ISSN: 0973-7510
E-ISSN: 2581-690X
Listeria monocytogenes is one of the most important foodborne pathogens in human worldwide. In present study, this bacterium was isolated from different animal products and human clinical samples. The isolates were characterized by antibiotic susceptibility tests, serotyping, virulence genes and 16SrRNA sequencing. Out of 1362 investigated samples, Listeria monocytogenes were identified in 48(3.5%) of samples. Seven samples 1.1% were from human, while 41(5.7%) were from food samples. The majority of food isolates were resistant to penicillin, cephalexin, doxycycline, ampicillin and vancomycin; while variable resistance to the other antibiotics was observed. Serotyping of food and human isolates found that 7 of human isolates and 28 of food isolates belonged to serogroup 1/2a (3a). While, 8 isolates from food samples belonged to the serogroup 4b. Five fresh red meat isolates belonged to the serogroup 1/2b. All food and human isolates contained virulence genes actA, hlyA, plcA and iap genes. Phylogenetic analysis based on 16SrRNA sequencing showed that the L. monocytogenes isolated from milk were not closely related to the meat and human isolates. This data suggests that the antibacterial resistant Listeria monocytogenes are widely spread within the animal products rather than the clinical samples. The most common serogroup within the isolated strains was 1/2a (3a). Surprisingly, all isolates found to be virulent strains depending on the virulence genes detection. Therefore, it is highly recommended to apply strict biosecurity measurements on food and food processing environment to avoid or to maintain the spread of the bacterial infection within the area.
Listeria monocytogenes, Multiplex PCR, Virulence factors, antibiotic sensitivity test, Genotyping characterization, serotyping.
Listeria monocytogenes is a facultative anaerobic gram-positive intracellular pathogen and mesophilic. This microorganism is considered as a fatal foodborne bacterium with a great impact on a public health; the bacterial infection in human is associated with a high mortality rate (20-30%)1,2. The disease is a major risk among immunocompromised persons due to the suppression of T cell3; and among old people, pregnant woman, neonates, transplant and AIDS patients4. Listeria causes septicemia, meningitis, encephalitis, stillbirth and abortion, in addition to gastrointestinal5,6. Transmission of Listeria to the human could be through ingestion of undercooked and contaminated food7,8.
Numerous virulence associated genes were reported to play important roles in L. monocytogenes pathogenicity9,10. Phosphatidy-linositol phospholipase C (plcA), invasive associated protein (iap), actin polymerization protein (actA), listeriolysin (hlyA) and internalin (inlA) genes were found to play a crucial role in the bacterial pathogenicity9,10. Studies found various serotypes of L. monocytogenes strain; about 13 serotypes; 1/2a, 1/2b, 1/2c, 3a, 3b, 3c, 4a, 4b, 4ab, 3c, 4d, 4e and 7) have been recognized11. These serotypes are distributed according to the different environmental niches into 4 strains (I, II, III and IV). The majority of L. monocytogenes isolates from food samples and among patients (approximately 98%) belonged to strains I and II12. The serotypes of strain I consists of 1/2b, 3b, 4b, 4d and 4e; serotypes 1/2b, and 4b were documented as a causative agent of human listeriosis13. Strain II consists of serotypes 1/2a, 1/2c and 3c; serotype 1/2a have been detected in food and found to be associated with listeriosis in animal and sporadic cases in human14. However, strain III consists of serotypes 1/2c, and 4c and strain IV consist of 4b, 4d and 4e serotypes15.
This organism is distributed widely in food products such as meat, poultry and seafood. Studies found that approximately 99% of listeriosis cases occur through consumption of contaminated food8. Different serogroups are responsible for the different epidemiological features such as outbreak, sporadic, and epidemic, therefore, the proper serotyping of human isolates is important in order to identify the proper serogroups and the source of infection of this microorganisms2,16. However, little information on the prevalence of clinical listeriosis and L. monocytogenes prevalence among food products in Duhok province of Iraq is available. To our best knowledge insufficient studies took place to identify this bacterium in food products17,18 and no study, as yet, has been done to detect the prevalence rate of L. monocytogenes among human in Duhok province. Therefore, the present study was set to determine the prevalence of L. monocytogenes from human clinical samples in Duhok province, and to isolate and determine their serotypes and virulence potential. Besides, antibiotics susceptibility profile and genetic diversity between isolates were investigated.
Collection of samples
A total of 1362 samples were obtained from various sources during July 2016 to May 2017 in Duhok province, Iraqi Kurdistan Region. Three hundred and nine (309) frozen chicken samples were taken from the directorate of prevention affairs, 167 raw goat and mutton meat samples were collected from local butcher shops. Furthermore, 239 samples from milk and milk products (118 of raw milk from local sheep, and 121 samples of local soft cheese) were collected from local shops. In addition to food samples, a total of 400 human samples were collected from third trimester pregnant women (Urine =200 and High vaginal swab=200). The rest were 247 blood samples which were taken from different groups of immunocompromised patients such as renal failures patients (N=101), septicemia cases (N=83), premature babies (N=44), meningitis cases (N=15) and Heart failure patients (N=4). The urine and high vaginal swabs samples were collected from the patients who visited Duhok Obstetrics and Gynecology Teaching Hospital, while the blood samples were collected from Azadi Teaching Hospital, Hevi Pediatric Teaching Hospital, and Duhok kidney and diseases transplantation center. The samples were collected under the aseptic condition and were delivered in cold box to the directorate of prevention affairs where processed shortly within 24 hrs of collection.
L. monocytogenes isolation and identification
L. monocytogenes were isolated from food samples according to the standard double enrichment method recommended by ISO 11290:1 with some changes19. A ratio of 1:9 of all samples were collected, 1 ml of milk was add to 9 ml of half fraser broth (pre-enrichment medium) and 25 g of meat and soft cheese were inoculated into 225 ml of half fraser broth (LabM, UK), and then incubated for 24h at 30°C. From overnight incubate broth, 100ul (0.1ml) was transferred into 10ml of fully concentrated fraser broth (LabM, UK) as a second enrichment then incubated at 37°C for 48h. Loop full of inoculum from second enrichment was subsequently, platted on PALCAM agar (LabM, UK) and re-incubated for 48h at 37°C. The Gray-greenish colonies with black center were picked and streaked on Harlequin™ Listeria Chromogenic Agar (LabM, UK), the blue/green colonies surrounded by an opaque halo were then chosen and confirmed using 13 biochemical reactions such as aesculin hydrolysis, acid production from rhamnose, xylose (Microgen, UK)20.
