ISSN: 0973-7510
E-ISSN: 2581-690X
Hepatocellular carcinoma (HCC) is the greatest traditional kind of pre-eminent cancer worldwide, which happens mainly in chronic liver disease and cirrhotic patients. The available surveillance strategies for suspected HCC patients include serum alpha-fetoprotein (AFP) and liver imaging have been mainly recommended. However, the sensitivity and selectivity of these diagnostic strategies especially in the early stages of HCC have many obstacles. MicroRNAs (miRNAs) are non-coding RNAs that are 18–25 nucleotides in length. Plasma miRNAs may be a promising new biomarker for cancer detection and prognosis in the early stages. Assessment of Plasma MicroRNA-21 (miRNA-21) significance as a noninvasive Hepatocellular carcinoma marker compared with AFP gold standard test to improve HCC early diagnostic power. This is a prospective research project that included 90 patients in total, split into three classes., liver cirrhosis patients (LC) without any malignancies and (HCC) patients in addition to the healthy control group. Patients and controls were subjected to the clinical studies, routine investigations, imaging studies, and detection of plasma miRNA-21 & AFP. miRNA-21 showed a highly significant difference in the 3 studied groups. Control group with LC group, control group with HCC group, and LC group with HCC group P value (P 0.0001, P1 0.0001, P2 0.0001and P3 0.0001) respectively. Also, a highly significant difference was observed between pre-TACE and post-TACE miRNA-21 in the HCC group P value (0.0001). Circulating miRNA-21 may be used as a noninvasive co biomarker with AFP to increase HCC diagnostic accuracy in its early stages.
Hepatocellular carcinoma, Early diagnosis, Alpha-fetoprotein, MicroRNA-21
The most common form of primary liver cancer is hepatocellular carcinoma (HCC), which occurs mainly in chronic liver disease and cirrhotic patients.1
It progresses with local expansion, intrahepatic spreading, and distant metastases.2 HCC is the fifth most common tumor worldwide and the third leading cause of cancer-related deaths, with over half a million new cases diagnosed each year.3 Mainly, HCC develops with a well-established chronic liver disease background. The majority of HCC cases occur in Eastern Asia and Sub-Saharan Africa, where hepatitis B virus (HBV), hepatitis C virus (HCV), and aflatoxin B1 exposure are the most common risk factors. On the other hand, in North America, Europe, and Japan, alcohol consumption and HCV are the main risk factors. However, in the USA, nonalcoholic fatty liver disease (NAFLD), or nonalcoholic steatohepatitis (NASH), and diabetes are the common risk factor for HCC.4
Inflammation, necrosis, fibrosis, and continuous regeneration characterize liver cirrhosis and take part in HCC development. HBV patients can develop HCC without any cirrhosis history; underlying fibrosis is usually present, with the suggestion of regeneration. By contrast, in HCV patients, HCC invariably presents, more or less, in the setting of cirrhosis.5 Besides, many nodules are mainly found in the cirrhotic liver, including regenerative and dysplastic nodules. Prospective studies came to the fact that the occurrence of small-cell dysplastic nodules increases HCC risk, on the other hand, large-cell dysplastic nodules aren’t related to HCC initiation. The relation of small-cell dysplastic nodules to HCC includes conserved proliferation markers presence in addition to the occurrence of nodule-in-nodule during pathological examination.6
Since surgical resection and liver transplantation, the two curative therapies for HCC, are only available to a small percentage of patients with early tumours, the HCC survival rate is very low. There is currently no successful systemic chemotherapy for HCC.7 The available surveillance strategies for suspected patients include serum alpha-fetoprotein (AFP) and liver imaging that have been mainly recommended. However, the sensitivity and selectivity of these diagnostic strategies especially in the early stages of HCC have many obstacles. Many imaging and serological markers have been tested as early diagnostic tools for HCC in recent decades with different sensitivity and selectivity but till now imaging with AFP remains the gold standard test for diagnosis, staging, and follow-up of HCC, despite their failure, especially in the early stages.8
MicroRNAs (miRNAs) are non-coding RNAs that are 18–25 nucleotides in length. The regulatory regions in the 31 or 51 untranslated region (UTR) of target messenger RNAs are complementary to miRNAs9 Specific miRNA binding inhibits the translation of target mRNAs or promotes mRNA degradation, resulting in a responsive and rapid gene-expression regulation cascade.10 miRNA expression in liver tissues has proven that miRNA is associated with different liver disease stages also, by comparing plasma miRNAs before and after resection of a tumor it was found that circulating miRNAs like miRNA-15b, miRNA-21, miR-130b, and miRNA-183 are overexpressed with malignancies so circulating miRNAs may be a good indicator for early prediction of tumors.11
Among the miRNAs that are dysregulated, miRNA-21 was identified as an oncogenic miRNA and its upregulation facilitates the proliferation, invasion, and metastasis of malignant cells.11 It was found that miRNA-21 increases cell proliferation and suppresses cancer cell apoptosis in the xenograft model, further defining miRNA-21 as an oncogenic miRNA.12
According to sources, miRNA-21, miRNA-155, and miRNA-210 were raised in the plasma of B-cell lymphoma patients in comparison to healthy people, and miRNA-21 elevation was linked to the rate of relapse-free survival.13
So, Plasma miRNAs may be a promising new biomarker for cancer detection and prognosis in the early stages.14
Consequently, the aim of this project was the evaluation of circulating plasma miRNA-21 significance as a noninvasive marker for hepatocellular carcinoma compared with AFP.
Design
This is a prospective research project. that was carried out at Sohag Centre of Cardiac and Digestive System, Sohag, Egypt between May 2018 and January 2019.
