ISSN: 0973-7510
E-ISSN: 2581-690X
Experiments were conducted to test the ability of chitin or chitosan to control anthracnose (Colletotrichum sp.) on cucumber (Cucumis sativus L.). Practical grade chitin and unpurified chitin (ground crab shells) were added to compost at concentrations of 0.05 and 0.1% and chitosan was applied to the leaves of the plant at the concentrations of 0.05 and 0.1%. A fungicide (penncozeb) was used as a positive control while water and untreated plants were used as negative controls. Conidia of Colletotrichum sp. were applied to all the plants to initiate even disease development. All three treatments of chitin, at various concentrations, resulted in control of Colletotrichum sp. that was comparable to the fungicide. These results showed that Colletotrichum sp. on cucumber could be controlled by the use of chitin or chitosan and that both gave comparable levels of control.
Anthracnose; Crab chitin; Nonchemical control; Biocontrol; Thailand.
Anthracnose caused by Colletotrichum spp. are economically important pathogens.1 Cucurbit anthracnose is distributed widely, wherever cucurbits are grown in the world and can cause serious losses in varieties of cucumber and melon.2 Anthracnose of cucumber is caused by the fungus Colletotrichum orbiculare (syn. C. lagenarium).3
The symptoms of cucumber anthracnose occur on all above ground parts of the plant. Lesions that appear on the cotyledons are small, water-soaked and pale in colour, chlorotic (yellow) or necrotic (brown). The lesions then progress, becoming larger and coalescing before the cotyledon dries up and dies. The symptoms on the leaves are similar. Other parts of the plant can also become infected, including the fruit. The conidia of C. orbiculare are oval or pill-shaped, clear and have no cross walls.3
Much work has been done on Colletotrichum spp. that cause anthracnose of chillies (Capsicum spp.) from within Thailand4,5 and from the rest of the world.6
Four mixtures of plant growth-promoting rhizobacteria (PGPR) strains were tested under field conditions in Thailand against anthracnose of long cayenne paper and mosaic disease of cucumber among other diseases and hosts.7 PGPR strains, not from Thailand, have been tested as possible means of inducing systemic resistance in cucumber to C. orbiculare.8 Wei et al.8 found that when PGRP strains were applied to cucumber seeds, some systemic resistance to C. orbiculare could be induced. A promising strain of endophytic Streptomyces sp. has also been identified for biological control of C. orbiculare on cucumber.9 A study on the extracts of 203 weed species found that several of the extracts inhibited C. orbiculare on cucumber while three of the extracts completely inhibited the anthracnose infection.10
Chitin is a major component of fungal cell walls and insect exoskeletons.11 Plants produce chitinase when under attack by fungal pathogens.11 When a mutant line has been produced that lacks the gene for production of chitinase the fungal infection is worse than in the wild type.12 Chitinases have been isolated from Streptomyces violaceusniger where they acted as antifungal compounds.13
Chitin, extracted from shellfish sources, is a unique biopolymer based on the N-acetyl-glucosamine monomer. When the number of N-acetyl-glucosamine units is higher than 50%, the biopolymer is called chitin, and when the number of N-glucosamine units is higher than that of the N-acetyl-glucosamine units, it is called chitosan. For over 40 years this biopolymer has been studied for use in biomedical applications.14 Chitin is a major constituent of waste from shellfish processing, the management of which is a pressing need.15
Chitin, added to the soil, has been used to control several different fungal pathogens of plants. The soil borne infection, Verticillium wilt, has been controlled by adding powdered crab shells to the soil at a concentration of 2%.16 Control of Fusarium oxysporum on peas, at a concentration of 0.3%, and celery was achieved by the addition of chitin to the soil.17,18 While rose blackspot has also been controlled by the addition of powdered chitin to the soil.19
Chitosan has been used to control fungal pathogens including Fusarium solani f. sp. glycines on soybean at concentrations ranging from 1-5%,20 Fusarium oxysporum f. sp. apii on celery17 and Blumeria graminis f. sp. hordei on barley at 2%.21 The post-harvest fungal pathogens Botrytis cinerea and Rhizopus stolonifer on strawberries at concentrations of 10% and 15%22 as well as B. cinerea and Penicillium expansum on tomatoes at concentrations ranging from 0.01-1%.23
On cucumbers chitosan has controlled B. cinerea at concentrations of 0.1%24 and Sphaerotheca fuliginea at concentrations of 1% and 2.5%.25 Pythium aphanidermatum has also been controlled by chitosan alone26 and when combined with an application of Lysobacter enzymogenes strain 3.1T8.27
Anthracnose, on post-harvest fruits, has been controlled by chitosan on papaya, at concentrations ranging from 0.1% to 3%,28,29 tomatoes and grapes at concentrations of 1% and 2.5%.30
Cucumber is an important crop in Thailand, particularly in the Northeast of Thailand.31 The possible use of chitin to control anthracnose would result in a reduction of the amount of fungicides used and would provide a use for a waste product. This study aims to test the effectiveness of applications of practical grade chitin, unpurified chitin and chitosan at a range of concentrations against a locally isolated Colletotrichum sp. on cucumber.
