ISSN: 0973-7510
E-ISSN: 2581-690X
In recent days, non albicans candida species are emerging, many of these species are inherently resistant to routinely used antifungals. Hence, the need for speciation of candida is important in the treatment point of view. Speciation of candida can be done by conventional methods, using chromogenic media, serological, molecular methods. Most of the laboratories use Hicrome for identification of yeast species considering its ease, rapidity . To determine the usefulness of Hicrome agar in the identification of different species of candida in comparison with corn meal agar. The candida isolated from cutaneous candidiasis were included. Species identification was done by colony color on Hicrome, and confirmed by morphology on corn meal agar for 80 candida isolates. The 10 different species of candida were identified. Emerging species of candida – Candida kefyr, C.zeylanoides, C.lusitaniae C.lipolytica were identified by morphology on corn meal agar which were misidentified as C.glabrata by Hicrome. Identification of C.albicans, C.tropicalis, C.krusei by colony colour on Hicrome correlated with the morphology on corn meal agar. So, the incorporation of corn mealagarin the routine yeast identification is more judicious than Hicromeas it increase the accuracy in the identification of candida species within in the same time span as that of Hicrome.
Hicrome, corn meal agar, Candida
Candida albicans remains the most common causative agent of both superficial and deep fungal infections1. But recent reports suggest that a shift has occurred in the distribution of infections, with NAC being increasingly detected2.
Due to the epidemiological alteration in the distribution of Candida species as well as significant increasing trend of either intrinsic or acquired resistance in some of these fungi, the precise identification of Candida species is necessary for effective antifungal therapy and also for prevention of nosocomial infections3. The strains of C. lusitaniae may show resistance to amphotericin B so, for example, the automatic prescription of amphotericin B in a patient with a septicemia due to C. lusitaniae 4.
A large variety of methods have been developed with the aim of facilitating rapid, accurate yeast identification4. Many tests with different techniques from conventional to molecular methods are available for yeast identification.
Clinical microbiology laboratories face an important challenge to select a system for yeast identification that is accurate, cost-effective, easily interpreted and reasonably rapid 4.
Now a days, chromogenic media are frequently used in the direct and rapid identification of yeasts because different Candida species produce unique colors on these media.5
Our study is to evaluate the usefulness of Hicrome agar for speciation of candida in comparison with the corn meal agar.
The samples from cutaneous candidiasis cases (nail clippings from onychomycosis, swabs from intertrigo/napkin rash ) were included in the study. A total number of 80 candida were isolated following culture on the SDA slants. Candida isolates were speciated using Hicrome. Hicrome Candida agar product code number M-677 was obtained commercially. Plates were prepared as per manufacturer’s instructions. 5-8 colonies from 24-48 hr old SDA slants were inoculated on Hicrome agar and incubated at 37°C for 48 hrs. Colony morphology and colour was noted and compared with HiMedia instructions6. As per HiMedia instructions, depending on the colour of colonies species were identified as follows
On Hicrome agar medium C.albicans appear as light green coloured smooth colonies, C.tropicalis appear as blue to purple coloured raised colonies. C.glabrata colonies appear as cream to white smooth colonies, while C.krusei appear as purple fuzzy colonies. C.parapsilosis appears as cream coloured colonies with mauve tinge6. Then the colony were inoculated on 1cm x 1 cm block of corn meal agar (CMA) block. The agar block was covered with sterile coverslip and placed in a sterile Petri dish moistened with filter paper and incubated at room temperature for 48 hours7,8.
After 48 hrs, the slide was placed on the microscopic stage and the edge of the cover slip was observed using 10x and 40x objectives for chlamydospores, pseudohyphae, hyphae, blastospores, blastoconidia etc and the Candida were speciated7,8
Table 1 Showing the identification of various candida species by Hicrome and cornmeal agar. In our study 10 different candida species were identified by each method. But Candida kefyr, C.zeylanoides, C.lusitaniae, C.lipolytica were not identified by Hicrome.
Table (1):
Showing the identification of various candida species by Hicrome and cornmeal agar.
Isolates |
Detected on CMA |
Detected on CA |
|---|---|---|
Candida albicans |
24 |
24 |
Candida tropicalis |
18 |
18 |
Candida parapsilosis |
17 |
12 |
Candida glabrata |
04 |
16 |
Candida guilliermondii |
05 |
0 |
Candida krusei |
06 |
06 |
Candida kefyr |
03 |
0 |
Candida zeylanoides |
01 |
0 |
Candida lusitaniae |
01 |
0 |
Candida lipolytica |
01 |
0 |
Total |
80 |
80 |
Table 2 Showing sensitivity and specificity of Hicrome agar against CMA 100% sensitivity of Hicrome agar was observed in identification of C.albicans, C.tropicalis, C. krusei, C.glabrata. Moderate sensitivity of 70.58% was observed in C.parapsilosis. 100% specificity was observed in C.parapsilosis, C. krusei. 96.77% & 96.42% specificity was observed in C. tropicalis, C.abicans respectively. 84.21% specificity was observed in C.glabrata.
