ISSN: 0973-7510
E-ISSN: 2581-690X
Blood is a sterile, liquid connective tissue. When infected with microbes, grave consequences can occur, such as shock, multiple organ failure, disseminated intravascular coagulation (DIC), and death. The World Health Organization reported 49 million cases of sepsis and 11 million sepsis-related deaths in 2017, accounting for approximately 20% of deaths annually worldwide. Rapid identification of the causative organism and timely, appropriate treatment are required to reduce mortality due to bloodstream infections. This study was conducted to analyze the patterns of various bacteria causing bloodstream infections and their antibiotic susceptibility patterns. All blood samples received for diagnosing bloodstream infections at the Microbiology Department of Sri Guru Ram Das Institute of Medical Sciences and Research, Amritsar, were included in the study, the duration of which was 1 year, from January to December 2020. Blood samples of 5–10 ml from adult and 5 ml from pediatric patients, were collected under aseptic conditions, stored in BACTEC bottles, and processed in an automated BACTEC system before antimicrobial therapy. After 7 days of incubation, if no microbial growth was observed, the sample was reported as sterile for aerobic organisms. When growth was observed, broth from positive blood culture bottles was subcultured on blood and MacConkey agar for identification and antimicrobial susceptibility testing using Vitek 2 according to CLSI (Clinical Lab Standard Institute) guidelines and the manufacturer’s instructions. A total of 441 (14.5%) bacteria were isolated from 3007 blood samples from patients with suspected bacteremia. Contamination was observed at a rate of 2.5%. Gram-positive cocci (49%) were the predominant organisms recovered, followed by Gram-negative bacilli (34%). Gram-positive cocci were coagulase-negative Staphylococci (46%), Staphylococcus aureus (7%), and Enterococcus spp. (6%). Among the Gram-negative bacilli, E.coli (14%), Klebsiella spp. (13%), Acinetobacter baumannii (7%), Pseudomonas spp. (7%), Salmonella typhi (2%), Enterobacter spp. (1%), and Serratia spp. (1%) and single isolates of Aeromonas spp., Morganella morgani, Pantoea spp., Proteus mirabilis, and Providentia rettgeri were identified. Linezolid, teicoplanin, and vancomycin were the most effective drugs for treating Gram-positive bacteremia. Tigecycline, carbapenems, and aminoglycosides were the most effective treatments for Gram-negative bacteremia. The results stress the need for continued screening and surveillance in routine blood culture techniques to start empiric therapy for bloodstream infections.
Bloodstream Infections, Vitek-2, Bactec
Despite advances in treatment modalities and supportive care, bloodstream infections remain a primary cause of morbidity and mortality.1 Bloodstream infections can cause health problems from asymptomatic transient bacteremia to fulminant septic shock, resulting in an increased mortality rate.2 The new definition for Sepsis and Septic shock, i.e., Sepsis-3, provided by the Third International Consensus 2016 is as follows:
a) Sepsis is a life-threatening organ dysfunction caused by a dysregulated host response to infection.
b) Septic shock is a subset of sepsis in which profound circulatory cellular and metabolic abnormalities are associated with a greater mortality risk than with sepsis alone.3
Blood is a sterile liquid connective tissue; however, once infected with microbes, grave consequences such as shock, multiple organ failure, disseminated intravascular coagulation (DIC), and death, may occur.4 A document released by the WHO reported 49 million cases of sepsis and 11 million sepsis-related deaths in 2017, accounting for approximately 20% of deaths annually worldwide.5 The highest incidence of septicemia is reported in low- and middle-income countries. In India, 11.3 million cases of sepsis were detected in 2017.6 Reducing morbidity and mortality due to bloodstream infections (BSIs) requires rapid identification of the causative organism and timely, appropriate treatment.7
Early Gram-negative organisms were predominantly known to cause bloodstream infections.8-9 By contrast, presently, Gram-positive organisms are predominant,7 especially among neonates and children.10 Moreover, excessive and irrational use of antibiotics has further complicated the scenario due to the increase in multidrug-resistant strains.
The gold standard for the detection of bacteremia is the blood culture method.7 Conventional blood culture method is time-consuming, and repeated subculturing may introduce contaminants. Many faster and more automated culture techniques have been developed. BACTEC is an automated blood culture method. It detects the growth of microorganisms by monitoring the consumption of carbon dioxide by using a calorimetric method. For species identification and antimicrobial susceptibility profiling, an automated system is available, i.e., the Vitek 2 system, which facilitates rapid, accurate identification, and minimum inhibitory concentration (MIC) evaluation, for these pathogens.9 This study aimed to analyze the pattern of microorganisms causing BSIs and examine their antimicrobial susceptibility profile for one year to guide clinicians in formulating antimicrobial policies for empirical therapy.