Fungal isolation
Cucumbers with visual symptoms of anthracnose were obtained from a market near Mahasarakham, Northeast Thailand. They had been grown locally.
Sections (1 cm2) of the samples, where the symptoms were visible, were cut using a scalpel and placed on a potato dextrose agar (PDA) (HiMedia Laboratories PvT. Ltd., India) plate. The samples were cultured at 25oC for up to two weeks. Individual colonies were sub-cultured onto further PDA plates and incubated at 25oC for two weeks again.
Colonies that were determined to be a species of anthracnose were re-plated to develop a stock from which test plants could be infected. They were also transferred to PDA slants and stored at 4oC.
Chemicals
Chitin was obtained from Sigma-Aldrich Co., USA, as ‘Chitin from crab shells, practical grade’. Chitosan was obtained from Fluka BioChenika, Japan. The chitosan was prepared for use following the method of Lui et al.23 It was dissolved in 1% HCL by stirring. This solution was then diluted to the required concentrations and the pH value was adjusted to 5.4 with NaOH.
Crab shells were ground to produce unpurified chitin. Crabs were obtained locally, the meat was removed and the shells were ground to produce a fine powder. This was stored in a sealed container at room temperature until use.
In-addition two products, available commercially in Maha Sarakham were also used. The first product (product 1) contained biological extracts and enzymes but no chitin. The label listed Colletotrichum as one of the species that it could control. The other product (product 2) contained 3% chitosan, from shrimps. This product did not list Colletotrichum as a species that it could control.
A fungicide (penncozeb, Elf Atochem) was used as a positive control.
Experimental design
Cucumber seeds, variety Ninja 179 (Chia Tai Seed, Thailand), were obtained from a local supplier. Three seeds were grown in each pot (14 cm diameter) containing locally sold compost. A controlled release fertilizer containing 14% nitrogen, 14% phosphorus pentoxide and 14% potassium oxide was added to the compost, as per the instructions on the container, before it was placed in the pots.
The treatments were practical grade chitin at 0.05 and 0.1% (w/w), unpurified chitin at 0.05 and 0.1% (w/w), chitosan at 0.05 and 0.1% (w/v), two commercial products (applied as per the label), fungicide (applied as per the label), water and untreated.
The practical grade chitin and the unpurified chitin were added to the compost as the seeds were being planted. The chitosan and two commercial products were applied, using a hand held pressurised sprayer, as the first true leaf was emerging on the seedlings. Four days after this the Colletotrichum sp. conidia were inoculated onto the plants. The conidia were harvested from the plate cultures and the conidial suspension was adjusted to a concentration of 50,000 +/- 5000 conidia per ml with a haemocytometer.32 Then four days after that the fungicide was applied. Each treatment was applied to three pots. The pots were arranged in a randomized block design and kept externally in a shaded location (average temperature 29.4oC).
