ISSN: 0973-7510
E-ISSN: 2581-690X
The development and evolution of viruses that cause disease have presented a formidable challenge to contemporary medicine and the global economy, not to mention a catastrophic risk to human health. Almost all of these viruses are zoonotic, meaning they were first identified in animals and then spread to humans. An emerging virus may cause only a few isolated instances, resulting in a limited outbreak, or it may cause widespread infection and spread to other parts of the world, triggering a full-blown epidemic. These kinds of emerging occurrences have occurred frequently and in many different forms during the past few decades. Monkeypox is a zoonotic disease caused by the monkeypox virus, a member of the orthopox family that also includes variola, cowpox, and vaccinia. Both animals and humans can get infected by this virus. Similar to smallpox this disease shows less severe rashes and lower mortality rate. The outbreak of monkeypox was declared a global public health emergency by the World Health Organization in July 2022. Unknown mutations and variations are linked to the recent epidemic. Presently, FDA approved tecovirimat, cidofovir and brincidofovir are there in market to treat monkeypox virus. But there are some side effects of these drugs as they are synthetic. So, scientists are working on natural remedies that can be used as alternative to these drugs. In the present study virtual screening of phytochemicals (N-(2-Allylcarbamoyl-4-chloro-phenyl)-3,4-dimethoxy-benzamide, 6-Dimethylaminonaphthene-1-sulfonicacid amide, Oleic Acid and dipentyl ester) from Allophylus serratus were employed against core viral cysteine proteases from monkeypox virus was done. The docking study revealed that selected ligands bind with target viral protein with binding affinity in the range of -5.0 to -6.7 kcal/mol. N-(2-Allylcarbamoyl-4-chloro-phenyl)-3,4-dimethoxy-benzamide showed the highest binding affinity of -6.7 kcal/mol which can be investigated in the future to design potential drugs against monkeypox virus. Thus, this study foresees the possibility of bioactive phytochemicals functioning as template molecules for further experimental evaluation of their efficiency against monkeypox virus.
Monkeypox, Tecovirimat, Docking Study, Drugs, Cysteine Proteases
Multiple viral illnesses have recently made a comeback, with the world seeing new cases of Nipah, influenza (H1N1, H5N1), Ebola, Zika, and coronaviruses in recent years (MERS-CoV and SARS-CoV-2). Recently included to this group are orthopoxviruses such as monkeypox virus (MPXV), variola, cowpox, and vaccinia, all of which can cause disease in humans.1,2 MPXV is an orthopox virus that leads to a viral zoonosis disease named as ‘monkeypox’, and is generally transmitted from monkeys to humans. This disease has similar symptoms to smallpox but it is less critical than it. But due to outbreak of this virus and increasing number of cases in July 2022, it was declared a public health emergency by World Health Organization (WHO).3 Starting with West and Central Africa, the outbreak led to around 80,328 cases all over the world, affecting 110 countries with 53 deaths, as reported by Centers for Disease Control and Prevention (CDC) on 19th November 2022.4 Monkeypox virus (MPXV) is a double helical DNA virus residing in animal hosts like rodents and non-human primates. From humans to humans, it is transmitted through semen contact and nasal droplets. Previously there were two different genetic clades based on geographic division that vary in severity and pathology of infection. The Central African trait was more severe and transmissible than the West African trait but due to general viral evolution a third multi country outbreak strain has been reported that led to spread of disease all over the world.5,6 Smallpox and monkeypox share somewhat same symptoms but monkeypox is less severe than smallpox. The same vaccination would have been effective against monkeypox but due to eradication of smallpox in 1980, the vaccine was discontinued. These vaccines can be first line of defense but their usage is limited due to ill effects working with live/ attenuated viruses.7 Many drugs have been reported for treating viruses from orthopox family. A p37 viral envelope protein inhibitor Tecovirimat, is generally administered that halts the spread of virus particles from infected cells. Another possible mechanism of action can be DNA polymerase inhibitors Cidofovir and Brincidofovir that can be used to inhibit the replication of viral particles.1,8 But the problem with these drugs is ample of side effects leading to many other ailments as they are synthetic. Efforts are needed to fill the gap of applicable science and basic science to stop the further outbreaks of new viruses that are emerging due to faster evolution and susceptibility of various hosts. So, the scientists are exploring something new and natural remedies including of medicinal herbs, plant metabolites and phytochemicals for these viruses that can be effective and safe alternative for these drugs as found useful against various important emerging and re-emerging pathogens.9-12 In addition, about 50 single nucleotide polymorphisms (SNPs) were discovered in the 2022 MPXV genome that were not present in the 2018-2019 MPXV strains that were circulating. This adds to the seriousness of the ongoing global pandemic and that MPXV is accelerating its progression. This highlights the critical need for research into new treatments for MPXV. In this respect, drug repurposing appears to be a useful alternative because it eliminates the requirement for drug development and shortens the timeline for drug discovery.
