The depletion of fossil fuel for energy production is one of the major problems being faced worldwide. As an alternative to fossil fuels, first and second generation biofuel was developed from corn, grains and lignocellulosic agricultural residues. These generations are inefficient in achieving the desired rate of biofuel production, climate change mitigation and economic growth. Therefore, third generation biofuel specifically derived from microalgae have proved to be a promising unconventional energy source. Microalgae are microscopic organisms that grow in salt or fresh water and have been used for producing metabolites, cosmetics and for energy production. The conventional approaches used for biofuel production include pyrolysis, gasification, direct combustion and thermomechanical liquefaction. The search for biological and eco-friendly approaches led to the emergence of Microbial Fuel Cell (MFC), which provide a new solution to energy crisis. Integration of photosynthetic organisms such as microalgae into MFC resulted in a new approach i.e. Microbial Solar Cell, which can convert solar energy into electrical energy via photosynthesis. Microbial solar cells have broad range application in wastewater treatment, biodiesel processing and intermediate metabolite production.
Microalgae, biofuel, microbial fuel cell, microbial solar cell, biomass conversion
Today’s world is facing many environmental problems; energy crisis is one of the major issues being faced globally. Population growth and fast industrialization has led to the overexploitation and depletion of non-renewable fossil fuels [1]. Thus, the scarcity of fossil fuels can be compensated by utilizing renewable energy sources such as solar, wind, hydro, tidal, and biomass [2,3]. International Energy Agency (IEA) compared the potential of renewable sources for energy production and reported wastes and combustible sources to be the most promising alternative. Biomass derived from terrestrial and aquatic sources have been exploited for biofuel production [4]. Biofuel production is associated with three generations, namely 1st, 2nd and 3rd generation. First and second generation biofuel were derived from the agricultural sources. These generations utilize food sources, slow processes and have low production capacity which limits its pilot scale use. Therefore, third generation biofuel derived from algal biomass are the best alternative owing to their rapid and higher production as compared to first and second generation biofuels. Unsustainable consumption resulted in scarcity of biomass, reduction of green cover and biodiversity [5,6].
So, nowadays bioenergy research paradigm has shifted towards algal biomass for production of biofuels. Microalgae are photosynthetic microbes which require sunlight, carbon dioxide and inorganic nutrients for growth. It produces huge amount of biomolecules like fatty acids and sugars, which can be converted into biofuels (bio-oil, bioethanol, biodiesel etc.) [7,8]. Microalgal biomass increases the process efficiency due to its low hemicellulosic and neglible lignin content [5]. Microalgae have wide range of application in the field of nutrition, environmental pollution, fertilizers and animal feed [9,10]. Biofuel production has been possible with the various conventional (chemical, physical and biological) and emerging (MFC, MSC) methods. The conventional methods have been used to produce biofuel but their requirement for energy, low efficiency and complex process limited their use. With the development in technology, new approaches for biofuel production were designed and used.
Microalgae
Algae are the oil-rich organisms which can efficiently use sunlight, remove contaminants from environment without competing for food or agricultural resources [11,12,13]. Their short generation time enables the rapid biomass recovery. The oil present in algae can be processed into biofuel [14]. Algae consist of macro and micro-algae. Macro-algae also known as “seaweeds” are multicellular rapidly proliferating plants inhabiting in saline or fresh water [15]. On the basis of pigmentation they have been classified into three subgroups: i) brown seaweed (Phaeophyceae); ii) red seaweed (Rhodophyceae) and iii) green seaweed (Chlorophyceae). They consist of a thallus and lack roots, stems and leaves. The algae cultivation for food production and the hydrocolloid extraction on commercial scale in Asia has been practiced since many years. The most cultivated macroalgae include brown algae Laminaria japonica and Undaria pinnatifida, the red algae Porphyra, Eucheum and Gracilaria, and the green algae Monostroma and Enteromorpha [16].
Micro-algae are very small in size and inhabit in both marine and freshwater. Their photosynthetic ability allows them to efficiently convert solar energy into biomass. On the basis of pigmentation, chemical nature, the organisation of photosynthetic membranes microalgae have been classified into two prokaryotic divisions and nine eukaryotic divisions. Cyanophyceae (blue-green algae), Chlorophyceae (green algae), Bacillariophyceae and Chrysophyceae are the most cultivated microalgae. These organisms can live phototrophically, heterotrophically and even mixotrophically [17]. Microalgae existed on Earth since its environment was formed. These organisms possess chlorophyll, perform photosynthesis for producing oxygen (O2) and remove carbon dioxide (CO2) from the atmosphere [18].
Microalgae consist of protein, lipids, pigments (carotene) and vitamins [19]. Microalgae are associated with three elemental attributes which provides commercial applications. They include genetically diverse group of organisms possessing physiological and biochemical characteristics and can produce carbohydrates, lipids and bioactive compounds, etc. They can also cost-effectively integrate the stable isotopes (13C, 2H and 15N) into their biomass. They contain many unexplored organisms which may provide novel unused product source [20].
Applications of Microalgae
Food and dietary supplements
World’s population is increasing exponentially but the food resources are not increasing at the same pace. So, there is an urgent need to provide safe food and food materials to meet the ever increasing demands for food. Microalgae can remove carbon dioxide using sunlight and produce organic matter. These form the primary level i.e. phytoplankton and are consumed by zooplanktons, which are further eaten by bigger fish and animals on earth. Therefore, microalgae are supporting life on this earth [18].
Microalgae have also been exploited as food owing to its significant nutritional content. For instance, Spirulina has been used as food in Mexico and Chad. Spirulina also plays an important role in Chad’s economy [21]. Countries like Thailand, China, US and India are also using Spirulina as a supplement to the regular diet. The global net production of Spirulina is nearly 4000 metric tons [22]. Spriulina contains proteins, g-linolenic acid, vitamins and minerals. It is also reported for its therapeutic applications in curing diabetes, arthritis, cardiovascular diseases and even cancer [23]. Antioxidant properties of Spirulina are due to phycocyanin and vitamin E present in it [24,25].
A different microalgae Nostoc is consumed in China [26]. It is rich in proteins, pigment and provides less fat content, thus making it a healthy choice. Nostoc flagelliforme is popularly known as “fa cai”. Pigments such as echinenone and myxoxanthophyll, allophycocyanin, phycocyanin and chlorophyll are present in it [27]. It also contains essential amino acids for human nutrition. It has been used in traditional Chinese medicine for the treating diarrhea, hypertension and hepatitis. N. sphaeroides (Ge-Xian-Mi) is specie being used as food and herbal ingredients [28].
Chlorella is also cultivated as health food and commercially available in the form of tablets and powder. The first large scale Chlorella production unit was setup in Boston, USA. The other countries (Israel, Japan, Czechoslavakia, Taiwan, Malaysia and Indonesia) also followed afterwards [29]. Chlorella consists of proteins (51-58% dry weight), carotenoids and vitamins which makes it a suitable health food [30]. It also contains an immunostimulator i.e. â-glucan and helps in reducing blood lipids [29].
Cosmetics
The evaluation of quantity of pigment in microalgae is an important feature to determine cell growth and to check the trophic level. In cosmetics, components of algae are used as thickening agents, water-binding agents and antioxidants. Arthrospira and Chlorella are the microalgal species utilized in skin care products [31]. Microalgae extracts are present in face, sun protection, hair care and skin care products. Mostly used microalgal species include Chondrus crispus, Mastocarpus stellatus, Alaria esculenta, S. platensis, C. vulgaris and D. salina [20].
