ISSN: 0973-7510
E-ISSN: 2581-690X
Gardenia obtusifolia, a traditional medicinal plant from Southeast Asia, was investigated for its phytochemical composition and bioactivity using methanol extracts and solvent-partitioned fractions from the leaves, fruit pulp, and rind. The plant material was authenticated morphologically and confirmed through matK gene analysis. Phytochemical screening revealed the presence of phenolic compounds, flavonoids, tannins, coumarins, and alkaloids. Among the fractions tested, the rind ethyl acetate fraction exhibited strong antibacterial activity against clinical pathogens, high phenolic content, and potent antioxidant capacity. GC-MS profiling identified key bioactive compounds, including vanillic acid, octadecane, and 5-hydroxymethylfurfural, which likely contribute to the observed bioactivities. Among the solvent fractions, the rind ethyl acetate fraction demonstrated strong anti-tyrosinase activity. This fraction consistently exhibited high phenolic content and potent antioxidant capacity, suggesting that these bioactivities are attributable to key phytochemicals known for antioxidant effects. Purification and GC-MS profiling identified bioactive compounds in the rind ethyl acetate fraction, revealing vanillic acid as a major contributor to antioxidant activity. G. obtusifolia shows great promise for application in the cosmetic industry as a natural ingredient for skin-whitening and anti-aging formulations. However, further in vivo and clinical studies are required, as in vitro results may not directly reflect clinical efficacy.
Gardenia obtusifolia, Anti-tyrosinase, Antibacterial, Antioxidant, Phytochemical Profiling, Ethyl Acetate Fraction, GC-MS Analysis
Within the Rubiaceae family, the genus Gardenia comprises more than 250 species, predominantly found in tropical and subtropical regions. Traditionally, Gardenia species have been used to treat fever, inflammation, infections, and digestive disorders. Phytochemical and pharmacological investigations have revealed a diverse range of secondary metabolites and demonstrated various bioactivities. Numerous Gardenia species have shown promising pharmacological potential. For example, G. sootepensis exhibits antibacterial and cytotoxic activity against small-cell lung carcinoma.1 G. latifolia has been documented to contain antibacterial and antioxidant constituents.2 Regarding cytotoxic potential, extracts from G. ternifolia have demonstrated selective effects against MCF-7 breast cancer cells.3 Additionally, fruit extracts of G. gummifera possess nitric oxide-inhibitory and antioxidant activities,4 and Nauclea latifolia, another member of the Rubiaceae family, has shown anti-tyrosinase properties.5
Despite these reports, many Gardenia species, including Gardenia obtusifolia Roxb. ex Kurz, remain underexplored in terms of their phytochemistry and biological activities. G. obtusifolia, native to Southeast Asia, particularly Thailand and Myanmar, has been the subject of only limited phytochemical investigations. Previous studies have reported a small number of isolated compounds, such as polymethoxylated flavones and triterpenoids, which exhibit cytotoxic and anti-HIV activities.6-8 However, most existing studies have focused predominantly on the cytotoxic effects of leaf and stem extracts, leaving other key biological activities largely unexplored. Traditionally, fresh leaves are used to treat wounds, and the ripe fruit is consumed as a brain tonic to enhance alertness. However, these ethnomedicinal uses remain scientifically underinvestigated. To address these gaps, in this study, we aimed to examine the phytochemical profile and biological activities of the leaves and ripe fruits of G. obtusifolia.
Methanol was used to obtain crude extracts for initial bioactivity screening, which were then subjected to solvent partitioning and purification using column chromatography to enable detailed chemical characterization and confirmation of their biological activities. This study evaluated the antimicrobial, antioxidant, anti-tyrosinase, nitric oxide-inhibitory, and cytotoxic properties and identified the major phytochemical constituents using gas chromatography-mass spectrometry (GC-MS).
DNA barcoding was used to confirm the identity of the specimen as G. obtusifolia, ensuring precise plant species authentication. These findings provide foundational data supporting the ethnomedicinal uses of G. obtusifolia and highlight its potential as a source of bioactive compounds for pharmaceutical, cosmeceutical, and other health-related applications.
Plant samples
Fresh leaves and ripe fruits of G. obtusifolia were collected from the Ong Phra Subdistrict, Dan Chang District, Suphan Buri Province, Thailand (Figure 1) during 2023-2024. The species was taxonomically verified by a botanist at the Bangkok Herbarium (BKF), Department of Agriculture, Bangkok, Thailand. A voucher specimen (BKF No. SN269026) was deposited for reference. The plant materials were collected for two distinct purposes. Ripe fruits and mature leaves were collected from multiple individuals at the collection site for phytochemical extraction and evaluation of their biological activities. For molecular identification, a separate set of young, fresh leaves was collected from a single individual (14°472 243 N, 99°242 553 E), immediately dried in silica gel, and stored for subsequent DNA extraction.
Figure 1. Morphological characteristics of Gardenia obtusifolia: (A) whole plant habit; (B) leaves; (C) flowers; (D) immature fruit with cross-section; (E) mature fruit with cross-section
Genomic DNA isolation and matK gene amplification
Genomic DNA was extracted from fresh young leaves of G. obtusifolia using a modified CTAB method,9 and its quality was verified prior to PCR. The chloroplast matK region was amplified by PCR using specific primers (forward: 5′-ATCCATCTGGAAATCTTAGTTC-3′; reverse: 5′-GTTCTAGCACAAGAAAGTCG-3′). Each 25 µL reaction contained buffer, MgCl2, dNTPs, primers, Taq DNA polymerase (New England Biolabs, Ipswich, MA, USA), and 50 ng of template DNA. The PCR program consisted of an initial denaturation (94 °C, 2 min), 35 cycles of denaturation (94 °C, 1 min), annealing (46 °C, 1 min), extension (72 °C, 2 min), and a final extension (72 °C, 5 min). Amplified products were resolved on 2% agarose gels and purified before sequencing on the FastNGS™ platform. The obtained sequences were aligned and compared with NCBI GenBank references using BLASTn to confirm species identity.
Extraction and fractionation
Fresh leaves and ripe fruits of G. obtusifolia were shade-dried at 50 °C and ground into coarse powder. The fruits were manually separated into pulp and rind before grinding. Each part, leaf (L), pulp (P), and rind (R), 30 g each, was macerated in 300 mL of methanol at a ratio of 1:10 (w/v) at room temperature for seven days to obtain crude methanol extracts (LM, PM, and RM) after filtration and concentration under reduced pressure. Each crude methanol extract was subjected to solvent-partitioning via liquid-liquid extraction. The extract was dissolved in water and successively extracted with hexane (H), dichloromethane (D), ethyl acetate (E), and n-butanol (B) in a 1:1 (v/v) ratio. The residual aqueous layer (W) was also collected, yielding solvent-partitioned fractions designated as LH-LW, PH-PW, and RH-RW for the leaf, pulp, and rind, respectively. The rind ethyl acetate extract (RE) was further separated using column chromatography (stationary phase: silica gel). The mobile phase consisted of solvent mixtures in specific ratios of H:D (4:6) and H:D:E (1:8:1). Thin-layer chromatography (TLC) was used to identify and combine similar fractions, which were then dried and prepared for detailed analysis. The resulting fractions were designated REF1-REF8 (REF = rind ethyl acetate fraction; numbers 1-8 indicate the fraction sequence).
Phytochemical screening
Major phytochemical classes, including alkaloids, tannins, saponins, coumarins, and flavonoids, were detected in crude methanolic extracts using standard qualitative assays with minor modifications.10 Saponins were assessed using froth and hemolysis tests, while the presence of other compounds was determined through colorimetric reactions and precipitation-based methods.
Total phenolic and flavonoid contents
The total phenolic content (TPC) of the extracts was assessed using the Folin–Ciocalteu colorimetric assay according to established protocols,10 and results were expressed as mg of gallic acid equivalents (GAE) per gram of extract, with gallic acid serving as the reference standard. The total flavonoid content (TFC) was measured using a modified aluminum chloride assay,11 and results were expressed as mg quercetin equivalents (QE) per gram of extract.
Antioxidant activity
The antioxidant potential of the extracts was assessed using DPPH and ABTS assays with slight modifications.12,13 For the DPPH assay, a 0.2 mM DPPH solution (Sigma-Aldrich, St. Louis, MO, USA) was prepared, while the ABTS•+ solution was generated from 7 mM ABTS and 2.45 mM potassium persulfate. Absorbance was measured at 517 nm for DPPH and 734 nm for ABTS, with Trolox used as the reference standard in both assays. The percentage of radical scavenging activity was calculated using the following formula:
Scavenging activity (%) = [(Acontrol – Asample) / Acontrol] × 100
where Acontrol and Asample represent the absorbance of the radical solutions in the absence and presence of the extract, respectively. The IC50 value, derived from dose-response curves, was used to compare antioxidant potency.