DNA extraction and Confirmation of L. monocytogenes isolates by PCR
Genomic DNA was recovered from L. monocytogenes isolates using the direct boiling method 21. The purity and the concentration of the DNA was evaluated using Nanodrop (Thermofisher, UK) through the calculation of optical densities ratio at 260/280 nm. L. monocytogenes isolates were confirmed using PCR primers complementary to the highly conserved 16S rRNA sequence as stated by22. The reaction conditions consisted of initial denaturation of DNA template (94°C for 3 min), then 35 cycles of denaturation (94°C for 1 min), annealing (60°C for 2 min) and extension (72°C for 1min). This primer amplified 938 bp of 16S rRNA which is considered the species specific primers depending on the conserved region for L. monocytoges detection. The positive control was obtained from College of Veterinary Medicine- Duhok Research Center23 .The primers used are listed in Table 1.
Table (1):
List of oligonucleotide used in this study.
| No. | Primers | Sequences 5’→3′ | bp | Reference |
|---|---|---|---|---|
| 1 | 16S rRNA | F 5′-CAG CAG CCG CGG TAA TAC-3′ | 938 | 22 |
| R 5′-CTC CAT AAA GGT GAC CCT-3′ | ||||
| 2 | Iap | F 5′-ACA AGC TGC ACC TGT TGC AG-3‘ | 131 | 28
28 28 28 |
| R 5′-TGA CAG CGT GTG TAG TAG CA-3′ | ||||
| 3 | hlyA | F5′-GCA GTT GCA AGC GCT TGG AGT GAA-3’ | 456 | |
| R5’-GCA ACG TAT CCT CCA GAG TGA TCG-3′ | ||||
| 4 | ActA | F5′-CGCCGCGGA AATTAA AAA AAG A-3′ | 839 | |
| R5′- ACG AAGGAACCGGGCTGC TAG – 3′ | ||||
| 5 | PlcA | F 5′-CTGCTTGAGCGTTCATGTCTCATCCCCC-3′ | 1484 | |
| R 5′-ATG GGT TTC ACT CTC CTT CTA C-3’ | ||||
| 6 | lmo0737 | F 5′-AGGGCTTCAAGGACTTACCC-3′ | 691 | 13 |
| R 5′-ACGATTTCTGCTTGCCATTC-3′ | ||||
| 7 | lmo1118 | F5′-AGGGGTCTTAAATCCTGGAA-3′ | 906 | 13 |
| R5′-CGGCTTGTTCGGCATACTTA-3′ | ||||
| 8 | ORF2819 | F5′-AGCAAAATGCCAAAACTCGT-3′ | 471 | 13 |
| R5′-CATCACTAAAGCCTCCCATTG-3′ | ||||
| 9 | ORF2110 | F5′-AGTGGACAATTGATTGGTGAA-3′ | 597 | 13 |
| R5′-CATCCATCCCTTACTTTGGAC-3′ | ||||
| 10 | Universal 16SrRNA | 27 F 5′- AGAGTTTGATCMTGGCTCAG-3′ | 1600 | 29 |
| 1492R 5′-TACGGYTACCTTGTTACGACTT-3′ |
Antibiotic sensitivity test
All the L. monocytogenes isolates were subjected to antimicrobial sensitivity test against thirteen most frequently used antibiotics in veterinary and human therapy24,25, using disc diffusion method. The following antibiotics discs (Oxoid, UK) with the following concentrations were tested in the study: Vancomycin (30 mcg), gentamycin (10 mcg), cephalexin (30 mcg), piperacillin (100 mcg), meropenem (10 mcg), doxycycline (10 mcg), chloramphenicol (10 mcg), ampillicin (10 mcg), penicillin (10 U), rifampin (5 mcg), clindamycin (2 mcg), co-trimoxazol (1.25/23.75 mcg). The zone of inhibition was recorded and interpreted after incubation at 37°C for 24h following clinical and Laboratory standards Institute guideline for Gram-positive bacteria26.
Virulence genes detection of L. monocytogenes
Virulence genes amplification (hylA, plcA, actA and iap) was carried out through Multiplex PCR using similar amplification conditions and primer sets as used earlier27. Briefly, PCR reaction was carried out in 25µl volume containing 12, 5µl ready to use master mix (1X) (Genet-Bio, South korea), primers (10ng/µl) each and DNA template (50 ng/ µl) under the following conditions: initial denaturation for 2 mints at 94°C, then 30 cycles for (20 s at 94°C, 20 s at 55°C and 50 s at 72°C) and final extension for 2 mints at 72°C. The details of oligonucleotides used in this study are shown in (Table 1).
Serogroup identification of L. monocytogenes
Serogrouping of L. monocytogenes (1/2a, 1/2b, 1/2c, and 4b) was performed using Multiplex PCR using primers Imo0737, Imo1118, ORF2819 and ORF2110 and the amplification conditions as reported before11. Amplification was carried out in a 25ul reaction volume containing 12.5µl ready to use master mix (1X) (Genet-Bio, South Korea), Primers were used at 0.1µM and about 50 ng/ul of DNA was added as template. Reaction was carried out with an initial denaturation for 5 min at 94°C, 35 cycles for (30 s at 94°C, 75 s at 54°C and 75 s at 72°C) and final extension for 10 min at 72°C in a thermal cycler (Applied Biosystems 9700, USA).
16S rRNA sequencing and phylogenetic construction
The sequencing of universal 16SrRNA29 was implemented using primer listed in (Table 1) at Macrogen Company, Korea. The qualities sequences were tested and aligned using BioEdit sequence alignment editor 7.0.0 (Isis Pharmaceuticals, Inc., Carlsbad, USA). The sequences of different isolates were submitted to GenBank to get the GenBank accession Numbers (Fresh red meat isolate MK968361, Frozen chicken meat isolate MK968368, Raw milk isolate MK968369, white soft cheese isolate MK968371, human-vaginal swab MK968366, human-blood MK968364 and human-urine MK968365). The sequence identity comparing with corresponding sequences submitted in GenBank was estimated using the “BLAST” tool on NCBI website. The sequences were aligned using clustalW. The Neighbor-Joining method with Jukes-Cantor model in MEGA7 with 1000 bootstrap replicates was used for construction of phylogenetic tree.