Table (2):
Showing sensitivity and specificity of Hicrome agar against the cornmeal agar.
Species |
Sensitivity % |
Specificity % |
|---|---|---|
Candida albicans |
100% |
96.42% |
Candida tropicalis |
100% |
96.77% |
Candida parapsilosis |
70.58% |
100% |
Candida glabrata |
100% |
84.21% |
Candida krusei |
100% |
100% |
Table 3 Showing sensitivity, specificity of Hicrome as observed in other similar studies. The sensitivity, specificity of C.albicans, C.tropicalis, C. krusei are in agreement with the similar studies done by VP Baradkar 5, Manisha 9.
Table (3):
Showing sensitivity, specificity of candida of Hicrome agar in comparison with other similar studies.
Species |
Our study |
VP Baradkar5 |
Manisha9 |
|---|---|---|---|
C albicans |
100%, 96.42% |
96.55, 96.42 |
100%,100% |
C tropicalis |
100%,96.77% |
100%,100% |
92.9,100% |
C parapsilosis |
70.58%,100% |
80%, 98.03% |
—– |
C glabrata |
100%,84.21% |
90.9%, 88.23% |
100%, 96.42% |
C krusei |
100%, 100% |
————— |
100%, 98.42 % |
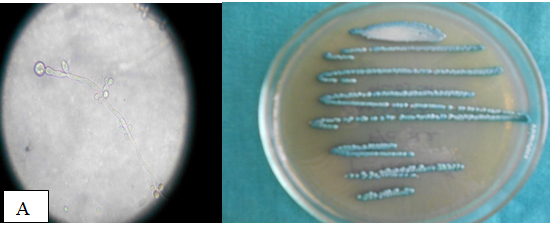
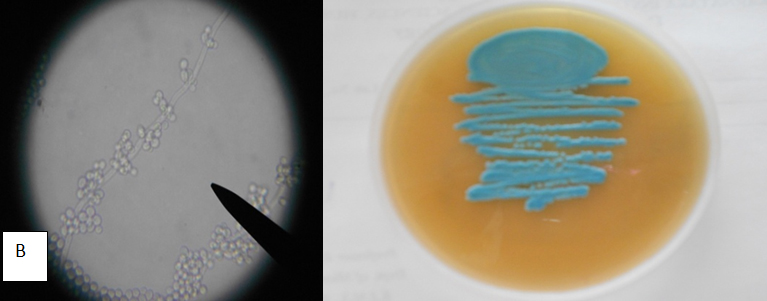
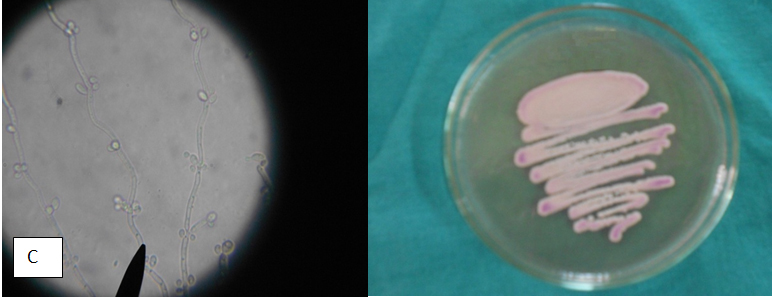
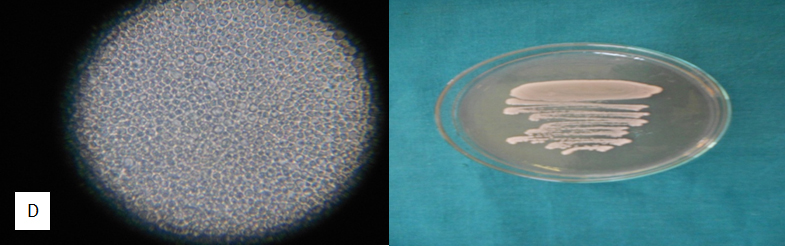
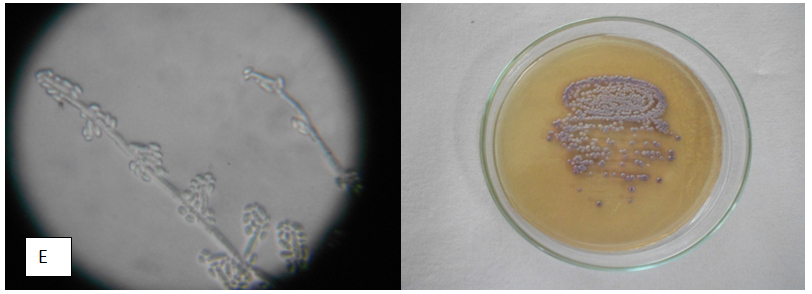
Table 4 Appearance of different candida isolates on Hicrome, Cornmeal agar Hicrome agar for candida falsely identified C.parapsilosis as C.glabrata based on colony colour. Candida kefyr, C.zeylanoides, C.lusitaniae C.lipolytica were identified by morphology on corn meal agar which were misidentified as C.glabrata by Hicrome. The appearance of candida isolates on Hicrome agar, microscopic appearance on Cornmeal agar are given in Fig. 1.