Tyrosinase inhibitory activity
The inhibitory effect on tyrosinase was evaluated using a modified DOPA-chrome method.14 Briefly, mushroom tyrosinase (654 U/mL; Sigma-Aldrich) was combined with phosphate buffer (pH 6.8) and the extract (1 mg/mL) in a 96-well microplate and incubated in the dark for 10 min. The reaction was initiated by adding 2.5 mM L-DOPA (Sigma-Aldrich) and further incubated for 30 min, after which the absorbance was recorded at 475 nm. Ascorbic acid served as the positive control. The percentage of inhibition was calculated as follows:
Tyrosinase inhibition (%) = [(Acontrol – Asample) / Acontrol] × 100
where Acontrol and Asample represent the absorbance of the reaction mixtures in the absence and presence of the extract, respectively.
Antibacterial activity
Antibacterial activity was evaluated against eight bacterial strains, including six Gram-positive species: Bacillus subtilis TISTR1248, Bacillus cereus DMST5040, Micrococcus luteus TISTR2374, Staphylococcus aureus TISTR746, Staphylococcus epidermidis TISTR2141, and Propionibacterium acnes DMST14916; and two Gram-negative species: Escherichia coli TISTR074 and Pseudomonas aeruginosa TISTR2370. Bacterial isolates were obtained from the Thailand Institute of Scientific and Technological Research (TISTR) and the Department of Medical Sciences, Thailand (DMST). All bacterial strains were cultured in Mueller–Hinton Broth (MHB) at 37 °C for 18-24 hrs under aerobic conditions, except P. acnes, which was cultivated anaerobically in Tryptic Soy Broth (TSB). According to the Clinical and Laboratory Standards Institute (CLSI) guidelines, antibacterial activity was evaluated using the disc diffusion method.15 Sterile paper discs (6 mm in diameter) were impregnated with each extract. Gentamicin (10 µg/disc) served as the positive control, while methanol or methanol:DMSO (100:10, v/v) was used as the negative control.
Nitric oxide (NO) radical scavenging activity
Nitric oxide (NO) radical scavenging activity was determined using the method described by Vinaykumar et al.4 with slight modifications. Briefly, 10 mM sodium nitroprusside (SNP) (Sigma-Aldrich) prepared in phosphate-buffered saline (PBS, pH 7.4) was added to the extract solution (1 mg/mL) and incubated in the dark for 30 min. Griess reagent (Sigma-Aldrich) was then added, followed by an additional 30 min incubation at room temperature. Absorbance was measured at 540 nm, with ascorbic acid used as the positive control. The percentage inhibition was calculated as follows:
Nitric oxide inhibition (%) = [(Acontrol – Asample) / Acontrol] × 100
where Acontrol and Asample are the absorbance values of the reaction mixtures in the absence and presence of the extract, respectively.
MTT cytotoxicity assay
Cytotoxicity was evaluated following ISO 10993-5 using human keratinocytes (HaCaT; iCell-h066), African green monkey kidney cells (Vero; CLS605372), and human breast cancer cells (MCF-7; ATC HTB-22).16 Cells were cultured in DMEM containing 10% FBS and 50 µg/mL gentamicin at 37 °C, in a 5% CO2 atmosphere. Mitomycin C and 1% DMSO were used as positive and negative controls, respectively. After 24 hrs of treatment with the extracts, 1 mg/mL MTT solution (Sigma-Aldrich) was added, and cells were incubated for 4 hrs. Formazan crystals were dissolved in DMSO, and absorbance was measured at 570 nm. Cytotoxicity (%) was calculated as follows:
Cytotoxicity inhibition (%) = [(Acontrol – Asample) / Acontrol] × 100
where Acontrol and Asample represent the absorbance of untreated and treated cells, respectively. IC50 values were determined from dose–response curves using GraphPad Prism 8 (GraphPad Software Inc., San Diego, CA, USA).
Gas chromatography-mass spectrometry (GC-MS) analysis
GC-MS profiling of the extracts was performed using an Agilent 8890 gas chromatograph coupled with a 5977B mass-selective detector (Agilent Technologies, Santa Clara, CA, USA). One microliter of each extract dissolved in methanol was injected in split mode (50:1) at 250 °C. Separation was carried out on an HP-5MS column (30 m × 0.25 mm, 0.25 µm film thickness) using helium as the carrier gas at a constant flow rate of 1.0 mL/min. The oven temperature was initially set to 50 °C (held for 3 min), increased to 250 °C at 10 °C/min, and held for 10 min. The mass spectrometer was operated with an interface temperature of 260 °C, a scan range of m/z 50-550, and a solvent delay of 3.0 min. Compounds were identified by matching the spectra with the NIST Mass Spectral Library (version 20, 2020).
Statistical analysis
Data are presented as mean ± SD of three independent experiments. Statistical significance was assessed using one-way ANOVA followed by Duncan’s multiple range test (DMRT). Pearson’s correlation coefficient (r) was used to evaluate the relationship between total phenolic/flavonoid content with bioactivity, with r = 0.5-0.7 indicating moderated correlation, r = 0.7-0.9 indicating high correlation, and r = 0.9-1.0 indicating very high correlation. Principal component analysis (PCA) was performed to relate GC-MS-identified compounds to bioactivity using SPSS Statistics version 26.0 (IBM Corp., Armonk, NY, USA). Half-maximal inhibitory concentration (IC50) values were calculated using GraphPad Prism 8 (GraphPad Software Inc.). A value of p < 0.05 or p < 0.01 was considered statistically significant.
Morphological and molecular identification of G. obtusifolia
The identity of the collected specimen as G. obtusifolia was confirmed through both morphological and molecular analyses. Morphologically, this species is an evergreen tree reaching 2-8 m in height, with simple, opposite-decussate leaves that are narrowly obovate to elliptical (5-8 cm wide and 13-18 cm long), with entire margins and obtuse apices. Young shoots exude a sticky latex. The flowers are solitary and terminal, white when young and turning yellow with age. The calyx consists of five light-green lobes, and the corolla is funnel-shaped with five fused lobes. The globose fruits have a rough, uneven exocarp; immature fruits are greenish-brown with a hard pericarp (2-4 mm thick) that ripens to yellow, orange, reddish-orange, or brown. The fleshy pulp is orange to yellow-orange and contains numerous ellipsoid seeds of varying sizes.
For molecular confirmation, the chloroplast matK gene was amplified and sequenced. The obtained sequence was subjected to BLAST analysis against the NCBI GenBank database and showed 100% sequence identity with G. obtusifolia (accession no. KJ815831), thereby confirming the taxonomic identity of the specimen.
Phytochemical profile and biological activities of methanol extracts
The extraction yield of the rind methanol (RM) extract was yielded 26.06%, while the leaf (LM) and pulp (PM) extracts yielded 22.22% and 21.54%, respectively.
Phytochemical screening of methanol extracts from G. obtusifolia revealed the presence of key secondary metabolites. The LM, PM, and RM extracts tested positive for tannins, coumarins, and flavonoids. Alkaloids were detected only in the LM extract. Similar classes of secondary metabolites have been reported in other species and are associated with antioxidant, anticancer, anti-inflammatory, and antibacterial activities.17
The quantitative profiles of TPC and TFC in methanol extracts of G. obtusifolia. The LM extract exhibited the highest TPC (134.48 ± 0.58 mg GAE/g extract) and TFC (45.48 ± 0.78 mg QE/g extract). In contrast, the PM extract contained substantially lower levels (TPC: 33.51 ± 1.06 mg GAE/g extract; TFC: 4.15 ± 0.56 mg QE/g extract), whereas the RM extract displayed intermediate values (TPC: 54.00 ± 0.96 mg GAE/g extract; TFC: 13.48 ± 0.78 mg QE/g extract) (n = 3; p < 0.05 ).