This study was conducted in the bacteriology section of the Microbiology Department in a tertiary care center in North India. All blood samples received in the bacteriology laboratory for diagnosing bloodstream infections were included in the study for one year, from January to December 2020. Blood samples of, 5–10 ml from adult and 5 ml from pediatric patients, were collected under aseptic conditions; stored in BacT/ALERT FA and PF plus-aerobic bottles (Biomerieux, Durham, NC, USA), respectively; and processed in an automated BACTEC system, before antimicrobial therapy.11 Sufficient microbial growth was automatically indicated by the BACTEC system. In the absence of microorganism growth, no signal was generated even after 7 days of incubation, and the sample was reported to be sterile for aerobic organisms. Positive growth broth from positive blood culture bottles was subcultured on blood and MacConkey agar. Next, 0.5 McFarland suspension was prepared from the growth on these culture plates for identification and antimicrobial susceptibility testing using Vitek 2 (bioMerieux, Durham, NC, USA) according to CLSI (Clinical Lab Standard Institute) guidelines and the manufacturer’s instructions.12 Data were compiled and statistically analyzed. Data were collected from institutional databases that contained patient information that required institutional ethics committee approval; however, informed consent was not required.
Ethical Clearance
Approval was obtained from the ethical committee of the institute for this study.
Statistical Analysis
Data collected was analyzed with Microsoft Office Excel 2016.
The bacteriology laboratory at the hospital received 3007 blood specimens from patients with clinically suspected septicemia from January to December 2020. From the blood samples of these patients, 441 aerobic bacterial isolates were isolated, showing a culture positivity of 14.5% and confirmed cases of septicemia. During blood culture, 76 (2.5%) isolates were considered contaminants, including skin commensals, mainly coagulase-negative Staphylococci (CONS), diphtheroids, Micrococcus, and Bacillus sp. Of the 441 culture-positive samples, 291 (66%) and 150 (34%) were from male and female patients, respectively. The sex-ratio was 1.94:1 and skewed in favor of males. The highest positive blood culture results were from patients between the ages of 46–60 years, followed by the ages 0–15 years. (Table 1). The distribution of patients with culture-positive bacteremia in the intensive care unit (ICU) and various wards is illustrated in Table 2. Among the ICUs, the maximum number of patients was from the main ICU 87 (20%). In the ward-wise distribution, the maximum number of patients, 147 (34%), was from the emergency ward.
Table (1):
Age-based distribution of patients.
Age group |
No. of Patients (%) |
|---|---|
0-15 years |
98 (22%) |
16-30 years |
30 (7%) |
31-45 years |
69 (16%) |
46-60 years |
122 (28%) |
61-75 years |
89 (20%) |
76-90 years |
33 (7%) |
Total |
441 (100%) |
Table (2):
Ward-wise distribution of patients.
Ward |
No. of Patients (%) |
|---|---|
E/W(Emergency ward) |
147 (34%) |
Medicine wards |
92 (21%) |
MICU (Main Intensive care unit) |
87 (20%) |
NICU (Neonatal intensive care unit) |
70 (16%) |
Pediatric ward |
17 (4%) |
Surgery ward |
11 (2%) |
PICU (Pediatric intensive care unit) |
7 (1%) |
Others (Orthopedics, Otorhinolaryngology, Gynaecology, Oncology, Burn intensive care unit) |
10 (2%) |
Total |
441 (100%) |
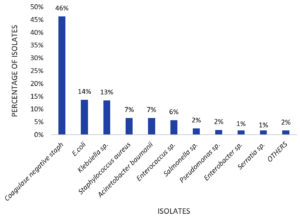
Of the 441 isolates, 214 (49%) were Gram-positive cocci, and 151 (34%) were Gram-negative bacilli. Gram-positive cocci comprised Staphylococcus aureus 24, 7%) and Enterococcus spp.21,6%). In addition, 169 (46%) isolates of CONS were considered clinically significant based on host factors, such as indwelling catheters, immunosuppression, extremes of age, and clinical correlation. The most commonly observed Gram-negative bacilli were E.coli 50(14%); followed by Klebsiella spp.49 (13%), Acinetobacter baumannii 24(7%), Pseudomonas spp.7 (2%), Salmonella sp.7 (2%), Enterobacter spp.4(1%), and Serratia spp. 2(1%); and single isolates of Aeromonas spp., Morganella morgani, Pantoea spp., Proteus mirabilis, and Providentia rettgeri. (Figure 1)
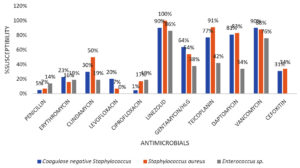
Figure 2 suggests that all Gram-positive bacteria had high susceptibility to linezolid, followed by teicoplanin, vancomycin, and daptomycin; moderate susceptibility to gentamicin and clindamycin; and low susceptibility to erythromycin and quinolones. Gram positive bacteria showed the lowest susceptibility to penicillin. Among Staphylococcus aureus, 66% were methicillin-resistant Staphylococcus aureus (MRSA), confirmed based on their resistance to cefoxitin, and 69% of CONS were methicillin-resistant Coagulase-negative Staphylococci (MRCONS). (Figure 2).