Patients
The study included 90 patients (53 males and 37 females).; the mean age of the HCC group was 64.2±7.73 years with a range between 51 to 81 years while the LC group mean of was 63.1±8.12 years with a range between 48 and 78 years and in control healthy group, age’s median was 41.73±13.59 years with range 18 years and 80 years. The studied participants were separated into three groups:
- Group, I included 30 healthy control.
- Group, II included 30 patients with liver cirrhosis (LC) without any malignancies.
- Group, III included Patients with Hepatocellular carcinoma (HCC).
Exclusion criteria
Patients with alcoholic liver cirrhosis, autoimmune liver cirrhosis, patients with any other malignancies, and HBsAg positive patients were not included in this report.
All patients underwent a comprehensive clinical review and a full history with particular focusing on the presence of jaundice, ascites, lower limbs edema, hepatomegaly, previous attacks of hepatic encephalopathy and splenomegaly. Complete blood count (CBC), liver function tests, specific HCV testing (anti-HCV antibodies by enzyme-linked immunosorbent assay (ELISA), and HCV RNA and viral load by real-time PCR were then done for all patients. Chronic HCV infection was defined by the persistence of HCV antibodies and HCV RNA in the patients’ serum for at least 6 months. The abdominal sonographic examination was then done to detect the presence of hepatic fibrosis, HCC, splenomegaly, and to measure the portal vein diameter. The severity of liver cirrhosis was determined using the Child-Pugh score.15
According to the American Association for the Study of Liver Diseases (AASLD) guidelines, the diagnosis of HCC was confirmed by measuring of AFP in serum and triphasic computed tomography (CT) and/or magnetic resonance imaging.16
The Child-pugh score (CTP) was used to determine the seriousness of the disease, which takes into account five clinical (hepatic encephalopathy and ascites) and laboratory (albumin, prothrombin duration, and bilirubin values) parameters. According to the degree of abnormality, each variable is given a score of 1-3. Patients with compensated cirrhosis (class A) get a score of 5–6, those with mildly moderate decompensated cirrhosis (class B) get a score of 7–9, and those with extremely decompensated disease (class C) get a score of 10–15. All patients subjected to this study didn’t receive any medications at the time of the study.
The BCLC classification has been tested in various settings and provides treatment guidelines for hepatocellular carcinoma at all levels.17
TACE is the treatment of choice for intermediate-stage tumours (BCLC stage B). TACE uses a chemotherapeutic agent and embolization of occluding particles at the same time. The existence of a hyper vascularized tumor is a requirement for effective TACE. The combination of chemotherapeutic agent and occluding particles is administered selectively, resulting in a high local concentration of the chemotherapeutic agent in the tumor and low systemic distribution. The chemotherapeutic agent remains in the tumor region due to the occlusion of tumor vessels, and the resulting hypoxia enhances the chemotherapeutic agent’s impact.18
Aside from a tumor size of less than 4 cm and improved vascularization, absorption of lipidol of at least 75% of the tumor volume is a favorable prognostic factor for good TACE.
Methodology
The following tests were performed on both patients and controls:
Clinical studies
including full medical history and complete clinical examination with focusing on the hepatobiliary system especially ascites, jaundice, previous attacks of hepatic encephalopathy, abdominal pain, or hematemesis
Laboratory studies
- Routine investigations included kidney functions,19 liver functions,20 and complete blood count (CBC).21
- Serological markers included HCVAb (22) and HBsAg.23
- Molecular studies included detection of plasma miRNA-21 expression by Taqman Real time-PCR: where Isolation and quantification of total RNA were performed according to Mariner et al.24 Briefly, EDTA blood samples were centrifuged for 15 minutes (1500 g) for plasma separation and storage at – 80°C.
Plasma samples were de-frozen in ice and centrifuged for 15 min at 5°C (16,000g). Total RNA was extracted from plasma samples using phenol, chloroform, and ethanol according to total RNA isolation protocol (ThermoFisher), centrifuged at 4°C for 30 minutes (16,000g) followed by washing and collection in preheated nuclease-free water. A Nanodrop spectrophotometer was used to determine RNA accuracy (NanoDrop Technologies, Wilmington). The absorbance ratio at 260nm has been used as a measure of RNA purity. 10 microliters of the extracted RNA were reverse transcribed into cDNA. The Step One TM Plus Real-Time PCR System (Applied Biosystems, USA) was used to conduct the real-time PCR reactions in triplicate.
The Syber-green PCR master mix (Applied Biosystems) was used to evaluate miRNA-21 expression by the following primers: miRNA-21 Forward primer: TAGCTTATCAGACTGATGTTGA and universal reverse: GCGAGCACAGAATTAATACGAC. U6 gene was used as internal control reference for miRNA-21 expression data normalization. Primers used for detection of the U6 gene were the U6-F: CGCTTCGGCAGCACATATAC and U6-R: TTCACGAATTTGCGTGTCAT.
- Immunological investigations for detection of serum AFP.25
Imaging studies included abdominal ultrasound confirmation of liver cirrhosis26 and Triphasic CT abdomen.27
Statistical analysis
- For statistical data analysis, IBM-SPSS (Statistical Package for Social Sciences), version 24 IBM-Chicago, USA (May 2016) was used.
- The mean, standard deviation (SD), number, and percentage are all used to express data. For quantitative data, the mean and standard deviation were used as descriptive values, while qualitative data was defined using numbers and percentages.
- The means of two groups were compared using the student t-test, and the means of more than two groups were compared using the one-way analysis of variance (ANOVA) test. In the case of non-parametric data, the Mann-Whitney test was substituted for the student t-test.
