ISSN: 0973-7510
E-ISSN: 2581-690X
Nanoparticles have become important scopes of research. The present study made insight into the using of marine bacteria as a new ecofriendly and low cost source for biosynthesis of silver nanoparticles. Fifteen marine bacteria were tested for their capabilities to synthesize silver nanoparticles. Characterization of silver nanoparticles synthesized by the most promising Pseudomonas sp. H64 was performed using UV–Visible spectrophotometer, Fourier transform-infrared spectroscopy, zeta potential, transmission electron microscopy and X-ray. The produced Ag NPs are spherical particles with size of 3-22 nm and negative charge (-14 mV). Different valuable applications of the biosynthesized AgNPs were investigated. The produced AgNPs exhibited promising antibacterial activity against human and fish pathogens (Vibrio parahaemolyticus, Aeromonas hydrophila, Escherichia coli, Streptococcus faecalis, Staphylococcus aureus, Bacillus subtilis) with inhibition zone diameters (10-37 mm). Moreover, antifungal activity against (Fusarium solani, Rhizoctonia solani; Rhizopus oryzae, Helminthosporium sp.) expressed as inhibition of growth was (77.8±0.68-100%), while no antifungal activity was detected against Aspergillus niger. Antifouling and antioxidant activities were also confirmed. AgNPs exhibited anticancer activity toward (HepG-2, MCF-7, CaCo-2) cell lines. Moreover, AgNPs exhibited bioremediation potential of heavy metals and dyes.
Silver nanoparticles, Antibacterial, Pseudomonas sp. H64
Nanobiotechnology is a miscellany of biotechnology and nanoscience. Nanoparticles with sizes below 100 nm are applicable in different fields of biomedicine 1. Traditional methods for production of nanoparticles cause harmful effect to the environment. This leads to a growing interest for the development of non-toxic and environmentally friendly sources for production of nanoparticles2. Metal nanoparticles can be produced using natural marine resources as alternatives to the physical and chemical methods including plants, fungi, bacteria and algae 3.
Metal ions can be grabbed by the microorganisms from their environment and then turn into the element metal through reduction/oxidation mechanism using some enzymes with reducing or antioxidant properties which act on the compounds and result in the production of nanoparticles 4.
Researchers and pharmaceutical companies are searching for new antimicrobial agents due to arising of antibiotic resistant pathogenic strains causing infectious diseases5,6. Silver nanoparticles are firmly the best used nanomaterials as antimicrobial agents. Besides possessing antibacterial property, silver nanoparticles (AgNPs) seem to be good conductor, catalysts, antifungal agents, antiviral agents, anticancer, anti-inflammatory and antifouling 7- 11. In addition, nano-bioremediation technology has been developed to facilitate the bioremediation of organic pollutants including synthetic dyes and water treatment12.
According to our knowledge, few literatures concerned with the green synthesis of silver nanoparticles by marine bacteria. Thus, in the current study an attempt has been made towards synthesis of silver nanoparticles by marine bacteria as a new and ecofriendly marine source. Further characterization and evaluation of the antimicrobial, antifouling, anticancer and antioxidant activities in addition to removal of dye and heavy metals will be investigated.
Microorganisms
The marine bacteria were provided by Dr. Gehan Abou Elela. National Institute of Oceanography and Fisheries, Alexandria, Egypt.
Pathogenic indicators
Different pathogenic indicators of Gram-negative bacteria (Vibrio parahaemolyticus, Aeromonas hydrophila, Escherichia coli), Gram-positive bacteria (Streptococcus faecalis, Staphylococcus aureus, Bacillus subtilis) and fungi (Fusarium solani, Rhizoctonia solani, Rhizopus oryzae, Helminthosporium sp. and Aspergillus niger) were kindly provided by Marine Microbiology Department, Environment division, National Institute of Oceanography and Fisheries, Alexandria, Egypt
Growth condition and AgNPs biosynthesis
Pure culture of Pseudomonas sp. H64 was cultured on nutrient broth medium of the following components (g/l): beef extract, 1; yeast extract, 3; peptone, 5 at pH 7 to synthesize the AgNPs13. The bacterial culture was incubated for 24 h under shake condition at 150 rpm and 30 oC. The supernatant of the overnight bacterial culture was collected after centrifugation at 5000 g for 10 min. and retained for AgNPs biosynthesis. Fifty millilitre of cell-free supernatant was added to individual reaction vessels each containing fifty milliliter of AgNO3 solution (1 mM final concentration). The reaction mixture was incubated at 30 oC in the dark for 24 h. Visual observation of change in colour of the reaction mixture was regarded as positive indication of AgNPs biosynthesis compared with control (Absence of Ag NO3). The formation of nanoparticle was examined under UV-visible spectrophotometer at 24h time interval 6.
Characterization of AgNPs
UV–Visible spectrometric analysis
Confirmation of AgNPs production was performed using UV–vis spectral analysis using UV–Vis spectrophotometer (Helios alpha, Unican) at wavelength of 300–800 nm 14.
Fourier- Transform Infrared (FTIR) spectroscopy analysis
FTIR spectroscopy was carried out to determine the functional groups on the biosynthesized silver nanoparticles15, which are responsible for stability of the nanoparticle. The purified dried silver nanoparticles were mixed with potassium bromide in the ratio of 1:100 then analyzed in a FTIR (Perkin–Elmer Spectrum RX1, Shelton, Connecticut) with spectrum (450–4000 cm-1 ) and resolution of 4 cm-1 .
Zeta potential
Zetasizer (ZS 90, Malvern, UK) was used for analysis of surface charge on AgNps. Samples were exposed to dilution using 0.15M phosphate buffer saline at pH 7.2, then were examined in dynamic light scattering cuvettes by zeta potential within the range of 0.1–10000 nm at a scattering angle of 90° and 25°. This was done at Faculty of Pharmacy, Alexandria University.