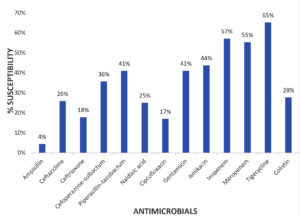
Antibiotic susceptibility tests of Gram-negative isolates showed that Enterobacteriaceae (Figure 3) were most sensitive to tigecycline, followed by carbapenems, and moderately sensitive to aminoglycosides, piperacillin-tazobactam, and cefoperazone-sulbactam. Gram-negative isolates showed low susceptibility to colistin, third-generation cephalosporins, and quinolones.
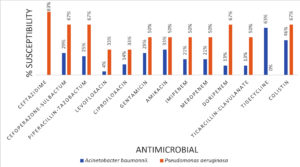
Among non-fermenters (Figure 4), Pseudomonas aeruginosa was more susceptible to antibiotics than Acinetobacter baumannii. P. aeruginosa was highly susceptible to cefoperazone-sulbactam, piperacillin-tazobactam, and colistin, followed by ceftazidime, and moderately susceptible to ticarcillin-clavulanate, aminoglycosides, and carbapenems. No isolate was sensitive to tigecycline. A. baumannii was most susceptible to tigecycline, followed by moderate susceptibility to colistin; however, the later had low susceptibility to aminoglycosides, followed by cefoperazone-sulbactam and piperacillin-tazobactam, and both P. aeruginosa and A. baumannii isolates had the least sensitivity to quinolones.
Rapid identification of the causative organism and antimicrobial treatment can reduce morbidity and mortality associated with bloodstream infections. This study attempted to analyze bacterial profiles and assess their antimicrobial susceptibility trends to formulate an antibiogram and effective empirical treatment of bloodstream infections.
In this study, all blood samples were collected from patients suspected to have septicemia; however, the blood culture showed positive results in 14.5% of patients, i.e., the confirmed septicemia cases. Patients in our institute are mainly referred from peripheral centers, where they receive antibiotic courses before being referred to our tertiary center. This aspect could be an relevant reason for the low culture positivity rate, which is consistent with the literature.5,9,13 Previously, higher culture positivity has been reported, ranging from 24.86% to 49.18%.7,14 Culture positivity rates vary because of differences in the geographical area, epidemiological disparities in etiological agents, and population characteristics.
The contamination rate in this study was 2.5%, which is within the permitted levels Hall et al. suggested15 Normally, human skin is colonized by commensals such as CONS, Corynebacterium sp., Micrococci, Bacillus sp. When they are introduced into the culture during specimen collection or processing and are not pathogenic to patients, they are considered contaminants.
The sex-ratio of 1.94:1 was skewed in favor of males in this study. This finding is consistent with those in the literature.2,16,17 A possible reason for this is that men in rural areas are more involved in outdoor activities to earn their livelihood than women, which predisposes the former to infections; additionally, the former is more privileged than the latter regarding physician visits for treatment.
In this study, bloodstream infections caused by Gram-positive organisms (49%) predominated over Gram-negative isolates (34%). Similar results were observed by Katyal et al.,7 Banik et al.,13 and Orsini et al.1 However, many national and international studies have reported a higher incidence of GNB than GPC as the causative organism of bloodstream infections.8,9,14
Among the Gram-positive pathogens, coagulase-negative Staphylococcus was the most commonly observed isolate, followed by Staphylococcus aureus and Enterococcus sp. This finding supports those in the literature.5,7,9 CONS has been considered the most common blood contaminant; however, the clinical significance of CONS was defined as at least two blood cultures positive for CONS within 5 days or one positive blood culture plus clinical evidence of infection, which comprise an abnormal leukocyte count and body temperature.18 Improper techniques of blood collection and the presence of longstanding intravascular devices are recognized possible causes of BSIs by CONS.
