ISSN: 0973-7510
E-ISSN: 2581-690X
Urinary tract infections (UTIs) are among the most common bacterial infections of the urinary tract, accounting for a high percentage of all infections. The insertion of a catheter into the bladder increases the susceptibility of the patient to tract infection and serves as the initial site of infection by introducing pathogenic organisms into the tract. Indwelling catheters in the bladder facilitate the colonization of uropathogens through various mechanisms. The objective of this study was to compare the different pathogenic bacteria causing UTIs and their antibiotic sensitivity patterns in catheterized and non-catheterized patients. The study included catheterized and non-catheterized patients, regardless of gender, exhibiting clinical signs or symptoms of urinary tract infection. Urine samples were examined and cultured for bacterial growth using standard microbiological procedures. After confirming the pathogens, antibiotic susceptibility testing was conducted on Mueller-Hinton agar using the Kirby-Bauer disc diffusion method. Out of the 200 samples, 88 were catheterized and 112 were non-catheterized. Catheterized patients exhibited a higher infection rate (39, 44.32%) compared to non-catheterized patients (31, 27.68%), and women had a higher infection rate than men. Various organisms were isolated, with Escherichia coli being the most common organism in both catheterized and non-catheterized patients. Among the tested drugs against gram-negative organisms, nitrofurantoin displayed higher sensitivity. The present study demonstrated a higher incidence of bacterial infection in catheterized patients compared to non-catheterized patients, highlighting the importance of avoiding unnecessary catheter insertion. To prevent antimicrobial resistance, it is crucial to implement various infection control policies, care bundle approaches, and regular surveillance.
UTI, CAUTI, Antibiotic Sensitivity, GNB, GPC
Healthcare-associated infections are infections acquired during a hospital stay and treatment for medical and surgical conditions.1 Urinary tract infections (UTIs) are the most common bacterial infections and are a major type of hospital-acquired infection.2 If the immune system fails to eliminate bacteria that have bypassed the first line of defense, it can result in a urinary tract infection or a more severe illness.3 As a foreign body, the catheter serves as the initial entry point for pathogenic organisms into the urinary tract.4 Infections occur due to the formation of bacterial colonies in the epithelial cells of the urinary tract.5 Bacterial entry can occur during catheter insertion, through its lumen, or along the catheter-urethral or catheter-skin interface.6 Inserted catheters provide a surface for the attachment of bacterial adhesion receptors recognized by host cells. Additionally, catheters can damage the protective mucosa of the uroepithelium, exposing new binding sites for bacterial adhesion.7 The presence of an indwelling urinary catheter disrupts the normal mechanical defenses of the host, leading to bladder overdistension and residual urine, which provides a breeding ground for microbial growth.8
Patients with UTIs present with symptoms such as fever, lower abdominal pain, dysuria, urinary urgency, pyuria, and leukocytes in urine.9 The presence of bacteria in urine, with a count of 105 CFUs/ml, indicates a urinary tract infection.10 Catheterized patients have a threefold higher risk of hospitalization, longer hospital stays, and increased antibiotic therapy compared to non-catheterized patients.11 Additionally, all iatrogenic illnesses significantly contribute to morbidity and mortality among hospitalized patients.12 Often, the etiological agents associated with urinary tract infections consist of multidrug-resistant pathogens. While the specific pathogen profile may vary from place to place, E. coli remains the most common causative pathogen.13
Therefore, this study aimed to compare the rate of UTI between catheterized and non-catheterized patients, as well as the antibiotic susceptibility of different isolated organisms.
Sample population
This retrospective study was conducted by the Department of Microbiology at our institute over three months. The study included a total of 200 patients, comprising 88 with catheter-associated UTI (CAUTI) and 112 with non-catheter-associated UTI (NON-CAUTI).
Sample collection
Urine samples (2 mL) were collected from each catheterized patient, either by puncturing the catheter tube with a needle or syringe using aseptic techniques for short-term catheterization or from freshly placed catheters for long-term indwelling urinary catheter cases. In symptomatic patients, urine samples were collected immediately before initiating antimicrobial therapy. For non-catheterized patients, midstream urine samples were collected in sterile urine containers and transported to the laboratory.