Transmission electron microscope (TEM)
The morphology and size of AgNPS were characterized by TEM (TECNAI SPIRIT). It was carried out at the National Institute of Oceanography and Fisheries.
Energy dispersive X-ray analysis (EDX)
EDX was achieved by X-ray microanalysis system joined with SEM to confirm the presence of AgNPs and to observe the elementary compositions of the particles 14. It was investigated at the Faculty of Science, Alexandria University.
Preparation of Ag NPs stock solution
AgNPs (5 mg/mL) stock solution was prepared using sterilized deionized water. It was saved at 4oC and used within seven days. Sonication of the stock solution for 10 min and vortex for 1 min was done before use.
Antibacterial activity
Disk diffusion test 16 was applied to estimate the antibacterial effect of AgNPs. Briefly, the tested bacterial cultures were grown for overnight in nutrient broth, then separately lawn cultured on the prepared nutrient agar plates. Sterile filter paper discs (5 mm) were saturated by AgNPs solution and placed above the culture and incubated at 37oC for 24h. The diameter of inhibition zone was measured in millimeters.
Antifungal activity
Antifungal activity of AgNPs against F. solani, R. solani; R. oryzae, Helminthosporium sp. and A. niger was detected by assay of growth inhibition of the tested fungi17 with some modifications. 50 µl of AgNPs solution was added to potato dextrose medium (PDA) to obtain the required concentration. A disc of each indicator fungus (8mm diameter) was put on the presterilised PDA medium and incubated at 30 oC. AgNPs free medium was taken as control. Colony diameter was measured after 72 h and compared with the control (without addition of AgNPS). Inhibition % of the fungal growth was estimated using the formula:
I = C – T/ C x 100
Where I = percentage inhibition; C = radial growth in control, T = radial growth in treatment (Test).
Antifouling activity
Antifouling activity was performed according to Abd-Elnaby et al. (2016)11 with some modifications. Briefly, 200 µl of AgNPs was added to 1ml of Seawater in conical flask (50 ml) containing cover glass and incubated at 28oC for 24h. Then the cover glass was exposed to dying with crystal violet solution (0.4%) for ten minutes, followed by washing by water, dried at room temperature and observed under the microscope . A control flask with out AgNPs was taken as control.
Antioxidant activity
In vitro free radical scavenging activity of the biosynthesized AgNPs, was estimated with the 2,2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging assay. Reduced AgNPs were dissolved in 10, 20, 30, 40, 50, 60, 70, 80, 90 and 100mg/mL methanol; an equal amount of methanol was added to the control. DPPH solution (2 mL) in ethanol (100 mmol/l) was individually mixed with 2 mL of each concentration of methanol 13 followed by dark incubation (15 min); thereafter, the optical density was indicated at 517 nm against the blank, and ascorbic acid was taken as control. The scavenging percentages of DPPH with the AgNPs were calculated using the following formula: Sfr (%) =(Ac -As/Ac) x100%, where Sfr is free radical scavenging, Ac is the absorbance of the control (ascorbic acid) and As is the absorbance obtained from the sample.
Anticancer activity
The anticancer activity of the AgNPs at concentration (0.39-25µg/ml) was tested against Intestinal carcinoma cells (CaCo-2); Hepatocellular carcinoma cells (HepG-2) and Breast carcinoma cells (MCF-7). Cell growth and survival was detected using MTT assay18. Proliferation reduction of cancer cells was estimated as follows:
Inhibition rate (%) = (1- Abs sample / Abs control) × 100
Bioremediation potential of Ag NPs (Dye decolorization using AgNPs)
Decolorization of four dyes namely Acid orange, Direct green, Acid black and Acid fast brown using the biosynthesized Ag NPs was investigated. Briefly, 1ppm concentration of each dye were prepared. Five test tubes, each containing 1ml of the prepared dye solutions were prepared and 0.1 mg of AgNPs were added to each tube, while one was maintained as blank. The test tubes were incubated at 30 oC for 2 h, then samples were centrifuged and the supernatant was used to measure the concentration of dye using UV-visible spectrophotometer according to the l max of each dye15. Decolorization ratio expressed as mean value was estimated according to the following equation:
Decolorization%= (Co-Ce) x100 / Co
Co= Initial absorbance before decolorization, Ce: final absorbance after decolorization
Bioaccumulation of heavy metal solutions
The uptake capacity of AgNPs to different heavy metals such as copper sulphate, nickel chloride, cadmium chloride and lead chloride with concentration of 100 ppm was investigated using batch adsorption experiment 19.
Bioremediation study
The biosynthesized AgNPs (0.1 mg) were added to 50 ml of metal solution and kept under shake condition at 120 rpm and 30 R”C for 24 h. The solution was centrifuged at 6900 g for separation of the absorbent. In the end of each experiment, the residual metal concentration in each solution was determined using an Atomic Absorption Spectrophotometer (Shimadzu-A-A—6800). Accumulation of metal ions was evaluated using the following equation: q=V(IC-FC)/W, where q, metal accumulation; V, volume of reactions; IC, initial concentration (mg/l); FC, final concentration (mg/l) and W, total biomass 19.
Bacterial mediated synthesis of silver nanoparticles
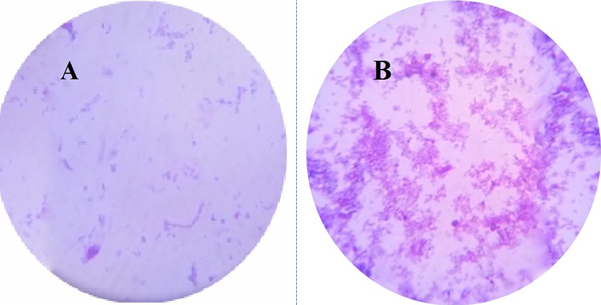
Fifteen bacterial species were tested for their ability to synthesize silver nanoparticles. Changes of the filtrate from yellow colour to yellowish-brown after addition of silver nitrate was used as indication of AgNPs formation14. The most promising isolate was Pseudomonas. sp. H64. It showed positive result in formation of AgNPs (Fig. 1). Development of surface plasmon resonance in the mixture of the reaction is the main cause of change in the colour as was previously reported 20.