Having comparable signs and symptoms to smallpox, monkeypox is typically a self-limiting infection. In most people, monkeypox causes only mild symptoms, but in a few, significant complications may arise. Since MPXV is so very similar to the smallpox virus, vaccinations and antiviral treatments developed for smallpox can be used successfully against monkeypox.1,13,14 Cross-immunity against poxviruses, however, has been on the wane worldwide ever since smallpox vaccination campaigns were abandoned. That is why in 2022, when monkeypox broke out in various parts of the world, public health officials began to worry about the possibility of a new global pandemic.15,16 In the cytoplasm of infected cells, MPXV employs several virally encoded proteins, including cysteine proteinase, to replicate, as is the case with other poxviruses.17 In the life cycle of most viruses, proteases catalyze the cleavage of viral precursor polyproteins, playing a pivotal part in the process. Therefore, cysteine proteinase and other proteases enzymes are promising targets for the development of new antiviral medicines.18,19 Unfortunately, the MPXV cysteine proteinase enzyme has not yet been crystallized, which can hinder structure-based drug design and screening efforts.20
Researchers are increasingly looking to plant-derived compounds and bioactive phytochemicals in contemporary medicine because of their widespread antibacterial, antiviral, antivirulence, and pharmacological characteristics. Phytochemicals and other medications have been projected to be effective against SARS-CoV-2, Ebola, and Nipah virus in numerous in silico and pharmacoinformatics-based investigations.10,12,21 In the present study, our team has explored various compounds of well-known Indian medicinal plant Allophylus serratus for its potential to spread and prevent monkeypox disease. This drug is known in Ayurveda for its anti-viral and anti-inflammatory properties.22 There are many bioactive compounds present in this plant that are rich in flavonoids, tannins, steroids, phenolics, alkaloids and saponins. Various anti-inflammatory, anti-ulcer, anti-bacterial and anti-viral properties have been reported.23 Using this ancient Ayurvedic knowledge we have explored the binding affinities of core protease of MPXV with different bioactive compounds of this plant. Core proteases are cysteine proteases that are responsible for replication and cleavage of major membrane and structural proteins.24 So, this is a well-known target for inhibiting molecular mechanisms of MPXV. After deriving this target our team has identified binding affinities of this protease with various compounds using molecular docking studies. The results show that phytochemicals of Allophylus serratus can be used as a natural pharmacophore for countering MPX.
Retrieval of MPXV viral core cysteine proteases amino acid sequence
The FASTA sequence of core cysteine proteases of MPXV was retrieved from Uniprot (https://www.uniprot.org/).
Secondary and tertiary structure prediction
Secondary structure was depicted by PHYRE (http://www.sbg.bio.ic.ac.uk/~phyre2/html/page.cgi?id=index). 3-D structure of core cysteine protease of monkeypox virus was modelled by online server SWISS-MODEL (https://swissmodel.expasy.org/). After modelling, the structure was further validated by Protein Structure Validation Software Suite (PSVS)(https://mybiosoftware.com/psvs-1-5-proteins.
Receptor preparation
The verified 3-D model was prepared for docking studies by removing water molecules and heteroatoms from modeled receptor.