Prevalence of L. monocytogenes in different samples
In the present study, 715 food samples were tested, out of these, a total of 309 frozen chicken samples and 167 fresh red meat samples were tested. L. monocytogenes was isolated at a rate of 8.73% from frozen chicken samples and 5.98% from fresh red meat. Our findings are in agreement with the prevalence rate of 8% and 7.1% in Iraq17,18 both in chicken samples. Similarly, a study conducted in Egypt reported 8.1% isolation which is very close to our findings30. However, other studies reported much higher values compared to our results, particularly 94.7% recorded in Turkey31 and 14.1% in Iran32. The lowest prevalence to our knowledge was 0.8% from a study conducted in South Korea33. Regarding to the raw red meat, the prevalence rate of L. monocytogenes in this study was close to what was found in France (5.0%) and South Korea (5.2%), which support our results22,34. In different studies conducted in the Kurdistan region/ Iraq, a higher prevalence was reported from red meat at 14%17. In contrast to all previously mentioned studies in red meat including the current study, lower prevalence rates were recorded in Spain, Turkey, and India 2.6% and 2.2%, respectively35,36. Furthermore, studies took place in India and reported the prevalence of L. monocytogenes 2.4 % and 2.7% in two separate studies37,38. High mortality and hospitalization rates are recorded from L. monocytogenes infections due to eating of contaminated and undercooked food. Different factors found to be associated with the incidence of L. monocytogenes in meat such as the ability of this microorganism to form biofilms on the exterior and interior part of the tissue and the optimum temperature and pH of meat39,40.
Raw milk and locally produced white soft cheese are other types of samples were investigated for L. monocytogenes identification. The results showed that out of 118 milk samples examined just 3 (2.54%) samples were positive for L. monocytogenes and only 1(0.82%) out of 121 white cheese samples were found to be positive for L. monocytogenes. This study found low prevalence of L. monocytogenes in dairy products. This is probably due to pasteurization of milk is an obligatory step during soft cheese preparation. However, most of L. monocytogenes contamination in cheese occurs after pasteurization41. The differences in prevalence rate in different studies and sources could be due to many reasons such as, sample size, study region, time of study and methods of L. monocytogenes isolation17. In addition to the slaughtering process of animals, personal hygiene and storage condition of food samples. The nutrient composition, water content, and pH of the environment are other significant factors that enable proliferation of numerous microorganisms in raw milk and dairy products and other food samples42. On the other hand, Gilbert and colleagues highlighted different factors that could affect the incidence rate of L. monocytogenes in milk in different countries; including differences in seasonal variation, location, differences between the milking devices used, and the bacterial capability to survive within different environmental conditions43. Fortunately, low frequency of L. monocytogenes was found in ready to eat food represented by cheese and milk compared to chicken and red meat. Although cooking is required for both meat types, dealing with contaminated meat can lead to listeriosis through handling and may cross contaminate other food sources as well44.
However 647 human clinical samples (Blood, Urine, and Vaginal swabs) were tested for L. monocytogene. This bacterium was detected from 7/647 (1.1%) samples. Out of 200 vaginal swabs collected from pregnant women only three samples (1.5%) were positive for L. monocyto-genes namely among women with history of at least one miscarriage. Whereas, only one (0.5%) L. monocytogenes was isolated from 200 urine samples. The positive sample from patients also had history of stillbirth delivery. The age’s distributions of these patients were between 18-38 years old. In addition to that, three samples (1.2%) were positive out of 247 tested immunocompromised patients for Listeriosis, which were diagnosed in their blood samples. Two of the patients were of age >70 years, who suffered from renal failure and experienced hemodialysis for a long time, the third positive case was a 3 years old and diagnosed as meningitis case. No significant relationship between clinical case and L. monocytogenes infection have been recognized (P value=0.21) (Fig. 1). These results are in agreement with previous investigation in human clinical samples that reported L. monocytogenes by 1.3% from vaginal swab30,45. The lower and higher infection rate were reported in India from the samples taken from vaginal swabs at 0.8% and 10.28%, respectively46,47. Generally, pregnant women were found more sensitive to Listeriosis than other people by 17 fold48. This may lead to spontaneous abortions, premature births and stillbirths. In agreement with our results48 reported 1.0%. Fewer than our outcomes, only 0.3% of clinical samples were found to be infected with L. monocytogenes in a study conducted by49. Recently, Al-dorri, 2018 reported 37.93% of positive cases in a study conducted in Tikrit province-Iraq which dis-consistent with the present study results50. The differences between the data of the present study and that reported by47,50 may be due to the time when the samples were collected from the pregnant women (pregnancy trimesters), variations in the samples size involved and the methods used for the bacterial isolation, the type of the samples used, personal hygiene and diet.
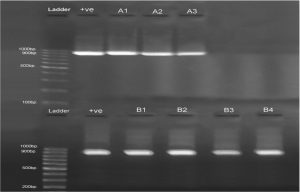
Electrophoresis was performed on 1.2% agarose gel at 85V for 1hour. Lane 1 is 100bp ladder, Lane 2 (+ve) positive control of 16SrRNA (938bp). Whereas A1 (Vaginal swab), A2 (Urine), and A3 (Blood) are positive samples isolated from human clinical sources, and B1(Frozen chicken meat), B2 (Fresh red meat), B3 (Milk), B4(White soft cheese) are positive samples detected from food sources.
Antibiotics sensitivity
L. monocytogenes isolated from the meat products, dairy products and human clinical samples were examined for their antibiotic sensitivity. Majority of the isolates, from food samples were resistant to ampicillin (65.85%), Cephalexin (65.85%), Penicillin (63.41%) and Doxycycline (60.97%), while, variable resistances have been noticed to Gentamicin (56.09%), Clindamycin (53.65%), Rifampin (51.21%), Chloramphenicol (41.46%) and Co-trimoxazol (39.02%). However, these isolates were mostly sensitive to Piperacillin (75.60%), Meropenem (73.17%) and Ciprofloxacin (70.73%). On the other side, the majority of human clinical isolates were resistant to Clindamycin (71.42%) and Doxycycline (71.42%), while some human L.monocytogenes isolates exhibited multi-drug resistance (MDR) to the Ampicillin, Cephalexin, Chloramphenicol, Gentamicin, Penicillin and Rifampin. These isolates were completely sensitive to Co-trimoxazol (85.71%), Meropenem (85.71%), Ciprofloxacin (71.42%) and Piperacillin (71.42%) (Table 2). Penicillin, ampicillin, gentamycin, rifampin, chloramphenicol, tetracycline, and co-trimoxazol were used widely in human and veterinary Listeriosis treatments25,47. Our results are supported by many studies conducted elsewhere25,47. In one study in Iraq, 100% of isolates were resistance to clindamycin and 100% of isolates were sensitive to gentamycin and Imipenem which were similar to our results to some extent23. Contrary, Osaili, Kalekar, Nצlkes, Jemal and their collegues reported lower rate of resistance of L. monocytogenes strain against the previous antibiotics51-53. According to the currently accepted standards if the bacterium resistant to three or more antibiotics of different classes this will be considered as a multi-drug resistant bacterium54. Multi-drug L. monocytogenes strains has been reported in different countries55,56. In the present study, 45/48(93.75%) strains were resistance to at least 4 antibiotics. In agreement with our findings, multidrug-resistant L. monocytogenes strains were reported in many clinical cases which indicated an extensive health considerations25,53,57. In general the antibiotics resistant could be due to the extensive and uncontrolled use of antibiotics for human and veterinary treatment58, as ampicillin or penicillin G combined with an aminoglycoside (gentamicin) is considered the standard and the first choice for listeriosis treatment. While, the second line of treatment of this microorganism is the co-trimoxazol59. The other possible reasons for antibiotics resistant are the acquisition of antibiotic resistant genes by insertion elements and integrons60. Thus, the outcome of current study revealed prevalence of multi-drug resistant isolates of L. monocytogenes in meat, human clinical and milk samples. The results also emphasize the necessity for active and continuous investigation of their antibiotic resistance.