Fig. 1. Cell filtrate of Pseudomonas sp. H64 (before addition of AgNO3) (A); treated with 1mM AgNO3 (B)
Characterization of the biosynthesized AgNPs
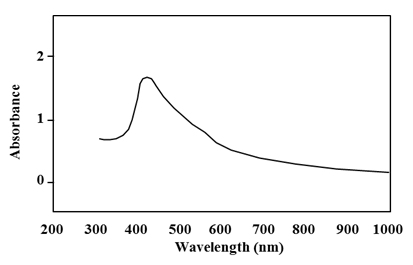
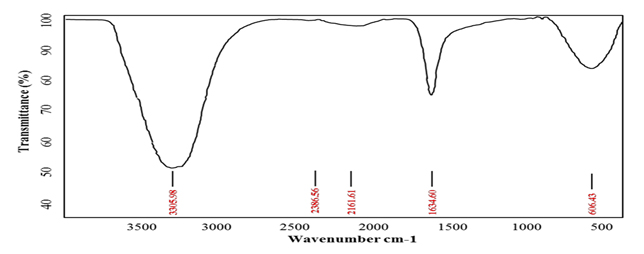
AgNPs synthesis by Pseudomonas. sp. H64 was first confirmed by UV analysis. The absorption obtained indicated a characteristic peak for silver nanoparticles at 450 nm (Fig. 2). As it was reported, reduction of the silver ions to silver metal occurred in nanometer range by the action of extracellular reductase enzymes excreted by the microorganisms 14. Some studies showed that the plasmon absorbance of AgNPs caused colour change of the reaction mixture to brown and the obtained peak was at 420 nm 21. Screening for the functional groups included in the biosynthesized AgNps was accomplished using FT-IR analysis aiming at understanding the transformation process of silver nitrate into elemental silver. FT-IR spectrum at array of absorbance bands from 400-4000 cm-1 (Fig. 3) showed three major peaks. Intense absorption bands at 3305. 98, which is characteristic to amine group(N–H stretch), 1634.60 cm-1 which is characteristic to protein and 606.4 cm-1 which indicates the presence of C-l stretching of halides. Thus the present study confirmed the presence of amines, halides and proteins, which have strong binding affinity with Ag and help in capping, reducing and conversion of Ag +ions to AgNPs. Similar results were previously reported 22,23. It was stated that fabrication and stabilization of metal nanoparticles is dependent on different molecules including proteins, amines, phenolics, carbonyl groups, terpenoids 24. Another study showed that proteins exhibit paired function of Ag+ reduction and shape-control during the synthesis of the silver nanoparticles14.
Zeta potential
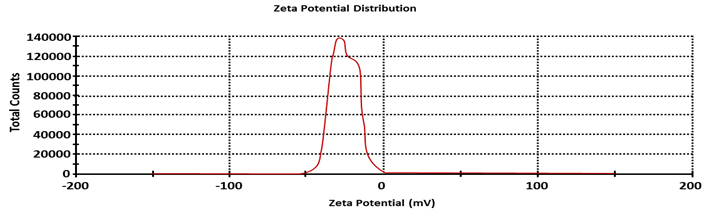
Zeta potential (f) is used to predict the interaction between particles in suspension. In the present study, the zeta potential value of Pseudomonas. sp. H64 mediated AgNPs was -14.4. mV (Fig. 4). Previous study reported that spherical Ag NPs showed potential of -15.3 mV22. Another study mentioned that charge of the AgNPs was -15.8 mV17. Disruption of charges on the surface plays a principal role in the assembly of the nanoparticles. Therefore, nanoparticles with reasonable zeta potential value may be applied as a drug providing a new horizon in avoiding resistance of bacteria and fungi 25.
TEM
TEM micrographs of the purified nanoparticles (Fig. 5) showed the presence of silver nanoparticles with spherical shape, and size from 3-22 nm. It was reported that Streptomyces ERI-3 mediated synthesis of silver nanoparticles were relatively spherical with size 110-100 nm 26. It can be concluded that, the size ranges of silver nanoparticles synthesized by the marine of Pseudomonas. sp. H64 in the current investigation seemed closer to the size of AgNPs synthesized by other bacteria, which enable their use in different applications27,28.
EDX
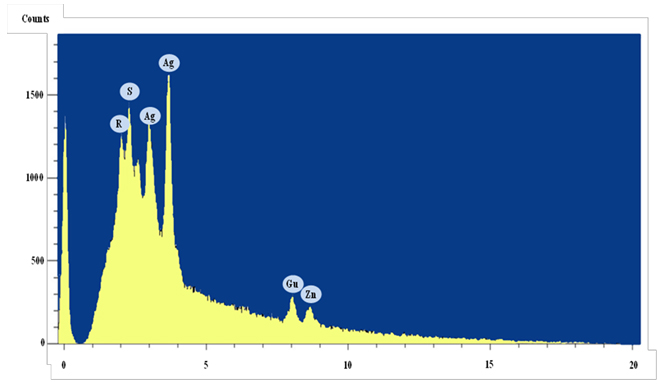
EDX analysis was done to study the X-ray analysis of silver peak in AgNPs. Results in Fig. 6 indicated that the absorption peak of AgNPs was observed at 3.5 keV, which represented a typical peak of metallic silver nanoparticles and confirmed that the main composition of the nanoparticles was silver as was reported 29,30. Appearance of other peaks such as copper, sulphur, phosphorus and zinc, may be produced from the chemical and biological molecules like proteins and enzymes bound to the surface of the AgNPs14,31.
