ISSN: 0973-7510
E-ISSN: 2581-690X
The mineral nutrients are absorb from the soil by the roots, these are either macro or micro nutrients. Zinc is micro nutrients that help in several physiological process of plants and help in phyto hormone synthesis. The present Study includes the synthesis of ZnOPs by using Zinc oxalate decomposing method and these particles are characterized by UV-vis spectroscopy, and size was analyzed by Zeta analyzer. ZnOPs size was analyzed 65 nm. Seed germination was done by paper towel method. Soaking of seed was done in 100ppm, 500ppm and 1000ppm of ZnOPs and 100 ppm of ZnO supplement solution. After 5 day germination, germination percentage and seed vigor index (SVI) was calculated. It was observed that seed germination at 100 ppm is maximum in both the seed of Lentil (Lens esculentum Linn) and Chick pea (Cicer arietinum Linn). Seed vigor index was found increased at concentration of 100ppm. Presented study inferred that zinc provided as nanoparticle absorb more prominently than the traditional zinc supplement available in market at low concentration as the ZnOPs concentration increases the germination and seed vigor index retarded so it can be proposed that micronutrient in nano particles based formulation at lower concentration can provide better result in agriculture production.
Micronutrients, Seed vigor index, ZnO nanoparticles (ZnOPs).
By the 2050 world population reached to 9.5 billion In present word micronutrient deficiency is one of the major problem because it is very difficult to find the symptom of mall nutrition in human population1.
Zn mall nutrition is one of the major problem that is associated basically to child malnutrition. According to WHO Zn is the fifth factor associated with the children diseases in developing countries. Food security is associated with the micronutrient also because food having the lack of trace minerals like, Zn, Fe, Cu, Mn, Se etc. leaded to certain diseases like anemia, diarrhoea, skin diseases, lack of immunity2. Zn has the effect on several physiological functions like in regulation of more than 200 enzymes, gene expression under stress condition and detoxify the free radicals and reactive oxygen species.
Significance of Zn was first reported in maize, barley and sunflower3. Zn act as antioxidant stimulator that increase the photosynthates and essential oil biosynthesis in growing leaves. It was also reported that fresh weight, dry weight, chlorophyll content also increase in plants supplied with optimum concentration of Zn and toxicity and deficiency also has negate effect on plant growth.
Zn is important for the biosynthesis of Auxin and tryptophan4. Zn is also required by the carbonic anhydrase enzyme the help in interconversion of carbonic acid and carbon dioxide in C4 pathway of photosynthesis5. Zn increase the carbonic anhydrage activity ten folds in algage also6. Zn shortage result in many physiological problem of plants7. Zn deficiency also affect the yields and productivity of crop plants.
It was observed that Indian soil lack the trace element Zn so that it cannot be present in crop product produced in these soil as a result it is also not present in human8. Zn deficiency in plant leads to genetic variation in each generation that result in low productivity, poor grain quality, poor germination rate and poor nutritional value. There are report9 that Zn deficiency increased 10 fold by 2025. There are several strategies to reduce the effect of Zn disorder that include the biofortification, supplementation and fortification of zinc salt that are cheaper method the other method include the using the zinc supplement in soil during the growth of crops that indirectly reaches to the plant cells and ultimately to human through food chain. Along with fertilizer zinc supplement require specific calculation of dose that depend on the crop, variety, soil type and mineral itself. The major advantages of this method are, it is simple, inexpensive and can be achieved in short period of time. By frequent use of zinc supplement along with fertilizer result in accumulation of Zn in soil that create some harmful toxic effect above certain limit on human and animals depend upon the these crop. So it was proposed to use of nano particles of zinc to enhance its uptake by plants that affect the agronomic effectiveness of zinc fertilizer. The present work was performed to evaluate the comparative effect of ZnO supplement and ZnO nanoparticles (ZnOPs) over the seed germination of Lentil (Lens esculentum Linn) and Chick pea (Cicer arietinum Linn).