A cotyledon and the first true leaf to emerge, on each plant, were tagged and repeatedly scored for the incidence and severity of symptoms twice a week using a seven point scale (0 = no visible symptoms, 1 = small pinprick lesions, 2 = 1-20% of leaf surface covered with lesions, 3 = 21-40%, 4 = 41-60%, 5 = 61=80% and 6= 81-100%).
Statistics
The three plants in each pot had their measurements combined to form an average for the pot. The combined measurements were used to determine the area under the disease progress curve (AUDPC) for each of the three pots. The AUDPCs were compared by analysis of variance (ANOVA) with Fisher’s least significance difference test (LSD) (OpenStat). P<0.05 was considered to be statistically significant.
The cotyledons showed no significant differences between any of the treatments and the fungicide or the untreated (data not shown).
The leaves of the untreated plants and the leaves treated with water had the highest amount of disease symptoms (Fig. 1). While the plants treated with the fungicide had the least amount of disease symptoms (Fig. 1). The symptoms that did develop appeared up to 8 d after the symptoms first appeared on the untreated plants.
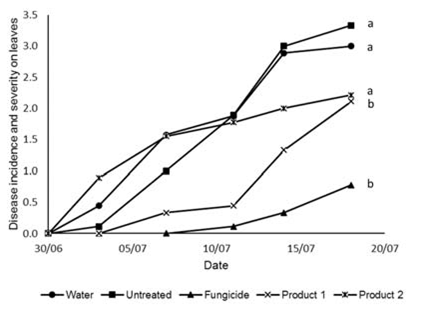 Fig. 1. Incidence and severity of Colletotrichum sp. lesions (measured on a scale from 0-6) on the leaves of cucumber plants inoculated with Colletotrichum sp. conidia and treated with one of two commercially available products (Product 1 and Product 2) compared to plants that were treated with a fungicide, had water applied or were untreated. Lower case letters indicate significant statistical differences as measured by AUDPC (P<0.05) according to LSD test following ANOVA (OpenStat).
Fig. 1. Incidence and severity of Colletotrichum sp. lesions (measured on a scale from 0-6) on the leaves of cucumber plants inoculated with Colletotrichum sp. conidia and treated with one of two commercially available products (Product 1 and Product 2) compared to plants that were treated with a fungicide, had water applied or were untreated. Lower case letters indicate significant statistical differences as measured by AUDPC (P<0.05) according to LSD test following ANOVA (OpenStat). Product 1 provided control that was statistically different from the untreated and similar to the level given by the fungicide over the period of this experiment. For the first 12 d the disease level was low but it started to increase rapidly after the twelfth day towards the end of the experiment (Fig. 1). Product 2 did not reduce the disease symptoms at all (Fig. 1).
Practical grade chitin at both concentrations provided control on the leaves that was statistically better than the untreated and no different from the fungicide. The control given closely followed that of the fungicide (Fig. 2).