The antioxidant activity was evaluated using DPPH and ABTS assays. Among the tested extracts, the LM extract exhibited the strongest activity, with IC50 values of 84.51 µg/mL (DPPH) and 159.83 µg/mL (ABTS). Meanwhile, the RM extract displayed moderate activity (IC50 = 254.94 µg/mL for DPPH), while the PM extract exhibited weaker activity (IC50 = 337.74 µg/mL for DPPH). Both PM and RM extracts showed ABTS IC50 values exceeding 1000 µg/mL, indicating low radical scavenging capacity. All extracts exhibited weaker activity compared to Trolox, which showed IC50 values of 37.65 µg/mL (DPPH) and 53.57 µg/mL (ABTS). In comparison with related species, G. gummifera fruit extract exhibited moderate antioxidant capacity with a DPPH IC50 of 131.11 µg/mL,4 whereas G. latifolia fruit extract demonstrated stronger activity with an IC50 = 65.82 µg/mL.2
At a concentration of 1000 µg/mL, the LM, PM, and RM extracts exhibited less than 50% inhibition of tyrosinase activity, as assessed by the DOPA-chrome assay and nitric oxide scavenging, indicating relatively weak activity under the tested conditions (n = 3; p < 0.05 ). In contrast, fruit extracts from related species demonstrated significantly stronger effects; N. latifolia (Rubiaceae family) fruit extract showed potent tyrosinase inhibition with an IC50 of 124.4 µg/mL,5 while G. gummifera fruit extract exhibited stronger nitric oxide scavenging activity with an IC50 of 175.95 µg/mL.4
At 500 µg/mL, the LM extract exhibited notable cytotoxicity against MCF-7 breast cancer cells as well as against normal Vero and HaCaT cell lines. In contrast, the PM and RM extracts showed lower cytotoxicity across all tested cell lines (n = 3; p < 0.05). Similarly, leaf ethanol extracts of G. latifolia and G. gummifera at the same concentration have exhibited cytotoxicity against MCF-7 cells, with inhibition rates of 74.01 ± 0.9% and 67.76 ± 1.0%, respectively.18 Meanwhile, G. ternifolia leaf extract showed low cytotoxicity against both MCF-7 and non-cancerous L6 cells, with CC50 values exceeding 100 µg/mL.3
At 2000 µg/disc, the LM extract exhibited significant antibacterial activity against B. cereus (11.20 ± 0.61 mm), B. subtilis (7.88 ± 0.26 mm), M. luteus (9.33 ± 0.15 mm), S. aureus (8.25 ± 0.41 mm), S. epidermidis (8.92 ± 0.58 mm), and P. acnes (7.20 ± 0.27 mm). In contrast, the PM and RM extracts showed inhibitory effects only against P. acnes, with inhibition zone diameters of 6.93 ± 0.05 mm and 6.42 ± 0.00 mm, respectively (n = 3; p < 0.05 ). However, all extracts showed weaker activity against the bacterial strains than gentamicin. These results highlighted the antibacterial potential of the LM extract, particularly against clinically relevant Gram-positive pathogens, underscoring its potential for further development as a natural antimicrobial agent.
GC-MS analysis of the LM, PM, and RM extracts identified a diverse array of phytochemicals previously reported to possess antioxidant, antibacterial, anticancer, and anti-inflammatory properties, including 5-hydroxymethylfurfural (19.54% and 50.32%),19 n-hexadecanoic acid (6.19% and 6.89%),20,21 b-Caryophyllene (5.99%),22 and a-humulene (20.39%).23 These compounds likely contribute to the observed bioactivities. Among the extracts, LM contained the highest levels of phenolics and flavonoids, which correlated with its strong antioxidant activity. In contrast, all extracts demonstrated weak anti-tyrosinase and nitric oxide-inhibitory activities, showing less than 50% inhibition at 1000 µg/mL. Although the LM extract exhibited cytotoxicity against MCF-7 breast cancer cells, it lacked selectivity, also affecting normal cell lines.
Among the crude methanol extracts from the leaf (LM), pulp (PM), and rind (RM) of G. obtusifolia, all exhibited varying levels of bioactive compounds and biological activities. The leaf extract showed the highest concentration of key phytochemicals and the most potent bioactivities, followed by the rind and pulp extracts. These results support further solvent partitioning and purification for detailed phytochemical characterization and confirmation of biological activities.
Biological activities of solvent-partitioned fractions
Sequential partitioning of crude methanol extracts from G. obtusifolia leaves, pulp, and rind with hexane, dichloromethane, ethyl acetate, n-butanol, and water yielded fractions that were assessed for biological activity. The fractions were labeled LH to LW for the leaf, PH to PW for the pulp, and RH to RW for the rind. The extraction yields varied among the fractions, as shown in Table 1.
Table (1):
Yield, TPC, TFC, antioxidant (DPPH and ABTS), anti-tyrosinase, nitric oxide (NO) inhibition, and cytotoxic activities of solvent-partitioned fractions from methanol extracts of Gardenia obtusifolia
| Codes extracts | Yield (%:w/w) | TPC | TFC | Antioxidant activity | Anti-tyrosinase activity | Nitric oxide inhibition | Cytotoxic activity | |||
|---|---|---|---|---|---|---|---|---|---|---|
| (mg GAE/g extract) | (mg QE/g extract) | IC50 (µg/mL) | Inhibition (%) at 1000 µg/mL | IC50 (µg/mL) | ||||||
| DPPH | ABTS | Vero | HaCaT | MCF-7 | ||||||
| LH | 6.67 | 117.02f ± 0.66 | 69.30b ± 0.56 | >400 | >400 | 27.58i ± 0.85 | 27.70f ± 0.38 | <250 | 334.42 | <250 |
| LD | 1.45 | 140.42d ± 0.82 | 103.19a ± 0.80 | 186.58 | 179.93 | 39.12g ± 0.42 | 32.19e ± 0.50 | 365.81 | 490.95 | 268.07 |
| LE | 22.50 | 203.27a ± 1.73 | 32.26d ± 1.22 | 18.73 | 50.51 | 74.45b ± 0.38 | 46.39a ± 0.90 | <250 | 366.67 | <250 |
| LB | 8.71 | 69.18g ± 0.30 | 4.44j ± 0.22 | 55.10 | 296.65 | 42.80f ± 0.74 | 46.53a ± 1.07 | 639.69 | 380.23 | 263.50 |
| LW | 19.35 | 26.78i ± 0.30 | 2.52k ± 0.13 | 224.54 | >400 | 14.52l ± 0.53 | 36.65c ± 0.49 | >1000 | >1000 | 347.38 |
| PH | 6.32 | 12.57m ± 0.44 | 10.30h ± 0.68 | >400 | >400 | 14.87l ± 0.85 | 24.23g ± 1.06 | >1000 | >1000 | >1000 |
| PD | 0.68 | 37.13h ± 0.10 | 53.56c ± 0.80 | >400 | >400 | 48.72e ± 0.30 | 32.07e ± 0.37 | 774.47 | 751.64 | <250 |
| PE | 0.52 | 138.13e ± 1.47 | 18.44g ± 0.44 | 35.52 | 181.21 | 63.73c ± 0.87 | 41.27b ± 0.88 | 298.65 | 533.82 | <250 |
| PB | 7.36 | 19.59j ± 0.18 | 6.00i ± 0.26 | >400 | >400 | 24.19j ± 0.87 | 25.18g ± 0.09 | >1000 | 942.17 | 981.36 |
| PW | 33.81 | 18.83jk ± 0.20 | 5.11ij ± 0.56 | >400 | >400 | 12.38m ± 0.48 | 31.63e ± 0.90 | >1000 | >1000 | 878.06 |
| RH | 2.18 | 17.72k ± 0.27 | 25.56f ± 1.11 | >400 | >400 | 18.77k ± 0.87 | 18.08h ± 0.47 | 372.44 | <250 | 441.51 |
| RD | 0.80 | 159.91c ± 1.00 | 30.30e ± 0.38 | 88.06 | 161.51 | 56.12d ± 1.04 | 33.90d ± 1.23 | <250 | <250 | <250 |
| RE | 23.02 | 185.36b ± 0.27 | 25.63f ± 1.56 | 43.57 | 138.35 | 76.01a ± 0.73 | 40.78b ± 0.75 | 274.38 | 434.45 | <250 |
| RB | 0.47 | 37.66h ± 0.51 | 10.52h ± 0.26 | >400 | >400 | 37.46h ± 0.59 | 31.32e ± 1.25 | >1000 | 936.13 | 299.90 |
| RW | 35.13 | 14.39l ± 0.62 | 5.26ij ± 0.68 | >400 | >400 | 12.35m ± 0.72 | 18.86h ± 0.56 | >1000 | >1000 | 398.83 |
| Trolox | – | – | – | 37.65 | 53.70 | – | – | – | – | – |
Abbreviations: L = leaf; P = pulp; R = rind; H = hexane; D = dichloromethane; E = ethyl acetate; B = n-butanol; W = water.