Table (2):
The sensitivity of different L. monocytogenes isolates to different types of antibiotics.
| Antibiotics | Isolates from different sources | |||||
|---|---|---|---|---|---|---|
| Human | Food | |||||
| S | I | R | S | I | R | |
| Ampicillin | 3 | 0 | 4 | 14 | 0 | 27 |
| Cephalexin | 2 | 1 | 4 | 11 | 3 | 27 |
| Chloramphenicol | 3 | 0 | 4 | 24 | 0 | 17 |
| Ciprofloxacin | 5 | 1 | 1 | 29 | 4 | 8 |
| Clindamycin | 1 | 1 | 5 | 18 | 1 | 22 |
| Doxycycline | 2 | 0 | 5 | 11 | 5 | 25 |
| Gentamycin | 3 | 1 | 3 | 16 | 2 | 23 |
| Meropenem | 6 | 0 | 1 | 30 | 2 | 9 |
| Penicillin | 3 | 0 | 4 | 15 | 0 | 26 |
| Piperacillin | 5 | 1 | 1 | 31 | 3 | 7 |
| Rifampin | 2 | 1 | 4 | 18 | 2 | 21 |
| Co-trimoxazol | 6 | 0 | 1 | 23 | 2 | 16 |
| Vancomycin | 3 | – | 4 | 12 | 7 | 21 |
| Total | 44 | 6 | 41 | 252 | 31 | 248 |
S= sensitive I=intermediate R=Resistant
Serotypes identification
Due to the importance of serotyping in determining the sporadic and epidemic strains of L. monocytogenes, we dedicated a major part of our study for that purpose. The method used by Doumith et al. (2004) was successfull in separating our 48 strains of L. monocytogenes into three distinguished groups based on specific multiplex PCR11. All food and human isolates gave positive results for species specific gene for L. monocytogenes (16sRNA). All human isolates (7) belonged to serogroup 1/2a (or 3a) which is known to be more prevalent in food and food related environments44. However, three different serogroups were found in food samples, sixteen frozen chicken meat, eight fresh red meat, three raw milk samples and one white soft cheese isolates belonged to serogroup 1/2a or 3a. The second serogroup was (4b) in which 6 frozen chicken and 2 fresh meat isolates were found to be in this serogroup. Five fresh red meat isolates belonged to third group, 1/2b (Table 3). All our strains belonged to serotypes (1/2a, 1/2b, and 4b) which are mostly associated with human listeriosis such as61. Serotyping profile of human isolates, revealed that about 98% of the strains diagnosed among patients and food samples belonged to serotype 1/2a, 1/2b, 1/2c and 4b12. These data are in contrast to the other study reported elsewhere that found most food born listerial strains belonging to serogroup 4d (or 4b, 4e) and particularly 4b62. These results show the necessity of proper handling to prevent outbreaks of listeriosis in Kurdistan. Also, routine sampling from supermarkets and butcher stores is recommended as this bacterium can survive for long periods if favorable temperature and nutrients are available61.
Table (3):
shows the serotypes and virulence genes distribution among food and human clinical isolates of L. Monocytogenes.
| Virulence associated genes | Serotypes | Sample Source | Date of Isolation | Code | |||
|---|---|---|---|---|---|---|---|
| Iap | plcA | actA | hlyA | ||||
| + | + | + | + | 1/2a (or 3a) | White soft cheese | 15- Oct -2016 | 1123 |
| + | + | + | + | 1/2a (or 3a) | Raw milk | 02-Nov -2016 | 6594 |
| + | + | + | + | 1/2a (or 3a) | Raw milk | 29-Apr -2017 | 1016 |
| + | + | + | + | 1/2a (or 3a) | Raw milk | 10-May -2017 | 506 |
| + | + | + | + | 1/2a (or 3a) | Frozen chicken meat | 30-Jul- 2016 | 7505 |
| + | + | + | + | 1/2a (or 3a) | Fresh red meat | 02-Aug -2016 | 1300 |
| + | + | + | + | 4b(or 4d,4e) | Frozen chicken meat | 04-Aug – 2016 | 8018 |
| + | + | + | + | 1/2a (or 3a) | Fresh red meat | 06-Aug – 2016 | 8211 |
| + | + | + | + | 1/2a (or 3a) | Frozen chicken meat | 08-Aug -2016 | 8436 |
| + | + | + | + | 4b(or 4d,4e) | Frozen chicken meat | 08-Aug -2016 | 8308 |
| + | + | + | + | 4b(or 4d,4e) | Frozen chicken meat | 09-Aug -2016 | 8616 |
| + | + | + | + | 1/2a (or 3a) | Frozen chicken meat | 10-Aug -2016 | 8734 |
| + | + | + | + | 1/2a (or 3a) | Frozen chicken meat | 10-Aug -2016 | 8731 |
| + | + | + | + | 1/2a (or 3a) | Frozen chicken meat | 10-Aug -2016 | 8740 |
| + | + | + | + | 1/2a (or 3a) | Frozen chicken meat | 10-Aug -2016 | 8730 |
| + | + | + | + | 1/2a (or 3a) | Frozen chicken meat | 13-Aug -2016 | 8910 |
| + | + | + | + | 1/2b (or 3b) | Frozen chicken meat | 15-Aug -2016 | 9106 |
| + | + | + | + | 1/2b (or 3b) | Frozen chicken meat | 15-Aug -2016 | 9102 |
| + | + | + | + | 1/2b (or 3b) | Frozen chicken meat | 15-Aug -2016 | 9132 |
| + | + | + | + | 4b(or 4d,4e) | Fresh red meat | 17-Aug -2016 | 9301 |
| + | + | + | + | 1/2a (or 3a) | Frozen chicken meat | 17-Aug -2016 | 9319 |
| + | + | + | + | 1/2a (or 3a) | Frozen chicken meat | 18-Aug -2016 | 9405 |
| + | + | + | + | 4b(or 4d,4e) | Frozen chicken meat | 18-Aug -2016 | 9408 |
| + | + | + | + | 1/2a (or 3a) | Frozen chicken meat | 20-Aug -2016 | 9608 |
| + | + | + | + | 1/2b (or 3b) | Frozen chicken meat | 21-Aug -2016 | 9714 |
| + | + | + | + | 1/2a (or 3a)) | Fresh red meat | 22-Aug -2016 | 5.00 |
| + | + | + | + | 4b(or 4d,4e) | Fresh red meat | 22-Aug -2016 | 17.00 |
| + | + | + | + | 4b(or 4d,4e) | Frozen chicken meat | 23-Aug -2016 | 9920 |
| + | + | + | + | 1/2b (or 3b) | Frozen chicken meat | 23-Aug -2016 | 9910 |
| + | + | + | + | 1/2a (or 3a) | Frozen chicken meat | 23-Aug -2016 | 9905 |
| + | + | + | + | 4b(or 4d,4e) | Frozen chicken meat | 27-Aug -2016 | 304 |