- The percentages of qualitative data were compared using Pearson Chi-square, and non-parametric data was compared using Fisher’s Exact Test.
- To compared two quantitative variables, a Pearson correlation test was used. The following is how the value of (r) was calculated:
r <0.2 negligible correlation, r 0.2-0.4 weak correlation, r 0.4-0.7 moderate correlation, r 0.7-1 strong correlation, r positive was considered as positive correlation and r negative was considered as a negative correlation.
- For all these tests, the level of significance (P-value) was explained as: No significance P > 0.05, Significance P < 0.05 and High significance P < 0.001.
- The best cut-off for AFP and MicroRNA 21 in HCC prediction was determined using ROC curve analysis. Also, sensitivity, accuracy, positive predicted value, and negative predictive value were measured.
Ethical Considerations
- The International Ethical Guidelines for Biomedical Research Involving Human Subjects (CIOMS/WHO, 1993) were used to conduct this analysis. Ethical approval by Research and Ethical Committee of Sohag Centre of Cardiac and Digestive System, Sohag, Egypt. All of the participants in this study gave their informed consent. Cases were assigned a serial number, and personal information was kept private.
Our study included 60 patients with chronic Hepatitis C virus (CHCV). We divided patients with CHCV into two groups: group HCV with liver cirrhosis (LC) and group HCV with hepatocellular carcinoma (HCC).
Age & Sex
A statistical significance in age difference between the studied groups. Control group with LC group, and control group with HCC group p-value (p <0.0001, p1 <0.0001 and p2 <0.0001) respectively but a non-significant difference in age when compared LC group with HCC group.
Also, sex, there was a significant difference between the studied groups, control with HCC group, and LC with HCC P value were (P1 0.02, P2 0.01, and P3 0.02) respectively (Table 1).
Table (1):
Demographic and Laboratory findings of the studied patients.
Variables |
Control N=30 |
LC N=30 |
HCC N=30 |
P |
P1 |
P2 |
P3 |
|---|---|---|---|---|---|---|---|
Age/years |
|||||||
Mean ± SD |
41.73±13.59 |
63.1±8.12 |
64.2±7.73 |
1.00 |
|||
Median (range) |
40.5 (18:80) |
63 (48:78) |
65 (51:81) |
||||
Gender |
|||||||
Female |
16 (53.33%) |
15 (50.00%) |
6 (20.00%) |
0.02 |
0.80 |
0.01 |
0.02 |
Male |
14 (46.67%) |
15 (50.00%) |
24 (80.00%) |
||||
HCV Ab |
|||||||
Negative |
30 (100%) |
0 |
0 |
1.00 |
|||
Positive |
0 |
30 (100%) |
30 (100%) |
||||
HBV Ag |
|||||||
Negative |
30 (100%) |
30 (100%) |
30 (100%) |
||||
Positive |
0 |
0 |
0 |
||||
TLC |
|||||||
Mean ± SD |
6.63±2.77 |
6.65±2.47 |
7.07±2.62 |
0.58 |
0.76 |
0.32 |
0.46 |
Median (range) |
5.95 (3.5:14.8) |
6.55 (2.7:12.3) |
6.95 (2.7:14) |
||||
HGB |
|||||||
Mean ± SD |
13.44±2.04 |
13.39±1.18 |
12.37±1.95 |
0.03 |
1.00 |
0.04 |
0.08 |
Median (range) |
12.95 (10.5:18.9) |
13 (11:16) |
12 (9.6:18.7) |
||||
PLT |
|||||||
Mean ± SD |
222.77±49.21 |
262.8±68.88 |
164.3±43.39 |
0.02 |
|||
Median (range) |
217 (155:338) |
251 (152:421) |
165.5 (102:289) |
||||
Albumin |
|||||||
Mean ± SD |
4.4±0.49 |
4.15±0.55 |
3.70±0.64 |
0.33 |
0.007 |
||
Median (range) |
4.3 (3.6:) |
4.05 (3.3:5.3) |
3.7 (2.6:5) |
||||
Total bilirubin |
|||||||
Mean ± SD |
0.83±0.19 |
0.95±0.36 |
1.02±0.30 |
0.054 |
0.46 |
0.01 |
0.16 |
Median (range) |
0.85 (0.5:1.1) |
0.85 (0.6:2.1) |
0.99 (0.5:1.5) |
||||
Direct bilirubin |
|||||||
Mean ± SD |
0.23±0.06 |
0.61±0.23 |
0.41±0.24 |
0.0001 |
0.0001 |
0.01 |
0.01 |
Median (range) |
0.2 (0.1:0.3) |
0.6 (0.3:1.6) |
0.4 (0.1:0.8) |
||||
ALT |
|||||||
Mean ± SD |
40.10±16.16 |
98.7±24.51 |
47.6±25.22 |
0.0001 |
0.0001 |
0.33 |
0.0001 |
Median (range) |
37 (7.5:82) |
92 (65:147) |
47 (8:108) |
||||
AST |
|||||||
Mean ± SD |
35.35±11.42 |
72.83±17.21 |
48±21.93 |
0.0001 |
0.0001 |
0.008 |
0.0001 |
Median (range) |
36.5 (13:59) |
72 (39:108) |
47.5 (9:99) |
||||
PT |
|||||||
Mean ± SD |
12.82±0.76 |
13.14±1.85 |
14.60±1.80 |
1.00 |
0.001 |
||
Median (range) |
12.9 (11.7:15.3) |
12.2 (11:18.2) |
14.25 (12:19) |
||||
INR |
|||||||
Mean ± SD |
1.04±0.07 |
1.21±0.28 |
1.27±0.21 |
0.0001 |
0.004 |
0.88 |
|
Median (range) |
1.0 (1.0:1.28) |
1.0 (1.0:1.7) |
1.26 (1.0:1.6) |
||||
Creatinine |
|||||||
Mean ± SD |
0.67±0.22 |
0.89±0.33 |
1.01±0.22 |
0.0001 |
0.003 |
0.0001 |
0.01 |
Median (range) |
0.6 (0.4:1.3) |
0.8 (0.5:1.8) |
0.98 |
||||
(0.7:1.6) |
|||||||
Ascites |
No [30 (100%)] |
Yes [30 (100%)] |
No [30(100%)] |
1.00 |
|||
Hepatomegaly |
|||||||
No |
100% |
100% |
26 (86.6%) |
0.02 |
1.00 |
0.11 |
0.11 |
Yes |
0% |
0% |
4 (13.3%) |
||||
Splenomegaly |
|||||||
No |
30 (100%) |
4 (13.3%) |
7 (23.3%) |
0.0001 |
|||
Yes |
0 |
26 (86.6%) |
23 (76.6%) |
P compared the three group, p1 compared control & LC, p2 compared control & HCC, p3 compared LC & HCC. LC : Liver cirrhosis.
