ISSN: 0973-7510
E-ISSN: 2581-690X
The indiscriminate use of pesticides in agricultural practice in recent years has generated various environmental problems, since these compounds tend to bioaccumulate through trophic chains presenting considerable levels of toxicity and conditioning potential risks in ecosystems and public health. In the present work, the ability of a bacterial consortium to degrade carbofuran, malathion and 2,4-D was evaluated. The bacteria were isolated from plots with application of pesticides located in Los Mochis, Sinaloa, Mexico. The bacterial consortium degraded carbofuran (34.99%), malathion (46.68%) and 2,4-D (48.42%) in soil samples contaminated with each of the pesticides, the bacterial consortium degraded in a greater proportion the 2,4-D.
Pesticides, degradation, soil, bacterial consortium, Mexico
Pesticides are widely used worldwide, using various pesticides in order to prevent pests from damaging crops, however, each pesticide has different chemical properties and toxicity mechanisms. Mexico is not the exception in terms of the use of a wide diversity of pesticides, using annually an approximate of 35,000 tons1.
Pesticides can be highly damaging to public health, ecosystems and causing intoxication of animals and plants2-4, so it has been suggested to use less aggressive methods for the control of pests and weeds, or in their case natural pesticides that cause minor effects on public health and the environment.
Pollution in soils generates alterations in the cultivation areas, such as salinization and alkalization5, in addition to the effect they cause in the reduction of microbiological diversity, as well as causing effects on the natural enemies of pests6.
To mitigate the problem in contaminated places, one of the best known methods is the microbial bioremediation with which the degradation and mineralization of multiple contaminants can be accelerated, since the microorganisms intervene in the decomposition of organic compounds7,8. In the present work, the ability of a bacterial consortium to degrade carbofuran, malathion and 2,4-D was evaluated.
Bacterial consortium preparation
We used a bacterial consortium composed of Pseudomonas sp and Enterobacter sp isolated samples of cultivation plots in Los Mochis-Sinaloa (Mexico), the isolates showed metabolic activity and resistance against carbofuran, malathion and 2,4-D9. Each of the bacterial isolates were reseeded in 1 mL of nutrient broth and incubated for 24 hours at 30°C, then each of the bacterial cultures was poured into 300 mL of nutrient broth (forming the bacterial consortium), to propagate it for 24 hours. hours at 30°C.
Ten tubes were prepared with 9 mL of nutrient broth and 1 mL of the bacterial consortium was poured into the first tube and then eight decimal dilutions were made. In addition, from each of the tubes, 10 ìL were resected by stria in nutritive agar plates to perform the CFU/mL count.
From each tube four samples of 200 ìL were taken and placed in 96-well microplates and the absorbance reading was made at 492 nm (PoweamWHYM201 plate reader), in order to adjust the bacterial inoculum to a concentration of 8X107 CFU / mL., correlating the number of CFU with respect to the optical density of the turbidity of bacterial consortium.
Degradation detection by UV-vis spectroscopy of pesticides
Soil was used with pH 5.6 and organic matter (1.24%), which was washed three times with distilled water in its equivalent weight, to remove impurities and placed in the muffle at 100°C for 6 hours to evaporate the excess water.
Of the washed soil, 300 grams were weighed and divided into 3 fractions of 100 grams, each fraction was added one of the pesticides (carbofuran, malathion and 2,4-D) in a 100 mL solution and a concentration of 100 ppm, the samples were then placed in the muffle at 100°C.
Eight sub-samples of 10 grams were prepared, processing a sample every third day for 24 days. 20 mL of acetone were eluted per sample to extract each pesticide and analyzed by UV-visible making a 1:200 dilution with deionized water. This procedure was repeated with inoculation of the bacterial consortium, after removing the samples from the muffle, adding 1mL to a concentration of 8X107 CFU/mL per subsample, which contained 10 grams of soil.
The UV-vis spectra were determined, with the absorbance observed in the maximum of each spectrum the concentrations were calculated using the calibration curve of each pesticide to compare the degradation in the absence and presence of bacterial consortium.
Statistic analysis
To analyze the data referring to the percentage of degradation of the pesticides against the bacterial consortium, the analysis of variance test was performed with its respective Tukey test with a significance level of 0.05 (Software Instat 2.0).
Through the reading of the optical density and the CFU/mL quantified on the plate, the inoculum was adjusted (bacterial consortium), considering the concentration of 8X107 CFU/mL to carry out the inoculation of consortium in the soil samples.
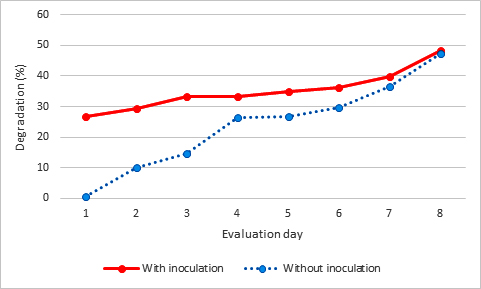
When comparing the degradation of 2,4-D, it was observed that the degradation of the pesticide predominated in the test, mainly during the first four dates of evaluation, where the bacterial consortium was inoculated. The 2,4-D on the date 1 of evaluation of degradation in the sample with inoculation of the bacterial consortium presented a 26.71% degradation with respect to the trial where there was no inoculation of bacterial consortium with a 0.44%, and in the last evaluation date there was only a difference of 1.03% degradation between both treatments (Fig. 1).