Urine Culture
All urine samples were cultured on blood and MacConkey agar plates. After inoculation, the plates were incubated at 37°C in an incubator for 16-18 hours, and bacterial growth was examined the following day. Bacterial growth was identified using standard microbiological procedures, including examination of bacterial colony morphology, Gram stain appearance, motility testing using a hanging drop preparation, and biochemical tests such as catalase, oxidase, indole, citrate utilization, urea hydrolysis, triple sugar iron, sugar fermentation, methyl red, Voges–Proskauer, oxidation fermentation, phenylpyruvic acid, coagulase, and bile esculin hydrolysis tests. Once the bacterial pathogen was confirmed, antibiotic susceptibility testing was conducted on Mueller–Hinton agar using the Kirby–Bauer disc diffusion method to determine the susceptibility pattern. The agar plates were incubated at 37°C for 16–18 hours, and after incubation, the susceptibility pattern was noted, measured, and interpreted following the guidelines of the CLSI. The antibiotics tested included ampicillin/sulbactam (20 mg), cotrimoxazole (25 mcg), cephalexin (30 mcg), tetracycline (30 mcg), and ciprofloxacin (5 mcg). Specific antibiotics for gram-negative bacteria were ceftizoxime (30 mcg), nitrofurantoin (300 mcg), sparfloxacin (10 mcg), gatifloxacin (10 mcg), norfloxacin (10 mcg), ofloxacin (5 mcg), piperacillin/tazobactam (100/10 mcg), imipenem (10 mcg), and for gram-positive bacteria were cefotaxime (30 mcg), levofloxacin (5 mcg), linezolid (30 mcg), cloxacillin (1 mcg), roxithromycin (15 mcg), lincomycin (2 mcg), gentamycin (10 mcg), cefoxitin (30 mcg). The sensitivity patterns of the organisms are reported as percentages of the total number of cases in each group.
Out of the 200 urine samples collected from patients, 88 were identified as catheter-associated UTI (CAUTI) cases, while 112 were categorized as non-catheter-associated UTI (NON-CAUTI) cases (Table 1). Among the catheterized patients, 39 (44.32%) samples exhibited bacterial growth, while 49 (55.68%) samples did not show any bacterial growth. In the non-catheterized group, out of the 112 samples tested, 31 (27.68%) displayed bacterial growth, while 81 (72.32%) were negative (Table 2).
Table (1):
Showing sample division in catheter-associated UTI (CAUTI) and non-catheter-associated UTI (NON-CAUTI).
Samples |
Percentage |
|
|---|---|---|
CAUTI |
88 |
44% |
NON-CAUTI |
112 |
56% |
Total |
200 |
100% |
Table (2):
Urine culture results in catheterized and non-catheterized patients.
Urine culture |
CAUTI |
NON-CAUTI |
Total |
|---|---|---|---|
Positive |
39 (44.32%) |
31(27.68%) |
70(35%) |
Negative |
49(55.68%) |
81(72.32%) |
130(65%) |
Total |
88 |
112 |
200 |
The present study revealed that among the 88 catheterized patients, 48 were women and 40 were men, while among the 112 non-catheterized patients, 68 were women and 44 were men (Table 3). Table 4 illustrates that the incidence of infection was higher in women compared to men, both in catheterized patients (27 cases, 69.23%) and non-catheterized patients (24 cases, 77.42%).
Table (3):
Gender-wise distribution of CAUTI and NON-CAUTI patients.