HCC : Hepatocellular carcinoma. HCV Ab : hepatitis C virus antibody. HBV Ag : hepatitis B virus antigen. TLC : Total leucocytes count. HGB : Hemoglobin PLT : Platelets. Alt : alanine transaminase. Ast : aspartate transaminase. Pt :prothrombin time. INR : international normalized ratio.
HCV Antibody Serology Test
By comparing between the 3 studied groups as regard HCV antibody (Ab), we found that there was a significant difference in HCV Ab, when compared control group with LC group and HCC group P value (p <0.0001, P1<0.0001, P2<0.0001) respectively but there was no significant difference between LC group and HCC group (Table 1).
Routine Laboratory Investigations
In terms of blood picture results of the studied groups included total Leukocytes Count (TLC), Hemoglobin (HGB), and platelets count (PLT). We found that there was no significant difference in TLC but there was a significant difference in HGB between the 3 studied groups, control with HCC group, LC group and HCC group P value (P 0.03, P2 0.04 and P3 0.08) respectively.
PLT count showed a significant difference when compared the 3 studied groups, control with LC group & HCC group in addition to LC group and HCC group P value (P < 0.0001, P1 0.02, P2 <0.0001, and P3 <0.0001) respectively (table 1).
Preposition liver function levels in the 3 studied groups including Albumin, total bilirubin and direct bilirubin, ALT (Alanine Transaminase), AST (Aspartate Transaminase), PT (Prothrombin Time), and INR (International Normalized Ratio).
Albumin results showed a significant difference between the studied group, when compared the control group and HCC and when compared LC group and HCC group P value (P < 0.0001, P2 <0.0001 and P3 0,007) respectively.
But total Bilirubin showed no significance when compared the control group and LC group and compared the LC group with the HCC group, otherwise, it showed a significant difference when compared the control group and HCC group P value (P 0.0001, P2 0.01) respectively.
As regards direct bilirubin, there was a significant difference between studied groups, control group with LC group, control group with HCC group, and LC group with HCC group P value (P 0.0001, P1 0.0001, P2 0.01and P3 0.01) respectively.
The recorded data denotes a significant difference in ALT between groups, control group with LC group and LC group with HCC group P value (P 0.0001, P1 0.0001and P3 0.0001) respectively.
Also, there was a significant difference in AST between groups, control group with LC group, control group with HCC group and LC group with HCC group P value (P 0.0001, P1 0.0001, P2 0.008 and P3 0.0001) respectively. Data demonstrated a significant difference regarding Prothrombin Time (PT) & (INR) in this study, control group with LC group, control group with HCC group and when compared LC group with HCC group P value (P < 0.0001, P2 < 0.0001and P3 0.001) respectively (Table 1).
Regarding serum creatinine in the three studied groups, it was noticed that there was a highly significant difference between the studied groups, control group with LC group, control group with HCC group and LC group with HCC group P value (P 0.0001, P1 0.003, P2 0.0001and P3 0.01) respectively (Table 1).
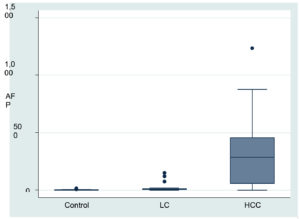
It was shown that there was a highly significant difference in AFP & MicroRNA-21 levels between groups, control group with LC group, control group with HCC group and by compared LC group with HCC group P value (P 0.0001, P1 0.0001, P2 0.0001, and P3 0.0001) respectively (Table 2) (Fig. 1).
Table (2):
Comparison of Alpha-fetoprotein level and Micro RNA-21 among 3 studied groups.
Variables |
Control N=30 |
LC N=30 |
HCC N=30 |
P |
P1 |
P2 |
P3 |
|---|---|---|---|---|---|---|---|
AFP |
|||||||
Mean ± SD |
4.35±3.34 |
18.54±34.65 |
315.08±295.7 |
0.0001 |
0.0001 |
0.0001 |
0.0001 |
Median (range) |
3.36 (0.4:14.1) |
7.25 (0.9:150) |
286.5 (1.49:1236) |
||||
MicroRNA-21 |
|||||||
Mean ± SD |
1.0±0 |
2.73±1.01 |
19.7±7.74 |
0.0001 |
0.0001 |
0.0001 |
0.0001 |
Median (range) |
1.0 (1.0:1.0) |
2.55 (1.2:4.8) |
19.2 (3.1:33) |
LC : Liver cirrhosis.HCC : Hepatocellular carcinoma. AFP : Alpha fetoprotein
Imaging studies including abdominal ultrasound and Triphasic CT abdomen
With relation to the sonographic findings of the studied groups, as regards to the liver, we found that appearance of course and irregular in 100% of LC group and Hepatic Focal Lesion (HFL) in 90% of HCC group and no HFL in10% of HCC group while liver appears homogenous in 70% of the control group and mild bright in 30% of them. As regards spleen and presence of ascites, it was normal in the 3studied groups (Table 1).