| + | + | + | + | 1/2a (or 3a) | Frozen chicken meat | 27-Aug -2016 | 312 |
| + | + | + | + | 1/2a (or 3a) | Fresh red meat | 21-Sep -2016 | 16.00 |
| + | + | + | + | 1/2a (or 3a) | Fresh red meat | 26-Sep -2016 | 3333 |
| + | + | + | + | 1/2a (or 3a) | Fresh red meat | 10-Oct -2016 | 406 |
| + | + | + | + | 1/2a (or 3a) | Frozen chicken meat | 5-Oct -2016 | 40 |
| + | + | + | + | 1/2a (or 3a) | Fresh red meat | 5-Oct -2016 | 25 |
| + | + | + | + | 1/2a (or 3a) | Fresh red meat | 5-Oct -2016 | 41 |
| + | + | + | + | 1/2a (or 3a) | Frozen chicken meat | 15-Oct -2016 | 5213 |
| + | + | + | + | 1/2a (or 3a) | Frozen chicken meat | 15-Oct -2016 | 5208 |
| + | + | + | + | 1/2a (or 3a) | Frozen chicken meat | 18-Oct -2016 | 5534 |
| + | + | + | + | 1/2a(or 3a) | Vaginal swab | 10-Nov-2016 | Human |
| + | + | + | + | 1/2a(or 3a) | Vaginal swab | 15-Nov-2016 | Human |
| + | + | + | + | 1/2a(or 3a) | Urine | 07-Dec-2016 | Human |
| + | + | + | + | 1/2a(or 3a) | Vaginal swab | 09-Jan-2017 | Human |
| + | + | + | + | 1/2a(or 3a) | Blood | 11-Feb-2017 | Human |
| + | + | + | + | 1/2a(or 3a) | Blood | 14-Apr-2017 | Human |
| + | + | + | + | 1/2a(or 3a) | Blood | 16-Apr-2017 | Human |
Detection of Virulence genes
Strains of L. monocytogenes vary in their pathogenicity according to the number of virulence genes. Potential correlation between hlyA, plcA, actA and iap virulence associated genes from L. monocytogenes and their pathogenicity was detected63. Virulent strains have been found to produce more phagosomal membrane disruptors particularly hlyA and plcA as compared to non-virulent strains64. Unexpectedly, all L. monocytogenes strains isolates from both food and human samples were positive towards four virulence genes tested (iap, hlyA, ActA and plcA), (Table 3). Three out of 4 milk samples isolated in a study from India were found to be positive towards all virulence genes tested which might also refer to human source contamination46. Such results were found only in pathogenic strains isolated from human samples and not from food samples55.
Sequencing of 16rRNA and phylogeny analysis
Out of 48 isolated strains from food and human samples, 15 isolates were selected for sequencing, analyzed and examined for detection of any genetic diversity within the isolated samples and compared with the data base isolates. All isolated samples exhibited amplification of 16sRNA up to expected size 1600bp. All food and human sequences were blasted (http://blast.ncbi.nlm.nih.gov/Blast.cgi) and showed a sequence identity of 98-100% for the 16S rRNA gene, based on the sequence similarity, more than 90 isolates from the data base were found to be strictly related to our L. monocytogenes isolates. These isolates were mainly from soil, vegetables, milk, water and human. However, genetic diversities were found within the isolated samples of food and human as show in (Fig. 2). Phylogenetic analysis of our results divided our isolates in to three different groups. On the basis of phylogeny analysis of 16SrRNA, it is found that there is a genetic relationship between human clinical isolates; the same pattern was determined between the foods isolates as a second group, while the third group which was of milk isolates was genetically further away and grouped out from both human and food samples. Comparing with the databases isolates, it is clear that the Listeria monocytogenes isolated from fresh red meat of this study is closely related to the strains isolated from prepared meat products in South Africa and to that isolated from patient in USA. L. monocytogenes isolates from different sources, food and clinical samples showed to be identical with the isolates from the milk product recorded in India and sludge, waste water reported in France65.
Taken together, the data in the present article confirmed that the virulent strains of L. monocytogenes are widely distributed within animals’ food products with a high incidence of the bacterial infection among the human population in Duhok province. Authorities should be notified to take their role in maintaining and controlling the further spreading of the diseases, strict hygienic measurements should be applied to control entrance of the contaminated food to the area. The current study confirmed the existence of potential virulent strains of L. monocytogenes in food and human clinical samples, and the study stated that the prevalence rate of L. monocytogenes in this study was higher in food samples compared with human samples. The prevalence rate of multi-drug resistant isolates of L. monocytogenes in meat, human clinical and milk samples, and highlighted the necessity for active and continuous investigation of their antibiotic resistance.
The most serotypes found in our isolates were 1/2a (or 3a); also, the molecular serotyping is irreplaceable for better understanding the routes of L. monocytogenes dissemination and the origin of the contamination. In term of antibiotic susceptibility profile, the study determined that the majority of our isolates from both food and human were resistant to the most antibiotics tested in this study. Furthermore, milk isolates were found to be genetically diverse from food and human isolates.
