB. Whereas increased IL-10 levels, is a possible indicator of persistent infection with bacteria possibly resistant to antibiotics which may potentially lead to diabetic renal disease. Role of cytokines as indicators of renal damage needs to be further confirmed by follow-up study in these patients.
ACKNOWLEDGMENTS
The authors would like to thank Department of Microbiology, GVPIHCMT for facilitating this work. Authors also thanks Dr. P. Sailaja and Dr. R. Poorani for their support during the study and Mr. N Hanumanth for the statistical analysis.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
KVSBVS designed the study and performed the experiments. KVSBVS and PSJ analysed and interpreted the data. KVSBVS wrote the manuscript. PSJ, NRB and KS reviewed, edited and approved the final manuscript for publication.
FUNDING
None.
DATA AVAILABILITY
All data sets generated or analysed during this study are included in the manuscript.
ETHICS STATEMENT
This study was approved by the Institutional Ethical Committee, Gayatri Vidya Parishad Institute of Health Care and Medical Technology, Visakhapatnam, Andhra Pradesh, with reference number GVPIHCMT/IEC/20201208/03.
INFORMED CONSENT
Written informed consent was obtained from the participants before enrolling in the study.
- Mehta SR, Kashyap AS, Das S. Diabetes Mellitus in India: The Modern Scourge. Med J Armed Forces India. 2009;65(1):50-54.
Crossref - Geerlings SE. Urinary tract infections in patients with diabetes mellitus: epidemiology, pathogenesis and treatment. Int J Antimicrob Agents. 2008;31(Suppl 1):S54-7.
Crossref - Bharti A, Chawla SPS, Kumar S, et al. Asymptomatic bacteriuria among the patients of type 2 diabetes mellitus. J Family Med Prim Care. 2019;8(2):539-543.
Crossref - Bowker N, Shah RL, Sharp SJ, et al. Meta-analysis investigating the role of interleukin-6 mediated inflammation in type 2 diabetes. EBioMedicine. 2020;61:103062.
Crossref - Navarro-Gonzalez JF, Mora-Fernandez C. The role of inflammatory cytokines in diabetic nephropathy. J Am Soc Nephrol. 2008;19(3):433-442.
Crossref - Mahadevan P, Larkins RG, Fraser JRE, Fosang AJ, Dunlop ME. Increased hyaluronan production in the glomeruli from diabetic rats: a link between glucose-induced prostaglandin production and reduced sulphated proteoglycan. Diabetologia. 1995;38(3):298-305.
Crossref - Ophori EA, Imade P, Johnny EJ. Asymptomatic bacteriuria in patients with type-2 diabetes mellitus. J Bacteriol Res. 2010;2:14-17.
- Zhanel, GG, Harding GK, Nicolle LE. Asymptomatic Bacteriuria in Patients with Diabetes Mellitus. Rev Infect Dis. 1991;13(1):150-154.
Crossref - Geerlings SE, Meiland R, Hoepelman AIM. Pathogenesis of Bacteriuria in Women with Diabetes Mellitus. Int J Antimicrob Agents. 2002;19(6):539-545.
Crossref - Celen S, Oruc AS, Karayalcin R, et al. Asymptomatic bacteriuria and antibacterial susceptibility patterns in an obstetric population. ISRN Obstet Gynecol. 2011:2011:721872.
Crossref - Donate-Correa J, Martin-Nunez E, Muros-de-Fuentes M, Mora-Fernandez C, Navarro-Gonzalez JF. Inflammatory cytokines in diabetic nephropathy. J Diabetes Res. 2015;2015:948417.
Crossref - Nosadini R, Velussi M, Brocco E, et al. Course of renal function in type 2 diabetic patients with abnormalities of albumin excretion rate. Diabetes. 2000;49(3):476-484.
Crossref - Vestra MD, Mussap M, Gallina P, et al. Acute-phase markers of inflammation and glomerular structure in patients with type 2 diabetes. J Am Soc Nephrol. 2005;16(Suppl 1):S78-S82.
Crossref - Ellingsgaard H, Hauselmann I, Schuler B, et al. Interleukin-6 enhances insulin secretion by increasing glucagon-like peptide-1 secretion from L cells and alpha cells. Nat Med. 2011;17(11):1481-1489.
Crossref - Suzuki T, Imai J, Yamada T, et al. Interleukin-6 enhances glucose-stimulated insulin secretion from pancreatic beta-cells: potential involvement of the PLC-IP3-dependent pathway. Diabetes. 2011;60(2):537-547.
Crossref - Kurauti M, Costa-Junior J, Ferreira S, et al. Interleukin-6 increases the expression and activity of insulin-degrading enzyme. Sci Rep. 2017;7(1):46750.
Crossref - Navarro JF, Milena FJ, Mora C, et al. Tumor necrosis factor-alpha gene expression in diabetic nephropathy: Relationship with urinary albumin excretion and effect of angiotensin-converting enzyme inhibition. Kidney Int. 2005;99:S98-S102.
Crossref - Salek F, Mirzaei H, Khandaghi J, Javadi A, Nami Y. Apoptosis induction in cancer cell lines and anti-inflammatory and anti-pathogenic properties of proteinaceous metabolites secreted from potential probiotic Enterococcus faecalis KUMS-T48. Sci Rep. 2023;13(1):7813.
Crossref - Drage LKL, Robson W, Mowbray C, et al. Elevated urine IL-10 concentrations associate with Escherichia coli persistence in older patients susceptible to recurrent urinary tract infections. Immunity & Ageing. 2019;16(1):1-11.
Crossref
© The Author(s) 2024. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.