Biotechnological applications of the biosynthesized AgNPs
Antibacterial activity
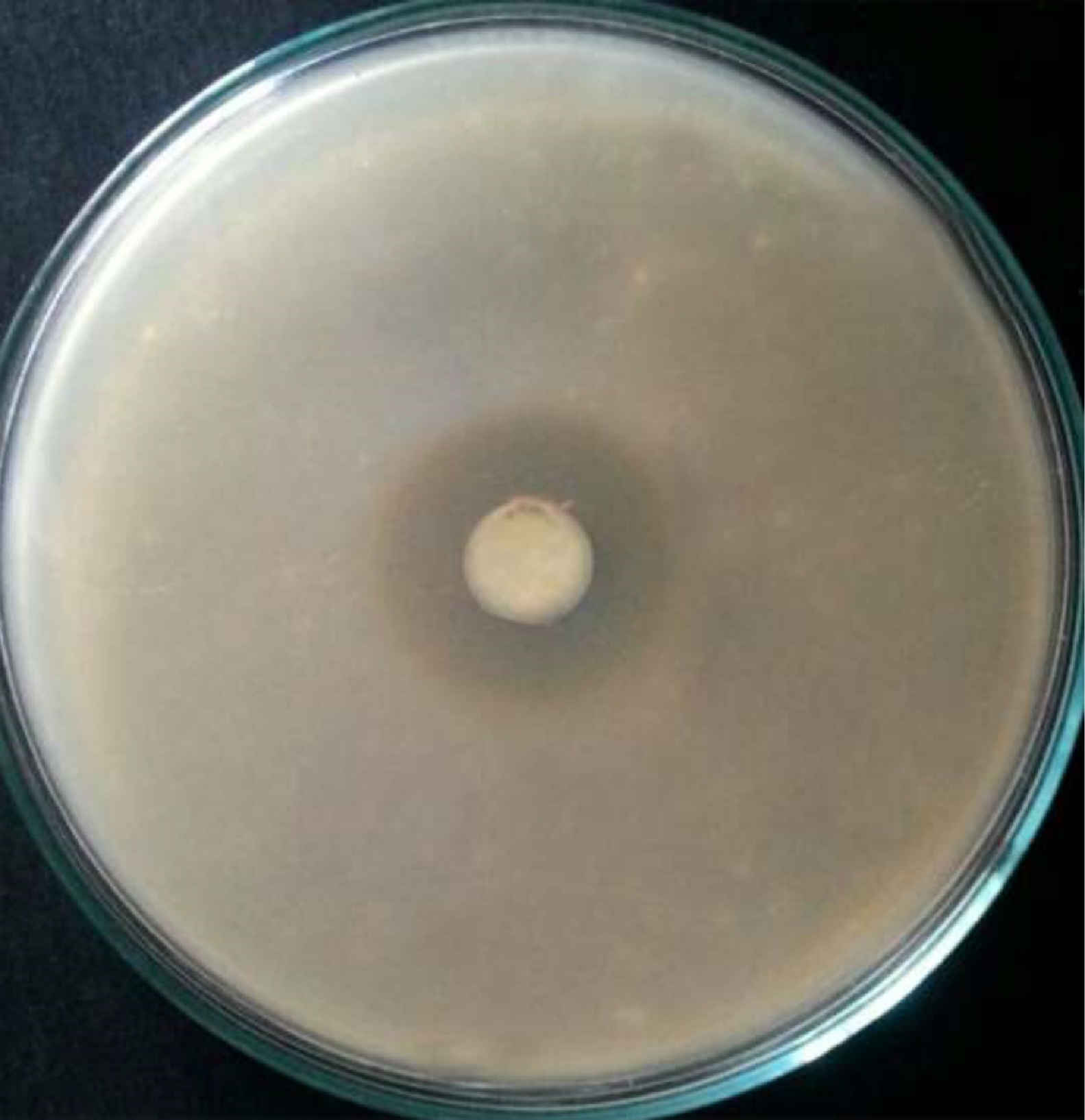
The biosynthesized AgNPs exhibited varied antibacterial activity against the tested pathogens (B. subtilis, S. faecalis, S. aureus, E. coli, A. hydrophil, V. parahaemolyticus) using disc diffusion method. As shown in Table 1, the highest antibacterial zone of inhibition (37±3.08 mm) was recorded against A. hydrophila (Fig. 7) followed by E.coli (32±2.67 mm). Abd-Elnaby et al.19 demonstrated the antibacterial effect of the AgNPS synthesized by marine Streptomyces rochei MHM13 against the test pathogens including Staphylococcus aureus 25923, Bacillus subtilis 6633, Escherichia coli 19404, Pseudomonas aeruginosa 9027, Bacillus cereus, Salmonella typhimurium 14028, Vibrio damsela and Vibrio fluvialis with inhibition zone diameter (16-19 mm). Antibacterial activity of Ag NPs was proven in other studies 32-35. Higher effect against Gram negative bacteria may be due to the existence of thin peptidoglycan layer compared to rigid peptidoglycan layer in Gram positive bacteria36. Moreover, the charged AgNPs exhibit better ability to bind the negative charges on cell wall of the Gram negative bacteria. The effect may also be attributed to binding of nanoparticles to bacterial membrane and release of hydrogen peroxide (H2O2) and super-oxide, which may lead to bacterial killing37.
Fig. 7. Zone of inhibition of silver nanoparticles synthesized by Pseudomonas sp. H64 against A. hydrophila
Table (1):
Antibacterial activity of AgNPs (expressed as inhibition zone diameter (mm) against different Gram positive and Gram negative bacterial pathogens.