Food Colorant
The pigment in microalgae can be used as natural food coloring agent. Some microalgae contain Carotene (â carotene), which is used as a coloring agent in margarine, food additive in enhancing the color of fish flesh and overcome sterility in cattle consuming grains [32]. â-carotene is used as a dye and also provides vitamin C. According to National Cancer Institute, â Carotene is anticarcinogenic and also maintains cholesterol level. It can also minimize the chances of heart disease. Thus, â Carotene production has become more important. D. salina is cultivated for â-carotene. However, its efficiency as a food colorant is limited to the instability in light and also the color is prone to bleaching on cooking [20].
High-Value Molecules
Microalgae can be a used for producing new compounds which can act as functional ingredients. Some microalgae live in unfavorable environments of high salinity, high and low temperature, etc. and adapt by producing some secondary metabolites explicitly. Marine microalgae have been exploited for producing polyunsaturated fatty acids (PUFA), which can effectively prevent several diseases. PUFA such as a-linolenic acid (ALA, C18:3n-3), docosapentaenoic acid (DPA, C22:5n-3), and docosahexaenoic acid (DHA, C22:6n-3), effectively provided protection against various diseases like cardiovascular disorders, cancer, type 2 diabetes, arthritis, kidney and skin disorders, depression and schizophrenia. Dunaliella species, Chlorella species and Spirulina species are most cultivated species for producing high value molecules including lipids, proteins, etc. [33,34].
Biofertilizer
Microalgae have also been used in agriculture as biofertilizers and soil conditioners. Cyanobacteria can fix atmospheric nitrogen and used as biofertilizers. Cyanobacteria as a natural biofertilizer maintain and increase soil fertility along with high rice growth and yield [35]. Application of Blue green algae (BGA) as a biofertilizer, yield increases and the soil physico-chemical properties also improve. On application of biofertilizer, the pH, electrical conductivity and nutritive (nitrogen and carbon) value of residual soil improved. The protein content was also increased in grain. Nostoc, Anabaena, Tolypothrix and Aulosira are mostly used for paddy fields. Anabaena when associated with water fern Azolla fixes about 60 kg/ha/season nitrogen and supplements the required organic matter in soil. Cyanobacteria also produce growth-promoting substances which can improve soil.
Pharmaceuticals
Algal organisms can produce biologically active primary and secondary metabolites which can be employed in the pharmaceutical industry [36]. These explicit bioactive compounds are produced by algae for their survival during the competition with neighboring competitor organisms. These algae derived bioactive molecules cannot be produced by chemical synthesis. The culture extracts of Chlorella vulgaris and Chlamydomonas pyrenoidosa have been reported for their antibacterial potency against both Gram-positive and Gram-negative bacteria. Extracts of green algae, diatoms and dinoflagellates can serve as antifungal agents. Toxins produced by microalgae such as Ochromonas sp., Prymnesium parvum can be of use in pharmaceutical industries [37,38]. Cyanobacterial strains produce various intracellular and extracellular metabolites which possess antimicrobial activity.
Environmental biotechnology
Bioremediation, bioassay and biomonitoring are the three basics of microalgal environmental biotechnology. The release of contaminated wastewater into the clean water bodies creates a risk to health of existing flora and fauna. The high rate algae pond (HRAP) system proved to be an efficient in treating contaminated water [39,40]. The HRAP system is most suitable for tropical climate where the sun light is available and the temperature is also warmer. HRAP reduces pollutants and produces algal biomass which can be further used as food for animals and feedstock for producing biodiesel. HRAP algal systems have been successful in treating rubber effluent, palm oil mill effluent (POME) and municipal wastewater. Anaerobic digestion of starch factory wastewater by Spirulina platensis reduced phosphate of more than 99% [41]. The HRAP system is also used as end treatment of the pretreated water before channelizing it for human use [39]. Chlorella vulgaris grown in HRAP has been used for color removal of the wastewater [42]. A group of five microalgal species grown in HRAP treated landfill leachate [43]. C. vulgaris immobilised in alginate effectively removed color from textile dyes [44]. Immobilised C. vulgaris and Scenedesmus obliquus effectively remove nitrogen and phosphorus from wastewater [45]. Toxic tolerant microalgae are used for first B i.e. bioremediation while sensitive species are employed for rest two B’s i.e. bioassay and biomonitoring of environmental pollutants [46].
Microalgae have also been used to detect the toxicity of contaminants such as heavy metals, pesticides and pharmaceuticals. The level of heavy metals in the aquatic ecosystems and even in aquatic organisms such as fish and mussel has been determined using microalgae [47]. The commonly used microalgal species for toxicity analysis include Pseudokirchneriella subcapitata, Dunaiella tertiolecta, Isochrysis galbana, Chlorela spp. [48]. Microalgae have also been used for evaluating nutrient enrichment due to nitrogen and phosphorus. For instance, C. vulgaris, Scenedesmus quadricauda and Ankistrodemsus convolutus efficiently helped in assessing nitrogen and phosphorus enrichment in freshwater ecosystems [49].
Biodiesel
Microalgae have been reported for biodiesel production. The microalgal biofuel production is advantageous over other agricultural feedstocks as it doesn’t affect food production, fodder and other feedstocks. In Malaysia, palm oil has been used for biodiesel production but the continuous use may lead to scarcity. Therefore, microalgae present an alternative of biodiesel and replace fossil diesel [50]. Amongst all the microalgal species known Chlorella has been mostly cultivated for biodiesel production. C. protothecoides efficiently produces highly viscous biodiesel with high heating value making it of superior quality. In comparison to the terrestrial plants, microalgae can perform photosynthesis more efficiently and can flourish normally even at high temperature environment with high CO2 level [51]. It has been reported that microalgae can use produce 280 tons of dry biomass per ha-1 yr-1 utilizing 9% of the sunlight and eliminating nearly 513 tons of CO2 [52]. Species which can survive at high levels of CO2 include Spirulina sp., Scenedesmus obliquus and Chlorella vulgaris [53]. Such microalgae can be used for bioremediation of flue gas which contains nearly 12% CO2 [54]. Thus, an integrated microlagal system for biofixation of CO2 along with biodiesel production can be the most fascinating and environmentally beneficial approach. Fawzy [55] reported Asteromonas gracilis (new microalgae from Egypt) as a potent feedstock for biodiesel production.
Cultivation of Microalgae
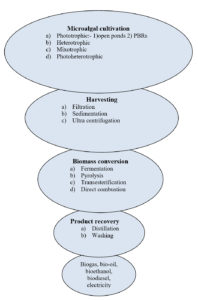
The cultivation of microalgae can be done by four different methods based on the type of metabolism and effect of environmental factors on the growth (figure 1). Four methods are: phototrophic (light as the only source of energy), heterotrophic (organic matter as the energy source), mixotrophic (organic and CO2 required for photosynthesis), and photoheterotrophic (use light to extract energy from organic matter) [56]. For the commercial biomass production of microalgae phototrophic approach is most viable, as scale up is easy and economical [57,1]. Several abiotic (light intensity, pH, temperature, salinity, nutrients and gaseous content) and biotic (fungus, other algae, bacteria etc.) factors affects the microalgal cultivation in all four methods. Among these temperature and light play vital role in growth of microalgae [58].