ACKNOWLEDGMENTS
This study was granted by the University of Duhok. The authors thank all member of Directorate of Prevention Affairs/ Foodstuff Analysis Laboratory, Duhok Obstetrics and Gynecology Hospital, Azadi Teaching Hospital, Hevi Pediatric Hospital, Hemodialysis Center in Duhok city and Central Public Health Laboratory for their help whenever needed.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
AMTA and IMAM designed the study, collected the data, performed the study, analyzed the data, written the manuscript.
FUNDING
The study is funded by the University of Duhok. Grant No. 10052017-4.
ETHICS STATEMENT
All procedures of current study were approved by the Local Ethics Committee of College of Medicine, University of Duhok, Kurdistan Region, Iraq and Duhok Directorate General of Health (Reference No.100520174).
AVAILABILITY OF DATA
All datasets generated or analyzed during this study are included in the manuscript.
- Mead, P.S.; Slutsker, L.; Dietz, V.; McCaig, L.F.; Bresee, J.S.; Shapiro, C.; Griffin, P.M.; Tauxe, R.V. Food-Related Illness and Death in the United States. Journal of Environmental Health, 2000; 62(7): 9.
- Ramaswamy, V.; Cresence, V.M.; Rejitha, J.S.; Lekshmi, M.U.; Dharsana, K.S.; Prasad, S.P.; Vijila, H.M. Listeria-Review of Epidemiology and Pathogenesis. Journal of Microbiology Immunology and Infection, 2007; 40(1):4.
- Janakiraman, V. Listeriosis in Pregnancy: Diagnosis, Treatment, and Prevention. Reviews in obstetrics & gynecology, 2008; 1(4): 179-185.
- Gregory, S.H.; Liu, C. CD8+ T-Cell-Mediated Response to Listeria Monocytogenes Taken up in the Liver and Replicating within Hepatocytes. Immunological Reviews, 2000; 174: 112-122.
- Fagerlund, A.; Langsrud, S.; Schirmer, B.C.T.; Maretin, T.; Heir, E. Genome Analysis of Listeria monocytogenes Sequence Type 8 Strains Persisting in Salmon and Poultry Processing Environments and Comparison with Related Strains. PLoS ONE, 2016; 11(3): e0151117.
Crossref - McLauchlin, J.; Mitchell, Rt.; Smerdon, W.J.; Jewell, K. Listeria Monocytogenes and Listeriosis: A Review of Hazard Characterisation for Use in Microbiological Risk Assessment of Foods. International Journal of Food Microbiology, 2004; 92(1): 15-33.
Crossref - Lunden, J.; Tolvanen, R.; Korkeala, H. Human Listeriosis Outbreaks Linked to Dairy Products in Europe. Journal of Dairy Science, 2004; 87(Table 1): E6-E12.
Crossref - Swaminathan, B.; Gerner-Smidt, P. The Epidemiology of Human Listeriosis. Microbes and Infection, 2007; 9(10): 1236-1243.
- Rawool, D.B.; Malik, S.V.S.; Shakuntala, I.; Sahare, A.M.; Barbuddhe, S.B. Detection of Multiple Virulence-Associated Genes in Listeria Monocytogenes Isolated from Bovine Mastitis Cases. International Journal of Food Microbiology, 2007; 113(2): 201-207.
Crossref - Portnoy, D.A.; Chakraborty, T.; Goebel, W.; Cossart, P. Molecular Determinants of Listeria Monocytogenes Pathogenesis. Infection and Immunity, 1992; 60(4): 1263-1267.
- Doumith, M.; Buchrieser, C.; Glaser, P.; Jacquet, C.; Martin, P. Differentiation of the Major Listeria Monocytogenes Serovars by Multiplex PCR. Journal of Clinical Microbiology, 2004; 42(8): 3819-3822.
Crossref - Liu, D. Handbook of Listeria Monocytogenes, 1st Editio.; CRC press: Boca Raton, 2008.
Crossref - Lee, S.; Ward, T.J.; Graves, L.M.; Wolf, L.A.; Sperry, K.; Siletzky, R.M.; Kathariou, S. Atypical Listeria Monocytogenes Serotype 4b Strains Harboring a Lineage II-Specific Gene Cassette. Applied and Environmental Microbiology, 2012; 78(3): 660-667.
Crossref - Perez-Trallero, E.; Zigorraga, C.; Artieda, J.; Alkorta, M.; Marimon, J.M. Two Outbreaks of Listeria monocytogenes Infection, Northern Spain. Emerging Infectious Diseases, 2014; 20(12): 2155-2157.
Crossref - Camargo, A.C.; Woodward, J.J.; Nero, L.A. The Continuous Challenge of Characterizing the Foodborne Pathogen Listeria Monocytogenes. Foodborne Pathogens and Disease, 2016; 13(8): 405-416.
Crossref - Henriques-Normark, B.; Normark, S. Commensal Pathogens, with a Focus on Streptococcus Pneumoniae, and Interactions with the Human Host. Experimental Cell Research. Elsevier, 2010; 316(8): 1408-1414.
Crossref - Said S, Said T, Tayeb BA, et al. Safety & Hygiene Isolation and Molecular Detection of Listeria monocytogenes in Minced Meat, Frozen Chicken and Cheese in Duhok Province, Kurdistan Region of Iraq, Journal of Food: Microbiology, 2017; 2(1): 10-13. doi:10.4172/2476-2059.1000118
- Alzubaidy, Z.M.; Kakey, S.I.; Ali, J.F. Isolation and Identification of Listeria Moncytogenes by PCR from Some Food Sources in Erbil City. Journal of Agriculture Science, 2013; 5(3): 14-26.
- Aznar, R.; Solis, I. PCR Detection of Listeria monocytogenes in Different Food Products Compared with the Mini-VIDAS LMO System and the Standard Procedure ISO 11290-1. Journal fur Verbraucherschutz und Lebensmittelsicherheit, 2006; 1(2): 115-120.
Crossref - Dominguez Rodriguez, L.; Vazquez Boland, J.A.; Fernandez Garayzabal, J.F.; Echalecu Tranchant, P.; Gomez-Lucia, E.; Rodriguez Ferri, E.F.; Suarez Fernandez, G. Microplate Technique to Determine Hemolytic Activity for Routine Typing of Listeria Strains. Journal of Clinical Microbiology, 1986; 24(1): 99-103.