Bacterial pathogen |
S. aureus |
S. faecali |
B. subtilis |
A. hydrophila |
V. parahaemolyticus |
E. coli |
|---|---|---|---|---|---|---|
Inhibition zone diameter (mm) |
25±1.92 |
21±1.5 |
17±1.21 |
37±3.08 |
10±0.77 |
32±2.67 |
Antifungal activity
The present study extended to investigate the potency of the biosynthesized Ag NPs by Pseudomonas. sp. H64 as antifungal agents against R. solani, R. oryzae F. solani, Helminthosporium sp. and A. niger. Results shown in Table 2 revealed complete inhibition of R.oryzae. On the other side, silver nanoparticles caused 89%±0.36 and 89±0.70 % inhibition of F.solani and R. solani, respectively, while the lowest antifungal activity (77.8%±0.68) was against Helminthosporium sp. Absence of antifungal activity was observed against A. niger. Ali et al.38 tested the antifungal activity of the biosynthesized AgNPs against Fusarium graminearum, Fusarium solani and Penicillium expansum and showed the inhibition of all tested fungi with the highest inhibition (91%) against F. solani. Efficiency of biosynthesized nanoparticles as fungicides was previously reported in different studies38,39. It was reported that the efficiency of AgNPs as antifungal agent is due to the presence of some components in the extract, which increase the reduction of the metals and stability of the resultant NPS8.
Table (2):
Antifungal activity of Ag NPs (expressed as inhibition % of fungal growth).
| Fungal pathogen | A. niger | R.oryzae | R.solani | Helminthos porium sp. |
F.solani | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Growth inhibition (%) | 0 | 100± 0.70 |
89± 0.70 |
77.8± 0.68 |
89± 0.36 |
||||||
Antifouling activity
Antifouling activity of the biosynthesized AgNPs was tested. Results (Fig. 8A) showed the inhibitory activity of AgNPs on biofilm formation compared to the control (Fig. 8B). This in agreement with that reported by Abd-Elnaby et al. (2016)11, who confirmed the potential use of silver nanoparticles synthesized by marine Strptomyces. rochei HMM13 as antifouling agent.
Fig. 8. Photographs showing the antifouling effect of AgNPs synthesized by Pseudomonas sp. H64 on biofilm formation (A), Control (without AgNPs) (B)
Antioxidant activity
Natural antioxidants exhibit protecting effect of cells from oxidative damage40. The antioxidant activity of the AgNPs was detected using DPPH radical scavenging assay. The AgNPs achieved antioxidant activity with 84% inhibition at 100µg/ml. In parallel study, the antioxidant activity of AgNPs produced by Bacillus aerius caused 66.23% inhibition at 100µg/ml30.
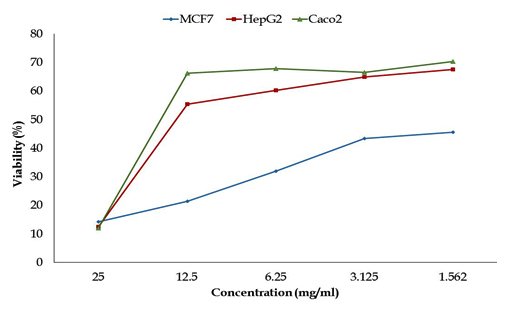
Fig. 9. Anticancer activity for different concentrations of silver nanoparticles produced by Pseudomonas sp. H64 against different cancer cell lines (measured by MTT assays after 24 h exposure)
Anticancer activity
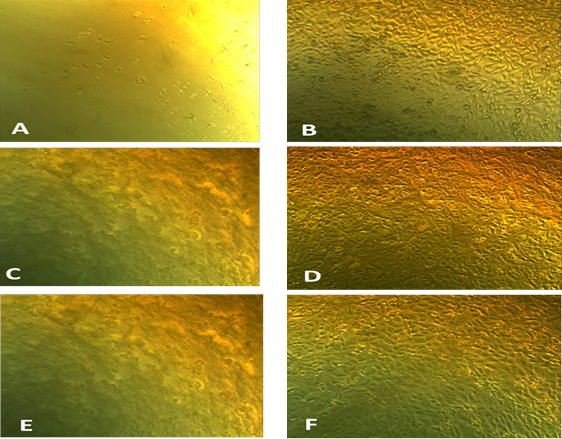
Anticancer activity of the Ag NPs (1.562-25mg/ml) was first detected by testing the toxicity against three cell lines: Intestinal carcinoma cells (CaCo-2); Hepatocellular carcinoma cells (HepG-2) and Breast carcinoma cells (MCF-7). Results in Fig. 9 showed that the viability of CaCo cells was (11.95-67.93 %), it was (12.40-60.24%) for HepG2 while the range of viability of MCF-7 was (14.24-32%) which means that the highest inhibitory activity was against MCF-7 and HepG2. The anticancer activity against the tested cell lines is shown in Fig. 10. The inhibition in the viability of the tested cell lines was dose dependent, which is in consistent with Abd -Elnaby et al.19 who reported that the highest anticancer efficiency of the biosynthesized AgNPs was against HCT-116, MCF-7, Hep-G2 and A-549 cell lines, while the lowest activity was against CACO. Parallel study reported 80% inhibition of MCF-7 cells with IC50 value less than 10 µg/ml. It was reported that the cytotoxic effect of silver nanoparticles is cell-type dependent 1 and change in morphology of the cell lines might be due to interaction between cell surface and AgNPs causing disturbance in cell composition7.