Phototrophic Cultivation
For microalgae cultivation, phototrophic mode is most efficient and economical approach as microalgae has higher growth and photosynthetic rates as compare to plants [12]. The ability of microalgae to act as potential sink of carbon by capturing free CO2 from atmosphere provides additional benefit. It can be done either in open ponds or enclosed photobioreactors (PBRs):
Open ponds
Being practiced from 1950’s, this is the oldest and simplest approach of phototrophic cultivation of microalgae for commercial purpose [57]. Now more than 95 % of the large scale algal cultivation is done by this approach [59]. Change in type of material, size, shape, agitation type and inclination, lead to the discovery of different types of ponds systems [60]. Among the various famous designs, raceways stirred by a paddle wheel is frequently used and others systems such as extensive shallow unmixed ponds, circular ponds mixed with a rotating arm, and sloping thin-layer cascade are also used for larger scale cultivation [61]. It is the economical and durable approach, with high productivity in large scale cultivation in comparison to enclosed PBRs. The maintenance and operational expenditure is also low, but energy input required for processing is high [62,2]. Mass transfer for the nutrient distribution and receiving light for metabolic processes, limit the depth of open pond upto 15cm. Design of open pond system is less technical, which make it more prone to factors like light, temperature, pH, gaseous content and biotic contaimants [63]. Due to above mentioned factors, the algal species must be grown in the selective conditions in open ponds [64,65,66].
Enclosed Photobioreactors (PBRs)
PBRs are available in various shapes (tube, bag and plate) and made up of different (glass, plastic etc.) materials. Mass distribution (supply of nutrients and CO2) and energy supply is efficient in PBRs, which is the major drawback of open pond system [67,68]. Annular, tubular and flat panel reactors are the familiar designs, but all of them are not commercially used for cultivation of microalgae [69,70]. Technical specificity of PBRs makes them suitable for cultivating species with specific requirements of nutrients, pH, temperature and photosynthetic gaseous [71]. The operational choice of continuous and batch mode in PBRs, provides additional benefit to algal producer. Continuous mode give higher control, better growth and biomass yield as compare to batch [72]. But biofouling in PBRs can adversely affect the productivity of biomass and biofuel. Biofouling decreases the penetration of light in PBRs as algal biomass adheres to the surface, which results in the reduction of photosynthetic efficiency. To overcome biofouling, material for PBRs surfaces should be appropriate or coated with functional groups to decrease adhesion [73].
Harvesting of microalgal biomass
After cultivation, separation of algal biomass is done for downstreaming processing to recover product. Near about 30% of production cost is invested in the recovery process of the biomass [74,58,1]. More than one approach is used for the solid-liquid separation to recover desired biomass, as there is no single efficient approach for harvesting [75]. Weissman and Goebel, [76] suggested the use of microstainer for better harvesting as it is a simple and effective approach. For larger quality of biomass, filter presses is method of choice. But slow and inadequate process of filter presses, limits their frequent use in biomass recovery [74]. For shear sensitive algal cells and small scale harvesting microfiltration and ultrafiltration can be applied. Use of these approaches for higher quantity is not feasible and economical, as maintenance cost of micro and ultrafiltration is high [58].Sedimentation, filtration and ultra-centrifugation are most frequently methods used for harvesting, but prior flocculation increase the efficiency of these methods. Microalgal biomass harvesting using fungi is successful in many cases but separation of fungi from algal cell is complicated and difficult [77]. Amalgamation of self-flocculating microalgal species with the desired non-flocculating cells is an alternative and efficient approach for increasing aggregation size, which results inefficient harvesting [78,79].
Microalgal biomass conversions
Conventional approaches
These are differentiated into four types (Table 1) as follow:
1). Biochemical approach (Anaerobic digestion and fermentation) for methanol and ethanol production.
2). Thermochemical approach (Pyrolysis, gasification, and liquefaction) for bio-oil, fuel gas and charcoal production.
3). Chemical approach (Transesterification) for biodiesel production.
4). Direct combustion for electricity production.
Biochemical approach
Fermentation
The microalgae have potential to supply sufficient nutrients (carbohydrates and proteins) to carry out fermentation by microbes such as bacteria, yeast etc. [80]. It can be carried out under anaerobic conditions, using pre-processed and saccharified biomass for bioethanol production [81]. Saccharomyces cerevisiae and Zymomonas mobilis are most frequently used microbes for bioethanol production. But, presence of mannitol in some algal species limits the use of anaerobic fermentation. It required oxygen supply and specific microbe (Zymobacter palmae) to carry out fermentation [4]. Hirano et al. [82] reported that Chlorella vulgaris is a suitable raw material for fermentation due to its high starch content, which is required for bioethanol production. 3.83 g l-1 of bioethanol was obtained using Chlorococum sp. as fermentation substrate [63]. Bioethanol can be produced via self-fermentation by microalgae. Ueno et al. [83] produced 450ìmolg-1 of ethanol by dark fermentation using Chlorococcum littorale (Green algae). Other algal species like Chlamydomonas perigranulata and agar weed (red seaweed) can also be used for production of bioethanol by fermentation [84,85].
Table (1):
Approaches involved in biomass conversion of microalgae to biofuel
| Approaches | Process | Product | ||
|---|---|---|---|---|
| Conventional approaches | Biochemical | Anaerobic digestion | Anaerbically converted into biofuel | Methanol |
| Fermentation | Aerobically converted in biofuel | Ethanol | ||
| Thermochemical | Pyrolysis | Thermal degradation in absence of oxygen | Bio-oil, Syngas | |
| Gasification | Incomplete oxidation of biomass | Syngas | ||
| liquefaction | Direct conversion of biomass to fuel oil | Bio-oil | ||
| Chemical | Transesterification | Chemical conversion of algal oil to biodiesel in presence of alcohols | Bio-diesel | |
| Direct combustion | Combustion | Burning of algal biomass | Electricity | |
| Novel approaches | Microbial fuel cell (MFC) | Chemical energy is converted into electric energy | Electricity | |
| Microbial solar cell (MSC) | Solar energy is converted into electric energy | Electricity | ||
Anaerobic digestion
The use of algae for biogas production gain interest due to low cellulose, negligible lignin content and high level of carbohydrate. Among the various species of algae sea weeds (Scenedesmus, Spirulina, Euglena, and Ulva) have higher potential to produce bio gas via anaerobic digestion [86,87,5,88]. The organic matter of the algal biomass is anaerobically converted in to gases (methane, carbon dioxide and hydrogen sulfide). This is carried out in four steps:
1). Insoluble organic compounds having high molecular weight converted to soluble form and this process is catalyzed by enzymes (from anaerobes present in digestor).
2). Acidogensis: – Carried out by acidogenic microbes, which convert soluble matter to fatty acids and alcohols.
3). Acetogensis: – Acetic acid and hydrogen is formed from fatty acids and alcohols by acetogenic microbes.
4). In last step methane and carbon dioxide is produced by methanogens using acetic acid and hydrogen.
Algal species like Chaetomorpha litorea [89], Macrocystis pyrifera [90], Chlamydomonas reinhardtii, Scenedesmus obliquus [91] are reported for bio gas production. The factors like heat, protein content of algae and salt level affect the growth of microbes, consequently affecting the bio gas yield. Sodium ions play vital role in inhibition of microbial growth, so salt tolerant microbes are preferred for anaerobic digestion [92,93]. Pretreatment with bacteria enhances the biogas production [94].
Thermochemical approaches
Pyrolysis
It is a process in which algal biomass is thermally degraded in absence of oxygen for biogas, bio-oil and charcoal production. Further classified into following
1). Conventional: – slow process occurs at the rate of 0.1–1 K/s with maximum temperature of 950K.
2). Fast: – rate (10–200 K/s) of process is fast with temperature maxima of 1250K.
3). Flash: – occurs at rate more than 1000 K/s at temperature range of 1050-1300K.
The abiotic factors which affect the efficiency of pyrolysis includes content of ash and water, temperature and time of vapor residence [95]. But algae pyrolysis is less frequently used in comparison to lignocellulosic matter [96]. Algal species like Nannochloropsis sp. [97], Saccharina japonica [98] found to produce bio-oil of superior quality.