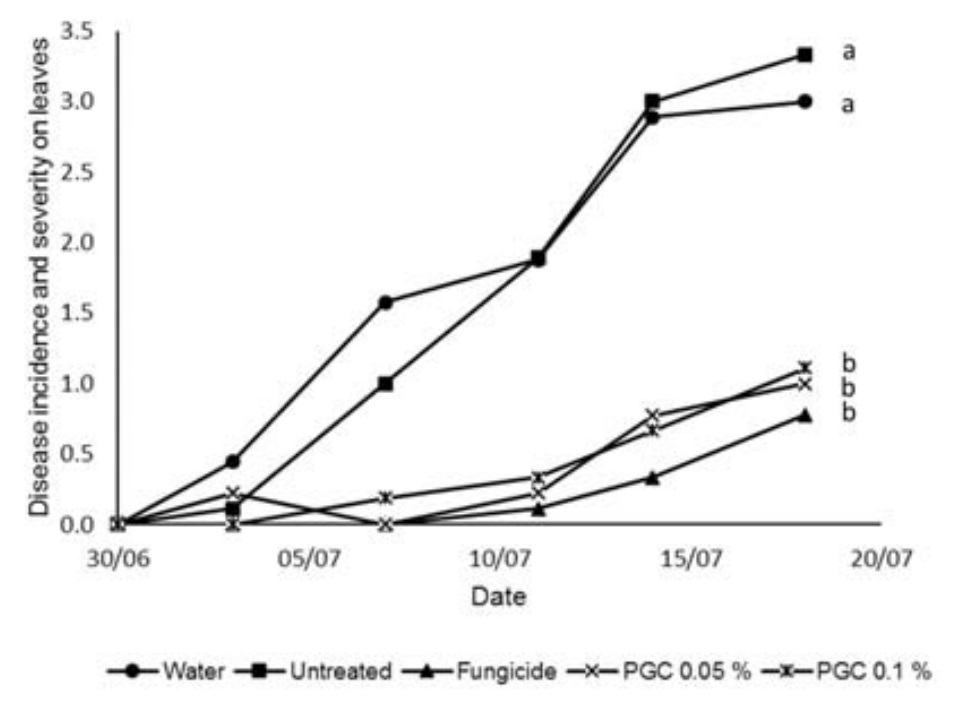 Fig. 2. Incidence and severity of Colletotrichum sp. lesions (measured on a scale from 0-6) on the leaves of cucumber plants inoculated with Colletotrichum sp. conidia and grown in compost with practical grade chitin (PGC) added at two different concentrations 0.05 and 0.1% compared to plants that were treated with a fungicide, had water applied or were untreated. Lower case letters indicate significant statistical differences as measured by AUDPC (P<0.05) according to LSD test following ANOVA (OpenStat).
Fig. 2. Incidence and severity of Colletotrichum sp. lesions (measured on a scale from 0-6) on the leaves of cucumber plants inoculated with Colletotrichum sp. conidia and grown in compost with practical grade chitin (PGC) added at two different concentrations 0.05 and 0.1% compared to plants that were treated with a fungicide, had water applied or were untreated. Lower case letters indicate significant statistical differences as measured by AUDPC (P<0.05) according to LSD test following ANOVA (OpenStat). Unpurified chitin at both concentrations provided control on the leaves that was statistically better than the untreated and no different from the fungicide. Here the control given did not follow that given by the fungicide as closely as with the practical grade chitin (Fig. 3).
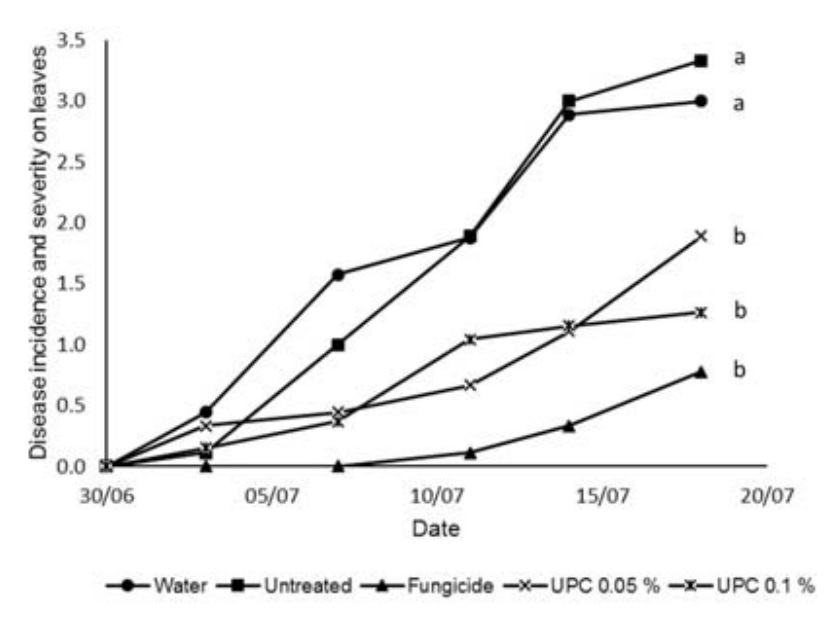 Fig. 3. Incidence and severity of Colletotrichum sp. lesions (measured on a scale from 0-6) on the leaves of cucumber plants inoculated with Colletotrichum sp. conidia and grown in compost with unpurified chitin (UPC) added at two different concentrations 0.05 and 0.1% compared to plants that were treated with a fungicide, had water applied or were untreated. Lower case letters indicate significant statistical differences as measured by AUDPC (P<0.05) according to LSD test following ANOVA (OpenStat)
Fig. 3. Incidence and severity of Colletotrichum sp. lesions (measured on a scale from 0-6) on the leaves of cucumber plants inoculated with Colletotrichum sp. conidia and grown in compost with unpurified chitin (UPC) added at two different concentrations 0.05 and 0.1% compared to plants that were treated with a fungicide, had water applied or were untreated. Lower case letters indicate significant statistical differences as measured by AUDPC (P<0.05) according to LSD test following ANOVA (OpenStat) Chitosan at the concentration of 0.05% gave control on the leaves that was statistically better than the untreated and no different from the fungicide. The concentration of 0.1% provided control that was statistically better than the untreated but not as good as the fungicide (Fig. 4).