Ligand library preparation and analysis of physiochemical properties of ligands
Four phytochemicals were selected from Allophylus serratus, viz.N-(2-Allylcarbamoyl-4-chloro-phenyl)-3,4-dimethoxy-benzamide, 6-Dimethylaminonaphthene-1-sulfonic acid amide, Oleic Acid and dipentyl ester. The 3-D structures of selected phytochemicals were downloaded from pubchem in sdf format. ADME profiling of ligands at pH 7 viz. Unfavorable absorption, distribution, metabolism, and elimination of ligands were studied by using online tool. Physiochemical properties or Lipinski’s rule of five was also determined, which include LogP (<5), molecular weight (<500 Da), H-bond donor (5), molar refractivity, H-bond acceptor (<10), and drug likeliness are mentioned in Table 1.
Table (1):
Physio-chemical Properties of ligands.
| S.No. | Ligands | Pubchem ID | ADME Properties (Lipinski’s Rule of Five) | Drug Likeliness | |
|---|---|---|---|---|---|
| Properties | Values | ||||
| N-(2-Allylcarbamoyl-4-chloro-phenyl)-3,4-dimethoxy-benzamide | 2898006 | Molecular weight (<500 Da) | 74.5 | Yes | |
| LogP (<5) | 2.7 | ||||
| H-bond donar (5) | 2 | ||||
| H-bond acceptor (<10) | 6 | ||||
| Molar Refractivity | 98.55 | ||||
| 1. | 6-Dimethylaminonaphthene-1-sulfonic acid amide | Molecular weight (<500 Da) | 336 | Yes | |
| LogP (<5) | 3.4 | ||||
| H-bond donar (5) | 2 | ||||
| H-bond acceptor (<10) | 5 | ||||
| Molar Refractivity | 94.4 | ||||
| 2. | Dipentyl ester | Molecular weight (<500 Da) | 306. | Yes | |
| LogP (<5) | 4.3 | ||||
| H-bond donar (5) | 0 | ||||
| H-bond acceptor (<10) | 4 | ||||
| Molar Refractivity | 86.05 | ||||
| Oleic Acid | Molecular weight (<500 Da) | 282 | Yes | ||
| LogP (<5) | 5.9 | ||||
| H-bond donar (5) | 1 | ||||
| H-bond acceptor (<10) | 2 | ||||
| Molar Refractivity | 87 | ||||
Molecular Docking studies of phytochemicals with viral core cysteine protease
For in silico docking studies, PyRxv0.8 was run (utilizes Auto dock Vina). Discovery Studio Visualizer was used to visualize docked structure with highest binding affinity.
The in-silico experiments analysis reveals best binding between ligand candidates and viral target associated with MPXV. The higher binding affinity was used to determine the docking strength of each ligand molecule with its receptor.
Firstly, Amino acid sequence (58 residues) of cysteine proteases of MPXV was retrieved from Uniport having accession number Q5IXV7 (Table 2).
Table (2):
Amino acid sequence of cysteine proteases.