Fig. 10. Photographs illustrating the difference between the tested silver nanoparticles on growth of CaCo-2 tumor cell line (A) compared to control (B); on growth of HepG2 (C) compared to control (D) and on growth of MCV tumor cell line (E) compared to control (F)
Bioremediation study
Biosorption of synthetic Azo dyes
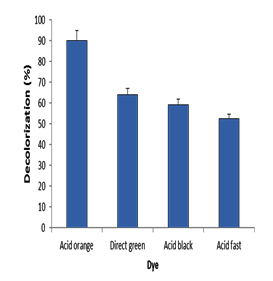
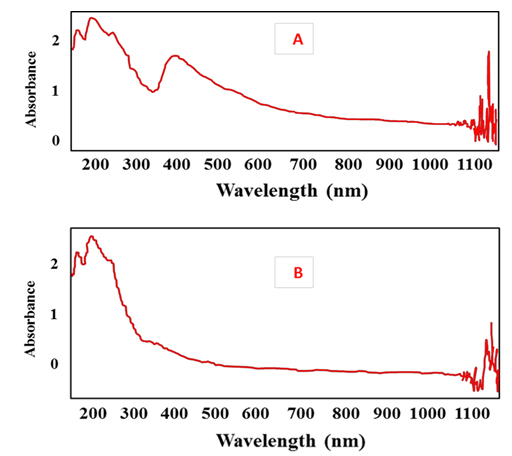
The biosynthesized nanoparticles by microorganisms showed excellent ecofriendly agent for catalytic activity and hence in dye effluent treatment 14. In the present experiment the potential of silver nanoparticles in decolorization of some Azo dyes including Acid orange, Direct green, Acid black and Acid fast brown was studied. decolorization of dyes was measured using UV-Vis spectrophotometer after 2h. Results (Fig. 11) revealed that the decolorization of Acid orange, Direct green, Acid black and Acid fast brown was 90%, 64%, 59% and 52.3%, respectively. The highest decolorization was for acid orange and was confirmed by UV spectrum analysis (Fig. 12 A, B). Results (Fig. 12 A) detected the presence of a strong sharp peak at 420 nm in visible region of the spectrum which diminished and disappeared after 2h of treatment with AgNPs synthesized by Pseudomonas. sp. H64 (Fig. 12 B). The present results are in harmony with previous study15 who showed 85% decolorization efficiency of Azo dye by Ag NPs synthesized by bacterial strain AN-1, after 32h. The highest decolorization efficiency of Phenol Red–PR, Methyl Orange–MO, Saffrain Stain Powder–SSP, Bromo Cresol Green–BCG by Ag nanoparticles synthesized using Pterocarpus Santilanus was 53%, 48%, 44%, 49% at the time of 30 min, 25 min, 40 min, 50 min, respectively41. Efficiency of Ag NPs synthesized by bacteria was also confirmed in other studies 42,43. pH is an important factor affecting the efficiency of dye adsorption and removal by AgNPs , where at lower pH, positively charged nano particles are formed, which are favourable for removal of the negative charged dye, providing more surface area and adsorbent sites41.
Fig. 12. UV spectra of Acid orange dye before treatment with silver nanoparticles (A); after treatment with silver nanoparticles (B)
Bioremediation of heavy metals
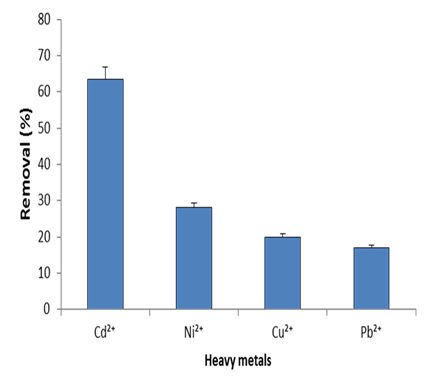
Results (Fig. 13) indicated that the highest removal efficiency (63.5 %) by AgNPs was for Cd2+, followed by 28%, 20% and 17% for Ni2+, Cu2+ and Pb2+, respectively. Al-Qahtani 44 confirmed the capability of the silver nanoparticles for removal of Pb2+ from aqueous solution. Also, removal efficiency for other metals including Pb2+, Zn2+, Cu2+ and Ni2+ was reported 45,46 showing that the removal efficiency depending on agitation speed, pH, contact time, initial metal ion concentration, and adsorption dosage. Difference in the removal efficiency for each tested metal may be also attributed to availability of binding sites on the surface of AgNPs45.
Fig. 13. Removal efficiency of different heavy metals by silver nanoparticles synthe-sized by marine Pseudomonas sp. H64
Statistical analysis
Triplicates experiments were done. Results were analyzed using IBM SPSS software package version 20.0. (Armonk, NY: IBM Corp). The results were shown as mean ± standard deviation47.
The evolution of efficient, easy and eco-friendly methods in the synthesis and application of nanoparticles are very promising and is beneficial for mankind. The present study encouraged the use of marine Pseudomonas. sp. H64 a natural and renewable source for biosynthesis of silver nanoparticles with different valuable applications. The biosynthesized AgNPs by Pseudomonas. sp. H64 could be considered as broad-spectrum antimicrobial agents. Moreover, antifouling, antioxidant and antitumor activities were proven. The bioremediation potential of heavy metals and Azo dyes also was confirmed. More studies concerning the optimization of Ag NPs will be conducted in the future.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
- Abdel-Fattah, W., Ali, G. On the anti-cancer activities of silver nanoparticles. J. Appl. Biotechnol. Bioeng. 2018; 5: 43-46.
- Sunkar, S., Nachiyar, V., Albert, K. Phytogenic silver nanoparticle synthesis with potential antibacterial activity and dye degrading ability. Res. J. Pharm. Biol. Chem. Sci., 2013; 4: 1085-1097.
- Buszewski, B., Railean-Plugaru, V., Pomastowski, P.,Rafiñska, K., Szultka-M lynska, M., Golinska, P., Wypij, M., Laskowski, D., Dahm, H. Antimicrobial activity of biosilver nanoparticles produced by a novel Streptacidiphilus durhamensis strain. J. Microbiol. Immunol. Infect., 2018; 51: 45-54.
- Yasmin, A., Ramesh, K ., Rajeshkumar, S. Optimization and stabilization of gold nanoparticles by using herbal plant extract with microwave heating. Nano Converg., 2014; 1: 1-7.