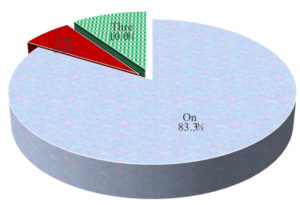
Regarding the distribution of the number and size of lesions in CT findings of the HCC group. We found that 25 patients (83.33%) had one lesion, 2 patients (6.67%) had 2 lesions and 3 patients (10%) had 3 lesions, according to the size of lesions by CT Mean ± SD4.81±2.95 with Median (range) 4 (1.5:16) (Table 3) (Fig. 2).
Table (3):
Distribution of number and size of lesions in CT finding of HCC group.
CT finding |
Number (%) |
|---|---|
Number |
|
1 |
25 (83.33%) |
2 |
2 (6.67%) |
3 |
3 (10.00%) |
Size |
|
Mean ± SD |
4.81±2.95 |
Median (range) |
4 (1.5:16) |
Size is measured by millimetre
Plasma miRNA-21
By comparing MicroRNA-21 in the 3 studied groups, it was found that there was a highly significant difference between the studied groups, control group with LC group, control group with HCC group and LC group with HCC group P value (P 0.0001, P1 0.0001, P2 0.0001and P3 0.0001) respectively (Table 4).
Table (4):
Comparison between number of lesions and MicroRNA-21 in HCC group, PreTACE and PostTACE.
Variables |
One N=25 |
Two N=2 |
Three N=3 |
P |
P1 |
P2 |
P3 |
|---|---|---|---|---|---|---|---|
MicroRNA-21pre |
|||||||
TACE |
|||||||
Mean ± SD |
19.75±8.18 |
21±10.46 |
18.4±2.68 |
0.94 |
0.85 |
0.74 |
1.00 |
Median (range) |
19.3 (3.1:33) |
21 (13.6:28.4) |
18.9 (15.5:20.8) |
||||
MicroRNA-21post |
|||||||
TACE |
|||||||
Mean ± SD |
2.26±0.93 |
3.55±0.35 |
3.03±1.07 |
0.10 |
0.08 |
0.17 |
0.56 |
Median (range) |
1.9 (1.1:4.1) |
3.55 (3.3:3.8) |
2.8 (2.1:4.2) |
TACE: Trans Arterial Chemo Emobolization
Number of lesions and MicroRNA-21 in HCC (Pre TACE and Post TACE)
In our study, by comparing the number of lesions and miRNA-21 in the HCC group, it was found that no statistical significance in PreTrans-Arterial Chemo Embolization TACE and post-TACE (Table 5).
Table (5):
Correlation between size of lesion and Micro RNA-21 in HCC group in preTACE and post TACE.
Correlation between |
Correlation coefficient |
P value |
|---|---|---|
Size & MicroRNA-21 pre– TACE |
0.01 |
0.95 |
Size & MicroRNA-21 post–TACE |
0.04 |
0.85 |
Correlation between size of lesion and MicroRNA-21 (Pre TACE and Post TACE)
Correlation between the size of lesion and MicroRNA-21 in the HCC. It was found that the correlation coefficient was 0.01 in pre-TACE and 0.04 in post-TACE; there was no significant correlation between the size of lesion and microRNA-21 in the HCC group in pre-TACE and post TACE (Table 5).
Comparison between pre-TACE and post-TACE MicroRNA-21
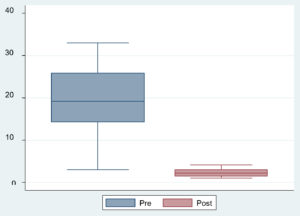
By comparing pre-TACE and post TACE MicroRNA-21 in the HCC group, It was found that there is highly significant when compared between pre-TACE and post TACE Micro RNA-21 in HCC group P value (0.0001) (Table 6) (Fig. 3).
Table (6):
Comparison between preTACE and post TACE Micro RNA-21 in HCC group.
Variables |
Summary statistics |
|---|---|
MicroRNA-21 pre – TACE |
|
Mean ± SD |
19.7±7.74 |
Median (range) |
19.2 (3.1:33) |
MicroRNA-21 post – TACE |
|
Mean ± SD |
2.42±0.97 |
Median (range) |
2.3 (1.1:4.2) |
P value |
HCC : Hepatocellular carcinoma. TACE: Trans Arterial Chemo Emobolization.
ROC curve analysis of circulating miR-21and AFP for diagnosis of HCC
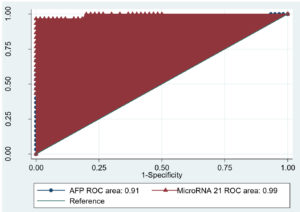
In this study, by ROC curve analysis of circulating miR-21 and AFP for diagnosis of HCC revealed that at a cut-off value of >4.8 in HCC with a sensitivity of 96.67%, the specificity of 100%; area under the curve (AUC) 0.99 (P<0.0001, 100% PPV and 98% NPV). However, the ROC curve analysis of AFP revealed that at a cut-off value of >21.4in HCC with a sensitivity of 80%, the specificity of 95%; AUC 0.91 (P< 0.0001, 88.9% PPV and 98% NPV) (Table 7) (Fig. 4).