- Adzitey, F.; Rahmat Ali, G.R.; Huda, N.; Cogan, T.; Corry, J. Prevalence, Antibiotic Resistance and Genetic Diversity of Listeria Monocytogenes Isolated from Ducks, Their Rearing and Processing Environments in Penang, Malaysia. Food Control, 2013; 32(2): 607-614.
Crossref - Park, S.; Jung, H.; Lee, M.; Choi, H.; Kim, J.; Jung, J.; Park, S.; Kim, M.; Kim, K.; Oh, Y.; et al. Detection of Listeria Monocytogenes in Foods and Characterization by PFGE. Advances in Microbiology, 2016; 06(04): 343-349.
Crossref - Ahmed, M.S.; Taha, Z.M.A.; Omer, L.T. Isolation and Molecular Identification with Resistant Profile Determination of Listeria Monocytogenes from Imported Chicken Carcasses in Duhok, Kurdistan Region, Iraq. Journal of Pure and Applied Microbiology, 2015; 9(Special Edition 1): 97-103.
- Lyon, S.A.; Berrang, M.E.; Fedorka-Cray, P.J.; Fletcher, D.L.; Meinersmann, R.J. Antimicrobial Resistance of Listeria Monocytogenes Isolated from a Poultry Further Processing Plant. Foodborne Pathogens and Disease, 2008; 5(3): 253-259.
Crossref - Nwachukwu, N.C., Orji, F.A., Iheukwumere, I. and Ekeleme, U.G. Antibiotic Resistant Environmental Isolates of Listeria Monocyto-genes from Anthropogenic Lakes in Lokpa-Ukwu, Abia State of Nigeria. Australian Journal of Basic and Applied Sciences, 2010; 4(7): 1571-1576.
- B. Patel, J. dkk. Performance Standards for Antimicrobial Susceptibility Testing CLSI Supplement M100S.; 2016; 29-50.
- Liu D, Lawrence ML, Austin FW, Ainsworth AJ. A multiplex PCR for species-and virulence-specific determination of Listeria Monocytogenes. Journal of Microbiological Methods, 2007; 71(2): 133-40.
- Manzano, M.; Cocolin, L.; Cantoni, C.; Comi, G. Temperature Gradient Gel Electrophoresis of the Amplified Product of a Small 16S RRNA Gene Fragment for the Identification of Listeria Species Isolated from Food. Journal of food protection, 2000; 63(5): 659-661.
Crossref - Harris, J.B.; Grubb, B.D.; Maltin, C.A.; Dixon, R. Diversity of Bacteria Associated with the Caribbean Coral Montastraea Franksi. Coral Reefs, 2001; 20(1): 85-91.
Crossref - El-Malek, A.M.A.; Ali, S.F.H.; Hassanein, R.; Mohamed, M.A.; Elsayh, K.I. Occurrence of Listeria Species in Meat, Chicken Products and Human Stools in Assiut City, Egypt with PCR Use for Rapid Identification of Listeria Monocyto-genes. Veterinary World, 2010; 3(8): 353-359.
- Elmali, M.; CAN, H.Y.; Yaman, H. Prevalence of Listeria Monocytogenes in Poultry Meat. Food Science and Technology, 2015; 35(4): 672-675.
- Fallah, A.A.; Saei-Dehkordi, S.S.; Rahnama, M.; Tahmasby, H.; Mahzounieh, M. Prevalence and Antimicrobial Resistance Patterns of Listeria Species Isolated from Poultry Products Marketed in Iran. Food Control, 2012; 28(2): 327-332.
- Heo, E.J.; Song, B.R.; Park, H.J.; Kim, Y.J.; Moon, J. S.; Wee, S.H.; Kim, J.-S.; Yoon, Y. Rapid Detection of Listeria Monocytogenes by Real-Time PCR in Processed Meat and Dairy Products. Journal of Food Protection, 2014; 77(3): 453-458.
Crossref - Granier, S.A.; Moubareck, C.; Colaneri, C.; Lemire, A.; Roussel, S.; Dao, T.T.; Courvalin, P.; Brisabois, A. Antimicrobial Resistance of Listeria Monocytogenes Isolates from Food and the Environment in France over a 10-Year Period. Applied and Environmental Microbiology, 2011; 77(8): 2788-2790.
Crossref - Aygun, O.; Pehlivanlar, S. Listeria Spp. in the Raw Milk and Dairy Products in Antakya, Turkey. Food Control, 2006; 17(8): 676-679.
Crossref - Gaya, P.; Saralegui, C.; Medina, M.; Nunez, M. Occurrence of Listeria monocytogenes and Other Listeria Spp. in Raw Caprine Milk. Journal of Dairy Science, 2010; 79(11): 1936-1941.
Crossref - Barbuddhe, S.B.; Chaudhari, S.P.; Malik, S.V.S. The Occurrence of Pathogenic Listeria Monocyto-genes and Antibodies against Listeriolysin?O in Buffaloes. Journal of Veterinary Medicine, Series B, 2002; 49(4): 181-184.
- Nayak, J.B.; Brahmbhatt, M.N.; Savalia, C.V.; Bhong, C.D.; Roy, A.; Kalyani, I.H.; Parmar, B.C. Detection and Characterization of Listeria Species from Buffalo Meat. Buffalo Bulletin, 2010; 29(2): 83-87.
- Meloni, D. Focusing on the Main Morphological and Physiological Characteristics of the Food-Borne Pathogen Listeria Monocytogenes. Journal of Veterinary Science and Research, 2014; 1(1:001): 1-2.
- Glass, K.A.; Doyle, M.P. Fate of Listeria Monocytogenes in Processed Meat Products during Refrigerated Storage. Applied and Environmental Microbiology, 1989; 55(6): 1565-1569.
- Brito, J.R.F.; Santos, E.M.P.; Arcuri, E.F.; Lange, C.C.; Brito, M.A.V.P.; Souza, G.N.; Cerqueira, M.M.P.O.; Beitran, J.M.S.; Call, J.E.; Liu, Y.; et al. Retail Survey of Brazilian Milk and Minas Frescal Cheese and a Contaminated Dairy Plant to Establish Prevalence, Relatedness, and Sources of Listeria Monocytogenes Isolates. Applied and Environmental Microbiology, 2008; 74(15): 4954-4961.
Crossref - Kells, J.; Gilmour, A. Incidence of Listeria Monocytogenes in Two Milk Processing Environments, and Assessment of Listeria Monocytogenes Blood Agar for Isolation. International Journal of Food Microbiology, 2004; 91(2): 167-174.