- Singh, R., Vora, J., Nadhe,, S.B., Wadhwani ,S.A., Shedbalkar, U.U, Chopade, B.A. Antibacterial activities of bacteriagenic silver nanoparticles against nosocomial Acinetobacter baumannii. J. Nanosci. Nanotechnol., 2018; 18: 3806-3815.
- Abo-State, M.A.M., Partila, A.M. Microbial production of silver nano particles by Pseudomonas aeruginosa cell free extract. J. Eco. Heal. Env., 2015; 3: 91-98.
- Ramkumar, V., Pugazhendhi, A., Gopalakrishnan, K., Sivagurunathan, P., Saratale, G., Dung, T., Kannapiran, E. Biofabrication and characterization of silver nanoparticles using aqueous extract of seaweed Enteromorphacompressa and its biomedical properties. Biotechnol. Rep., 2017; 14: 1–7.
- Shaik, M., Khan , M., Kuniyil, M., Warthan, A., Alkhathlan, H., Mohammed, R., Siddiqui, Shaik, J., Ahamed, A., Mahmood, A., Khan, M., Adil, S. Plant-extract-assisted green synthesis of silver nanoparticles using origanumvulgare L. extract and their microbicidal activities. Sustainability., 2018; 10: 1-14.
- Kalaivani, R., Maruthupandy, T., Muneeswaran, A., HameedhaBeevi, M., Anand, C.M., Ramakritinan, A., Kumaraguru, K. Synthesis of chitosan mediated silver nanoparticles (Ag NPs) for potential antimicrobial applications. Front. Lab. Med., 2018; 2: 30-35.
- Khan,S.U., Anjum, S.I., Ansari, M. J., Khan, M.H., Kamal, S.,Rahman, K., Shoaib, M., Man, S., Khan, A. J., Khan, S.U., Khan, D. Antimicrobial potentials of medicinal plant’s extract and their derived silver nanoparticles: A focus on honey bee pathogen. Saudi J. Biol. Sci., 2018; 1-20.
- Abd-Elnaby, H.M., Abo-Elala, G.H., Abdel-Raouf, U.M., Hamed, M.M. Antibacterial and anticancer activity of extracellular synthesized silver nanoparticles from marine Streptomyces rochei MHM13. Egyp. J. Aquat. Res., 2016; 42: 301-312.
- Syafiuddin, A., Salmiati, S., Hadibarata, T., Kueh, A., Salim, M.R., Zaini, M.A. Silver nanoparticles in the water environment in Malaysia: Inspection, characterization, removal, modeling, and future perspective. Sci .Rep., 2018; 8: 986.
- Elbeshehy, E.K.F., Elazzazy, A.M., Aggelis, G. Silver nanoparticles synthesis mediated by new isolates of Bacillus spp., nanoparticle characterization and their activity against Bean yellow mosaic virus and human pathogens. Front. Microbiol., 2015; 6: 453.
- El-Sheekh, M.M., El Kassas, H. Biosynthesis, characterization and synergistic effect of phytogenic gold nanoparticles by marine picoeukaryotePicochlorum sp. in combination with antimicrobials. Rend. Fis. Acc. Lincei., 2014; 25: 513–521.
- Amabye, T.G., Hagos, K. Green synthesis of silver nanoparticles and evaluation of its potential for decolourization of a synthetic dye and antibacterial activity. J. Medicinal. Chemi.Toxicol., 2016; 1: 2-8.
- Katva, S., Das, S., Moti,H.,Jyoti, A., Kaushik, S.Antibacterial synergy of silver nanoparticles with gentamicin and chloramphenicol against Enterococcus faecalis. Pharmacogn.Mag., 2017; 13: S828–S833.
- Kumar, P., Dhiman, S., Bhatt, R.P., Singh, L. In-vitro antifungal activity of Sapiumsebiferum L. against Aspergillusniger and Aflatoxigenic Aspergillus flavus. J. Appl. Pharm .Sci., 2011; 01: 108-110.
- Xie, C., Zhou, J., Guo, Z., Diao, X., Gao, Z., Zhong, D., Jiang, H., Zhang, L., Chen, X. Metabolism and bioactivation of famitinib, a novel inhibitor of receptor tyrosine kinase, in cancer patients. Br. J. Pharmacol., 2013; 168: 1687–1706.
- Abd-Elnaby, H.M., Abou-Elela, M.G., Ghozlan, H.A., Hussein, H., Sabry, S.A. Characterization and bioremediation potential of marine Psychrobacter species. Egypt. J .Aqua. Res., 2016; 42: 193–203.
- Zarina, A., Nanda, A. Green approach for synthesis of silver nanoparticles from marine Streptomyces-MS 26 and their antibiotic efficacy. J. Pharm. Sci. Res., 2014; 6: 321-327.
- Busi, S., Rajkumari, J., Ranjan, B., Karuganti, S. Green rapid biogenic synthesis of bioactive silver nanoparticles (AgNPs) using Pseudomonas aeruginosa. IET Nanobiotechnol., 2014; 8: 267-274.
- Singh, S., Bharti, A., Meena, V.K. Structural, thermal, zeta potential and electrical properties of disaccharide reduced silver nanoparticles. J. Mater. Sci. Mater Electron., 2014; 25: 3747–3752.
- Lakhsmi, S.Y.S., Lakhsmi, H., Sharmila, S. Isolation, screening, identification, characterization and applications of green synthesized silver nanoparticle from marine Actinomycetes-streptomyces althioticus. World. J. Pharm. Res., 2015; 4 -7: 1592–1611.
- Gomaa, E.Z. Silver nanoparticles as an antimicrobial agent: A case study on Staphylococcus aureus and Escherichia colias models for Gram-positive and Gram-negative bacteria. J. General. Appl. Microbiol., 2017; 63: 36–43.