Table (7):
Diagnostic performance of miR-21 and AFP for diagnosis of HCC.
Cutoff |
AUC |
Sensitivity |
Specificity |
PPV |
NPP |
P value |
|---|---|---|---|---|---|---|
AFP >21.4 |
0.91 (0.84-0.99) |
80.0 |
95.0 |
88.9 |
90.5 |
|
Micro RNA21>4.8 |
0.99 (0.98–1.00) |
96.67 |
100 |
100 |
98.0 |
P value comparing AFP & MicroRNA-21 =0.03
HCC is the fifth most common cancer worldwide and the third leading cause of cancer-related deaths.28
HCC has a high morbidity rate due to a lack of early warning markers and a poor prognosis. As a result, new frontiers in HCC diagnosis and therapeutics are still high- priority research areas.29
In Egypt, HCC is a public health concern. It accounts for 70.48 percent of all liver tumours in Egyptians, making it the second most common cancer after bladder cancer in men and breast cancer in women, as well as the second leading cause of death in men.30
The most commonly used tumor biomarker for HCC diagnosis is AFP. It has a low sensitivity and specificity, however. This emphasizes the need for other minimally invasive, quick, and effective methods.31
miRNAs are a type of short non-coding RNA that plays a key role in post-transcriptional gene attenuation based on the sequence.32
MiRNAs play a role in a variety of cellular processes, including carcinogenesis. Furthermore, due to their resistance to RNase, extreme pH, and temperature, miRNAs are extremely stable in the plasma. As a result, they’ve been identified as potential biomarkers for detecting pre-symptomatic diseases in their early stages, such as HCC.33,34
The study’s goal was to the evaluation of plasma miRNA -21 significance as a noninvasive Hepatocellular carcinoma marker and compared its diagnostic accuracy to AFP, to improve HCC diagnostic power at an early stage.
This study included 60 chronic HCV patients attending the Tropical Department at Centre of Cardiac and Digestive System, Sohag, Egypt, and 30 individuals as a control group during the period from May 2018 to January 2019. We divided patients with HCV into two groups, HCV Group with LC and HCV Group with HCC.
Our recorded data denoted a significant difference in routine laboratory investigations when compared to the 3 studied groups. Our results were similar to the results of Tarek et al.35 as they found that in terms of necro-inflammatory markers (AST & ALT), liver synthetic markers INR, albumin, bilirubin, and alkaline phosphatase, there was a highly significant difference between the three classes. These results were in line with those of other studies.36,37
In the current study, there was a highly significant difference when comparing miRNA -21 levels in the 3 studied groups. The study of Tarek
et al35 revealed that plasma miRNA -21 values were significantly different in the HCC group compared to both chronic liver disease and healthy control groups (p <0.01). Another study by Fang et al.38 analyzed the expression of miRNA -7 using qRT-PCR in 10 pairs of clinical HCC tissue samples and reported repression of miRNA -7 expression in 7 of 10 HCC cases compared to paired normal tissues.
So, miRNA -21 was found to be down-regulated in HCC tissues and inhibit proliferation and metastasis in HCC cells in vitro and in vivo, according to our findings, which were consistent with other studies. MiRNA -21 was found to be down-regulated in HCC tissues and inhibit proliferation and metastasis in HCC cells in vitro and in vivo, through different target genes and signalling pathways.39,40
According to our results, Correlation between numbers of lesions in CT & Sonographic findings with plasma miRNA -21 level it is clear that miRNA -21 is associated with HCC degree that was confirmed by the reduction of plasma miRNA -21 after TACE as the size of the tumor was reduced.
Furthermore, Guo et al.41 and Huang et al.42 compared the expression of miRNA -21 in HCC and noncancerous samples and found that HCC had higher miRNA -21 expression, while normal samples had lower miRNA -21 plasma levels than HCC. Taken together, these results indicated that miRNA -21 could be used to diagnose HCC as compared to healthy people.
Diagnostic performance of miRNA -21 and AFP for diagnosis of HCC showed that miRNA-21 has high sensitivity and specificity compared with AFP and this was in agreement with Liao et al.43 who suggested that miRNA -21 had a combined sensitivity and specificity of 62.4 percent and 84.4 percent for distinguishing HCC from chronic hepatitis volunteers, respectively. Also, the presence of an AFP level of 400 ng/ml is thought to be an indication of HCC, but it is not seen in the early stages of the disease. Approximately one-third of all HCC cases with small lesions (less than 3cm) cannot be detected early.44
Finally, according to our results, miRNA -21 is a potential noninvasive early diagnostic marker for HCC, circulating miRNA -21 has many unique advantages. First, plasma miRNA -21 possesses minimal invasion compared with histopathological examinations. Second, plasma microRNAs are stable and reproducible.45 Third, plasma miRNA -21 cannot be influenced by cirrhosis or viral status. Fourth, high significant overexpression of plasma miRNA-21 was noticed even in early-stage HCC and after treatment.46,47
So, circulating miRNA -21 may be used as a new co-biomarker with AFP to increase the accuracy of early-stage HCC diagnosis.
From this study we conclude that circulating miRNA -21 may be used as a noninvasive co biomarker with AFP to increase the precision of early-stage hepatocellular carcinoma diagnosis especially the sensitivity and selectivity of the current HCC early diagnostic strategies have many limitations where miRNA -21 possesses many unique properties including, minimal invasion compared with histopathological examinations, its stability, and reproducibility doesn’t influenced by cirrhosis or viral status, and has a high significant overexpression even in early-stage HCC and after treatment. For sure this study is a single centre study that has some limitations including the small sample size and need to be validated using a multicentre study.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
LY, KH and AN designed the research. AO and LY performed the research. AO, LY and AN analyzed data. All authors designed the figures and wrote the text. All authors read and edited the manuscript.