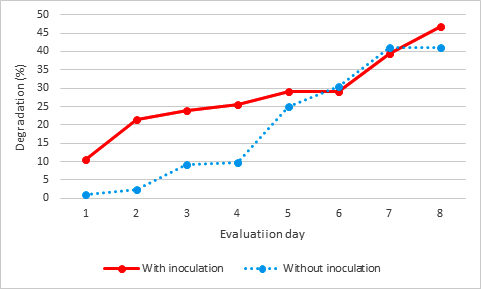
Malathion in the presence of the bacterial consortium presented a greater degradation in the first four evaluation dates, observing in the first date a difference of 9.64% with respect to the sample without inoculum of bacterial consortium, and during the last date there was greater degradation with a 5.6% compared to the one not inoculated with the bacterial consortium (Fig. 2).
On the one date of evaluation of the degradation of carbofuran, a greater degradation (10.81%) was observed in the sample with inoculation of bacterial consortium with respect to the sample without inoculation of the consortium with 0.58% degradation of the pesticide. It should be noted that on evaluation dates 3 and 7 without inoculation of the bacterial consortium, a greater degradation of Carbofuran was found compared to that inoculated with the consortium, however the tendency to greater degradation was observed in the sample that was inoculated with the consortium, which is observed towards the last evaluation date with 5% more pesticide degradation (Fig. 3).
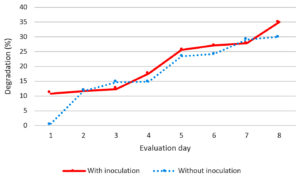
Fig. 3. Carbofuran degradation in presence and absence of bacterial consortium
Statistical analysis showed significant difference only between the values of degradation of 2,4-D in the presence of bacterial consortium compared to the group that did not receive consortium inoculation, and between the values of degradation of carbofuran compared to 2, 4-D in the presence of bacterial consortium (P <0.05).
The inoculum with bacterial consortium showed greater degradation activity in the presence of 2,4-D, followed by malathion and carbofuran. The 2,4-D in 24 days showed a maximum degradation of 48.42%. This is different from the results of Han et al., (2015) in which Cupriavidus campinensis BJ71 degraded 2,4-D by 87.13% in non-sterile soil and in sterile soil by about 60%10. These differences may be due to the fact that the soil used in the present work was washed and subjected to temperatures of 100°C for 24 hours.
The malathion at 24 days of monitoring of this work had a maximum of extracted from a salt marsh, and that degraded malathion as the only carbon source, degrading it by 48% in 30 days and monitoring the bacteria after 10 days, the 54% degraded malathion in less than 40%11. On the contrary, there is a greater difference with that reported by Ibrahim et al., (2014), where their isolates showed the following degradation activity, Nostoc muscorum with 91%, Aspergillus oryzae 65% and Streptomyces platensis 54%, highlighting that these tests were in liquid culture with a duration of 20 days in different percentages, which contrasts since the results presented here were directly in the soil12.
The results may vary depending on the bacterial species used to degrade the pesticide, such as the example of Alicyclobacillus tengchongensis, from which an enzyme can be extracted that efficiently degrades the malathion and has a potential use for the detoxification of malathion residues13. The results vary because the malathion tends to be unstable by changing its original structure to one of its degradation subproducts in short periods of time.
It should be noted that the initial concentration of the malathion when extracted from the muffle before inoculation was very low, this may be due to the fact that the malathion could be decomposed in the muffle even though it was at a relatively low temperature, since the malathion is highly sensitive to temperature and its chemical structure is transformed into a short time interval since it is not very stable14.
The carbofuran had a maximum degradation of 34.99%, in contrast to that reported by Castro et al., (2018), which through BPS (Pesticide Biopurification Systems), achieved the degradation of carbofuran in average of 2-3 days, using bioaugmentation and normal inoculation15.
In other studies the degradation of carbofuran by means of Arthrobacter sp in a mineral medium in a range of 72-120 hours has been reported, being the degradation more rapid at a temperature of 35°C16. similarly in another of his works carbofuran was hydrolyzed in 42 hours in a medium with glucose and its phenolic structure was mineralized in five days17.
Similarly, the results of the present work differ with Pseudomonas sp which can degrade up to 95% to carbofuran in nutritive broth in 30 days at 30°C18,19. These differences may be due to the fact that in most of the works cited degradation occurs under conditions more conducive to bacterial growth, such as liquid culture media and in the present work the degradation of the pesticide was carried out with inoculation in soil samples.