Gasification
The process involves the conversion of algal biomass into a mixture of gases which are flammable. The conversion is carried by incomplete oxidation and the resulting gas mixture (Carbon monoxide, carbon dioxide, methane, hydrogen and nitrogen) is termed as syngas [68,99,100]. The process is being used from more than thirty years for biofuel production. The gasification occurs in presence of oxygen and at temperature more than 1000ºC. It can be carried by two methods: catalytic and non catalytic. The catalytic mode requires less temperature and further research can be done to decreases the temperature requirement and make it more economical [101]. The syngas has wide range of application in fuel and chemical industry. Syngas can be further converted to fuels like hydrogen (water gas shift reaction), hydrocarbons (Fisher-tropsch synthesis) and liquid fuels [102,103,104].
Liquefaction
It produces bio-oil having high viscosity in presence of organic solvents (propanol, butanol, glycerin etc.), gases (carbon dioxide or hydrogen) and catalyst for conversion of biomass [105,104]. The direct conversion of algal biomass to fuel oil can be achieved by using synthetic gas mixture with specific catalyst [106]. It is further categorized into different types. The disintegration of primary structure and then decomposition of constituents of biomass occurs in aqueous liquefaction. Whereas in alkali liquefaction, decarboxylation of ester bonds formed in between hydroxyl group and formate ion occurs. Salts like sodium carbonate and potassium carbonate are suitable catalyst for depolymeration of lager biomolecules (polysaccharides). The use of solvents like acetone, butanol, propanol etc. ease the handling to tarry oil formed in the reaction. High pressure liquefaction can be used to produce bio-oil, but yield is low as compare to pyrolysis and handling is complicated [105]. The use of biodiesel as an extractant has proved to be an efficient method for lipid extraction [107].
Transesterification
This is chemical process of production of biodiesel from oil extracted from algal biomass. It is a multistep process, in which triglycerides (fatty acids) reacts with alcohols in presence of catalyst to produce esters (biodiesel). Among the various alcohols, which can be used for transesterification ethanol and methanol are preferred for larger scale purposes [4]. The catalyst needed for conversion of algal oil to biodiesel can be inorganic (acid and bases) or biological (enzymes). For the production of 3 moles of biodiesel, one mol trigyceride reacts with three moles of alcohol [108]. The removal of organic solvents from product is very critical, as traces of byproduct and solvents results in failure of machines [109]. Mazuber et al. [110] compared acid catalysis to basic for biodiesel production and reported that the basic catalysis is four thousand times faster than acid catalysis. Lipase enzymes are found to be more efficient than other catalyst due specificity, adaptability and high catalytic activity [111]. The genetical modification of Escherichia coli BL21 by integrating LipB68 in bacteria for production of lipase was done by Luo et al. [112]. More than 90% of biodiesel was produced using lipase as catalyst at temperature of 293.15K, which decrease the energy input of process [4]. The factors like triglycerides content, water level, presence and type of catalyst, temperature and molar ratios of substrates affect the yield [113]. With the advancement in technology nanocatalysts are available for conversion, which have potential to improve condition of process and product quality [114]. It has been reported that cell rupture prior to transesterification enhances the rate of conversion [115].
Direct combustion
The electricity can be produced by direct combustion of the algal biomass [116]. It is generally termed as burning; it is a chemical reaction occurring between biomass and oxygen. Heat is produced primarily, whereas byproducts such as carbon dioxide and water are also there. Direct combustion can be a good alternative of burning of nonrenewable fuel in boilers and stoves for domestic use [101].
Novel approaches
Microbial fuel cell (MFC)
Fossil fuels negatively influence the nature owing to the emission of carbon dioxide leading to global warming and atmospheric pollution [117]. However, many countries are finding a piece of cogent way-out for overcoming energy crisis by utilizing renewable sources. As an upshot of all the efforts, one of the latterly proposed alternative energy sources was fuel cell (FC) which generated energy using high value metal catalysts conventionally. FC is better than other energy generators as it doesn’t produce polluting gases (such as SOx, NOx, CO2, etc.) and do not require movable parts [118]. One type of FCs is microbial fuel cell (MFC) which involves microorganism as a biocatalyst compartment for production of bioelectricity in anaerobic condition [119,120]. Although, Potter, [121] described the electrical current generation by bacteria, but its limited applicability was observed even after five decades [122]. However, in the early 1990s, FCs became far more appealing devices; consequently, MFCs were considered as promising technology [123].
MFC is a novel technology for generating electricity using biomass which is otherwise of no use. MFC is a bio electrochemical system which produces electricity by mimicking natural bacterial interactions. MFC can convert chemical energy to electrical energy using the catalytic reaction of microorganisms during catabolism of contaminants from wastewater [124,125,126]. A typical MFC is composed of anode and cathode compartments. Microbes present in the anode compartment oxidize fuel to generate electrons and protons. An external electric circuit provides the medium for transfer of electrons to the cathode while the protons are transported via separator. Electrons and protons combine with oxygen to form water in the cathode compartment. MFC’s can be used simultaneously to generate clean energy and treat wastewater.
There are two types of MFC: with mediator and without mediator (mediator-less). In case of mediator MFC, the bacteria lacks the ability to use the electrode, therefore, chemical mediators such as neutral red or anthraquinone-2,6- disulfonate (AQDS) are added. For mediator-less MFC, no mediators are added exogenously. The microorganisms can produce metabolic intermediates or end products of anaerobic respiration [127]. In presence of oxygen, microorganisms feed on sugar and produce carbon dioxide along with water. However, in anaerobic environment carbon dioxide, protons and electrons are produced [128].
C12H22O11 + 13H2O ->12CO2 + 48H+ + 48e-
Then the cells further utilize inorganic mediators for accepting the electrons produced after crossing the lipid membrane and plasma wall. The mediator starts releasing electrons from the electron transport chain of cell. These electrons will be assimilated by oxygen and other intermediates. The mediator after being reduced transfers the electrons to a depositing electrode and leaves the cell. Therefore, the mediator returns to its original oxidized state after losing electrons and can repeat the process only in anaerobic conditions. On deposition of electrons, the depositing electrode becomes negatively charged. In presence of oxygen, it will collect all the electrons due to its higher electronegativity than the mediator. This forms the basis for generation of electron flow via micro-organisms [129].
Construction
A complete circuit should be designed for generating current using MFC. The mixture solution of mediator and the micro-organism is added to an appropriate substrate such as glucose. This mixture will be kept in an airtight chamber (anaerobic) to allow anaerobic respiration by microbes. An electrode kept in the solution would serve as anode. The second chamber contains another solution and an electrode (cathode). The cathode being positively charged is equivalent to the oxygen sink as it accepts electron. The solution can act as an oxidizing agent but would need large volumes of circulating gas. Therefore, a solid oxidizing agent can be employed to overcome this problem. The two electrodes should be connected through a wire or any other electrically conductive path. A salt bridge should be attached for completing the circuit and connecting the two chambers. This connection would allow the transfer of protons from the anode to the cathode. Then the reduced mediator carrying electrons would reoxidize after depositing the electrons on the electrode. These electrons then flow through the wire to the second electrode and then to an oxidising material completing the process [129].