TPC = Total phenolic content; TFC = Total flavonoid content; IC50 = Half-maximal inhibitory concentration
Data are expressed as mean ± SD (n = 3). Different superscript letters (a-m) within the same column indicate statistically significant differences at P < 0.05
TPC and TFC of the solvent-partitioned fractions ranged from 12.57 to 203.27 mg GAE/g extract and 2.52 to 103.19 mg QE/g extract, respectively (Table 1). Among these, the leaf ethyl acetate (LE) fraction exhibited the highest TPC (203.27 ± 1.73) and the leaf dichloromethane (LD) fraction exhibited the highest TFC (103.19 ± 0.80). In contrast, the lowest TPC and TFC values were observed in the pulp hexane (PH; 12.57 ± 0.44 mg GAE/g extract) and leaf aqueous (LW; 2.52 ± 0.13 mg QE/g extract) fractions, respectively. These results reflect the influence of solvent polarity, with ethyl acetate (intermediate polarity) yielding the richest phenolic content, consistent with reports on G. jasminoides.11 However, no direct correlation between TPC and TFC was observed, suggesting an independent distribution based on plant part and solvent system, as previously noted in other Gardenia species.24
DPPH and ABTS assays were used to assess the antioxidant potential of G. obtusifolia fractions at concentrations ranging from 12.5 to 400 µg/mL (Table 1). The LE fraction exhibited the strongest activity, with IC50 values of 18.73 µg/mL (DPPH) and 50.51 µg/mL (ABTS), surpassing Trolox and representing the only fraction with an ABTS IC50 below 100 µg/mL. The PE (35.52 µg/mL), RE (43.57 µg/mL), LB (55.10 µg/mL), and RD (88.06 µg/mL) fractions also showed strong DPPH scavenging activity. Other fractions displayed moderate to low activities (IC50 >100 µg/mL). These results highlighted the efficiency of ethyl acetate in extracting antioxidant-rich compounds from different tissues. The high activity of LE fraction suggests the presence of potent phenolic or polyphenolic antioxidants. Similar findings have been reported for other Gardenia species. For example, in G. jasminoides, the ethyl acetate extract exhibited stronger antioxidant activity (IC50 = 62.50 ± 1.45 ppm) than the aqueous fraction (IC50 = 435.40 ± 5.40 ppm).11 Likewise, the ethyl acetate extract of G. sootepensis leaves showed even greater potency, with an IC50 of 6.36 ± 0.02 mg/mL.1 These data support ethyl acetate as an effective solvent for extracting antioxidant compounds in Gardenia species. However, the lack of a consistent correlation between TPC, TFC, and IC50 values suggests that other compounds, such as non-flavonoid phenolics (e.g., tannins, phenolic acids), may also contribute significantly.25 These findings underscore the need for comprehensive phytochemical profiling to evaluate antioxidant potential accurately.
Tyrosinase inhibition by G. obtusifolia fractions at 1000 µg/mL ranged from 12.35% to 76.01% (Table 1). The highest activities were observed in the RE (76.01 ± 0.73%), LE (74.45 ± 0.38%), and the PE (63.73 ± 0.87%) fractions, indicating that ethyl acetate effectively concentrates tyrosinase-inhibiting constituents. In contrast, the aqueous fractions exhibited minimal activity (<15%), which was consistent with their low phenolic content and weak antioxidant activity. These results suggested that moderately polar compounds, possibly non-flavonoid phenolics or related polyphenols, primarily contributed to the observed inhibitory effects.
The antibacterial activity of G. obtusifolia fractions (2 mg/mL) was evaluated using the disc diffusion method (Table 2). The LD and RD fractions displayed the broadest spectrum of activity, inhibiting six bacterial strains: B. cereus, B. subtilis, M. luteus, P. acnes, S. aureus, and S. epidermidis. The LD fraction showed the most significant inhibition of B. cereus, whereas the RD fraction was more effective against other strains. The LE fraction inhibited the growth of all five strains, highlighting the antimicrobial potential of the semi-polar constituents. However, all extracts showed weaker activity against the bacterial strains than gentamicin. These results are consistent with previous findings in G. sootepensis, where hexane and ethyl acetate leaf extracts were active against S. aureus.1 The observed activities may be attributed to lipophilic compounds such as terpenoids and phenolic derivatives, often concentrated in the dichloromethane and ethyl acetate fractions.
Table (2):
Antibacterial activity of solvent-partitioned fractions of Gardenia obtusifolia (2000 µg/disc) against six bacterial strains using the disc diffusion method
| Codes extracts | Mean diameter of inhibition zone (mm) | |||||
|---|---|---|---|---|---|---|
| B. cereus | B. subtilis | M. luteus | S. aureus | S. epidermidis | P. acne | |
| LH | 7.43cd ± 0.29 | 0.00 | 6.70c ± 0.20 | 7.27c ± 0.66 | 6.48c ± 0.24 | 6.43c ± 0.29 |
| LD | 13.48a ± 0.81 | 8.85b ± 0.79 | 7.62bc ± 0.28 | 8.85b ± 0.71 | 9.02b ± 0.73 | 9.88b ± 0.53 |
| LE | 9.85b ± 0.13 | 7.58c ± 0.40 | 11.65a ± 1.19 | 6.90c ± 0.52 | 7.28c ± 0.28 | 0.00 |
| LB | 7.73c ± 0.47 | 0.00 | 8.10bc ± 0.10 | 0.00 | 0.00 | 0.00 |
| LW | 6.62d ± 0.10 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| PH | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| PD | 7.17cd ± 0.76 | 6.42d ± 0.24 | 8.93b ± 1.19 | 0.00 | 0.00 | 6.65c ± 0.07 |
| PE | 6.58d ± 0.33 | 0.00 | 0.00 | 0.00 | 0.00 | 6.79c ± 0.10 |
| PB | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| PW | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| RH | 6.82cd ± 0.10 | 6.73cd ± 0.29 | 0.00 | 9.22b ± 0.26 | 0.00 | 0.00 |
| RD | 9.12b ± 0.82 | 12.55a ± 0.79 | 12.15a ± 1.23 | 11.82a ± 0.68 | 11.93a ± 0.90 | 11.78a ± 1.42 |
| RE | 6.97cd ± 0.57 | 6.30d ± 0.56 | 0.00 | 7.47c ± 0.31 | 0.00 | 7.09c ± 0.19 |
| RB | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| RW | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 |
| Gentamicin | 23.26 ± 1.05 | 22.77 ± 1.42 | 24.40 ± 0.27 | 22.30 ± 0.90 | 24.96 ± 1.81 | 27.14 ± 1.30 |
Abbreviations: L = leaf; P = pulp; R = rind; H = hexane; D = dichloromethane; E = ethyl acetate; B = n-butanol; W = water.
Data are expressed as mean ± SD (n = 3). Different superscript letters (a–d) within the same column indicate statistically significant differences at P < 0.05
The nitric oxide (NO) inhibitory activity of G. obtusifolia fractions at 1000 µg/mL is summarized in Table 1. The LB fraction exhibited the highest inhibition (46.53 ± 1.07%), followed by the LE (46.39 ± 0.90%), the PE (41.27 ± 0.88%), and the RE (40.78 ± 0.75%) fractions. The remaining fractions showed moderate to low inhibition (18.08%-36.65%). These results indicate that semi-polar solvents such as ethyl acetate and n-butanol effectively concentrate NO-scavenging constituents with potential anti-inflammatory properties.
The cytotoxicity of G. obtusifolia fractions was assessed against Vero, HaCaT, and MCF-7 cell lines after 24 hrs exposure (250-1000 µg/mL), with IC50 values interpreted as follows: less than 250 µg/mL indicating high cytotoxicity and greater than 1000 µg/mL indicating low or negligible toxicity (Table 1). Six fractions, LH, LE, PD, PE, RD, and RE, exhibited potent activity against MCF-7 cells (IC50 < 250 µg/mL), indicating potential anticancer properties.