FUNDING
None.
ETHICS STATEMENT
This study was approved by the ethical committee of Center of Cardiac and Digestive System, Sohag, Egypt. (No.SCCDS/HU102/Pros18/TCU/2/21,2018). The study was carried out according to Internatinal Ethical Guidelines for Biomedical research involving human subjects (CIOMS/WHO, 1993), and as per the Helsinki Declaration of 1975, as revised in 2008. (http://www.wma.net/en/20activities/10ethics/10helsinki/).
INFORMED CONSENT
Written informed consent was obtained from all the participants involved in the study.
AVAILABILITY OF DATA
All datasets generated or analyzed during this study are included in the manuscript.
- Alison MR. Liver stem cells: implications for hepatocarcino-genesis. Stem Cell Rev. 2005;1(3):253-260.
Crossref - Forner A, Llovet JM, Bruix J. Hepatocellular carcinoma. Lancet. 2012;379(9822):1245-1255.
Crossref - Parkin DM, Bray F, Ferlay J, Pisani P. Estimating the world cancer burden: Globocan 2000. Int J Cancer. 2001;94(2):153-156.
Crossref - Bugianesi E. Non-alcoholics tea to hepatitis and cancer. Clin Liver Dis. 2007;11(1):191-207.
Crossref - Zhang X, Zhang H, Ye L. Effects of hepatitis B virus X protein on the development of livercancer. J Lab Clin Med. 2006;147(2):58-66.
Crossref - Cameron RG, Greig PD, Farber E, et al. Small encapsulated hepatocellular carcinoma of the liver. Provisional analysis of pathogenetic mechanisms. Cancer. 1993;72(9):2550-2559.
Crossref - Poon RT, Fan ST. Resection prior to liver transplantation for hepatocellular carcinoma: a strategy of optimizingthe role of resection and transplantation in cirrhotic patients with preserved liver function. Liver Transpl. 2004;10(6):813-815.
Crossref - Kanwal F, Kramer JR, Mapakshi S, et al. Risk of hepatocellular cancer in patientswith non-alcoholic fatty liver disease. Gastroenterology. 2018;155(6):1828-1837.e2.
Crossref - Calin GA, Croce CM. MicroRNA-cancer connection: the beginning of a new tale. Cancer Res. 2006;66(15):7390-7394.
Crossref - Lau NC, Lim LP, Weinstein EG, Barten DP. An abundant class of tiny RNAs with probable regulatory roles in Caenorhabditisl egans. Science. 2001;294(5543):858-862.
Crossref - Connolly E, Melegari M, Landgraf P, et al. Elevated expression of the miR-17-92 polycistron and miR-21 in hepadnavirus-associated hepatocellular carcinoma contributes to the malignant phenotype. Am J Pathol. 2008;173(3):856-864.
Crossref - Asangani IA, Rasheed SA, Nikolova DA, et al. MicroRNA-21 (miR-21) post-transcriptionally down regulates tumor suppress or Pdcd4 and stimulates invasion, intravasation and metastasis in colorectal cancer. Oncogene. 2008;27(15):2128-2136.
Crossref - Lawrie CH, Gal S, Dunlop HM, et al. Detection of elevated levels of tumour-associated micro RNAs in serum of patients with diffuse large B-celllymphoma. Br J Haematol. 2008;141(5):672-675.
Crossref - Cortez MA, Calin GA. MicroRNA identification in plasma and serum: A new tool to diagnose and monitor diseases. Expert Opin Biol Ther. 2009;9(6):703-711.
Crossref - Pugh R, Murray-Lyon I, Dawson J, Pietroni M, Williams R. Transection of the oesophagus for bleeding oesophageal varices. Br J Surg. 1973; 60(8):646-649.
Crossref - Bruix J, Sherman M. American Association for the Study of Liver Diseases (AASLD) Practice Guidelines. Management of Hepatocellular Carcinoma: An Update. Hepatology. 2011:53(3):1020-1022.
Crossref - European Association For The Study Of The Liver, & European Association for the Study of Diabetes (EASD): EASL-EASD-EASO Clinical practice guidelines for the management of non-alcoholic fatty liver disease. Obesity Facts. 2016;9(2):65-90.
Crossref - Lencioni R, Kudo M, Ye Sl, et al. GIDEON (Global Investigation of therapeutic Decisions in hepatocellular carcinoma and its treatment with sorafenib): second interim analysis. Int J Clin Pract. 2014;68(5):609-617.
Crossref - Stevens PE, Levin A. Evaluation and management of chronic kidney disease: synopsis of the kidney disease: improving global outcomes 2012 clinical practice guideline. Ann Intern Med. 2013;158(11):825-830.
Crossref - Lee M. Basic Skills in interpreting Laboratoary Data. ASHP. 2009:259- ISBN 978-1-58528-180-0.
- Buttarelo M, Plebani M. Automated blood cell counts: state of the art. Am J Clin Pathol. 2008;130(1):104-116.
Crossref - Brian M, Marc R. Desk Encyclopedia of Human and Medical Virology. 2010. ISBN: 9780123751478
- Raimondo G, Pollicino T, Squadrito G. Clinical virology of hepatitis B virus infection. J Hepatol. 2003;39(Suppl 1):26-30.
Crossref - Mariner PD, Korst A, Karimpour-Fard A, Stauffer BL, Miyamoto SD, Sucharov CC. Improved Detection of Circulating miRNAs in Serum and Plasma Following Rapid Heat/Freeze Cycling. Microrna. 2018;7(2):138-147.