Methodology
Synthesis of Zinc oxide nano particles (ZnOPs)
Zinc oxide nano particles was synthesized by using Zinc oxalate decomposing method as describe by Prasad et al. (2012). Zinc oxalate was prepared by mixing the Zinc acetate (0.1M) and oxalic acid (0.1M) in equal amount as result zinc oxalate was precipitated that was rinsed by distilled water and allow in air for decomposition dried in oven at 280°C and these particles are characterized by UV-vis spectroscopy and size was analyzed by Zeta analyzer. ZnOPs size was analyzed 65 nm. Suspension of ZnO of 100 ppm and ZnOPs of 100, 500 and 1000 ppm was prepared by dissolving 100, 500 and 1000 mg/L in distilled water.
Germination of Seed
Fifty seeds each of Lentil (Lens esculentum Linn) and Chick pea (Cicer arietinum Linn) were soaked in 100 ppm of ZnO supplement solution that was taken as control and in100ppm, 500ppm and 1000ppm of ZnOPs solution for 12 hours. After soaking seeds were placed on paper towel for germination at 37°C under moist and dark condition for five days. Germination was done After five days shoot length, root length was measured in centimeter, germination percentage and Seedling Vigour Index (SVI) was calculated.
Seedling Vigour Index (SVI) was calculated by measuring the length of root and shoot length by measuring scale in centimeter using the following formula described by
Seed Vigour Index = Germination%× (root length + shoot length) cm
Ten seeds (S1 to S10) of Lentil and Chick pea were soaked in ZnO and ZnONp as described in methodology and their root (radical) length and shoot (Plumule) length were measured in centimeter (cm) by measuring scale Table 1, 2, 3 and 4. Fifty seeds was soaked in ZnO and ZnONp for the germination and germination percentage was calculated Table 5 and 6. Seed vigour index was calculated in cm it show that at 100 ppm concentration of ZnONp it is more than the control while at 500ppm and 1000ppm concentration of ZnONp it is less than control Table 7 and 8.
Table (1):
Effect of ZnONp on shoot Length( cm) of Lentil (Lens esculentum).
S1 |
S2 |
S3 |
S4 |
S5 |
S6 |
S7 |
S8 |
S9 |
S10 |
Average (cm) |
|
|---|---|---|---|---|---|---|---|---|---|---|---|
(Control ZnO) 100 PPM |
2.1 |
3.2 |
3.6 |
3.9 |
4.5 |
4.5 |
4.2 |
3.2 |
3.2 |
2.5 |
3.49 |
ZnONp 100 PPM |
6.2 |
8.1 |
7.3 |
5.4 |
6.2 |
7.2 |
8.3 |
11.2 |
6.2 |
3.1 |
6.92 |
ZnONp 500 PPM |
3.2 |
3.1 |
3.3 |
3.2 |
3.1 |
3 |
4.5 |
3.6 |
3.3 |
3 |
3.33 |
ZnONp 1000 PPM |
3.1 |
3.2 |
2.7 |
2.2 |
2.4 |
3.2 |
4.2 |
3.2 |
4.1 |
5.1 |
3.34 |
Table (2):
Effect of ZnONp on Root Length(cm) of Lentil (Lens esculentum).
S1 |
S2 |
S3 |
S4 |
S5 |
S6 |
S7 |
S8 |
S9 |
S10 |
Average (cm) |
|
|---|---|---|---|---|---|---|---|---|---|---|---|
(Control ZnO) 100 PPM |
1.1 |
0.7 |
1.2 |
1.1 |
1.1 |
1.2 |
1.3 |
1.3 |
1.2 |
0.9 |
1.11 |
ZnONp 100 PPM |
1.2 |
3.3 |
1.8 |
1.2 |
2.2 |
3.2 |
1.1 |
3.3 |
1.2 |
0.9 |
1.94 |
ZnONp 500 PPM |
0.2 |
0.3 |
0.3 |
0.3 |
0.2 |
0.2 |
0.2 |
0.3 |
0.3 |
0.3 |
0.26 |
ZnONp 1000 PPM |
0.2 |
0.1 |
0.3 |
0.2 |
0.1 |
0.1 |
0.1 |
0.2 |
0.1 |
0.2 |
0.16 |
Table (3):
Effect of ZnONp on shoot Length (cm) of Gram (Cicer arietinum).