The RH and RD fractions were active against HaCaT cells, whereas LH, LE, and RD fractions inhibited Vero cell growth. In contrast, water and hexane fractions (e.g., LW, PH, PB, PW, RB and RW) showed weak or no cytotoxicity (IC50 >1000 µg/mL). These results suggest that the dichloromethane and ethyl acetate fractions are particularly effective in concentrating the cytotoxic constituents. Although the selective activity toward MCF-7 cells is promising, further studies on normal cell selectivity are warranted. Comparable findings have been reported: 5,3’-dihydroxy-3,6,7,8,4’-pentamethoxyflavone from G. obtusifolia leaves inhibited MCF-7 Neo and MCF-7 HER2 cells.7 Similarly, G. ternifolia ethyl acetate leaf extract exhibited cytotoxicity against MCF-7 and L6 cells (CC50 = 45.44 and 63.21 µg/mL, respectively.3 In contrast, G. sootepensis leaf extracts (n-hexane and ethyl acetate) showed no cytotoxic effect on MCF-7 cells,1 highlighting variation by species, solvent, and compound profile.
The relationship between the phytochemical content and biological activities of the solvent-partitioned fractions from G. obtusifolia was evaluated using Pearson’s correlation analysis. The total phenolic content of solvent-partitioned fractions from G. obtusifolia showed a significantly high negative correlation (DPPH assay, r = -0.842; p < 0.01) and a very high negative correlation (ABTS assay, r = -0.988; p < 0.01) with antioxidant activity. In contrast, TPC showed a significantly high positive correlation with tyrosinase inhibition (r = 0.837; p < 0.01) but no significant correlation with nitric oxide inhibitory activity. TPC was positively correlated with antibacterial activity, showing a moderate positive correlation with B. cereus (r = 0.677; p < 0.05) and a significantly high positive correlation with S. aureus (r = 0.768; p < 0.05). At the same time, the total flavonoid content showed no significant correlation with antioxidant activity, anti-tyrosinase activity, or nitric oxide inhibition. However, TFC demonstrated a high positive correlation with the antibacterial activity against B. cereus (r = 0.720; p < 0.05).
GC-MS analysis of the solvent-partitioned fractions from G. obtusifolia revealed a variety of volatile and semi-volatile compounds (Tables 3-5). Fractions obtained using hexane (LH, PH, and RH) and water (LW, PW, and RW) were excluded from the tables because of their low bioactivities. For ease of comparison, the GC-MS results were organized according to plant tissue origin: leaf-derived, pulp-derived, and rind-derived fractions.
Table (3):
GC-MS profiles of dichloromethane, ethyl acetate, and n-butanol fractions derived from Gardenia obtusifolia leaves, including retention time, compound name, and relative peak area (%)
| Extracts | Retention Time (min) | Compound Name | % Peak Area |
|---|---|---|---|
| LD | 5.01 | Cyclopentanol, 1-methyl | 8.71 |
| 7.04 | 2-Hexanone, 6-hydroxy | 9.10 | |
| 8.57 | 2-Penten-1-ol, 2-methyl-, (Z)- | 13.96 | |
| 11.89 | Benzoic acid | 41.14 | |
| 13.08 | Benzeneacetic acid | 5.79 | |
| 15.80 | trans-Cinnamic acid | 2.69 | |
| 16.42 | Methylparaben | 2.05 | |
| 18.40 | 8-Nonenoic acid | 3.66 | |
| 21.68 | 2-Butanone, 4-(2,6,6-trimethyl-1-cyclohexen-1-yl)- | 5.21 | |
| 23.62 | Loliolide | 7.70 | |
| LE | 8.79 | Phenol | 5.65 |
| 9.14 | Decane | 4.13 | |
| 12.41 | Hydroxylamine, O-decyl | 9.78 | |
| 15.39 | Tetradecane | 14.07 | |
| 20.29 | Hexadecane | 10.24 | |
| 24.03 | Octadecane | 16.29 | |
| 24.42 | Isopropyl myristate | 8.00 | |
| 26.10 | n-Hexadecanoic acid | 9.96 | |
| 26.58 | Eicosane | 9.52 | |
| 28.44 | Hexadecanamide | 12.36 | |
| LB | 8.78 | Phenol | 9.66 |
| 12.41 | Hydroxylamine, O-decyl | 13.46 | |
| 15.39 | Tetradecane | 17.26 | |
| 20.29 | Hexadecane | 13.18 | |
| 26.09 | n-Hexadecanoic acid | 13.18 | |
| 28.44 | Hexadecanamide | 33.26 |
Abbreviations: LD = leaf dichloromethane; LE = leaf ethyl acetate; LB = leaf n-butanol fractions
Table (4):
GC-MS profiles of dichloromethane, ethyl acetate, and n-butanol fractions derived from Gardenia obtusifolia pulp, including retention time, compound name, and relative peak area (%)
| Extracts | RetentionTime (min) | Compound Name | % Peak Area |
|---|---|---|---|
| PD | 7.04 | 2-Hexanone, 6-hydroxy | 1.22 |
| 11.30 | Butanedioic acid, hydroxy-, dimethyl ester | 1.45 | |
| 11.80 | Benzoic acid | 1.58 | |
| 12.28 | Ethanol, 2-(2-butoxyethoxy)- | 17.47 | |
| 12.41 | Thiophene, 2,3-dihydro- | 7.47 | |
| 12.81 | 5-Hydroxymethylfurfural | 6.16 | |
| 12.85 | (1R,2S,4R)-2,7,7-Trimethylbicyclo[2.2.1]heptan-2-ol | 6.43 | |
| 13.79 | Salicylic acid | 1.33 | |
| 14.13 | 2-Methoxy-4-vinylphenol | 1.84 | |
| 14.80 | Limonene oxide, cis- | 6.11 | |
| 15.50 | 3-Hydroxy-4-methoxybenzaldehyde, O-ethoxycarbonyl- | 16.98 | |
| 15.65 | trans-Chrysanthenol | 7.16 | |
| 15.83 | 2-Propenoic acid, 3-phenyl- | 3.91 | |
| 15.88 | (+)-cis-Verbenol, acetate | 4.87 | |
| 16.53 | Isolongifolol | 1.92 | |
| 19.21 | Vanillic acid | 3.05 | |
| 21.70 | Benzaldehyde, 4-hydroxy-3,5-dimethoxy- | 1.73 | |
| 26.10 | n-Hexadecanoic acid | 0.83 | |
| 26.21 | 1,2-Benzenedicarboxylic acid, butyl octyl ester | 2.17 | |
| 35.87 | Decanedioic acid, bis(2-ethylhexyl) ester | 5.47 | |
| PE | 7.38 | Ethanol, 2-butoxy- | 4.87 |
| 8.78 | Phenol | 2.29 | |
| 10.74 | Butanedioic acid, monomethyl ester | 2.90 | |
| 12.07 | Levomenthol | 2.02 | |
| 12.41 | Hydroxylamine, O-decyl- | 3.69 | |
| 12.50 | 3-Hydroxydecanoic acid | 5.21 | |
| 12.80 | 5-Hydroxymethylfurfural | 45.01 | |
| 19.14 | Vanillic acid | 2.76 | |
| 20.29 | Hexadecane | 5.94 | |
| 24.03 | Octadecane | 6.24 | |
| 24.42 | Isopropyl myristate | 4.60 | |
| 26.10 | n-Hexadecanoic acid | 5.81 | |
| 26.58 | Eicosane | 5.11 | |
| PB | 12.41 | Hydroxylamine, O-decyl- | 27.13 |
| 15.39 | Hydroxylamine, O-decyl- | 37.50 | |
| 20.29 | Hexadecane | 35.38 |
Abbreviations: PD = pulp dichloromethane; PE = pulp ethyl acetate; PB = pulp n-butanol fractions
Table (5):
GC-MS profiles of dichloromethane, ethyl acetate, and n-butanol fractions derived from Gardenia obtusifolia rind, including retention time, compound name, and relative peak area (%)
| Extracts | RetentionTime (min) | Compound Name | % Peak Area |
|---|---|---|---|
| RD | 12.27 | 2-Propanol, 1-(2-butoxyethoxy)- | 4.06 |
| 12.80 | 5-Hydroxymethylfurfural | 2.44 | |
| 15.49 | Vanillin | 15.96 | |
| 19.16 | Vanillic acid | 12.14 | |
| 21.70 | Benzaldehyde, 4-hydroxy-3,5-dimethoxy- | 6.50 | |
| 25.73 | Hexadecanoic acid, methyl ester | 7.94 | |
| 26.10 | n-Hexadecanoic acid | 17.30 | |
| 27.62 | Linoelaidic acid | 2.46 | |
| 27.68 | 9,12-Octadecadienoyl chloride, (Z,Z)- | 3.68 | |
| 28.01 | Oleic acid | 6.70 | |
| 28.23 | Octadecanoic acid | 8.55 | |
| 35.86 | Decanedioic acid, bis(2-ethylhexyl) ester | 12.28 | |
| RE | 9.07 | Cyclohexene, 1,5,5-trimethyl-3-methylene- | 2.34 |
| 10.74 | Butanedioic acid, ethyl methyl ester | 3.61 | |
| 11.68 | Camphor | 1.73 | |
| 12.07 | Levomenthol | 3.43 | |
| 12.34 | Resorcinol | 2.84 | |
| 12.48 | Malic acid | 4.87 | |
| 12.80 | 5-Hydroxymethylfurfural | 3.99 | |
| 13.39 | Resorcinol | 3.40 | |
| 13.87 | ortho tert-Butyl cyclohexyl acetate | 3.17 | |
| 19.18 | Vanillic acid | 21.76 | |
| 20.29 | Nonadecane | 5.78 | |
| 24.04 | Octadecane | 7.01 | |
| 24.42 | Isopropyl myristate | 7.02 | |
| 24.92 | Luvangetin | 4.58 | |
| 26.10 | n-Hexadecanoic acid | 10.82 | |
| 27.48 | 1-Octadecanol | 5.64 | |
| 29.79 | 2-Propenoic acid, 3-(4-methoxyphenyl)-, 2-ethylhexyl ester | 8.00 | |
| RB | 25.72 | 7,9-Di-tert-butyl-1-oxaspiro(4,5)deca-6,9-diene-2,8-dione | 46.56 |
| 26.10 | n-Hexadecanoic acid | 53.44 |
Abbreviations: RD = rind dichloromethane; RE = rind ethyl acetate; RB = rind n-butanol fractions
Sixteen compounds were identified in the LH fraction, with humulenol-II (43.03%), 14-hydroxycaryophyllene (11.21%), n-hexadecanoic acid (11.27%), and squalene (6.06%) being the predominant constituents. The LD fraction contained ten compounds, primarily benzoic acid (41.14%), 2-penten-1-ol, 2-methyl-, (Z)- (13.96%), and 2-hexanone, 6-hydroxy (9.10%). The LE fraction also yielded ten compounds, with octadecane (16.29%), tetradecane (14.07%), and hexadecanamide (12.36%) as the major components. The LB fraction shared several components with LE, and was dominated by hexadecanamide (33.26%) and tetradecane (17.26%). In contrast, the aqueous fraction (LW) showed the lowest chemical diversity, with four detectable compounds: 9-octadecenamide (Z)- (48.18%), hexadecanamide (39.08%), dibutyl phthalate (7.95%), and citrusol (4.80%). A complete list of the compounds is provided in Table 3.