| >tr|Q5IXV7|Q5IXV7_MONPV Core protease I7 OS=Monkeypox virus MERYTDLVISKIPELGFTNLLCHIYSLAGLCSNIDVSKFLTNCNGYVVEKYDKSTTAGKV SCIPIGMMLELVESGHLSRPNSSDELDQKKELTDELTTRYHSIYDVFELPTSIPLAYFFK PQLREKVSKAIDFSQMDLKIDDLSRKGIHTGENPKVVKMKIEPERGAWMSNRSIKNLV SQFAYGSEVDYIGQFDMRFLNSLAIHEKFDAFMNKHILSYILKDKIKSSTSRFVMFGFCY LSHWKCVIYDKKQCLVSFYDSGGNIPTEFHHYNNFYFYSFSDGFNTNHRHSVLDNTNC DIDVLFRFFECTFGAKIGCINVEVNQLLESECGMFISLFMILCTRTPPKSFKSLKKVYTFFKFL ADKKMTLFKSILFNLQDLSLYITETDNAGLKEYKRMEKWTKKSINVICDKLTTKLNRIVDDDE |
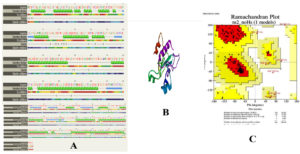
Then, the primary amino acid sequence was uploaded in secondary and tertiary structure prediction servers i.e. PHYRE and SWISS-MODEL respectively. Based on Phyre secondary structure modelling the alpha helix and beta strand represent 58% and 12% respectively. The template used for 3-D modeling was other cysteine proteases with PDB ID 6ups. The 3-D structure of cysteine protease was visualized by the PyMOL visualization tool. The secondary and tertiary model of cysteine proteases of MXOV is depicted in Figure 1. Ramachandran plot was drawn from PROCHECK and it was observed that 84.8% residues were present in most favoured regions followed by 11.0% residues in additionally allowed regions and 2.4% residues in generally allowed and 1.8% residues in disallowed regions. The overall G-factor score was found to be−0.98 (recommended value −0.5) for the probable model and the overall model average also had positive scores (cut-off score was >0.2) as established during verification of 3D structure. All of these programs together verified the probable 3-D structure of cysteine proteases of MPXV.
Figure 1. Secondary structure (A), Modelled 3-D structure of Cysteine proteases (B), Ramachandran plot (C)
Molecular Docking Analysis
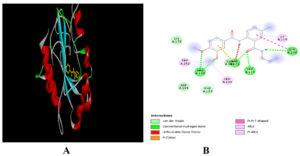
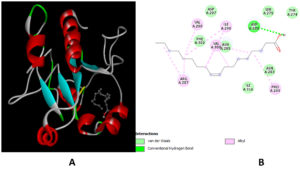
Molecular docking results revealed that N-(2-Allylcarbamoyl-4-chloro-phenyl)-3,4-dimethoxy-benzamide,6-Dimethylaminona-phthene-1-sulfonic acid amide, Oleic Acid and dipentyl ester showed the binding affinity in the range of -6.7 to -5.0 kcal/mol with target protein. Among four selected ligands N-(2-Allylcarbamoyl-4-chloro-phenyl)-3,4-dimethoxy-benzamide showed the highest binding affinity of -6.7 kcal/mol (Table 3). It formed five H-bonds with the amino acid residues of cysteine protease as shown in Figure 2.
Table (3):
Binding affinity and RMSD value range of N-(2-Allylcarbamoyl-4-chloro-phenyl)-3,4-dimethoxy-benzamide.
Ligand |
Binding Affinity (∆G Kcal/mol) |
rmsd/ub |
rmsd/lb |
|---|---|---|---|
N-(2-Allylcarbamoyl-4-chloro-phenyl)-3,4-dimethoxy-benzamide _uff_E=517.53 |
-6.7 |
0 |
0 |
N-(2-Allylcarbamoyl-4-chloro-phenyl)-3,4-dimethoxy-benzamide _uff_E=517.54 |
-6.1 |
13.726 |
10.913 |
N-(2-Allylcarbamoyl-4-chloro-phenyl)-3,4-dimethoxy-benzamide _uff_E=517.55 |
-5.8 |
14.907 |
11.879 |
N-(2-Allylcarbamoyl-4-chloro-phenyl)-3,4-dimethoxy-benzamide _uff_E=517.56 |
-5.7 |
14.663 |
11.851 |
N-(2-Allylcarbamoyl-4-chloro-phenyl)-3,4-dimethoxy-benzamide _uff_E=517.57 |
-5.6 |
8.212 |
3.433 |
N-(2-Allylcarbamoyl-4-chloro-phenyl)-3,4-dimethoxy-benzamide _uff_E=517.59 |
-5.6 |
29.831 |
26.371 |
N-(2-Allylcarbamoyl-4-chloro-phenyl)-3,4-dimethoxy-benzamide _uff_E=517.60 |
-5.6 |
8.475 |
3.438 |
N-(2-Allylcarbamoyl-4-chloro-phenyl)-3,4-dimethoxy-benzamide _uff_E=517.61 |
-5.3 |
13.281 |
10.247 |
N-(2-Allylcarbamoyl-4-chloro-phenyl)-3,4-dimethoxy-benzamide _uff_E=517.62 |
-5.3 |
14.3799 |
12.509 |
Figure 2. Docking of N-(2-Allylcarbamoyl-4-chloro-phenyl)-3,4-dimethoxy-benzamide with cysteine protease of MPXV. 3-D view (A) 2-D view (B) of binding of ligand with target protein
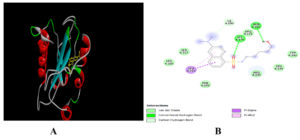
Similarly, 6-Dimethylaminonaphthene-1-sulfonic acid amide, dipentyl ester and oleic acid interact with binding affinity of -6.3, -5.1 and -5.0 kcal/mol (Table 4, 5 and 6). 6- Dimethylaminonaphthene-1-sulfonic acid amide interacts with target protein via two hydrogen bonds (Figure 3).