Performance of MFCs depends on various factors such as supply and consumption of oxygen in cathode chamber, oxidation of substrates in anode chamber, electron shuttle from anode compartment to anode surface and permeability of PEM [130]. Recently, MFC technology has improved significantly but it is still facing challenges in scale-up and practical application. It encounters various problems such as compartmental turbulence and membrane resistance in proton transportation [131]. MFCs have two major issues in power generation. Firstly, the power production and substrate concentrations are directly related, i.e. concentration of substrate beyond threshold will restrict the power generation [132]. Secondly, high internal resistance due to PEM uses a significant proportion of power generated [130]. To overcome these problems biocathodes have been developed which will improve oxygen oxidation in MFCs [131]. Novel designs of MFCs (single-chamber MFC, stacked MFC and up flow MFC) have been proposed and implemented for amplifying the power generation [123].
Microbial solar cells (MSCs)
This is a novel technology for production of electricity from the solar energy. It exploits the potential of photosynthetic microbes (microalgae) and plants for harvesting solar energy [133]. The electrochemically active microbes are then employed to generate electricity. These cells provide a fixed electric current and can be used in vast areas such as wastewater treatment plant, biodiesel and metabolite production [134]. Yoon, [135] reported a micro-sized MSC, in which anode is made of cyanobacteria (Synechocystis). The power (7.09 nW/cm2) obtained by micro-sized MSC was much higher than previously reported. As solar energy is renewable source of energy, enhancement in MSCs which depend on sunlight for energy production is offering an important and interesting area for future research.
Employment of alternative sustainable energy resources to replace conventional non renewable fuels is the thirst area of research. Due to high productivity and low land requirement make microalgae the suitable candidate for the purpose. It offers wide range of biofuels including ethanol, diesel and biogas. One of the limiting factors of its large scale production is high input cost of harvesting. So, there is a need for efficient and economical method of harvesting to reduce the input cost. With the advancement in the technology the new approaches for biomass conversion are being discovered and employed in biofuel production. MFC is one such invention, which allows the clean energy production along with the waste water treatment. MSCs are extended form of MFC, where electricity is produced by using biological pathways such as photosynthesis. The future research can be focused on the enhancement of MSCs for energy production.
- Medipally, S.R., Yusoff, F.M., Banerjee, S., Shariff, M. Microalgae as Sustainable Renewable Energy Feedstock for Biofuel Production. BioMed. Res. Int., 2015; 519513: 1-13.
- Dragone, G., Fernandes, B., Vicente, A.A., Teixeira J.A. Third generation biofuels from microalgae. In: Current research, technology and education topics in applied microbial biotechnology. (Ed, A. Mendez Vilas) Madrid:Formatex, 2010; pp1355-1366.
- Rajkumar, R., Yaakob, Z., Takriff, M.S. Potential of the micro and macroalgae for biofuel production: a brief review. Bioresour., 2014; 9: 1606-1633.
- Behera, S., Singh, R., Arora, R., Sharma, N.K., Shukla M., Kumar, S. Scope of algae as third generation biofuels. Front. Bioeng. Biotechnol., 2015; doi: 10.3389/fbioe.2014.00090
- Saqib, A., Tabbssum, M.R., Rashid, U., Ibrahim, M., Gill, S.S., Mehmood, M. A. Marine macroalgae Ulva: a potential feed-stock for bioethanol and biogas production. Asian J. Agri. Biol., 2013; 1:155-163.
- Razack, S.A., Duraiarasan, S., Mani, V. Biosynthesis of silver nanoparticle and its application in cell wall disruption to release carbohydrate and lipid from C. vulgarisfor biofuel production. Biotechnol. Rep. (Amst)., 2017; 11:70-76.
- Brennan, L., Owende, P. Biofuels from microalgae are view of technologies for production, processing, and extractions of biofuels and co-products. Renew. Sustain. Energ. Rev., 2010; 14: 557-577.
- Nigam, P.S., Singh, A. Production of liquid biofuels from renewable resources. Prog. Energy Combust. Sci., 2011; 37:52-68.
- Hsueh, H.T., Chu, H., Yu, S.T. A batch study on the bio-fixation of carbon dioxide in the absorbed solution from a chemical wet scrubber by hot spring and marine algae. Chemosphere., 2007, 66: 878-886.
- Choi, W., Han, J., Lee, C., Song, C., Kim, J., Seo, Y. Bioethanol production from Ulvapertusa kjellman by high-temperature liquefaction. Chem. Biochem. Eng., 2012; 26:15-21.
- Pirt, S.J. The thermodynamic efficiency (quantum demand) and dynamics of photosynthetic growth. The New Phytologist., 1986; 102: 3-37.
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv., 2007; 25: 294-306.
- Searchinger, T., Heimlich, R., Houghton, R.A., Dong, F., Elobeid, A., Fabiosa, J., Tokgoz, S., Hayes, D., Yu, T.H. Use of U.S. croplands for biofuels increases greenhouse gases algae cultivation algae harvesting & drying lipid extraction transesterification biodiesel through emissions from land-use change. Science., 2008; 319:1238-1240.
- Sudhakar, K., Premalatha, M. Micro-algal technology for sustainable energy production: state of the art. J. Sustainable Energy & Environment, 2012; 3:59-62.
- McHugh, D.J. A guide to the seaweed industry. Rome, FAO. 2003, FAO Fisheries Technical Paper No. 441.
- Luning, K., Pang, S.J. Mass cultivation of seaweeds: current aspects and approaches. J. Appl. Phycology., 2003; 15:115-119.
- Carlsson, A.S., van Beilen, J.B., Möller, R., Clayton, D. Micro- and Macro-algae: utility for industrial applications. (ed. D. Bowles) CPL Press, UK, 2007; pp 1-86.
- Sumi, Y. Microalgae Pioneering the Future- Application and Utilization. 2009; Quarterly review no. 34: 1-13.
- Avagyan, A.B. Microalgae: big feed potential in a small package. Feed Int., 2008; 16-18.
- Priyadarshani, I., Rath, B. Commercial and industrial applications of micro algae – A review. J. Algal Biomass Utln., 2012; 3:89-100.
- Abdulqader, G., Barsanti, L., Tredici, M.R. Harvest of Arthrospira platensis from Lake Kossorom (Chad) and its household usage among the Kanembu. J. Appl. Phycol., 2000; 12: 493-498.
- Belay, A. “Spirulina (Arthrospira): production and quality assurance. In: Spirulina in human nutrition and health. (eds. M. E. Gershwin, A. Belay) Boca Raton: CRC Press, 2008; pp 1-25.
- Mani, U.V., Iyer, U.M., Dhruv, S.A., Mani, I.U., Sharma, K.S. Therapeutic utility of Spirulina. In; Spirulina in human nutrition and health, (eds. M. E. Gershwin, A. Belay) Boca Raton: CRC Press, 2008; pp 71-99.
- Chu, W.L., Lim, Y.W., Radhakrishnan, A.K., Lim, P.E. Protective effect of aqueous extract from Spirulina platensis against cell death induced by free radicals. BMC Complement Altern. Med., 2010; 10: 53.
- Chu, W.L. Potential applications of antioxidant compounds from algae. Curr. Top. Nutraceut. Res., 2011; 9: 83-98.
- Han, D., Bi, Y., Hu, Z. Industrial production of microalgal cell-mass and secondary products- species of high potential Nostoc. In: Handbook of microalgal culture: biotechnology and applied phycology, (ed. A. Richmond) Oxford: Blackwell Science, 2004; pp 304-311.
- Lu. R.S., Liu, B., Zhang, Z.D. Studies on the isolation of and spectral characteristics of phycobilisomes from Nostoc flagelliforme. Chinese Bull Bot., 1990; 7: 27-30.
- Barsanti, L., Gualtieri, P. Algae: anatomy, biochemistry and biotechnology. Florida: Taylors R Francis Group, 2006; pp 301.