The GC-MS analysis of the five pulp-derived fractions revealed distinct chemical profiles (Table 4). The PH fraction contained 12 compounds, which were predominantly fatty acid esters, including ethyl oleate (34.24%), methyl linoleate (31.50%), and methyl palmitate (21.89%). The PD fraction exhibited the highest compound diversity with 20 constituents, including ethanol, 2-(2-butoxyethoxy)-(17.47%), 3-hydroxy-4-methoxybenzaldehyde, O-ethoxycarbonyl- (16.98%), trans-chrysanthenol (7.16%), and limonene oxide, cis- (6.11%). The PE fraction contained 13 compounds, notably 5-hydroxymethylfurfural (45.01%) and octadecane (6.24%). In contrast, the PB fraction contained only two detectable compounds: hydroxylamine O-decyl ( 64.63%) and hexadecane (35.38%). The PW fraction yielded six polar constituents, namely stigmasterol (33.76%), quinic acid (23.07%), and dl-2-amino-1-pentanol (13.43%).
GC-MS analysis of the rind-derived fractions, with the complete compound lists provided in Table 5, revealed distinct chemical compositions. The RH fraction contained ten compounds, dominated by n-hexadecanoic acid (32.40%) and cis-vaccenic acid (22.28%). The RD fraction contained 12 compounds: n-hexadecanoic acid (17.30%), vanillin (15.96%), decanedioic acid, bis(2-ethylhexyl) ester (12.28%), and vanillic acid (12.14%). The RE fraction showed the highest compound diversity (17 constituents), with vanillic acid (21.76%) and n-hexadecanoic acid (10.82%) being the main components. In contrast, the RB and RW fractions exhibited limited diversity, each containing only two major constituents. RB contained n-hexadecanoic acid (53.44%) and 7,9-di-tert-butyl-1-oxaspiro(4,5)deca-6,9-diene-2,8-dione (46.56%), whereas RW contained hexadecanoic acid, 2-hydroxy-1-(hydroxymethyl)ethyl ester (75.44%) and n-hexadecanoic acid (24.56%).
Overall, GC–MS profiling combined with bioactivity data indicated that moderately non-polar solvents, particularly dichloromethane and ethyl acetate, were the most effective in extracting bioactive and related constituents from the leaf-, pulp-, and rind-derived fractions of G. obtusifolia. Several key constituents identified in the solvent-extracted fractions of G. obtusifolia exhibit well-documented pharmacological properties. Humulenol-II, a sesquiterpene prevalent in essential oil-bearing plants, exhibits anticancer, anti-inflammatory, and antimicrobial effects.23 Benzoic acid, a natural preservative with established safety, demonstrates broad-spectrum antibacterial, antifungal, and anticancer properties.26 Vanillin has been reported to antibacterial, antioxidant, and anticancer activities.27 Hexadecanamide has been reported to exert anti-inflammatory effects and reinforce epithelial barrier integrity in mastitis models.28 Similarly, 9-octadecenamide (Z) is reported to suppress inflammatory responses,29 while ethyl oleate, previously isolated from Phyllanthus, possesses notable antibacterial activity.30 The presence of these bioactive compounds provides a chemical basis for the observed pharmacological effects and underscores the therapeutic potential of G. obtusifolia fractions.
Several identified compounds have been well documented for their bioactivity and may underlie the observed antioxidant and antimicrobial effects. 5-Hydroxymethylfurfural (5-HMF), a common thermal degradation product, exhibits antioxidant and cytotoxic activity, particularly against melanoma cells.19 Hydroxylamine, O-decyl-, has been reported to possess antibacterial properties.31 Octadecane contributes to antioxidant and anti-inflammatory activities,32 while stigmasterol, a phytosterol isolated from Sargassum aggregatum, is known for its antioxidant potential.33 One of the most consistently detected constituents, n-hexadecanoic acid (palmitic acid), is widely reported for its cytotoxic, antioxidant, and antimicrobial actions, and it is commonly found in medicinal plants and marine algae.20,21 Vanillic acid, a benzoic acid derivative, exhibits antioxidant, anti-inflammatory, and neuroprotective effects.34 Decanedioic acid, bis(2-ethylhexyl) ester, has been reported to have anticancer, antioxidant, and antibacterial activities.35 7,9-di-tert-butyl-1-oxaspiro(4,5)deca-6,9-diene-2,8-dione exhibits antioxidant activity.36 Additionally, hexadecanoic acid, 2-hydroxy-1-(hydroxymethyl)ethyl ester, has demonstrated antioxidant, anti-inflammatory, and anthelmintic activities.32 The presence of these bioactive metabolites highlights the therapeutic partitioned fractions from methanol extracts of Gardenia obtusifolia.
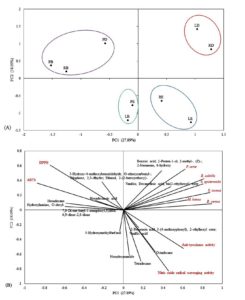
Principal component analysis (PC1 27.89%, PC2 18.09%) (Figure 2) revealed a relationship between GC-MS profiling and the biological activity of the solvent-partitioned fractions from G. obtusifolia. The PCA score plot (Figure 2A) revealed that the solvent-partitioned fractions clustered distinctly into four main groups: (1) PB, RB, and PD; (2) LD and RD; (3) LB and PE; and (4) LE and RE. This grouping reflects variations in chemical composition and biological activities. Based on the loading plot (Figure 2B), the top three compounds were selected for the correlation analysis based on the correlations. Antibacterial activity positively correlated with benzoic acid, 2-penten-1-ol, 2-methyl-, (Z)-, 2-hexanone, 6-hydroxy, vanillin, and decanedioic acid, bis(2-ethylhexyl) ester, indicating that these compounds potently inhibited bacterial growth. In contrast, 2-propenoic acid, 3-(4-methoxyphenyl)-, 2-ethylhexyl ester, vanillic acid, octadecane, tetradecane, and hexadecanamide were positively correlated with anti-tyrosinase and nitric oxide-scavenging activities. Furthermore, the PCA loading plot revealed a negative correlation between these metabolites and antioxidant activity (IC50 values), suggesting that these compounds contribute to the antioxidant activity.