- Salem,H.F., Eid, K., Sharaf, M.A. Formulation and evaluation of silver nanoparticles as antibacterial and antifungal agents with a minimal cytotoxic effect. Int. J. Drug Deliv., 2011; 3: 293- 304.
- FaghriZonooz, N., Salouti, M. Extracellular biosynthesis of silver nanoparticles using cell filtrate of Streptomyces sp. ERI-3. Sci. Iranica., 2011; 18: 1631–1635.
- Nayak, D., Pradhan, S., Ashe, S., Rauta, P.R., Nayak, B. Biologically synthesized silver nanoparticles from three diverse family of plant extracts and their anticancer activity against epidermoid A431 carcinoma. J. Collo. Interface. Sci., 2015; 457: 329–338.
- Dauthal, P., Mukhopadhyay, M. Noble metal nanoparticles: plantmediated synthesis, mechanistic aspects of synthesis, and applications. Industrial Engin. Chem. Res., 2016; 55: 9557–9577.
- Mohamedin, A., El-Naggar, N.E., Hamza, S.S., Sherief, A.A. Green synthesis, characterization and antimicrobial activities of silver nanoparticles by Streptomyces viridodiastaticus SSHH-1 as a living nanofactory: statistical optimization of process., 2015; 137: 1-4.
- Mahmoud,W.,Elazzazy, A.M.,Danial, E.N. In vitro evaluation of antioxidant, biochemical and antimicrobial properties of biosynthesized silver nanoparticles against multidrug-resistant bacterial pathogens. Biotechnol.Biotechnol. Equip., 2017; 31; 373–379.
- Mishra, S., Singh, B.R., Naqvi, A.H., Singh, H.B. Potential of biosynthesized silver nanoparticles using Stenotrophomonassp. BHU-S7(MTCC 5978) for management of soil-borne and foliar phyto pathogens. Sci. Rep., 2017; 7: 45154.
- Al-Hulu, S.M. Detection on sliver nanoparticles production by Streptomyces spp. isolated from soil samples in Hilla city. Int.J. Chem. Tech. Res., 2017; 10: 577-582.
- Alabdulmohsen, Z.A., Saad, A.Y. Antibacterial effect of silver nanoparticles against Enterococcus faecalis. Saudi.endod. J., 2017; 7: 29-35.
- Javani,S., Marín,. I., Amils, R., Abad, J.P. Four psychrophilic bacteria from Antarctica extracellularly biosynthesize at low temperature highly stable silver nanoparticles with outstanding antimicrobial activity. Colloids Surfaces A: Physicochemical and Engineering Aspects., 2015; 483: 60–69.
- Avila, E.M., Ferro-Flores, G, Ocampo-García, B.E., López-Téllez, G., López-Ortega, J., Rogel-Ayala, D.G., Sánchez-Padill, D. Antibacterial efficacy of gold and silver nanoparticles functionalized with the ubiquicidin (29–41) antimicrobial peptide. J. Nanomaterials. 2017; 2017: 1-10.
- Kokila, T., Ramesh, P.S., Geetha, D. Biosynthesis of silver nanoparticles from Cavendish banana peel extract and its antibacterial and free radical scavenging assay: a novel biological approach. Appl. Nanosci., 2015; 5: 911–920.
- Vidya, R., Venkatesan, K. Preparation and characterization of zinc ferrite (ZnFe2O4) nanoparticles using self-propagated combustion route and evaluation of antimicrobial activity. Res. J. Pharm. Biol. Chem. Sci., 2015; 6: 537-542.
- Ali, S.M., Yousef, N.M.H, Nafady, N.A. Application of biosynthesized silver nanoparticles for the control of land snail Eobaniavermiculata and some plant pathogenic fungi. J. Nanomat., 2015; 1-10.
- Mallmann, E.J., Cunha, F.A., Castro, B., NMF, Maciel, A.M., Menezes, E.A., Fechine, P.B. Antifungal activity of silver nanoparticles obtained by green synthesis. Rev. Inst. Med. Trop. Sao. Paulo., 2015; 57: 165-167.
- Gridharathan, T., Masis, C., Sindhu, S., Arumugam, P. Studies on green synthesis characterization and anti- proliferative potential of silver nanoparticle using Dodonaea viscose and Capparisdeciduas. Biosci.Biotechnol.Res.Asia., 2014; 11: 665-673.
- Raju, C.A.I., Bharghavi, D., Satyanandam, K., Prem, K., TukaramBai, M. Removal of dyes from synthetic wasters using biosynthesized silver nanoparticles. Int. J. adv. Res. Eng. Technol., 2016; 7: 16–25.
- Abdel-Ghani1, N. T, El-Chaghab, G. A. Biosorption for metal ions removal from aqueous solutions: A review of recent studies. Int. J. Latest Res. Sci. Technol., 2014; 3: 24-42.
- Bykkam, S., Ahmadipour, M., Narisngam, S., Chidurala, S. Extensive studies on X-ray diffraction of green synthesized silver nanoparticles. Adv. Nanoparticles., 4: 1-10.
- Al-Qahtani, K.M. 2015. Water purification using different waste fruit cortexes for the removal of heavy metals. J. Taib. Univ. Sci., 2014; 10: 700-708.
- Suman,K. A., Gera,M., Jain, V.K. A novel reusable nanocomposite for complete removal of dyes, heavy metals and microbial load from water based on nanocellulose and silver nano-embedded pebbles. Environ. Technol., 2015; 36: 706-714.
- Igberase, E., Osifo, P., Ofomaja, A. The adsorption of Pb, Zn, Cu, Ni, and Cd by modified ligand in a single component aqueous solution: equilibrium, kinetic, thermodynamic, and desorption studies. Int. J. Anal. Chem., 2017; 1-5.
- Kirkpatrick, L.A., Feeney, B.C. A simple guide to IBM SPSS statistics for version 20.0. Student ed. Belmont, Calif.: Wadsworth, Cengage Learning. 2013.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.