- Iwamoto, H. Industrial production of microalgal cell-mass and secondary products – major industrial species Chlorella. In: Handbook of microalgal culture: biotechnology and applied Phycology. (ed. A. Richmond) UK: Blackwell Science, 2004; pp 255-263.
- Becker, W. Microalgae in human and animal nutrition. In: Handbook of microalgal culture: biotechnology and applied phycology, (ed. A. Richmond) Oxford: Blackwell Science, 2004; pp 312-351.
- Stolz, P., Obermayer, B. Manufacturing microalgae for skin care. Cosmetics Toiletries, 2005; 120: 99-106.
- Borowitzka, M.A., Borowitzka, L.J. “Vitamins and fine chemicals from micro-algae,” in Micro-Algal Biotechnology, eds. M. A. Borowitzka and L. J. Borowitzka (Cambridge University Press),1987.
- El- Baz, F.K., Aboul-Enein, M.A., El-Baroty, G.S. ,Youssef, A.M., Abd El-Baky, H.H. Accumulation of antioxidant vitamins in Dunaliella salina. Online J. Biol. Sci., 2002; 2:220-223.
- Abd El-Baky, H.H., Moawd, A., El-Behairy, A.N., El-Baroty, G.S. Chemoprevention of benzo[a]pyrene-induced carcinogen and lipid peroxidation in mice by lipophilic algae extracts (phycotene). J. Med. Sci., 2002; 2:185-93.
- Song, T., Martensson, L., Eriksson, T., Zheng, W., Rasmussen, U. Biodiversity and seasonal variation of the cyanobacterial assemblage in a rice paddy field in Fujian, China. FEMS, 2005; 54:131-140.
- Rania, M.A., Hala, M.T. Antibacterial and antifungal activity of Cynobacteria and green microalgae evaluation of medium components by Plackett-Burman design for antimicrobial activity of Spirulina platensis. GJBBR, 2008; 3:22-31.
- Borowitzka, M.A., Borowitzka, L.J. “Microalgal Biotechnology,” (Cambridge University Press USA), 1992; 179.
- Katircioglu, H., Beyatli, Y., Aslim B., Yuksekdag, Z., Atici, T. Screening for antimicrobial agent production in fresh water. Internet J. Microbiol., 2006; 2:2.
- Hoffmann, J.P. Wastewater treatment with suspended and nonsuspended algae. J. Phycol., 1988; 34:757-763.
- Phang, S.M., Chui, Y.Y., Kumaran, G., Jeyaratnam, S., Hashim, M.A. High rate algal ponds for treatment of wastewater: a case study for the rubber industry. In: Photosynthetic microorganisms in environmental biotechnology, eds. H. Kojima, Y. K. Lee (Hong Kong: Springer-Verlag), 2001; pp 51-76.
- Phang, S.M., Miah, M.S., Yeoh, B.G., Hashim, M.A. Spirulina cultivation in digested sago starch factory wastewater. J. Appl. Phycol., 2000; 12:395-400.
- Lim, S.L., Chu, W.L., Phang, S.M. Use of Chlorella vulgaris for bioremediation of textile wastewater. Bioresour. Technol., 2010; 101:7314-7322.
- Mustafa, E.M., Phang, S.M., Chu, W.L. Use of an algal consortium of five algae in the treatment of landfill leachate using the high-rate algal pond system. J. Appl. Phycol., 2012; 24: 953.
- Chu, W.L., See , T.C., Phang, S.M. Use of immobilised Chlorella vulgaris for the removal of colour from textile dyes. J. Appl. Phycol., 2009; 21:641-648.
- Ruiz-Marin, A., Mendoza-Espinosa, L., Stephenson, T. Growth and nutrient removal in free and immobilized green algae in batch and semi-continuous cultures treating real wastewater. Bioresour. Technol., 2010; 101:58-64.
- Ismail, M. Phytoplankton and heavy metal contamination in the marine environment. In: Biomonitoring in tropical coastal ecosystems, eds. S. M. Phang, M. Brown [Kuala Lumpur: University of Malaya Maritime Research Centre (UMMrec)], 2004; pp 15-96.
- Irwandi, J., Farida, O. Mineral and heavy metal contents of marine fin fish in Langkawi Island, Malaysia. Int. Food Res. J., 2009; 16:105-112.
- Vannini, C., Domingo, G., Marsoni, M., De Mattia, F., Labra, M., Castiglioni, S., Bracale, M. Effects of a complex mixture of therapeutic drugs on unicellular algae Pseudokirchneriella subcapitata. Aquat., 2011; 101:459-465.
- Chu, W.L., Ramadhona, M.S., Phang, S.M. Assessment of three tropical chlorophytes as bioassay organisms for nitrogen and phosphorus enrichment in freshwater ecosystems. Mal. J. Sci., 2007; 26:15-25.
- Chisti, Y. Biodiesel from microalgae beats bioethanol. Trends Biotechnol., 2008; 26:126-131.
- Papazi, A., Makridis, P., Divanach, P., Kotzabasis, K. Bioenergetic changes in the microalgal photosynthetic apparatus by extremely high CO2-concentrations induce an intense biomass production. Physiol. Plant., 2008; 132:338-349.
- Bilaovic, D., Andargatchew, A., Kroeger, T., Shelef, G. Freshwater and marine microalgae sequestering of CO2 at different C and N concentrations-response surface methodology analysis. Energy Conver. Mgmt., 2009; 50:262-267.
- De Morais, M.G., Costa, J.A.V. Carbon dioxide fixation by Chlorella kessleri, C vulgaris, Scenedesmus obliquus and Spirulina sp. cultivated in flasks and vertical tubular photobioreactors. Biotechnol. Lett., 2007; 29:1349-1352.
- Douskova, I., Doucha, J., Livansky, K., Machar, J., Novak, P., Umysova, D., Zachleder, V., Vitova, M. Simultaneous flue gas bioremediation and reduction of microalgal biomass production costs. Appl. Microbiol. Biotechnol., 2009; 82:179-185.
- Fawzy, M, A. Fatty acid characterization and biodiesel production by the marine microalga Asteromonas gracilis: Statistical optimization of medium for biomass and lipid enhancement. Mar. Biotechnol., 2017. doi 10.1007/s10126-017-9743-y
- Wang, J., Yang, H., Wang, F. Mixotrophic cultivation of microalgae for biodiesel production: status and prospects. Appl. Biochem. Biotechnol., 2014; 7:3307-3329.
- Borowitzka, M. A. Commercial production of microalgae: ponds, tanks, tubes and fermenters. J. Biotechnol., 1999; 70:313-321.
- Mata, T.M., Martins, A.A., Caetano, N.S. Microalgae for biodiesel production and other applications: A review. Renew. Sustain. Energ. Rev. 2010; 14:217-232.
- Sheehan, J., Camobreco, V., Duffield, J., Graboski, M., Shapouri, H. “An overview of biodiesel and petroleum diesel life cycles” US Department of Agriculture and Energy Report, 1998.
- Borowitzka, M.A. “Culturing microalgae in outdoor ponds” in Algal Culturing Techniques, Ed. R. A. Anderson (Elsevier, London, UK), 2005; pp 205-18.
- Jimenez, C., Cosso, B. R., Labella, D., Niell, F. X. The feasibility of industrial production of Spirulina (Arthrospira) in Southern Spain. Aquaculture., 2003; 217:179-190.
- Rodolfi, L., Zittelli, G.C., Bassi, N. Microalgae for oil: strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol. Bioeng., 2009; 102:100-112.
- Harun, R., Singh, M., Forde, G.M., Danquah, M.K. Bioprocess engineering of microalgae to produce a variety of consumer products. Renew. Sustain. Energ. Rev., 2010a; 14:1037-1047.