S1 |
S2 |
S3 |
S4 |
S5 |
S6 |
S7 |
S8 |
S9 |
S10 |
Average(cm) |
|
|---|---|---|---|---|---|---|---|---|---|---|---|
(Control ZnO) 100 PPM |
3.1 |
0.8 |
3.3 |
1.1 |
1.6 |
3.1 |
3.5 |
3.1 |
2.9 |
2.2 |
2.47 |
ZnONp 100 PPM |
3.4 |
3.5 |
3.3 |
1.1 |
3.5 |
3.2 |
2.5 |
1.1 |
1.3 |
0.8 |
2.37 |
ZnONp 500 PPM |
2.1 |
0.9 |
1 |
1.3 |
0.9 |
0.8 |
0.7 |
1.2 |
1 |
0.2 |
1.01 |
ZnONp 1000 PPM |
0.8 |
0.5 |
0.8 |
1.1 |
0.9 |
0.8 |
1.2 |
0.8 |
0.7 |
0.3 |
0.79 |
Table (4):
Effect of ZnONp on root Length (cm) of Gram (Cicer arietinum).
S1 |
S2 |
S3 |
S4 |
S5 |
S6 |
S7 |
S8 |
S9 |
S10 |
Avearge (cm) |
|
|---|---|---|---|---|---|---|---|---|---|---|---|
(Control ZnO) 100 PPM |
2.8 |
0.8 |
0.9 |
0.5 |
3.2 |
5.4 |
2.8 |
3.3 |
1.1 |
0.9 |
2.17 |
ZnONp 100 PPM |
3.4 |
4.2 |
4.8 |
0.3 |
4.5 |
4.1 |
0.2 |
0.3 |
0.1 |
0.2 |
2.21 |
ZnONp 500 PPM |
0.2 |
0.3 |
0.3 |
0.3 |
0.2 |
0.2 |
0.2 |
0.3 |
0.3 |
0.3 |
0.26 |
ZnONp 1000 PPM |
0.2 |
0.1 |
0.3 |
0.2 |
0.1 |
0.1 |
0.1 |
0.2 |
0.1 |
0.2 |
0.16 |
Table (5):
Effect of ZnONp on Seed Germination % of Lentil (Lens esculentum).
Concentration |
Sown |
Germinated |
% |
|---|---|---|---|
(Control ZnO) 100 PPM |
50 |
45 |
90 |
ZnONp 100 PPM |
50 |
49 |
98 |
ZnONp 500 PPM |
50 |
44 |
88 |
ZnONp 1000 PPM |
50 |
46 |
92 |
Table (6):
Effect of ZnONp on Seed Germination % of Gram (Cicer arietinum).
Concentration |
Sown |
Germinated |
% |
|---|---|---|---|
(Control ZnO) 100 PPM |
50 |
43 |
86 |
ZnONp 100 PPM |
50 |
48 |
96 |
ZnONp 500 PPM |
50 |
41 |
82 |
ZnONp 1000 PPM |
50 |
39 |
78 |
Table (7):
Effect of ZnONp on Seedling Vigour Index of Lentil (Lens esculentum).
Concentration |
% |
Root Length (cm) |
Shoot Length (cm) |
SVI (cm) |
|---|---|---|---|---|
(Control ZnO) 100 PPM |
94 |
1.11 |
3.49 |
432.4 |
ZnONp 100 PPM |
98 |
1.94 |
6.92 |
868.28 |
ZnONp 500 PPM |
88 |
0.26 |
3.33 |
315.92 |
ZnONp 1000 PPM |
92 |
0.16 |
3.34 |
322 |
Table (8):
Effect of ZnONp on Seedling Vigour Index of Gram (Cicer arietinum).