Table (4):
Appearance of different candida isolates on Hicrome, cornmeal agar.
Species |
Colony colour on Hicrome |
Morphology on Corn meal agar(CMA) |
Identification with Hicrome |
Identification with Hicrome vs CMA |
|---|---|---|---|---|
C.albicans (24) |
Light green colored smooth colonies |
Pseudohyphae with terminal chlamydospores; clusters of blastoconidia at septa. |
Identified all strains as C.albicans |
Accurate identification all 24 |
C.tropicalis (18) |
Blue to purple coloured raised colonies |
Blastoconidia anywhere along pseudohyphae |
Identified all strains as C.tropicalis |
Accurate identification all 18 |
C.parapsilosis (17) |
Cream colored colonies with mauve tinge |
Blastoconidia along curved pseudohyphae; giant mycelial cells |
Only 12 identified, other 5 strains misidentified as C.glabrata |
Identified all 17 |
C. glabrata(4) |
Cream to white smooth colonies |
No pseudohyphae; cells small; terminal budding |
16 strains were identified, morphology essential |
4 strains correlated by both methods |
C.krusei (6) |
Purple fuzzy colonies |
Pseudohyphae with cross–match sticks or treelike blastoconidia |
Identified all strains as C.krusei |
Accurate identification all 6 |
C.guillerimondii(5) |
Cream colonies |
Fairly short, fine pseudohyphae, clusters of blastoconidia at septa |
misidentified as C.glabrata |
5 strains correlated by both methods |
C.kefyr(3) |
Cream colonies |
Elongated blastoconidia resembling logs in a stream along pseudohyphae |
misidentified as C.glabrata |
3 strains correlated by both methods |
C.zeylanoides(1) |
Cream colonies |
Pseudohyphae give feather- like appearance at low power |
misidentified as C.glabrata |
1 strains correlated by both methods |
C.luistaniae(1) |
Cream colonies |
Short chains of elongate blastoconidia along curved pseudohyphae |
misidentified as C.glabrata |
1 strains correlated by both methods |
C.lipolytica(`1) |
Cream colonies |
Elongated blastoconidia in short chains along pseudohyphae |
misidentified as C.glabrata |
1 strains correlated by both methods |
Approximately five Candida species were considered pathogenic in the 1960s, recent reviews listed at least 17 Candida species as being pathogenic 4. When studies are limited to this genus, the most frequently isolated species were C.albicans, C.tropicalis, C.glabrata, C.parapsilosis 5,9.
Isolation of other Candida species, such as C. krusei, C. guilliermondii, C.lipolytica, C. kefyr, along with other unspecified species were also increased10. These results are reflected by increase in the case reports concerning new and emerging yeasts11.
Many tests with different techniques from conventional to molecular methods are available for yeast identification. But selection of the method by a lab depends on its affordability (sample size etc.), reliability of the test result, and also the time factor.
Many studies states that the potential advantage of chromogenic media is the straightforward identification of mixed yeast infections12. Now a day’s Hicrome agar is most commonly employed for the yeast identification in clinical microbiology laboratories.
In our study 10 different species of candida were identified by morphological study oncorn meal agar. On Hicrome only 5 species of candida were identified ie, C.albicans, C.tropicalis, C.glabrata, C.krusei , C. parapsilosis. (Table 1)
Hicrome agar was useful in identification of C.albicans, C.tropicalis, C.glabrata, C.krusei with 100 % sensitivity, moderatesensitivityof70.58 % was observed in C.parapsilosis. 100 %specificitywas observed inC.parapsilosis, C.krusei. Moderate specificity was observed inC.glabrata84.21%. (Table 2)
The sensitivity, specificity of C.albicans C.tropicalis , C. krusei C. glabrata, C.parapsilosisare in agreement with the similar studies done by VP Baradkar 5, Manisha9 (Table 3). As observed in the previous studies done on speciation of candida using Hicrome 5,9 only the above-mentioned species were identified, new emerging candida species identification were not stated.