- Cysewski G.R., Lorenz, R.T. “Industrial production of microalgal cell-mass and secondary products-species of high potential: Haematococcus,” in Microalgal Culture: Biotechnology and Applied Phycology, Ed. A. Richmond (Blackwell Science, Oxford, UK), 2004; pp 281-288.
- Moheimani N.R., Borowitzka, M.A. The long-term culture of the cocco lithophore Pleurochrysis carterae (Haptophyta) in outdoor raceway pond. J. Appl. Phyco., 2006; 18:703-712.
- Borowitzka, M. A. “Carotenoid production using microalgae,” in Single Cell Oils: Microbial and Algal Oils, Eds. Z. Cohen and C. Ratledge (American Oil Chemists Society, Urbana, USA), 2010; pp 225-240.
- Pulz, O. Photobioreactors: production systems for phototrophic microorganisms. Appl. Microbio. Biotechnol., 2001; 57:287-293.
- Carvalho, A.P., Meireles, L. A., Malcata, F.X. Microalgal reactors: a review of enclosed system designs and performances. Biotechnol. Progr. 2006; 22:1490-1506.
- Chisti, Y. Microalgae as sustainable cell factories. Environ. Eng. Manag. J., 2006; 5:261-274.
- Ugwu C.U., Aoyagi, H. Microalgal culture systems: an insight into their designs, operation and applications. Biotechnol., 2012; 11:127-132.
- Manzanera, M. “Biofuels from Oily Biomass” in Carbon-neutral fuels and energy carriers, Eds, M. Muradov and N. Veziroghu (CRC, Taylor & Francis Group, Orlando, USA), 2011; pp 635-663.
- Picazo-Espinosa, R., Gonzalez-Lopez, A., Manzanera, M. “Bioresources for Third-Generation Biofuels” in Biofuel’s Engineering Process Technology, Ed. M.A.D.S. Bernardes, (Intech), 2011; pp 115-140.
- Zeriouh, O., Reinoso-Moreno, J.V., López-Rosales, L., Cerón-García, M.D.C, Sánchez-Mirón, A., García-Camacho, F., Molina-Grima, E. Biofouling in photobioreactors for marine microalgae. Crit Rev Biotechnol., 2017; 20:1-18.
- Grima, M.E., Belarbi, E.H., Fernandez, F.G.A., Medina, A.R., Chisti, Y. Recovery of microalgal biomass and metabolites: process options and economics. Biotechnol. Adv., 2003; 20:491-515.
- Harith, Z.T., Yusoff, F.M., Shariff, M., Ariff, A.B. Effect of different separation techniques and storage temperatures on the viability of marine microalgae, Chaetoceros calcitrans, during storage. Biotechnol., 2010; 9:387-391.
- Weissman, J.C., Goebel, R.P. Design and analysis of microalgal open pond systems for the purpose of producing fuels: a subcontract report. 1987, US DOESERI.
- Alam, M. A., Vandamme, D., Wan, C., Zhao, X., Foubert, I., Wang, Z., Muylaert, K. Yuan, Z. Bioflocculation as an innovative harvesting strategy for microalgae. Rev. Environ. Sci. Biotechnol., 2016; 15(4):573-83.
- Ummalyma, S. B., Gnansounou, E., Sukumaran, R. K., Sindhu, R., Pandey, A., Sahoo, D. Bioflocculation: an alternative strategy for harvesting of microalgae- an overview. Bioresour. Technol., 2017. doi:10.1016/j.biortech.2017.02.097.
- Liu, C., Hao, Y., Jiang, J. Liu, W. Valorization of untreated rice bran towards bioflocculant using a lignocellulose degrading strain and its use in microalgal biomass harvest. Biotechnol. Biofuels., 2017; 10:90.
- Harun, R., Danquah, M.K., Forde, G.M. Microalgal biomass as a fermentation feedstock for bioethanol production. J. Chem. Tech. Biotechnol., 2010b; 85:199-203.
- Nguyen, T.H.M., Vu, V.H. Bioethanol production from marine algae biomass: prospect and troubles. J. Viet. Env., 2012; 3:25-29.
- Hirano, A., Ueda, R., Hirayama, S., Ogushi, Y. CO2 fixation and ethanol production with microalgal photosynthesis and intracellular anaerobic fermentation. Energy., 1997; 22:137-142.
- Ueno, Y., Kurano, N., Miyachi, S. Ethanol production by dark fermentation in the marine green alga, Chlorococcum littorale. J. Ferment. Bioeng., 1998; 86:38-43.
- Hon-Nami, K. A unique feature of hydrogen recovery in endogenous starch to alcohol fermentation of the marine microalga Chlamydomonas perigranulata. Appl. Biochem. Biotechnol., 2006; 131:808-828.
- Yanagisawa, M., Nakamura, K., Ariga, O., Nakasaki, K. Production of high concentrations of bioethanol from seaweeds that contain easily hydrolysable polysaccharides. Process Biochem., 2011; 46:2111-2116.
- Ras, M., Lardon, L., Bruno, S., Bernet, N., Steyer, J.P. Experimental study on a coupled process of production and anaerobic digestion of Chlorella vulgaris. Bioresour.Technol., 2011; 102:200-206.
- Zhong, W., Zhang, Z., Luo, Y., Qiao, W., Xiao, M., Zhang, M. Biogas productivity by co-digesting Taihu blue algae with corn straw as an external carbon source. Bioresour.Technol., 2012;114:181-186.
- Tedesco, S., Stokes, J. Valorisation to biogas of macroalgal waste streams: a circular approach to bioproducts and bioenergy in Ireland. Chem Zvesti., 2017; 71(4): 721-728.
- Sangeetha, P., Babu, S., Rangasamy, R. Potential of green alga Chaetomorpha litorea (Harvey) for bio gas production. Int. J. Curr. Sci., 2011;1:24-29.
- Vergara-Fernandez, A., Vargas, G., Alarcon, N., Velasco, A. Evaluation of marine algae as a source of biogas in a two-stage anaerobic reactor system. Biomass Bioenergy., 2008; 32:338-344.
- Mussgnug, J.H., Klassen, V., Schluter, A., Kruse, O. Microalgae as substrates for fermentative biogas production in a combined biorefinery concept. J. Biotechnol., 2010; 150:51-56.
- Brennan, L., Owende, P. Biofuels from microalgae are view of technologies for production, processing, andextractions of biofuels and co-products. Renew. Sustain. Energ. Rev., 2010; 14:557-577.
- Jones, C.S., Mayfield, S.P. Algae biofuels: versatility for the future of bioenergy. Curr.Opin.Biotechnol., 2012; 23:346-351.
- Kavitha, S., Subbulakshmi, P., Rajesh Banu, J., Gobi, M., Tae Yeom, I. Enhancement of biogas production from microalgal biomass through cellulolytic bacterial pretreatment. Bioresour. Technol., 2017; 233:34-43.
- Fahmi, R., Bridgwater, A.V., Donnison, I., Yates, N. The effect of lignin and inorganic species in biomass on pyrolysis oil yield, quality and stability. Fuel., 2008; 87:1230-1240.
- Oyedun, A.O., Lam, K.L., Gebreegziabher, T., Lee, H.K.M., Hui, C.W. Optimisation of operating parameters in multi-stage pyrolysis. Chem. Engg.Trans., 2012; 29:655-660.
- Porphy, S.J., Farid, M.M. Feasibility study for production of biofuel and chemicals from marine microalgae Nannochloropsis sp. based on basic mass and energy analysis. ISRN Renew. Energ., 2012; 156824.
- Choi, J.H., Woo, H.C., Suh, D.J. Pyrolysis of seaweeds for bio-oil and bio-char production. Chem.Eng.Trans., 2014; 37:121-126.