Figure 2. Principal Component Analysis (PCA) of GC-MS compounds and biological activities of solvent-partitioned fractions of Gardenia obtusifolia: (A) Score plot of solvent-partitioned fractions; (B) Loading plot showing the correlation between GC-MS compounds with antioxidant (DPPH and ABTS), anti-tyrosinase, nitric oxide (NO) scavenging, and antibacterial activities
As these antioxidant compounds were found in the ethyl acetate extract, the results clearly indicated that the rind ethyl acetate (RE) fraction exhibited the most potent tyrosinase inhibitory activity, along with its potent antioxidant capacity. Consequently, this fraction was further purified by column chromatography to confirm the antioxidant and anti-tyrosinase activities of the obtained subfractions.
Antioxidant and Anti-tyrosinase activities of fractions isolated from the rind ethyl acetate fraction
The antioxidant activity was evaluated using DPPH and ABTS assays at concentrations ranging from 62.5 to 500 µg/mL (Table 6). Among the tested extracts, the REF8 fraction exhibited the strongest activity, with IC50 values of 116.81 µg/mL (DPPH) and 305.46 µg/mL (ABTS). In contrast, the REF1 to REF7 fraction showed low activity (IC50 values >500 µg/mL). However, all the extracts exhibited lower activity than Trolox.
Tyrosinase inhibition of REF1 to REF8 fraction at 1000 µg/mL ranged from 3.23% to 79.03% (Table 6). The REF8 exhibited the highest inhibition (79.03 ± 0.18%). The remaining fractions showed moderate to low inhibition (3.23% to 25.99%).
Table (6):
Antioxidant (DPPH and ABTS) and anti-tyrosinase activities of fractions isolated from the RE fraction
| Codes extracts | Antioxidant activity | Anti-tyrosinase activity | |
|---|---|---|---|
| IC50 (µg/mL) | Inhibition (%) at 1000 µg/mL | ||
| DPPH | ABTS | ||
| REF1 | >500 | >500 | 3.23g ± 0.72 |
| REF2 | >500 | >500 | 5.38f ± 1.43 |
| REF3 | >500 | >500 | 14.52d ± 0.54 |
| REF4 | >500 | >500 | 10.22e ± 0.74 |
| REF5 | >500 | >500 | 15.89d ± 1.62 |
| REF6 | >500 | >500 | 19.18c ± 0.90 |
| REF7 | >500 | >500 | 25.99b ± 0.18 |
| REF8 | 116.81 | 305.46 | 79.03a ± 0.18 |
| Trolox | 37.65 | 53.70 | – |
Abbreviations: R = rind; E = ethyl acetate; F = fraction. IC₅₀ = Half-maximal inhibitory concentration. Data are expressed as mean ± SD (n = 3). Different superscript letters (a–g) within the same column indicate statistically significant differences at p < 0.05
GC-MS analysis of the fractions isolated from the RE fraction revealed that the fractions obtained from REF1 to REF7 were excluded from the tables owing to their low bioactivity. However, the highest activity was observed in the REF8 fraction (Table 7). The most notable finding was the presence of vanillic acid (65.21%), followed by 9-Phenyl-2,3,4a,4-tetrahydro-1H-benz[f]isoindol-1-one (20.28%), and phenol, 2-methoxy- (7.44%). Vanillic acid has been reported to possess antioxidant properties and to act as an effective tyrosinase inhibitor.34,37 Similarly, phenol, 2-methoxy- exhibits antioxidant activity and inhibits the COX-2 enzyme.38 Besides, the chemical composition of the major compounds reveals the presence of an antimicrobial compound, benzoic acid. Trace amounts (<1%) were detected. Although, benzoic acid contributes to the antibacterial activity, its limited abundance suggests that the antibacterial potential of this fraction is weak and may not reach the effective threshold required for pathogen inhibition.
Table (7):
GC-MS profiles of fractions isolated from the RE fraction, including retention time, compound name, and relative peak area (%)
| Codes extracts | RetentionTime (min) | Compound Name | % Peak Area |
|---|---|---|---|
| REF8 | 6.69 | Carbolic acid | 1.38 |
| 8.58 | Phenol, 2-methoxy- | 7.44 | |
| 9.93 | Benzoic acid | 0.74 | |
| 10.67 | Benzofuran, 2,3-dihydro- | 0.78 | |
| 15.57 | Vanillic acid | 65.21 | |
| 25.73 | Hexanedioic acid, bis (1,3-dimethylbutyl) ester | 4.16 | |
| 26.05 | 9-Phenyl-2,3,4a,4-tetrahydro-1H-benz[f]isoindol-1-one | 20.28 |
Abbreviations: REF8 = rind ethyl acetate fraction, fraction sequence 8
The ethyl acetate fraction isolated from the rind of G. obtusifolia exhibited key antioxidant and anti-tyrosinase activities. In addition, the fruit of G. obtusifolia has traditionally been used in folk medicine as a brain tonic to improve alertness. Although its traditional use is mainly for its neurological benefits, this study found that the fruit also showed notable biological activities, particularly strong antioxidant properties. These antioxidant effects may help reduce oxidative stress in nerve cells, which could indirectly support its traditional use for brain nourishment and overall health, highlight the plant’s potential for natural cosmetic and pharmaceutical applications, and support its traditional use for brain nourishment and overall health. However, further investigations are required to evaluate the cytotoxicity of the ethyl acetate fraction isolated from the rind. Subsequently, in vivo and clinical studies are required, as the in vitro results may not directly reflect clinical efficacy.
This study presents the first comprehensive comparison of the phytochemical composition and bioactivities of solvent-partitioned fractions of Gardenia obtusifolia leaves, pulp, and rind. Among the fractions tested, the methanol, ethyl acetate, and dichloromethane fractions showed the most potent antioxidant and antibacterial activities. However, a correlation with high phenolic content was observed only in the ethyl acetate and dichloromethane fractions. Notably, the ethyl acetate fraction exhibited antibacterial activity against clinical pathogens, including B. cereus, S. epidermidis, and M. luteus, as well as strong antioxidant and anti-tyrosinase activities, highlighting its potential therapeutic applications. The GC-MS analysis identified several key bioactive constituents, including benzoic acid, vanillic acid, hexadecanamide, and octadecane, which may have contributed to the observed effects. The selective cytotoxicity against MCF-7 cells underscores the anticancer potential of specific fractions. The ethyl acetate fraction of the rind contained compounds with strong anti-tyrosinase and antioxidant activities. These findings suggest that G. obtusifolia is a promising source of natural bioactive agents with pharmaceutical, cosmetic, and antimicrobial applications. Future studies should focus on elucidating the detailed molecular mechanisms and in vivo studies to assess the therapeutic efficacy and safety profile of the rind ethyl acetate fraction. Furthermore, bioactivity-guided isolation should be expanded to other bioactive fractions, particularly the leaf ethyl acetate fraction, to identify the specific compounds responsible for the potent antioxidant and antibacterial activities.
ACKNOWLEDGMENTS
The authors gratefully acknowledge King Mongkut’s Institute of Technology Ladkrabang (KMITL), Ong Phra Subdistrict Campus, Dan Chang District, Suphan Buri Province, Thailand, for facilitating sample collection. The authors also sincerely thank Assistant Professor Thanarak Chantaraprasit for his assistance in capturing high-quality photographs during the sampling process.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
PN and SP conceptualized, designed the study, applied methodology, funding acquisition and performed analysis and data interpretation. PN performed investigation and visualization. PN wrote the manuscript. PN and SP revised the manuscript. SP read and approved the final manuscript for publication.
FUNDING
This study was funded by a Research Assistant/Teaching Assistant Scholarship of the School of Science, King Mongkut’s Institute of Technology Ladkrabang (KMITL) (RA/TA-2566-M-014).
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
Not applicable.
- Warinthip N, Liawruangrath B, Natakankitkul S, et al. Chemical constituents from leaves of Gardenia sootepensis and Pseudomussaenda flava biological activity and antioxidant activity. CMU J Nat Sci. 2022;21(1):e2022004.
Crossref - Reddy YM, Kumar SPJ, Saritha KV, Gopal P, Reddy TM, Simal-Gandara J. Phytochemical profiling of methanolic fruit extract of Gardenia latifolia Ait. by LC-MS/MS analysis and evaluation of its antioxidant and antimicrobial activity. Plants. 2021;10(3):545.