Table (4):
Binding affinity and RMSD value range of 6-Dimethylaminonaphthene-1-sulfonic acid amide.
Ligand |
Binding Affinity (∆G Kcal/mol) |
rmsd/ub |
rmsd/lb |
|---|---|---|---|
6-Dimethylaminonaphthene-1-sulfonic acid amide_uff_E=613.42 |
-6.3 |
0 |
0 |
6-Dimethylaminonaphthene-1-sulfonic acid amide_uff_E=613.43 |
-6.2 |
12.237 |
8.915 |
6-Dimethylaminonaphthene-1-sulfonic acid amide_uff_E=613.44 |
-6.2 |
12.171 |
6.97 |
6-Dimethylaminonaphthene-1-sulfonic acid amide_uff_E=613.45 |
-6.1 |
26.281 |
25.01 |
6-Dimethylaminonaphthene-1-sulfonic acid amide_uff_E=613.46 |
-5.9 |
28.455 |
26.562 |
6-Dimethylaminonaphthene-1-sulfonic acid amide_uff_E=613.47 |
-5.8 |
7.837 |
5.12 |
6-Dimethylaminonaphthene-1-sulfonic acid amide_uff_E=613.48 |
-5.8 |
27.684 |
25.753 |
6-Dimethylaminonaphthene-1-sulfonic acid amide_uff_E=613.49 |
-5.8 |
7.579 |
4.889 |
6-Dimethylaminonaphthene-1-sulfonic acid amide_uff_E=613.50 |
-5.7 |
11.442 |
9.925 |
Table (5):
Binding affinity and RMSD value range of dipentyl ester.
Ligand |
Binding Affinity (∆G Kcal/mol) |
rmsd/ub |
rmsd/lb |
|---|---|---|---|
dipentyl ester_uff_E=125.96 |
-5.1 |
0 |
0 |
dipentyl ester_uff_E=125.97 |
-4.7 |
8.108 |
1.768 |
dipentyl ester_uff_E=125.98 |
-4.7 |
26.741 |
25.441 |
dipentyl ester_uff_E=125.99 |
-4.6 |
6.874 |
0.95 |
dipentyl ester_uff_E=125.100 |
-4.5 |
2.135 |
1.476 |
dipentyl ester_uff_E=125.101 |
-4.1 |
9.863 |
4.012 |
dipentyl ester_uff_E=125.102 |
-4.1 |
3.202 |
1.844 |
dipentyl ester_uff_E=125.103 |
-3.9 |
7.125 |
2.026 |
dipentyl ester_uff_E=125.104 |
-3.8 |
27.473 |
25.867 |
Table (6):
Binding affinity and RMSD value range of Oleic acid.