- Prins, M.J., Ptasinski, K.J., Janssen, F.J.J.G. More efficient biomass gasification via torrefaction. Energy., 2006; 31:3458-3470.
- Lv, P., Yuan, Z., Wu, C., Ma, L., Chen, Y., Tsubaki, N. Biosyngas production from biomass catalytic gasification. Energy. Convers. Manag., 2007; 48: 1132-1139.
- Lee, S., Speight, J.G., Loyalka, S.K. Hand book of alternative fuel technologies. USA: CRC Taylor and Francis Group. 2007.
- Steen, E.V., Claeys, M. Fischer–Tropsch catalysts for the biomass-to-liquid (BTL) process. Chem. Eng. Technol., 2008; 31:655-60.
- Balat, M. Sustainable transportation fuels from biomass materials. Energy Educ. Sci. Technol., 2006; 17:83-103.
- Rowlands, W.N, Masters, A., Maschmeyer, T. The biorefinery-challenges, opportunities, and an Australian perspective. Bull. Sci. Technol. Soc., 2008; 28:149-58.
- Demirbas, A. Current technologies for the thermo-conversion of biomass into fuels and chemicals. Energy Sources., 2004; 26:715-730.
- Naik, S.N., Goud, V.V., Rout, P.K., Dalai, A.K. Production of first and second generation biofuels: A comprehensive review. Renew. Sustain. Energy. Rev. 2010; 14:578-597.
- Huang, W.C., Park, C.W., Kim, J.D. A novel microalgal lipid extraction method using biodiesel (fatty acid methyl esters) as an extractant. Bioresour. Technol., 2017; 226:94-98.
- Stergiou, P.Y., Foukis, A., Filippou, M., Koukouritaki, M., Parapouli, M.,Theodorou, L. G. Advances in lipase-catalyzed esterification reactions. Biotech-nol. Adv., 2013; 31; 1846-1859.
- Munir, N., Sharif, N., Naz, S., Saleem, F., Manzoor,F. Harvesting and processing of microalgae biomass fractions for biodiesel production (a review). Sci. Tech. Dev., 2013; 32:235-243.
- Mazubert, A., Poux, M., Aubin, J. Intensified processes for FAME pro-duction from waste cooking oil: a technological review. Chem. Eng. J., 2013; 233:201-223.
- Knezevic, Z. D., Siler-Marinkovic, S. S., Mojovic, L.V. Immobilized lipases as practical catalysts. Acta Period. Technol., 2004; 35:151-164.
- Luo, Y., Zheng, Y., Jiang, Z., Ma, Y., Wei, D. A novel psychrophilic lipase from Pseudomonas fluorescens with unique property in chiral resolution and biodiesel production via transesterification. Appl. Microbiol. Biotechnol., 2006; 73: 349-355.
- Sohpal, V.K., Singh, A. Optimization of alkali catalyst for transesterification of jatropha curcus using adaptive neurofuzzy modeling. Biofuel Res. J., 2014; 2:70-76.
- Akia, M., Yazdani, F., Motaee, E., Han, D., Arandiyan, H. A review on conversion of biomass to biofuel by nanocatalysts. Biofuel Res. J., 2014; 1:16-25.
- Gouveia, L., Janelas, J., Tropecêlo, AI., Oliveira, A.C. Microalga Nannochloropsis sp. biomass for biodiesel production: conventional (cell disruption) and in situ transesterification. J. Mar. Biol. Oceanogr., 2016; 5:1.
- Pena, N. Biofuels for Transportation: A Climate Perspective, Pew Centre on Global Climate Change. 2008; http://www.pewclimate.org/biofuelstransportation.
- Rahimnejad, M., Ghoreyshi, A., Najafpour, G., Younesi, H., Shakeri, M. A novel microbial fuel cell stack for continuous production of clean energy. Int. J. Hydrogen Energy., 2012; 37:5992-6000.
- Peighambardoust, S., Rowshanzamir, S., Amjadi, M. Review of the proton exchange membranes for fuel cell application. Int. J. Hydrogen Energy., 2010; 35:9349-9384.
- Rahimnejad, M., Ghoreyshi, A.A., Najafpour, G., Jafary, T. Power generation from organic substrate in batch and continuous flow microbial fuel cell operations. Appl. Energ., 2011; 88:3999-4004.
- Tardast, A., Rahimnejad, M., Najafpour, G., Ghoreyshi, A.A., Zare, H. Fabrication and operation of a novel membrane-less microbial fuel cell as a bioelectricity generator. Int. J. Environ. Eng. 2012; 3:1-5.
- Potter, M.C. Electrical effects accompanying the decomposition of organic compounds, Proc. Royal Soc. London, Ser. B, Containing Pap. Biol. Charact., 1911; 84:260-276.
- Lewis, K. Symposium on bioelectrochemistry of microorganisms. IV. Biochemical fuel cells. Bacteriol. Rev., 1966; 30:101-113.
- Rahimnejad, M., Adhami, A., Darvari, S., Zirepour, A., Sang-Eun, O. Microbial fuel cell as new technology for bioelectricity generation: A review. Alexandria Engineering J., 2015; 54:745-756.
- Allen, R.M., Bennetto, H.P. Microbial Fuel Cells-Electricity Production from Carbohydrates. Appl. Biochem. Biotechnol., 1993; 39(40):27-40.
- Rabaey, K., Verstraete, W. Microbial Fuel Cells: Novel Biotechnology for Energy Generations. Trends Biotechnol., 2005; 23:291-298.
- Ucar, D., Zhang, Y., Angelidaki, I. An Overview of Electron Acceptors in Microbial Fuel Cells. Front. Microbiol. 2017; 8:643.
- Eugenii, K., Shipway A.N., Willner, I. Biochemical fuel cells. IN: Handbook of Fuel Cells -Fundamentals, Technology and Applications, (ed. V. Wolf, A. G. Hubert and A. Lamm) John Wiley & Sons, Ltd, 2010.
- Bennetto, H.P. Electricity generation by micro-organisms. Biotechnol Edu., 1990; 1:163-168.
- Barua, P.K., Deka, D. Electricity generation from biowaste based Microbial Fuel Cells. Int J Energ Info Commun., 2010; 1: 77-92.
- Sharma, Y., Li, B. The variation of power generation with organic substrates in single-chamber microbial fuel cells (SCMFCs). Bioresour. Technol., 2010; 101:1844-1850.
- Chen, G.W., Choi, S.J., Lee, T.H., Lee, G.Y., Cha, J.H., Kim, C.W. Application of biocathode in microbial fuel cells: cell performance and microbial community. Appl. Microbiol. Biot., 2008; 79:379-388.
- Choudhury, P., Prasad, Uday, U. S., Bandyopadhyay, T. K., Ray, R.N., Bhunia, B. Performance improvement of microbial fuel cell (MFC) using suitable electrode and Bioengineered organisms: A review. Bioengineered., 2017; 28:1-17.
- Lee, D.J., Chang, J.J., Lai, J.Y. Microalgae–microbial fuel cell: A mini review. Bioresour. Technol., 2015; 198: 891-895.
- Guo, X., Yu, C., Zheng, T. Advances in microbial solar cells—A review. Wei Sheng Wu Xue Bao., 2015; 4:961-970.
- Yoon, S., Lee, H., Fraiwan, A., Dai, C., Cho, S. A micro-sized microbial solar cell in Nano/Micro Engineered and Molecular Systems (NEMS), in 9th IEEE International Conference (Waikiki Beach, HI), 2014; 265-268.
© The Author(s) 2017. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.