Female |
Male |
Total |
|
|---|---|---|---|
CAUTI |
48 |
40 |
88 |
NON-CAUTI |
68 |
44 |
112 |
Total |
116 |
84 |
200 |
Table (4):
Gender-wise culture positivity in catheterization and non-catheterization patients.
| Catheterization | Gender | Positive by culture |
|---|---|---|
| CAUTI | Male | 12 (30.77%) |
| Female | 27 (69.23%) | |
| NON-CAUTI | Male | 07 (22.58%) |
| Female | 24 (77.42%) |
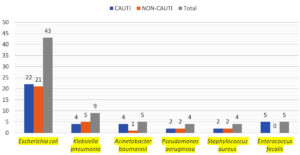
The isolated organisms included E. coli, Acinetobacter baumannii, Klebsiella pneumoniae, Pseudomonas aeruginosa, Staphylococcus aureus, and Enterococcus faecalis. Among these, E. coli was the most common pathogen in both, catheterized patients (22 cases, 56.40%) and non-catheterized patients (21 cases, 67.74%). Table 5 displays the organisms isolated from samples of catheterized and non-catheterized patients, while Figure provides a graphical representation of these findings.
Table (5):
Organisms isolated in catheterized and non-catheterized patient samples.
Bacterial Isolates |
CAUTI |
NON-CAUTI |
Total |
|---|---|---|---|
Escherichia coli |
22 (56.40%) |
21 (67.74%) |
43 (61.43%) |
Klebsiella pneumonia |
4 (10.26%) |
5 (16.13%) |
9 (12.86%) |
Acinetobacter baumannii |
4 (10.26%) |
1 (3.23%) |
5 (7.15%) |
Pseudomonas aeruginosa |
2 (5.13%) |
2 (6.45%) |
4 (5.71%) |
Staphylococcus aureus |
2 (5.13%) |
2 (6.45%) |
4 (5.71%) |
Enterococcus fecalis |
5 (12.82%) |
0 |
5 (7.14%) |
Total |
39 (100%) |
31 (100%) |
70 (100%) |
Antimicrobial resistance (AMR) has become a substantial clinical challenge in treating infections caused by numerous bacterial pathogens, with its prevalence continually rising. Table 6 enlists the antibiotic susceptibility patterns of 14 commonly utilized drugs against various gram-negative organisms. Notably, nitrofurantoin exhibited a higher sensitivity rate in patients with both CAUTI and NON-CAUTI. Additionally, Table 7 displays the sensitivity patterns of different antibiotics against gram-positive organisms in patients with CAUTI and NON-CAUTI, revealing linezolid as the most frequently employed antibiotic against Gram-positive organisms.
Table (6):
Sensitivity pattern of a gram-negative organism to various antibiotic agents.
| Antibiotic Tested | E. coli | K. pneumonia | A. baumannii | P. aeruginosa | ||||
|---|---|---|---|---|---|---|---|---|
| Catheter-Associated UTI | Non-Catheter-Associated UTI | Catheter-Associated UTI | Non-Catheter-Associated UTI | Catheter-Associated UTI | Non-Catheter-Associated UTI | Catheter-Associated UTI | Non-Catheter-Associated UTI | |
| Ampicillin/ Sulbactam | 27.27% | 28.57% | – | – | – | – | – | – |
| Co-trimoxazole | 45.45% | 52.38% | 50% | 40% | 75% | 100% | – | – |
| Cephalexin | 27.27% | 28.57% | 25% | 40% | – | – | – | – |
| Tetracycline | 45.45% | 47.62% | 50% | 40% | 50% | – | – | – |
| Ciprofloxacin | 27.27% | 28.57% | 25% | 20% | 50% | 100% | – | – |
| Ceftizoxime | 27.27% | 28.57% | 25% | 25% | 25% | – | – | – |
| Nitrofurantoin | 90.91% | 90.48% | 75% | 80% | 75% | 100% | 50% | 100% |
| Sparfloxacin | 77.27% | 90.48% | 75% | 80% | 75% | 100% | 50% | 50% |
| Gatifloxacin | 45.45% | 47.62% | 50% | 40% | 75% | 100% | 50% | 50% |
| Norfloxacin | 18.18% | 28.57% | 25% | 20% | 75% | 100% | – | – |
| Ofloxacin | 18.18% | 28.57% | 25% | 20% | 75% | 100% | – | – |
| Piperacillin/ Tazobactam | 68.18% | 66.67% | 75% | 100% | 100% | 100% | 100% | 100% |
| Imipenem | 77.27% | 76.19% | 75% | 80% | 75% | 100% | 100% | 100% |
(- Resistant to antibiotics)
Table (7):
Sensitivity pattern of a gram-positive organism to various antibiotic agents.