Ligand |
Binding Affinity (∆G Kcal/mol) |
rmsd/ub |
rmsd/lb |
|---|---|---|---|
Oleic Acid_uff_E=80.35 |
-5 |
0 |
0 |
Oleic Acid_uff_E=80.36 |
-4.7 |
2.089 |
1.285 |
Oleic Acid_uff_E=80.37 |
-4.6 |
22.272 |
20.718 |
Oleic Acid_uff_E=80.38 |
-4.6 |
28.375 |
24.898 |
Oleic Acid_uff_E=80.39 |
-4.6 |
24.375 |
22.692 |
Oleic Acid_uff_E=80.40 |
-4.5 |
24.302 |
1.497 |
Oleic Acid_uff_E=80.41 |
-4.5 |
3.312 |
1.817 |
Oleic Acid_uff_E=80.42 |
-4.5 |
2.355 |
1.399 |
Oleic Acid_uff_E=80.43 |
-4.5 |
3.393 |
10.913 |
Figure 3. Docking of 6-Dimethylaminonaphthene-1-sulfonic acid amide with Cysteine protease of MPXV. 3-D view (A) 2-D view (B) of binding of ligand with target protein.
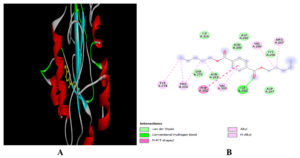
Further, dipentyl ester and Oleic acid also interacts via hydrogen bonds. Dipentyl ester and Oleic acid formed only one H-bonds via ILE and ASP residues of cysteine protease respectively (Figure 4 and 5). Moreover, all four ligands also formed Van Der Waals, alkyl and p- alkyl interactions with cysteine protease of MPXV.
Figure 4. Docking of dipentyl ester with Cysteine protease of MPOV. 3-D view (A) 2-D view (B) of binding of ligand with target protein
Figure 5. Docking of Oleic acid with Cysteine protease of MPXV. 3-D view (A) 2-D view (B) of binding of ligand with target protein
To stop the spread of MPXV infections, a successful strategy involving the repurposing of bioactive phytochemicals and existing pharmaceuticals is required.25-27 Saho et al.28 identified that Tipranavir, Cefiderocol, Doxorubicin, and Dolutegravir were the potential drugs among the FDA approved list for monkeypox virus.28 Similarly, Odhar et al.20 identified that tetracycline (antibiotics) strongly inhibits cysteine proteases of monkeypox virus.20 A natural biomolecule Glycocin F showed the highest binding with MPXV.12 High docking scores were also attained by lactococcin G and plantaricin ASM1 with the majority of related cell surface receptors and viral surface proteins. Study with phytochemicals is missing in literature; however, our team has published several reviews and editorial paper on monkeypox outbreak with promising insight into future perspectives.29-34 Our present study cannot be compared with previous studies as till date no in silico docking of MPXV with phytochemicals is reported.
A member of the same orthopoxvirus genus as the virus responsible for smallpox, MPXV has long been established as an endemic threat in Africa. Vaccination against orthopox viruses has been suspended for the past four decades, since smallpox illness was ultimately eradicated. The resurgence of MPXV in an under-vaccinated population is a global health emergency that requires swift action from the scientific community. Patients with monkeypox disease may benefit from long-term remedies provided by monkeypox-specific therapies rather than impractical mass immunization campaigns within a limited time frame. Reusing existing drugs to find new uses against MPXV saves time and money over developing and testing completely new drugs. Given the growing danger posed by zoonotic illnesses to human health, especially in light of the emergence of monkeypox and other poxviruses, it is crucial that the pharmacopeia be further investigated in order to combat these infections. Computationally designed docking studies are effective, stable, specific and low costing as compared to traditional study used for vaccine development. In the current study we use cysteine protease as target molecule and naturally occurring phytochemicals from medicinal plant as ligands (N-(2-Allylcarbamoyl-4-chloro-phenyl)-3,4-dimethoxy-benzamide,6-Dimethylaminonaphthene-1-sulfonic acid amide, Oleic Acid and dipentyl ester) and found that N-(2-Allylcarbamoyl-4-chloro-phenyl)-3,4-dimethoxy-benzamide has the highest binding affinity with target molecule. Therefore, after in vivo and in vitro study naturally occurring plant-based metabolites can be further used to design novel drugs against MPXV. The authors also believe that recent pandemics and epidemics, such as those caused by COVID-19 and monkeypox, may help bring much-needed attention to diseases that are now neglected or otherwise understudied, and hence motivate much-needed research.