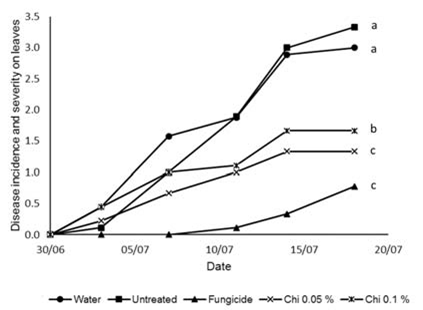 Fig. 4. Incidence and severity of Colletotrichum sp. lesions (measured on a scale from 0-6) on the leaves of cucumber plants inoculated with Colletotrichum sp. conidia and treated with chitosan (Chi) at two different concentrations 0.05 and 0.1% compared to plants that were treated with a fungicide, had water applied or were untreated. Lower case letters indicate significant statistical differences as measured by AUDPC (P<0.05) according to LSD test following ANOVA (OpenStat)
Fig. 4. Incidence and severity of Colletotrichum sp. lesions (measured on a scale from 0-6) on the leaves of cucumber plants inoculated with Colletotrichum sp. conidia and treated with chitosan (Chi) at two different concentrations 0.05 and 0.1% compared to plants that were treated with a fungicide, had water applied or were untreated. Lower case letters indicate significant statistical differences as measured by AUDPC (P<0.05) according to LSD test following ANOVA (OpenStat) There were no significant differences between the levels of control, on the leaves, provided by the three different types of chitin that were tested in this work (Table 1).
Table (1):
Significant statistical differences as measured by AUDPC (P<0.05) according to LSD test following ANOVA (OpenStat) between cucumber plants that were either grown in compost amended with practical grade chitin (PGC) or unpurified chitin (UPC) at the concentrations of 0.05 and 0.1% and plants which were treated with chitosan (Chi) at the concentrations of 0.05 and 0.1%. * = statistically different, NSD = not statistically different.
UPC 0.05% |
UPC 0.1% |
|
|---|---|---|
PGC 0.05% |
NSD |
NSD |
PGC 0.1% |
NSD |
NSD |
UPC 0.05% |
UPC 0.1% |
|
Chi 0.05% |
NSD |
NSD |
Chi 0.1% |
NSD |
NSD |
PGC 0.05% |
PGC 0.1% |
|
Chi 0.05% |
NSD |
NSD |
Chi 0.1% |
NSD |
NSD |
This study found that chitin, as either practical grade or unpurified, and chitosan provided control of this Colletotrichum sp. on cucumber. The level of control given by both the concentrations, which the different forms of chitin were used at, was significantly better than the untreated and in all but one case that was the same as that given by the fungicide.