| Antibiotic Tested | Staphylococcus aureus | Enterococcus fecalis | |
|---|---|---|---|
| CAUTI | NON-CAUTI | CAUTI | |
| Ampicillin/ Sulbactam | 50% | 50% | 20% |
| Co-trimoxazole | 100% | 100% | 60% |
| Cephalexin | 50% | 50% | 20% |
| Tetracycline | 50% | 100% | 40% |
| Ciprofloxacin | 50% | 50% | 40% |
| Cefotaxime | 50% | 50% | – |
| Levofloxacin | – | 50% | 20% |
| Linezolid | 100% | 100% | 100% |
| Cloxacillin | 50% | 50% | – |
| Roxithromycin | 50% | 50% | 20% |
| Lincomycin | 100% | 100% | 20% |
| Gentamycin | 50% | 100% | 20% |
| Cefoxitin | 50% | 50% | 20% |
(- Resistant to antibiotics)
UTIs rank as the second most prevalent infectious disease in the community, following respiratory tract infections.14 Among the complications commonly associated with the use of indwelling urinary catheters, CAUTIs are the most frequent.15 This can be attributed to the fact that a sterile bladder is directly connected to the heavily colonized perineum through the urinary catheter, providing a pathway for bacteria to enter both the external and internal surfaces of the catheter.16 The pooling of urine in the bladder or catheter, along with urinary stasis, promotes bacterial growth. Additionally, catheter obstruction can cause bladder distension and damage to the bladder mucosa, heightening the vulnerability to bacterial invasion.17
In the current study, CAUTIs were observed in 39 patients (44.32%), which is consistent with previous studies conducted by Shukla S et al.18 and Kathak R et al.,19 who reported CAUTI rates of 40% and 42%, respectively. Furthermore, the prevalence rate of NON-CAUTIs in the present study was 31 (27.68%), which aligns with the findings of research conducted by Shukla S et al.18 and Khan R et al.20 who reported rates of 31% and 33.4%, respectively.
The present study findings indicate that both CAUTI and NON-CAUTI cases show a higher prevalence of infection in women. Among women with CAUTI, 69.23% had an infection compared to 30.77% infection in men. Similarly, in NON-CAUTI cases, women had a higher infection rate of 77.42% compared to 22.58% in men. These results are consistent with the findings reported by Shukla S et al.,18 where women with CAUTI had a 70.58% infection rate and men had a 29.41% infection rate. In NON-CAUTI cases, women had a 77.77% infection rate, while men had a 22.22% infection rate.
In the current study, the most frequently isolated organism from patients with CAUTI was E. coli (56.40%), followed by E. faecalis (12.82%), K. pneumoniae (10.26%), A. baumannii (10.26%), P. aeruginosa (5.13%), and S. aureus (5.13%). A similar study conducted by Bhatia N et al.21 on CAUTI reported similar findings, with E. coli accounting for 59.10% of cases, followed by K. pneumoniae (19.69%), E. faecalis (15.15%), and S. aureus (6.06%). Another study by Karthikeya et al.22 reported E. coli as the predominant organism (50.54%), followed by K. pneumoniae (15.45%), P. aeruginosa (10%), E. faecalis (9.09%), Candida (8.18%), Acinetobacter species (3.63%), Proteus mirabilis (2.27%), and S. aureus (0.9%).
In the current study on NON-CAUTI cases, the most common organism isolated was E. coli (67.74%), followed by K. pneumoniae (16.13%), P. aeruginosa (6.45%), S. aureus (6.45%), and A. baumannii (3.23%). In a study conducted by Shukla S et al.,18 among patients with NON-CAUTI, Escherichia coli (55.55%) was the most common bacterium, followed by Klebsiella (11.11%), Enterococcus (11.11%), Proteus (7.40%), Pseudomonas (7.40%), S.aureus (3.70%), and Serratia fanticola (3.70%).