ACKNOWLEDGMENTS
The author would like to thank Maharishi Markandeshwar (Deemed to be University) for providing facilities to conduct the work.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
PB, MG, SS, and GKB conceptualized the idea, performed experimentation and wrote the manuscript. SR, DC and AD performed formal analysis. KD and HST revised and approved the manuscript for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
Not applicable.
- Chakraborty S, Chandran D, Mohapatra RK, et al. Clinical management, antiviral drugs and immunotherapeutics for treating monkeypox. An update on current knowledge and futuristic prospects. Int J Surg. 2022d;105:106847.
Crossref - Mohapatra RK, Tuli HS, Sarangi AK, et al. Unexpected sudden rise of human monkeypox cases in multiple non-endemic countries amid COVID-19 pandemic and salient counteracting strategies: Another potential global threat? Int J Surg. 2022;103:106705.
Crossref - Kozlov M. Monkeypox goes global: Why scientists are on alert. Nature. 2022;606(7912):15-16.
Crossref - Centers for diseases control and prevenation (CDC). https://www.cdc.gov/poxvirus/monkeypox/response/2022/world-map.html
- Abrahim M, Guterres A, da Costa Neves PC. The emergence of new lineages of the Monkeypox virus could affect the 2022 outbreak. bioRxiv. 2022;
Crossref - Luna N, Ramirez AL, Munoz M, et al. Phylogenomic analysis of the monkeypox virus (MPXV) 2022 outbreak: Emergence of a novel viral lineage? Travel Med Infect Dis. 2022;49:102402.
Crossref - Adler H, Gould S, Hine P, et al. Clinical features and management of human monkeypox: a retrospective observational study in the UK. Lancet Infect Dis. 2022;22(8):1153-1162.
Crossref - Rizk JG, Lippi G, Henry BM, Forthal DN, Rizk Y. Prevention and Treatment of Monkeypox. Drugs. 2022;82(12):957-963.
Crossref - Dhama K, Karthik K, Khandia R, et al. Medicinal and Therapeutic Potential of Herbs and Plant Metabolites / Extracts Countering Viral Pathogens – Current Knowledge and Future Prospects. Curr Drug Metab. 2018;19(3):236-263.
Crossref - Anand AV, Balamuralikrishnan B, Kaviya M, et al. Medicinal plants, phytochemicals, and herbs to combat viral pathogens including SARS-CoV-2. Molecules. 2021;26(6):1775.
Crossref - Singh S, Kola P, Kaur D, et al. Therapeutic Potential of Nutraceuticals and Dietary Supplements in the Prevention of Viral Diseases: A Review. Front Nutr. 2021;8:679312.
Crossref - Dassanayake MK, Khoo TJ, Chong CH, Martino PD. Molecular Docking and In-Silico Analysis of Natural Biomolecules against Dengue, Ebola, Zika, SARS-CoV-2 Variants of Concern and Monkeypox Virus. Int J Mol Sci. 2022;23(19):11131.
Crossref - Tiecco G, Degli Antoni M, Storti S,Lina Tomasoni R, Castelli F, Quiros-Roldan E. Monkeypox, a Literature Review: What Is New and Where Does This concerning Virus Come From? Viruses. 2022;27;14(9):1894.
Crossref - Abdi SAH, Ali A, Sayed SF, Abutahir, Ali A, Alam P. Multi-Epitope-Based Vaccine Candidate for Monkeypox: An In Silico Approach. Vaccines (Basel). 2022;19;10(9):1564.
Crossref - Abduljalil JM, Elfiky AA. Repurposing antiviral drugs against the human monkeypox virus DNA-dependent RNA polymerase; in silico perspective. J Infect. 2022;8(6):702-769.