There were significant differences between the treatments on the leaves. The untreated and the water were the same, showing that water alone does not have an inhibitory effect on the development of this Colletotrichum sp. on cucumber and so any inhibitory effects seen from the treatments must be due to the treatment. The fungicide significantly inhibited the development of this Colletotrichum sp. when compared to the untreated control. Since biocontrol methods, such as the use of chitin, are often less effective, as a single treatment, than a traditional chemical control method any results that give control comparable to that given by the fungicide would show great promise for future commercial use.
Product 1 gave control of this Colletotrichum sp. that was statistically comparable to the fungicide but the level of diseased leaf area was starting to increase towards the end of the experiment, possibly suggesting that the control given was not long lasting and the product would need reapplications. Product 2 did not give significant control of this Colletotrichum sp. on cucumber. Product 1 listed Colletotrichum spp. on the label as one of the suitable targets for the product but Product 2 did not, but it did contain chitosan at 3% and so was included in the study. Any other benefits that the products might have given to the plants were not investigated in this study.
The practical grade chitin and the unpurified chitin gave control of this Colletotrichum sp., at both concentrations, that was the same as that given by the fungicide. These results are comparable to the results of Bell et al.,17 Guy and Baker,18 Hall et al.,19 and Huang et al.,16 who all found that applications of chitin to the soil could result in reduced fungal disease symptoms.
There were no significant differences between the control given by the different concentrations of the practical grade chitin and the unpurified chitin. This suggests that if there was large scale adoption of chitin as a biocontrol product it would be possible to use the unrefined waste from shellfish processing that poses an environmental problem.15 This would hopefully result in a lower price and so a greater adoption by farmers.
The chitosan at a concentration of 0.05% gave control of this Colletotrichum sp. that was similar to that given by the fungicide. Unlike the chitin, the chitosan also resulted in a treatment that was significantly different from the untreated and the fungicide (0.1%). As has been shown by many authors previously, but not on anthracnose of cucumber, chitosan can result in significant control of fungal pathogens.17,20,21,22,23,24,26,27,28,29,30 The addition of chitin to the soil is something that could only be archived pre-planting, so the use of a foliar application of chitosan would be able to increase the amount of time that it would be possible for plant protection to be derived from chitin or its derivative.
The chitosan was applied to the leaves before the inoculation of the leaves as Bautista-Banos et al.28 and Ben-Shalom et al.24 had both shown that this was the most effective way to use chitosan.
There were no differences between the applications of chitin, either as practical grade or unpurified, and chitisan showing that both forms give comparable levels of control.
There were no significant differences between the AUDPCs of the treatments or controls for the cotyledons. Inagaki et al.10 found that cotyledons were suitable for the identification of weed extracts that could inhibit anthracnose lesions on cucumbers caused by C. lagenarium. In-addition chitosan significantly inhibited the powdery mildew incidence on cucumber cotyledons.25 Concentrations of 10-6 spores mm-1 and concentrations of 4×104 and 4×105 conidia ml-1 were used by Inagaki et al.10 and Moret et al.,25 respectively. Both authors added Tween-20 to the conidial suspensions at a low level, while in this work the method of Monroe et al.32 was followed where the conidial concentration was between the amounts used by the previous authors but without Tween-20. Monroe et al.32 worked on the leaves rather than the cotyledons. In addition, the treatments were applied at the optimal time for application to the leaves rather than to the cotyledons as in the previous papers.
This work has shown that chitin as either practical grade or unpurified and chitosan can all control Colletotrichum sp. on cucumber plants. This is the first report of these three types of chitin being used to control a Colletotrichum sp. isolated from samples obtained in Thailand. This work has shown that it would be possible to use chitin to control anthracnose on cucumber plants. It has also shown that unpurified chitin is comparable to practical grade chitin or chitosan so the waste products from shellfish processing could be use directly with minimal processing to help reduce the amount of fungicides used in agriculture.