Concentration |
% |
Root Length(cm) |
Shoot Length(cm) |
SVI(cm) |
|---|---|---|---|---|
(Control ZnO) 100 PPM |
86 |
2.17 |
2.47 |
399.04 |
ZnONp 100 PPM |
90 |
2.21 |
2.37 |
412.2 |
ZnONp 500 PPM |
82 |
0.26 |
1.01 |
104.14 |
ZnONp 1000 PPM |
78 |
0.16 |
0.79 |
74.1 |
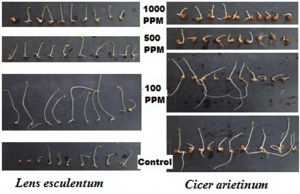
Fig.1 . Effect of ZnONp on seed germination of Lens esculentum Linn and Cicer arietinum
Efficacy of seed germination in bulk ZnO and ZnONp was evaluated by soaking the seeds of Lens esculentum Linn and Cicer arietinum in suspension of 100 ppm of ZnO supplement solution that was selected as control and in100ppm, 500ppm and 1000ppm solution of ZnONp and it was inferred that percentage germination of seeds increased 90 to 98 percentage for Lens esculentum Linn and 86 to 96 percent for Cicer arietinum in 100 ppm solution of ZnONp takes place while percentage seed germination decreases at higher concentration as compare to control. The reason behind it that nano particles easily penetrate the seed pore as compare to bulk ZnO10. The effect of Zn nano particle also described by fact that it increase the higher precursor activity for producing the higher biomolecules. Similar findings have also reported positive impact of ZnO NPs in different crops11-16. Effects of ZnONp was also observed on shoot length, root length and on seed vigor index. In Lens esculentum Linn shoot length and root length increased at lower concentration of Zn nano particle solution while at higher concentration it was retarded as compare to control. It was observed that no significant difference was observed on root and shoot length of germinating seed of Cicer arietinum while higher concentration of nano solution of Zn retard the growth of root and shoot of gram.
The probable reason for decreased germination at higher concentration could be the increased absorption and accumulation of these ZnONPs both in extracellular space and within the cells resulted in reduction in cell division, cell elongation and inhibition of the hydrolytic enzymes involved in food mobilization during the process of seed germination. Similar results were noticed by several workers who observed that ZnONPs at higher concentration had inhibitory effect on growth and development in different crops including maize17,18.
Effect of ZnONp on Seedling Vigour Index of Lentil was observed around two fold increased as compare to control while at higher concentration of ZnONp it was observed slight lower. In the case of gram not much difference was observed on Seedling Vigour Index. Higher seedling vigour under natural condition represent that soil have the optimum concentration of Zn19.
Future Prospects
Present study support the role of nano particles in plant development up to certain limit. This study open the future prospect for exploring the Role of different micro nutrient and macronutrient must be evaluated on plant physiology and anatomy by forming their nano particles. It may help reduce the toxicity of elements on plant metabolism.
ACKNOWLEDGMENTS
We are very thankful to Head of Department, Department of Biotechnology GLA University, for providing all the necessary requirements for this research work.
CONFLICT OF INTEREST
There is no conflict of interest between the authors.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
ETHICS STATEMENT
None.
AVAILABILITY OF DATA
All datasets generated or analyzed during this study are included in the manuscript.
- Girgis H, Hamed R and Osman M. Testing the equality of growth curves of independent populations with application. Am. J. Biostat. 2010; (1): 46-61.
Crossref - Zhao FJ and Mcgrath SP. Biofortification and phytoremediation. Curr Opin Plant Biol. 2009; (12): 373-380.
Crossref - Misra A and Srivastava N K. Value addition of essential monoterpene oil(s) in Geranium (Pelargonium graveolens) on leaf positions for commercial exploitation. Afr J Agric Res. 2010; (15): 2077-2079.
- Spiegel-roy P and Goldschmidt E. Biology of Citrus. Cambridge University Press, 2008; 140-184.