Crossref - Chadha J, Khullar L, Gulati P, et al. Insights into the monkeypox virus: Making of another pandemic within the pandemic? Environ Microbiol. 2022;24(10):4547-4560.
Crossref - Gulati P, Chadha J, Harjai K, et al. Targeting Envelope Proteins of Poxviruses to Repurpose Phytochemicals against Monkeypox: An In Silico Investigation. Preprints. 2022;2022100302.
Crossref - Banerjee R, Perera L, Tillekeratne LMV. Potential SARS-CoV-2 main protease inhibitors. Drug Discov Today. 2021;26(3):804-816.
Crossref - Tsai SC, Lu CC, Bau DT, et al. Approaches towards fighting the COVID 19 pandemic (Review). Int J Mol Med. 2021;47(1):3-22.
Crossref - Odhar HA. Computational Repurposing of FDA Approved Drugs Against Monkeypox Virus Cysteine Proteinase: A Molecular Docking and Dynamics Simulation Study. OSF Preprints. 2022;
Crossref - Burkhanova TM, Krysantieva AI, Babashkina MG, et al. In silico analyses of betulin: DFT studies, corrosion inhibition properties, ADMET prediction, and molecular docking with a series of SARS-CoV-2 and monkeypox proteins. Struct Chem. 2022;27:1-12.
Crossref - Dharmani P, Mishra PK, Maurya R, Chauhan VS, Palit G. Allophylus serratus: A plant with potential anti-ulcerogenic activity. J Ethnopharmacol. 2005;99(3):361-366.
Crossref - Jemal K, Sandeep BV Pola SR. A Review On Medicinal Importance Of Allophylus Serrattus And Premna Tomentosa. Int J Curr Res. 2015;7(10):21034-21039.
- Lam HYI, Guan JS, Mu Y. In Silico Repurposed Drugs against Monkeypox Virus. Molecules. 2022;27(16):5277.
Crossref - Khoo YW, Li S, Chong KP. In-silico primer designing and PCR for detection of monkeypox virus (MPXV). J Infect Public Health. 2022;15(12):1378-1380.
Crossref - Chandran D, Dhama K, MK, M A, et al. Monkeypox: An Update on Current Knowledge and Research Advances. J Exp Biol Agric Sci. 2022; 10(4):679-688.
Crossref - Zephyr J, Kurt Yilmaz N. Schiffer CA. Viral Proteases: Structure, Mechanism And Inhibition. Enzymes. 2021;50:301-333.
Crossref - Sahoo AK, Augusthian PD, Muralitharan I, et al. In Silico Identification Of Potential Inhibitors Of Vital Monkeypox Virus Proteins From FDA Approved Drugs. Mol Divers. 2022:1-16.
Crossref - Sah R, Mohanty A, Hada V, et al. The Emergence of Monkeypox: A Global Health Threat. Cureus. 2022;18;14(9):e29304.
Crossref - Chakraborty C, Bhattacharya M, Sharma AR, Dhama K. Monkeypox virus vaccine evolution and global preparedness for vaccination. Int Immunopharmacol. 2022a;20;113(Pt A):109346.
Crossref - Chakraborty C, Bhattacharya M, Pandya P, Dhama K. Monkeypox in South-East Asia: Is an alarming bell for this region? – Correspondence. Int J Surg. 2022b;106:106917.
Crossref - Ahmed SK, Abdulqadir SO, Hussein SH, et al. The impact of monkeypox outbreak on mental health and counteracting strategies: A call to action. Int J Surg. 2022;106:106943.
Crossref - Chakraborty S, Mohapatra RK, Chandran D, et al. Monkeypox vaccines and vaccination strategies: Current knowledge and advances. An update – Correspondence. Int J Surg. 2022c;105:106869.
Crossref - Sah R, Mohanty A, Siddiq A, et al. Monkeypox reported in India – South East Asia Region: Health and economic challenges. Lancet Reg Health Southeast Asia. 2022;4:100063.
Crossref
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.