ACKNOWLEDGMENTS
The authors would like to thank the staff and students of the Department of Biology, Faculty of Science, Mahasarakham University, especially Mr. Nitis Smantong and Miss. Jeerapar Noiseeluang. The authors would also like to acknowledge the National Research Council of Thailand for the funding (fiscal year 2010) to carryout the work and the British Society of Plant Pathology (BSPP) Undergraduate Vacation Bursary Fund (2010) for the funding for Mr. Nitis Smantong.
- Bailey, J.A., Jeger, M.J. Colletotrichum: Biology, pathology and control. CABI Publishing, Wallingford, UK, 1992.
- Averre, C.W. Anthracnose of cucurbits. Plant Pathology Extension, Collage of agriculture and Life Sciences, North Carolina State University, 1991. http://www.ces.ncsu.edu/depts/pp/notes/Vegetable/vdin011/vdin011.htm Date accessed 20/08/2010.
- Palenchar, J., Treadwell, D.D., Datnoff, L.E., Gevens, A.J. Cucumber anthracnose in Florida (PP266). Gainesville: University of Florida Institute of Food and Agricultural Sciences, 2009. http://edis.ifas.ufl.edu/pp266 Date accessed 20/08/2010.
- Oanh, L.T.K., Korpraditskul, V., Rattanakreetakul, C. A pathogenicity of anthracnose fungus, Colletotrichum capsici on various Thai chilli varieties. Kasetsart Journal, 2004; 38: 103-108.
- Than, P.P., Jeewon, R., Hyde, K.D., Pongsupasamit, S., Mongkolporn, O., Taylor, P.W.J. Characterization and pathogenicity of Colletotrichum species associated with anthracnose on chilli (Capsicum spp.) in Thailand. Plant Pathology, 2008; 57: 562-572.
- Than, P.P., Prihastuti, H., Phoulivong, S., Taylor, P.W.J., Hyde, K.D. Chilli anthracnose disease caused by Colletotrichum species. Journal of Zhejiang University Science B, 2008; 9: 764-778.
- Jetiyanon, K., Fowler, W.D., Kloepper, J.W. Broad-spectrum protection against several pathogens by PGPR mixtures under field conditions in Thailand. Plant Disease, 2003; 87: 1390-1394.
- Wei, G., Kloepper, J.W., Tuzun, S. Induction of systemic resistance of cucumber to Colletotrichum orbiculare by select strains of plant growth-promoting rhizobacteria. Phytopathology, 1991; 81: 1508-1512.
- Shimizu, M., Yazawa, S., Ushijima, Y. A promising strain of endophytic Streptomyces sp. for biological control of cucumber anthracnose. Journal of General Plant Pathology, 2009; 75: 27-36.
- Inagaki, H., Yamaguchi, A., Kato, K., Kageyama, C., Iyozumi, H., Oki, Y. Screening of weed extracts for antifungal properties against Colletotrichum lagenarium, the causal agent of anthracnose in cucumber. Weed Biology and Management, 2008; 8: 276-283.
- Ramonell, K., Berrocal-Lobo, M., Koh, S., Wan, J., Edwards, H., Stacey, G., Somerville, S. Loss-of-function mutations in chitin responsive genes show increased susceptibility to the powdery mildew pathogen Erysiphe cichoracearum1[w]. Plant Physiology and Biochemistry, 2005; 138: 1027-1036.
- Kojimn, M., Yoshiknwa, T., Ueda, M., Monomura, T., Matsuda, Y., Toyoda, H., Miyntake, K., Arai, M., Fukamizo, T. Family 19 chitinase from Aeromonas sp. No.10S-24: Role of chitin-binding domain in the enzymatic activity. Journal of Biochemistry, 2005; 137: 235-242.
- Shekhar, N., Bhattacharya, D., Kumar, D., Gupta, R.K. Biocontrol of wood-rotting Fungi with Streptomyces violaceusniger XL-2. Canadian Journal of Microbiology, 2006; 52: 805-808.
- Khor, E., Lim, L.Y. Implantable applications of chitin and chitosan. Biomaterials, 2003; 24: 2339-2349.