In the present study, E. coli isolates showed the highest sensitivity to nitrofurantoin (90.91%), followed by imipenem and sparfloxacin (77.27%) in CAUTI. A study conducted by Acharya A et al.23 reported similar findings with nitrofurantoin showing a sensitivity of 78% and imipenem showing a sensitivity of 52.1%.
Regarding ciprofloxacin, in the current study, it exhibited sensitivity rates of 27.27% and 28.57% against E. coli in patients with CAUTI and NON-CAUTI, respectively. In a study conducted by Hossain et al.,24 ciprofloxacin demonstrated sensitivity rates of 37.38% and 52.95% in E. coli CAUTI and NON-CAUTI patients, respectively.
Among the isolated Klebsiella species in CAUTI, 75% exhibited resistance to ciprofloxacin, which aligns with the findings of Arina S,25 where 74.35% resistance to ciprofloxacin was observed. In contrast, Pseudomonas, which is sensitive to imipenem, demonstrated a 100% sensitivity rate, consistent with the study conducted by Almalki S. et al.26 in patients with CAUTI.
Regarding gram-positive organisms, S. aureus exhibited 100% sensitivity to co-trimoxazole, which is in line with the findings reported by Acharya A et al.23 where a sensitivity rate of 100% to co-trimoxazole was also observed.
The present study highlights that urinary tract infections occurred more frequently in catheterized patients, with a higher incidence observed in women compared to men. These findings emphasize the importance of regular screening and surveillance for antimicrobial resistance among the pathogens causing UTIs. The use of indwelling catheters is associated with an increased risk of symptomatic UTIs. Therefore, it is crucial to develop, implement, and monitor infection control policies and clinical practices aimed at minimizing infections associated with the use of such devices. One of the primary objectives of these infection control policies should be to reduce the utilization of indwelling urinary catheters and to promptly remove catheters when they are no longer necessary.
ACKNOWLEDGMENTS
The authors would like to thank all the staff members of the microbiology department for their help and support.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made direct and intellectual contributions to the work and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This study was approved by the Institutional ethics committee, M. P. Shah Govt. Medical College and Guru Gobind Singh Hospital, Jamnagar, with reference number IEC/CERTI/163/04/2022.
- Collins AS. Preventing Health Care-Associated Infections. In: Hughes RG, ed. Patient Safety and Quality: An Evidence-Based Handbook for Nurses. Rockville (MD): Agency for Healthcare Research and Quality (US); April 2008.
- Patil AB, Nadagir SD, Praveen J, Dhaduti R, et al. Catheter-associated urinary tract infection: Aetiology, ESBL production, and risk factors. Journal, Indian Academy of Clinical Medicine. 2014;15(1):23.
- Feneley RC, Hopley IB, Wells PN. Urinary catheters: history, current status, adverse events and research agenda. J Med Eng Technol. 2015;39(8):459-470.
Crossref - Adegun PT, Odimayo MS, Olaogun JG, Emmanuel EE. Comparison of uropathogens and antibiotic susceptibility patterns in catheterized ambulant middle-aged and elderly Nigerian patients with bladder outlet obstruction. Turk J Urol. 2018;45(1):48-55.
Crossref - Sheerin NS. Urinary tract infection. Medicine. 2011;39(7):384-389.
Crossref - Geerlings SE. Clinical Presentations and Epidemiology of Urinary Tract Infections. Microbiol Spectr. 2016;4(5).
Crossref - Hashmi S, Kelly E, Rogers SO, Gates J. Urinary tract infection in surgical patients. Am J Surg. 2003;186(1):53-56.
Crossref - Garibaldi RA, Burke JP, Britt MR, Miller MA, Smith CB. Meatal colonization and catheter-associated bacteriuria. N Engl J Med. 1980;303(6):316-318.