In our study we noted that while identifying candida species based on the color description by manufacturer 8, 21 isolates which appeared as white colonies were interpreted as C. glabrata. When morphological identification by corn meal agar was done on these isolates, C. parapsilosis(5) , Candida kefyr(3) , C.zeylanoides(1), C.lusitaniae(1), C.lipolytica(1), C. guilliermondii (5) were identified. (Table 4)
Hicrome agar falsely identified C.parapsilosis asC.glabrata was stated in the study done by Sagar et al13 which is similar to our study.
The advantage of Hicrome agar in identification of candida species i e., ease of the test method compared to conventional methods and rapidity of identification cannot be ignored in the era of emergence of NAC. Identification on Hicrome agar poses a problem as it is based on colour, features like fuzzy, hue etc and variations in intensity of color with passage of time. The interpretation becomes subjective, besides the media only recommends identification of C.albicans, C.tropicalis, C. glabrata, C.krusei, C.parapsilosis.
The study by Hazen 10 states the variable efficacy for fluconazole is evident with C.glabrata, C.parapsilosis, C.rugosa,. C.tropicalis, S. cerevisiae, and T.beigelii which adds to the importance of accurate identification.
As observed in our study many new species identifications were missed when identification only Hicrome considering its, ease and rapidity.
The turnaround time taken for identification by morphology on corn meal agar is 48hr 7is similar to that on Hicrome as per the manufacturer’s instructions 6.
But identification by morphological study on corn meal agar demands the skill from the lab personnel, which can be mastered.
Koehler14 et al opine that careful observation of the yeast morphology on corn meal agar, adds confidence in the identification of candida species, which will also alert the microbiologist about the presence of unusual isolates.
In view of accurate identification, the limitations of Hicrome in yeast identification not to be ignored by a clinical microbiology laboratory. The incorporation of corn meal agar in routine yeast identification prevents misidentification, adds confidence, improves the mycology skills among the lab personnel without compromising cost, or time factor.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
- Jagdish Chander. Candidiasis. Textbook of Medical Mycology, 3rd edn. India: Mehta, 2009 ;pp 267-290.
- Capoor RM, Nair D, Deb M, Verma KP, Srivastava L, Aggarwal P. Emergence of Non-albicans Candida species and Antifungal Resistance in a Tertiary care hospital. Jpn. J. Infect. Dis., 2005; 58: 344-348.
- Fallahi AA, Korbacheh P, Zaini F, Mirhendi H, Zeraati H, Noorbakhsh F et.al.Candida Species in Cutaneous Candidiasis Patients in the Guilan Province in Iran; Identified by PCR-RFLP Method. Acta Medica Iranica., 2013; 51( 11):799 -804.
- Freydiere AM, Guinet R, Boiron P. Yeast identification in the clinical microbiology laboratory: phenotypical methods. Medical Mycology., 2001; 39: 9-33.
- Baradkar V P, Mathur M, Kumar S. Hichrom candida agar for identification of candida species. Indian J Pathol Microbiol., 2010; 53:93-5.
- HiCrome Single Streak rapid Differentiation Series. HIMEDIALaboratories.
- LaroneDH. Medically important fungi, A guide to identification, 4th edn. Washington DC :AmericanSocietyforMicrobiologyPress, 2002; 104-142.
- Medical mycology laboratory procedures. Centre of advanced research in medical mycology (ICMR)PGI, Chandigarh.
- Chauhan Dhamecha NM, Shah MU, Rajput AH, Shah UV, Kadam M. Use of Hichrom Candida Agar for the rapid identification of Candida species directly from clinical species. Forum 99
- Hazen CK. New and Emerging Yeast Pathogens. Clinical Microbiology Reviews., 1995; 8(4): 462–478.
- Banerjee SN, Emori TG, Culver DH, Gaynes RP, Jarvis WR, Horan T, Edwards JR et. secular trends in nosocomial primary bloodstream infections in the United States, 1980-1989. National Nosocomial Infections Surveillance System. Am J Med., 1991; 91(3B):86S-89S.
- Borg von-Zepelin, M., H. Eiffert, M. Kann, and R. Ru¨chel. Changes in the spectrum of fungal isolates: results from clinical specimens gatherein 1987/1988 compared with those in 1991/1992 in the University Hospital, Go¨ttingen, Germany. Mycoses.,1993; 36: 247–253.
- Sagar K, Mangalkar S, Gohel T Isolation of Different Candida Species from Clinical Samples and Comparison between Conventional and Hicrome Technique of Identification International Journal of Science and Research (IJSR) ISSN (Online): 2319-7064.
- Koehler PA, Chu CK, Houang TSE , Cheng BFA. Simple, Reliable, and Cost-Effective Yeast Identification Scheme for the Clinical Laboratory. Journal of Clinical Microbiology., 1999; 37(2): 422–426.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.