- Gohel, V., Singh, A., Vimal, M., Ashwini, P., Chhatpar, H.S. Bioprospecting and antifungal potential of chitinolytic microorganisms. African Journal of Biotechnology, 2006; 5: 54-72.
- Huang, J., Li, H., Yuan, H. Effect of organic amendments on Verticillium wilt of cotton. Crop Protection, 2006; 25: 1167-1173.
- Bell, A.A., Hubbard, J.C., Liu, L., Davis, R.M., Subbarao, K.V. Effects of chitin and chitosan on the incidence and severity of fusarium yellows of celery. Plant Disease, 1998; 82: 322-328.
- Guy, S.O., Baker, R. Inoculum potential in relation to biological control of fusarium wilt of peas. Ecology and Epidemiology, 1977; 67: 72-78.
- Hall, A.M., Ali, A., Pascoe, B. Can crab protect roses from blackspot? Proceedings of the BCPC Conference – Crop Science and Technology 2003, 2: 925-928.
- Parpagdee, B., Kotchadat, K., Kumsopa, A., Visarathanonth, N. The role of chitosan in protection of soybean from sudden death syndrome caused by Fusarium solani f. sp. glycines. Bioresource Technology, 2007; 98: 1352-1358.
- Faoro, F., Maffi, D., Cantu, D., Iriri, M. Chemical-induced resistance against powdery mildew in barley: the effects of chitosan and benzothiadiazole. Biocontrol, 2008; 53: 387-401.
- Ghaouth, A.E., Arul, J., Grenier, J., Asselin, A. Antifungal activity of chitosan on two postharvest pathogens of strawbery fruits. Phytopathology, 1992; 82: 398-402.
- Liu, J., Tian, S., Meng, X., Xu, Y. Effects of chitosan on control of postharvest diseases and physiological responses of tomato fruit. Postharvest Biology and Technology, 2007; 44: 300-306.
- Ben-Shalom, N., Ardi, R., Pinto, R., Aki, C., Fallik, E. Controlling gray mould caused by Botrytis cinerea in cucumber plants by means of chitosan. Crop Protection, 2003; 22: 285-290.
- Moret, A., Munoz, Z., Garces, S. Control of powdery mildew on cucumber cotyledons by chitosan. Journal of Plant Pathology, 2009; 91: 375-380.
- Ghaouth, A.E., Arul, J., Grenier, J., Benhamon, N., Asselin, A., Belanger, R. Effect of chitosan on cucumber plants: Suppression of Pythium aphanidermatum and induction of defense reactions. Phytopathology, 1994; 84: 313-320.
- Postma, J., Stevens, L.H., Wiegers, G.L., Davelarr, E., Nijhuis, E.H. Biological control of Pythium aphanidermatum in cucumber with a combined application of Lysobacter enzymogenes strain 3.1T8 and chitosan. Biological Control, 2009; 48: 301-309.
- Bautista-Banos, S., Hernandez-Lopez, M., Bosquez-Molina, E., Wilson, C.L. Effects of chitosan and plant extracts on growth of Colletotrichum gloeosporioides, anthracnose levels and quality of papaya fruit. Crop Protection, 2003; 22: 1087-1092.
- Hewajulige, I.G.N., Sultanbawa, Y., Wijeratnam, R.S.W., Wijesundara RLC. Mode of action of chitosan coating on anthracnose disease control in papaya. Phytoparasitica, 2009; 37: 437-444.
- Munoz, Z., Moret, A., Garces, S. Assessment of chitosan for inhibition of Colletotrichum sp. on tomatoes and grapes. Crop Protection, 2009; 28: 36-40.
- Nath, P., Papademetriou, M., Piluek, K., Herath, E.M. The vegetable sector in Thailand: A review. FAO Regional Office for Asia and the Pacific, 1999. http://www.fao.org/docrep/004/ac145e/AC145E09.htm#app Date accessed 20/08/2010.
- Monroe, J.S.. A model defining the relationship between temperature and leaf wetness duration, and infection of watermelon by Colletotrichum orbiculare. Plant Disease, 1997; 81.
© The Author(s) 2017. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