Crossref - Hooton TM, Bradley SF, Cardenas DD, et al. Diagnosis, prevention, and treatment of catheter-associated urinary tract infection in adults: 2009 International Clinical Practice Guidelines from the Infectious Diseases Society of America. Clin Infect Dis. 2010;50(5):625-663.
Crossref - Schmiemann G, Kniehl E, Gebhardt K, Matejczyk MM, Hummers-Pradier E. The diagnosis of urinary tract infection: a systematic review. Dtsch Arztebl Int. 2010;107(21):361-367.
Crossref - Kunin CM, Douthitt S, Dancing J, Anderson J, Moeschberger M. The association between the use of urinary catheters and morbidity and mortality among elderly patients in nursing homes. Am J Epidemiol. 1992;135(3):291-301.
Crossref - Jain P, Parada JP, David A, Smith LG. Overuse of the indwelling urinary tract catheter in hospitalized medical patients. Arch Intern Med. 1995;155(13):1425-1429.
Crossref - Moue A, Aktaruzzaman S, Ferdous N, Karim RM, Khalil MMR, Das AK. A. Prevalence of urinary tract infection in both outpatient department and patient department at a medical college setting of Bangladesh. Int J Biosci. 2015;7(5):146-152.
Crossref - Esposito S, Noviello S, Leone S. Catheter-associated urinary tract infections: epidemiology and prevention. Infez Med. 2008;16(3):130-143.
- Sayal P, Sandhu R, Singh K, Devi P. Bacterial colonization associated with prolonged catheterization: Who is at risk? Int J Res Med Sci. 2017;5(1):166-170.
Crossref - Nickel JC, Costerton JW, McLean RJ, Olson M. Bacterial biofilms: influence on the pathogenesis, diagnosis and treatment of urinary tract infections. J Antimicrob Chemother. 1994;33(Suppl A):31-41.
Crossref - Warren JW. Catheter-associated urinary tract infections. Int J Antimicrob Agents. 2001;17(4):299-303.
Crossref - Shukla S, Sharma V, Singh VK et al Comparison of bacterial urinary tract infection in catheterized and non-catheterized patients. IP Int J Med Microbio Trop Dis. 2021;7(4):288-291.
Crossref - Karthik R. A Study of Risk Factors for Catheter Associated Urinary Tract Infection. Thesis . 2014. http://repository-tnmgrmu.ac.in/id/eprint/11941
- Khan R, Saif Q, Fatima K, Meher R, Shahzad HF, Anwar KS. Clinical and bacteriological profile of Uti patients attending a north Indian tertiary care center. J Integr Nephrol Androl. 2015:2(1):29-34.
Crossref - Bhatia N, Daga MK, Garg S, Prakash SK. Urinary catheterization in medical wards. J Glob Infect Dis. 2010;2(2):83-90.
Crossref - Karthikeya P, Thota K S, Giduturi S R, Vedala S B, Urinary Tract Infections In Catheterized Patients And Antibiotic Sensitivity Patterns. Eur J Mol Clin Med. 2022;9(1):1200-1211.
- Acharya A, Pattnaik D, Jena J. Bacteriology and Antibiotic Sensitivity Pattern of Uropathogens in Patients with Catheter-Associated Urinary Tract Infections in a Tertiary Care Hospital, Bhubaneswar, Odisha. Indian J Public Health Res Dev. 2020;11(8):1-10.
Crossref - Hossain MD, Ahsan S, Kabir MS. Antibiotic resistance patterns of uropathogens isolated from catheterized and noncatheterized patients in Dhaka, Bangladesh. Tzu Chi Medical Journal. 2014;26(3):127-131.
Crossref - Arina S, Shamsuzzaman SM. Antibiotic sensitivity patterns of uropathogens isolated from catheterized patients in a tertiary care hospital in Dhaka. Bangladesh. Urol Nephrol Open Access J. 2021:9(3):61-66.
Crossref - Almalki MA, Varghese R, Prevalence of catheter-associated biofilm-producing bacteria and their antibiotic sensitivity pattern. Journal of King Saud University – Science. 2020;(2):1427-1433.
Crossref
© The Author(s) 2023. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.