The discrepancies between the results can also vary due to the effect of different variables in the soil, since the adsorption, persistence and mobility of some pesticides increases when the pH becomes acidic and in the presence of a higher content of clays and organic matter20. Of course, temperature also plays a key role in the degradation of the malathion, which greatly influences the temperature at which the experiments are carried out, for example the thermo-stability of the malathion dissipates rapidly at temperatures between 27 and 32°C21.
In conclusion, bacterial consortium degraded carbofuran by 34.99%, malathion 46.68% and 2,4-D 48.42%, efficiently degrading the pesticides in the first six days, which is a function of the bacterial growth rate. It is emphasized that the ability to degrade the 2,4-D more efficiently by bacterial consortium, and being related to its affinity for chlorinated organic compounds.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
- Ortíz, I, Avila MA, Torres LG. Plaguicidas en México: usos, riesgos y marco regulatorio. Rev Latinoam Biotecnol Amb Algal. 2013; 4: 26-46.
- Del Puerto AM, Suárez S, Palacio DE. Efectos de los plaguicidas sobre el medio ambiente y la salud. Rev Cubana de Higiene y Epidemiología. 2014; 52: 372-387.
- Zhao Y, Pei Y. Risk evaluation of groundwater pollution by pesticides in China: a short review. Procedia Environ Sci. 2012; 13: 1739-1747.
- Socorro J, Durand A, Temime B, Gligorovski S, Wortham H, Quivet E. The persistent organic pollutants (POPs particulate phase: An emerging air quality issue. Sci Rep. 2016; 6: 1-7.
- García C, Rodríguez GD. Problemática y riesgo ambiental por el uso de plaguicidas en Sinaloa. Ra Ximhai. 2012; 8: 1-10.
- Paoletti MG. The role of earthworms for assessment of sustainability and as bioindicators. Agric Ecosyst Environ. 1999; 74: 137-155.
- Martínez A, Pérez ME, Pinto J, Gurrola BA, Osorio AL. Biorremediación de suelo contaminado con hidrocarburos empleando lodos residuales como fuente alterna de nutrientes. Rev Int Contam Ambie. 2011; 27: 241-252.
- Lee SH, Lee S, Kim DY, Kim JG. Degradation characteristics of waste lubricants under different nutrient conditions. J Hazard Mater. 2007; 143: 65-72.
- Garcia-Saavedra Y, Rivera A, Romero O, Hernandez F, Sanchez-Morales P. Bacterial isolations of plots with different agricultural management, metabolic evaluation and viability with different pesticides. ARPN J Agric Biol Sci. 2018; 13: 25-33.
- Han L, Zhao D, Li C. Isolation and 2,4-D-degrading characteristics of Cupriavidus campinensis BJ71. Braz J Microbiol. 2015; 46: 433-441.
- Bourquin AW. Degradation of malathion by salt-marsh microorganisms. Appl Environ Microbiol. 1997; 33: 356-362.
- Ibrahim WM, Karam MA, El-Shahat RM, Adway AA. Biodegradation and utilization of organophosphorus pesticide malathion by Cyanobacteria. Biomed Res Int. 2014; 17: 1-6.
- Xie Z, Xu B, Ding J, Liu L, Zhang X, Li J, Huang Z. Heterologous expression and characterization of a malathion-hydrolyzing carboxylesterase from a thermophilic bacterium Alicyclobacillus tengchongensis. Biotechnol Lett. 2013; 35: 1283-1289.
- Kumar S, Kaushik G, Dar MA, Nimesh S, López-Chuken UJ, Villareal-Chiu JF. Microbial degradation of organophosphate pesticide: A review. Pedosphere. 2018; 28: 190-208.
- Castro GV, Masís MM, Carazo RE, Mora LM, Rodríguez RCE. Impact of oxytetracycline and bacterial bioaugmentation on the efficiency and microbial community structure of a pesticide-degrading biomixture. Environ Sci Pollut Res. 2018; 25: 11787-11799.
- Ramanand K, Panda S, Sharmila M, Adhya TK, Sethunathan N. Development and acclimatization of Carbofuran-degrading soil enrichment cultures at different temperatures. J Agric Food Chem. 1988; 36: 200-205.
- Ramanand K, Sharmila M, Sethunathan N. Mineralization of Carbofuran by a soil bacterium. Appl Environ Microbiol. 1988; 54: 2129-2133.
- Felsot AS, Maddox JV, Bruce W. Enhanced microbial degradation of carbofuran in soils with histories of furadan use. Bull Environ Contam Toxicol. 1981; 26: 781-788.
- Singh BK, Walker A. Microbial degradation of organophosphorus compounds. FEMS Microbiol Rev. 2006; 30: 428-471.
- Loux MM, Liebl RA, Slide FW. Adsorption of imazapyr and imazethapyr on soils, sediments, and selected adsorbents. Weed Science. 1989; 37(1): 712-718.
- Abraham J, Silambarasan S, Logeswari P. Simultaneous degradation of organophosphorus and organochloride pesticides by bacterial consortium. J Taiwan Inst Chem Eng. 2014; 45: 2590-2596.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.