Crossref - Tshibangu DS, Divakar S, Ramanathan M, et al. In vitro screening of the leaf extracts from Gardenia ternifolia (Forest Gardenia) for their anticancer activity. JOCAMR. 2016; 1(2):1-7.
Crossref - Vinaykumar NM, Mahmood R, Krishna V, Ravishankara B, Shastr SL. Antioxidant and in vivo hepatoprotective effects of Gardenia gummifera L.f. fruit methanol extract. Clin Phytosci. 2020;6:47.
Crossref - Akinwunmi OA, Popooola OK, Nwozo SO, Olanipekun AD, Faleye FJ. Total antioxidant and anti-tyrosinase activities of methanol extract of ripe Nauclea latifolia fruits and its chromatographic fraction. TJNPR. 2022; 6(5):806-810.
- Tuchinda P, Pompimon W, Reutrakul V, et al. Cytotoxic and anti-HIV-1 constituents of Gardenia obtusifolia and their modified compounds. Tetrahedron. 2002;58(40):8073-8086.
Crossref - Phromnoi K, Reuter S, Sung B, Limtrakul P, Aggarwal BB. A dihydroxy-pentamethoxyflavone from Gardenia obtusifolia suppresses proliferation and promotes apoptosis of tumor cells through modulation of multiple cell signaling pathways. Anticancer Res. 2010;30(9):3599-3610.
- Tanamatayarat P, Limtrakul P, Chunsakaow S, Duangrat C. Screening of some rubiaceae plants for cytotoxic activity against cervix carcinoma (KB-3-1) cell line. Thai J Pharm Sci. 2003;27 (3):167-172.
Crossref - Doyle JJ, Doyle JL. A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull. 1987;19(1):11-15.
- Thuion T, Poeaim S, Poeaim A. Phytochemical screening, antioxidant activity and total phenolic content of methanolic extract of Phak Wan Ton (Crotalaria medicaginea Lam.). IJAT. 2023;19(1):277-290.
- Yoga IBKW, Suprapta DN, Jawi IM, Permana IDGM. A study on the antioxidant and active compounds of Gardenia jasminoides Ellis (GJE) leaves extract. JAS-SL. 2022;17(3):445-457.
Crossref - Lee KJ, Oh YC, Cho WK, Ma JY. Antioxidant and anti-inflammatory activity determination of one hundred kinds of pure chemical compounds using offline and online screening HPLC assay. Evid Based Complement Alternat Med. 2015;2015:165457.
Crossref - Xiao F, Xu T, Lu B, Liu R. Guidelines for antioxidant assays for food components. Food Front. 2020;1:60-69.
Crossref - Saewan N, Koysomboon S, Chantrapromma K. Anti-tyrosinase and anti-cancer activities of flavonoids from Blumea balsamifera DC. J Med Plant Res. 2011;5(6):1018-1025.
- Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing. 30th ed. CLSI supplement M100. CLSI; 2020. https://www.nih.org.pk/wp-content/uploads/2021/02/CLSI-2020.pdf
- International Organization for Standardization. Biological evaluation of medical devices —Part 5: Tests for in vitro cytotoxicity. ISO 10993-5:2009. ISO; 2009. https://www.dent.chula.ac.th/wp-content/uploads/2025/02/file_1_0015.pdf
- Bouafir Y, Bouhenna MM, Nebbak A, et al. Algal bioactive compounds: a review on their characteristics and medicinal properties. Fitoterapia. 2025;183:106591.
Crossref - Vindhya K, Leelavathi S. In-vitro cytotoxic activities of leaf extract of Gardenia latifolia Ait. and Gardenia gummifera Linn. IJPSR. 2014;5(11):4977-4980.
Crossref - Zhao L, Chen J, Su J, et al. In vitro antioxidant and antiproliferative activities of 5-hydroxymethylfurfural. J Agric Food Chem. 2013;61(44):10604–10611.
Crossref - Ravi L, Krishnan K. Cytotoxic potential of n-hexadecanoic acid extracted from Kigelia pinnata leaves. Asian J Cell Biol. 2017;12(1):20-27.
Crossref - Bharath B, Perinbam K, Devanesan S, AlSalhi MS, Muthupandian S. Evaluation of the anticancer potential of hexadecanoic acid from brown algae Turbinaria ornata on HT-29 colon cancer cells. J Mol Struct. 2021;1235:130229.
Crossref - Younis NS, Mohamed ME. b-Caryophyllene as a potential protective agent against myocardial injury: the role of toll-like receptors. Molecules. 2019;24(10):1929.
Crossref - de Lacerda Leite GM, de Oliveira Barbosa M, Pereira Lopes MJ, et al. Pharmacological and toxicological activities of a-humulene and its isomers: a systematic review. Trends Food Sci Technol. 2021;115:255-274.
Crossref - Xiang Z, Liu L, Xu Z, et al. Solvent effects on the phenolic compounds and antioxidant activity associated with Camellia polyodonta flower extracts. ACS Omega. 2024;9(25):27192-27203.
Crossref - Salehi B, Azzini E, Zucca P, et al. Plant-derived bioactives and oxidative stree-related disorders: a key trend towards healthy aging and longevity promotion. Appl Sci. 2020;10(3):947.
Crossref - Kumar A, Kamal YKTK, Mishra AK, et al. Potential role of benzoic acid and its synthetic derivatives to alleviate cancer: an up-to-date review. Curr Drug Discov Technol. 2025;22(4):e15701638311865.
Crossref - Olatunde A, Mohammed A, Ibrahim MA, Tajuddeen N, Shuaibu MN. Vanillin: A food additive with multiple biological activities. EJMCR. 2022;5.
Crossref - Bao L, Sun H, Zhao Y, et al. Hexadecanamide alleviates Staphylococcus aureus-induced mastitis in mice by inhibiting inflammatory responses and restoring blood-milk barrier integrity. PLoS Pathog. 2023;19(11):e1011764.
Crossref - Khongpiroon C, Buakaew W, Brindley PJ, et al. Anti-fibrotic effect of oleamide identified from the Moringa oleifera Lam. leaves via inhibition of TGF-b1-induced SMAD2/3 signaling pathway. Int J Mol Sci. 2025;26(7):3388.
Crossref - Akin-Osanaiye CB, Gabriel AF, Alebiosu RA. Characterization and antimicrobial screening of ethyl oleate isolated from Phyllanthus Amarus (Schum and Thonn). Ann Biol Res. 2011;2(2):298-305.
- Kumar SR, Chozhan K, Murugesh KA, Rajeswari R, Kumaran K. Gas chromatography-Mass spectrometry analysis of bioactive compounds in chloroform extract of Psoralea corylifolia L. J Appl & Nat Sci. 2021;13(4):1225 – 1230.
Crossref - AI-Marzoqi AH, Hameed IH, Idan SA. Analysis of bioactive chemical components of two medicinal plants (Coriandrum sativum and Melia azedarach) leaves using gas chromatography-mass spectrometry (GC-MS). Afr J Biotechnol. 2015;14(40):2812-2830.
Crossref - Chen L, Chen P, Liu J, et al. Sargassum fusiforme polysaccharide SFP-F2 activates the NF-kB signaling pathway via CD14/IKK and P38 axes in RAW264.7 cells. Mar Drugs. 2018;16(8):264.
Crossref - Ullah R, Ikram M, Park TJ, et al. Vanillic acid, a bioactive phenolic compound, counteracts LPS-induced neurotoxicity by regulating c-Jun N-terminal kinase in mouse brain. Int J Mol Sci. 2021;22(1):361.
Crossref - Kanjana M, Kanimozhi G, Udayakumar R, Panneerselvam A. GC-MS analysis of bioactive compounds of endophytic fungi Chaetomium globosum, Cladodporium tenuissmum and Pennicillium janthinellum. J Biomed Pharm Sci. 2019;2:123.
- Al Bratty M, Hafiz AM, Hassan AA, et al. Phytochemical, cytotoxic, and antimicrobial evaluation of the fruits of miswak plant Salvadora persica L. J Chem. 2020;11:4521951.
Crossref - Chatatikun M, Tedasen A, Pattaranggoon NC, et al. Antioxidant activity, anti-tyrosinase activity, molecular docking studies, and molecular dynamic simulation of active compounds found in nipa palm vinegar. Peer J. 2023;11:e16494.
Crossref - Fujisawa S, Ishihara M, Murakami Y, Atsumi T, Kadoma Y, Yokoe I. Predicting the biological activities of 2-methoxyphenol antioxidants: effects of dimers. In vivo. 2007;21(2):181-188.
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.