Crossref - Sell S. Alpha-fetoprotein, stemcells and cancer: how study of the production of alpha-fetoprotein during chemical hepatocellularcarcinogenesis led to reaffirmation of the stem cell theory of cancer. Tumor Biol. 2008;29(3):161-180.
Crossref - Lorusso A, Quaia E, Poillucci G, Stacul F, Grisi G, Cova MA. Activity-based cost analysis of contrast-enhanced ultrasonography (CEUS) related to the diagnostic impact in focal liver lesion characterization. Insights Imaging. 2015;6(4):499-508.
Crossref - Xiaodong Wang, Joseph P, Erinjeri, et al. Pattern of Retained Contrast on Immediate Post procedure Computed tomography (CT) After Particle Embolization of Liver Tumors Predicts Subsequent Treatment Response. Cardiovasc Intervent Radiol. 2013;36(4):1030-1038.
Crossref - Ferlay J, Soerjomataram I, Dikshit R, et al. Cancer incidence and mortality world wide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136(5):E359-E386.
Crossref - Callegari E, Gramantieri L, Domenicali M, D’Abundo L, Sabbioni S, Negrini M. MicroRNAs in livercancer: a model fo rinvestigating pathogenesis and novel therapeutic approaches. Cell Death Differ. 2015;22(1): 46-57.
Crossref - Baghdady I, Fouad F, Sayed M, et al. Serum markers for the early detection of hepatocellular carcinoma in patients with chronic viral hepatitis C infection. Menoufia Med J. 2014;27(3):544-550.
Crossref - Marrero JA, Feng Z, Wang Y, et al. Alpha-feto protein, des gamma carboxy prothrombin, and lectin-boundalpha-fetoprotein in earlyhepato cellular carcinoma. Gastroenterology. 2009;137(1):110-118.
Crossref - Ha M, Kim VN. Regulation of microrna biogenesis. Nat Rev Mol Cell Biol. 2014;15(8):509-524.
Crossref - Mizuguchi Y, Takizawa T, Yoshida H, Uchida E. Dys regulated microRNAs in progression of hepatocellular carcinoma: a systematic review. Hepatol Res. 2016;46(5):391-406.
Crossref - Yang N, Ekanem NR, Sakyi CA, et al. Hepatocellular carcinoma and microRNA: new perspectives on the rapeutics and diagnostics. Adv Drug Deliv Rev. 2015;81:62-74.
Crossref - Tarek M, Louka ML, Khairy E, Ali-Labib R, Zaky DZ, Montasser IF. Role of microRNA-7 and selenoprotein P in hepatocellular carcinoma. Tumor Biol. 2017:39(5):1-10.
Crossref - Jiang L, Li X, Cheng Q, Zhang BH. Plasma microRNA might as a potential biomarker for hepatocellular carcinoma and chronic liver disease screening. Tumour Biol. 2015;36(9):7167-7174.
Crossref - Kamel MM, Matboli M, Sallam M, Montasser IF, Saad AS, El-Tawdi AHF. Investigation of long non coding RNAs expression profile as potential serum biomarkers in patients with hepatocellular carcinoma. Transl Res. 2016;168:134-145.
Crossref - Fang Y, Xue JL, Shen Q, Chen J, Tian L. MicroRNA-7 inhibits tumor growth and metastasis by targeting the phosphoinositide 3-kinase/Aktpathway in hepatocellular carcinoma. Hepatology. 2012;55(6):1852-1862.
Crossref - Ma C, Qi Y, Shao L, Liu M,Li X, Tang H. Downregulation of miR-7 upregulates Cullin 5 (CUL5) to facilitate G1/S transition in human hepatocellular carcinoma cells. IUBMB Life. 2013;65(12):1026-1034.
Crossref - Ning BF, Ding J, Liu J, et al. Hepatocyte nuclear factor 4α-nuclear factor-κ Bfeed back circuit modulates liver cancer progression. Hepatology, 2014;60(5):1607-19.
Crossref - Zhang X, Hu S, Zhang X, et al. MicroRNA-7 arrests cellcycle in G1 phase by directly targeting CCNE1 in human hepatocellular carcinoma cells. Biochem Biophys Res Commun. 2014;443(3):1078-1084.
Crossref - Guo X, Lv X, Lv Xing, Ma Yueyun, Chen L, Chen Y. Circulating miR-21 serves as a serum biomarker for hepatocellular carcinoma and correlated with distant metastasis. Oncotarget. 2017;8(27):44050-44058.
Crossref - Huang CS, Yu W, Cui H, et al. Increased expression of miR-21 predicts poor prognosis in patients with hepatocellular carcinoma. Int J Clin Exp Pathol. 2015;8:7234-7238.
- Liao Q, Han P, Huang Y, et al. Potential Role of Circulating microRNA-21 for Hepatocellular Carcinoma Diagnosis: A Meta-Analysis. PLoS ONE. 2015;10(6):e0130677.
Crossref - Chen X, Ba Y, Ma L, et al. Characterization of microRNAs in serum: a novel class of biomarkers for diagnosis of cancer and other diseases. Cell Res. 2008;18(10):997-1006.
Crossref - Tomimaru Y, Eguchi H, Nagano H, et al. Circulating microRNA-21 as a novel biomarker for hepatocellular carcinoma. J Hepatol. 2012;56(1):167-175.
Crossref - Corvalan AH. Early diagnosis of hepatocellular carcinoma bymicroRNAs: shining a light from the genome’s “darkmatter”. Dig Dis Sci. 2012;57(11):2737-2739.
Crossref
© The Author(s) 2021. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.