Crossref - Gilbert, R.J.; Mclauchlin, J.; Velani, S.K. The Contamination of Pate by Listeria monocytogenes in England and Wales in 1989 and 1990. Epidemiology and Infection, 1993; 110(3):543-551.
Crossref - Cordano, A.M.; Rocourt, J. Occurrence of Listeria Monocytogenes in Food in Chile. International Journal of Food Microbiology, 2001; 70(1-2): 175-178.
- Wang, X.M.; L , X. F.; Yin, L.; Liu, H.F.; Zhang, W.J.; Si, W.; Yu, S.Y.; Shao, M.L.; Liu, S.G. Occurrence and Antimicrobial Susceptibility of Listeria Monocyto-genes Isolates from Retail Raw Foods. Food Control, 2013; 32(1): 153-158.
Crossref - Soni, D.K.; Singh, D.V.; Dubey, S.K. Pregnancy – Associated Human Listeriosis: Virulence and Genotypic Analysis of Listeria Monocytogenes from Clinical Samples. Journal of Microbiology, 2015; 53(9): 653-660.
Crossref - Zuraini, M.I.; Elexson, N.; Son, R.; Marian, M.N.; Wong, W.C.; Maimunah, M.; Lee, H. Y.; Sharifah Aminah, S.M. MPN-PCR Detection and Antimicrobial Resistance of Listeria Monocyto-genes Isolated from Raw and Ready-to-Eat Foods in Malaysia. Food Control, 2012; 28(2): 309-314.
Crossref - Kaur, S.; Malik, S.V.S.; Vaidya, V.M.; Barbuddhe, S.B. Listeria Monocytogenes in Spontaneous Abortions in Humans and Its Detection by Multiplex PCR. Journal of Applied Microbiology, 2007; 103(5): 1889-1896.
Crossref - Dhanashree, B.; Otta, S.K.; Karunasagar, I.; Goebel, W. Incidence of Listeria Spp. in Clinical and Food Samples in Mangalore, India. Food Microbiology, 2003; 20(4): 447-453.
Crossref - Al-dorri, A. Study of Bacteria Listeria Monocytogenes in Spontaneous Aborted Women in Salah Al-Deen Province. Tikrit Journal of Pure, 2018; 21(1): 12-17.
- Osaili, T.M.; Al-Nabulsi, A.A.; Taha, M.H.; Al-Holy, M.A.; Alaboudi, A.R.; Al-Rousan, W.M.; Shaker, R.R. Occurrence and Antimicrobial Susceptibility of Listeria Monocytogenes Isolated from Brined White Cheese in Jordan. Journal of Food Science, 2012; 77(9): 528-532.
Crossref - Kalekar, S.; Doijad, S.; Poharkar, K.V; Rodriguez, S.; Kalorey, D.R.; Kurkure, N.V; Rawool, D.B.; D’Costa, D.; Bhosle, S.; Barbuddhe, S.B. Characterization of Listeria Monocytogenes Isolated from Human Clinical Cases. International Journal of Medical and Health Sciences, 2015; 4(2): 206-212.
- Nilkes, D.; Jemal, T. Prevalence and Antibiotic Susceptibility of Listeria Species in Raw Milk and Dairy Products from North Shewa Zone, Oromia Regional State, Ethiopia. 2014. http://institutional_repository.haramaya.edu.et/jspui/handle/123456789/1139. Accessed August 5, 2019.
- Exner, M.; Bhattacharya, S.; Christiansen, B.; Gebel, J.; Goroncy-Bermes, P.; Hartemann, P.; Heeg, P.; Ilschner, C.; Kramer, A.; Larson, E.; et al. Antibiotic Resistance: What Is so Special about Multidrug-Resistant Gram-Negative Bacteria? GMS Hygiene and Infection Control, 2017; 12: Doc05.
Crossref - Soni, D.K.; Singh, R.K.; Singh, D.V.; Dubey, S.K. Characterization of Listeria Monocytogenes Isolated from Ganges Water, Human Clinical and Milk Samples at Varanasi, India. Infection, Genetics and Evolution, 2013; 14(1): 83-91.
Crossref - Saran Tirumalai, P.; Tirumalai, P.S. Listeriosis and Listeria monocytogenes in India Increasing Trend of Infectious Diseases,. Wudpecker Journal of Food Technology, 2013; 1(September): 98-103.
- Abbas, B.A.; Jaber, G.M. Occurrence of Listeria Monocytogens in Raw Milk of Ruminants in Basrah Province. Iraqi Journal of Veterinary Sciences, 2012; 26(1): 47-51.
- Poros Gluchowska, J.; Markiewicz, Z. Antimicrobial Resistance of Listeria monocytogenes. Acta Microbiologica Polonica, 2003; 52(2): 113-129.
- Charpentier, E.; Courvalin, P. Antibiotic Resistance in Listeria Spp. Antimicrobial agents and chemotherapy, 1999; 43(9): 2103-2108.
- Mazel, D.; Dychinco, B.; Webb, V.A.; Davies, J. Antibiotic Resistance in the ECOR Collection: Integrons and Identification of a Novel Aad Gene. Antimicrobial Agents and Chemotherapy, 2000; 44(6): 1568-1574.
Crossref - Farber, J.M.; Peterkin, P.I. Listeria Monocyto-genes, a Food-Borne Pathogen. Microbiological Reviews, 1991; 55(3): 476-511.
- Hofer, E.; Falavina Dos Reis, C.M.; Hofer, C.B. Serovars of Listeria Monocytogenes and Related Species Isolated from Human Clinical Specimens. Revista da Sociedade Brasileira de Medicina Tropical, 2006; 39(1): 32-37.
Crossref - Liu, D.; Lawrence, M.L.; Austin, F.W.; Ainsworth, A.J.A Multiplex PCR for Species- and Virulence-Specific Determination of Listeria Monocyto-genes. Journal of Microbiological Methods, 2007; 71(2): 133-140.
Crossref - Vazquez-Boland, J.A.; Kuhn, M.; Berche, P.; Chakraborty, T.; Dominguez-Bernal, G.; Goebel, W.; Gonzalez-Zorn, B.; Wehland, J.; Kreft, J. Listeria Pathogenesis and Molecular Virulence Determinants. Clinical Microbiology Reviews, 2001; 14(3): 584-640.
Crossref - Soni, D.K.; Dubey, S.K. Phylogenetic Analysis of the Listeria Monocytogenes Based on Sequencing of 16S RRNA and HlyA Genes. Molecular Biology Reports, 2014; 41(12): 8219-8229.
Crossref
© The Author(s) 2019. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.