- Hacisalihoglu G, Hart JJ, Wang YIH, Cakmak I and Kochian VL. Zinc efficiently is correlated with enhanced expression and activity of zinc-requiring enzymes in wheat. Plant Physiol. 2003a; (131): 595-602.
Crossref - Karim W, Kaswadji R, Prartono T and Panggabea NLMG. Growth and Extracellular Carbonic Anhydrase Activity of Zooxanthellae Symbiodinium sp. In Response of Zinc Enrichment. Hayati. 2011; 18(4): 157-163.
Crossref - Grusak ML. Enhancing mineral content in plant food product. J Am Coll Nutr., 2002; (21): 178-183.
Crossref - Singh MV. Micronutrient nutritional problems in soil of indian and improvement for human and animal health. Indian J Agr Sci., 2010; 5(4): 11-56.
- Pramod M. Dhoke SK and Khanna AS. Effect of Nano-ZnO ParticleSuspension on Growth of Mung (Vigna radiata) and Gram (Cicer arietinum) Seedlings Using Plant Agar Method. J. Nanotechnol., 2011.
Crossref - Avinash C Pandey, Sharda S Sanjay and Raghvendra S Yadav. Application of ZnO nano particles in influencing the growth rate of Cicer arietinum. J. Exp. Nanosci. 2010; 5(6): 488-497.
Crossref - Korishettar P, Vasudevan SN, Shakuntala NM, Doddagoudar SR, Hiregoudar S and Kisan B. Seed polymer coating with Zn and Fe nanoparticles:An innovative seed quality enhancement technique in pigeon pea. J. Appl. Nat. Sci, 2016; 8(1): 445-450.
Crossref - Pandey AC, Sanjay SS and Yadav RS. Application of ZnO nanoparticles in influencing the growth rate of Cicer arietinum. J. Exp. Nanosci, 2010; 5(6): 488-497.
Crossref - Panwar J, Jain N, Bhargaya A, Akhatar SA and Yun YS. Positive effectof ZnO nanoparticles on Tomato plants. Conference paper: 3rd International Conference on Environmental Research and Technology (ICERT), University of Sains, Pinang, Malaysia, 2012.
- Prasad TNVKV, Sudhakar P, Sreenivasulu Y, Latha P, Munaswamy Y, Raja Reddy K, Sreeprasad T S, Sajanlal PR and Pradeep T. Effect of nanoscale zinc oxide particles on the germination, growth and yield of peanut. J. Plant Nutr., 2012; 35(6): 905-927.
Crossref - Shailesh KD, Pramod M, Rajashree K and Anand K. Effect of nanoparticles suspensions on the growth of mung (Vigna radiata) seedlings by foliar spray method.Nanotechnology Development. 2013; 3(1): 1-5.
Crossref - Laware SL and Raskar S. Influence of zinc oxide nanoparticles on growth,flowering, and seed productivity in onion. Int J Curr Microbiol Appl Sci, 2014; 3(7): 874-881.
- Shyla KK and Natarajan N. Customizing zinc oxide, silver and titanium dioxide nanoparticles for enhancing groundnut seed quality. Int J Sci Environ Technol., 2014; 7(9): 1376-1381.
- Lee C W, Mahendra S, Zodrow K, Li D, Tsai Y C and Braam J. Developmental phytotoxicity of metal oxide nanoparticles to Arabidopsis thaliana. Environ Toxicol Chem, 2010; (29): 669-675.
Crossref - Yang Z, Chen J, Dou R, Gao X, Mao C and Wang L. Assessment of the phytotoxicity of metal oxide nanoparticles on two crop plants, maize (Zea mays L.) and rice (Oryza sativa L.). Int J Environ Res Public Health, 2015; (12): 15100-15109.
Crossref - Yilmaz A, Ekiz H, Gultekin I, Torun B, Barut H, Karanlik S and Cakmak I. Effect of seed zinc content on grain yield and zinc concentration of wheat growth in zinc-deficient calcareous soils. J. Plant Nutr, 1998; (21): 2257-2264.
Crossref
© The Author(s) 2020. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.