In this study of Gram-negative organisms, Enterobacteriaceae was responsible for the greatest number of BSI cases: 25% of overall cases, with E.coli predominating (14%), followed by Klebsiella spp. (13%), Salmonella typhi (2%), and Enterobacter spp. (1%). Comparable findings have been observed in the literature.2,13 By contrast, Katyal A7 showed that A.baumannii was the predominant gram-negative organism. In our study, we observed 2% BSI due to Salmonella sp., which is less than the 4.42% reported by Khara R14 and 42.7% by Pandey S et al., 8 but more than the 1% and 0.2% reported by Abrahamsen et al. 19 and Sudaramano et al.20 from South East Asia, respectively.
In this study, all Gram-positive isolates showed high susceptibility to linezolid and vancomycin and low sensitivity to quinolones and erythromycin. Penicillin was the least sensitive antibiotic. A high percentage of Staphylococcus aureus and CONS were susceptible to teicoplanin, daptomycin, and vancomycin, as reported in the literature,2,9,17 and a moderate percentage of these two GPCs were susceptible to gentamicin and clindamycin. Vancomycin is the primary drug of choice for treatment of MRSA infections, and according to the institutional antibiotic policy, it is reserved for the treatment of MRSA infections, which explains the high susceptibility of Staphylococcus aureus to this drug. However, a moderate number of Enterococcus spp. are susceptible to teicoplanin and daptomycin. The overall rate of MRSA in our study was 69%, detected based on resistance to cefoxitin; Palewar et al2 observed similar results, but our results are much higher than the 4% reported by the ICMR-AMRSN.21 The high prevalence of MRSA could be attributed to multiple risk factors such as the carriage of MRSA by health care workers and patients, misuse and abuse of antimicrobials, and prolonged hospitalization. In addition, our study reported 66% MRCONS, which is higher than the 40% reported by Mamotra et al.22 This variation could be attributed to different patient profiles and local antibiotic regimens that can influence the prevalence.
Among Gram-negative organisms isolated from blood cultures, tigecycline and carbapenem were the most effective drugs against Enterobacteriaceae; a moderate number of Enterobacteriaceae members were susceptible to aminoglycoside and the cephalosporin-beta-lactamase inhibitor combination; fewer were susceptible to colistin, third-generation cephalosporins, and quinolones; and ampicillin was effective against the least number of Enterobacteriaceae. That third-generation cephalosporins and quinolones are the most commonly prescribed drugs could be the cause of their low susceptibility levels. Similar results have been reported by Katyal A,7 Banik A, et al.,13 and Palewar et al.2 Colistin has been shown to be susceptible to the maximum percentage of Enterobacteriaceae isolates,2,7,13 but our study reported moderate sensitivity, possibly because colistin is used indiscriminately on patients in ICUs.
Among the non-fermenters isolated from patients with sepsis, P. aeruginosa was highly susceptible to beta-lactam combinations and colistin. The antibiotic susceptibility pattern of P. aeruginosa in our study was comparable to that in the annual report by the ICMR-AMRSN. No isolate of P. aeruginosa was susceptible to tigecycline; according to an analysis by Stein and Craig, 90% of the strains of P. aeruginosa have an MIC value of > 4 µg/ml and would be considered resistant to tigecycline. The maximum susceptibility of Acinetobacter baumannii isolates was for tigecycline, although not much reference is available, but a review by Stein and Craig explains that A.baumannii has the lowest tigecycline MIC values.23 In our study, colistin was moderately susceptible to A.baumannii. Other studies have reported high susceptibility,21 and that the susceptibility to aminoglycosides, beta-lactam combinations, and carbapenems was higher than those reported by the ICMR AMRSN.21
The automated methods used in this study helped reduce the contamination rate by eliminating the need for repeated subcultures. Vitek 2 provides standardized detection of microbial growth and increases the sensitivity and specificity of blood cultures. Moreover, the initial specimen diversion technique described by Binkhamis et al.24 can be applied to reduce the contamination rate and hence the burden on labs.
CONS and E.coli were the most predominant blood-borne pathogens isolated in our tertiary care hospital in North India. Most of the Gram-positive cocci are susceptible to linezolid, teicoplanin, and vancomycin. The majority of Gram-negative bacilli are sensitive to tigecycline and carbapenems. Surveillance of the local BSI etiology is necessary for the formulation of hospital antibiograms and effective empirical treatment of sepsis in that particular area.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct, and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
The data analysed during the study are available from the corresponding author upon reasonable request.
ETHICS STATEMENT
This article does not contain any studies with human participants or animals performed by any of the authors.
- Orsini J, Mainadi C, Muzylo E, Karki N, Cohen N. Microbiological Profile of Organisms Causing Bloodstream Infection in Critically Ill Patients. J Clin Med Res. 2012;4(6):371-377.
Crossref - Palewar M, Mudshingkar S, Dohe V, Kagal A, Karyakarte R. Bacteriological profile and antibiogram of blood culture isolates from a tertiary care hospital of Western India. J Datta Meghe Inst Med Sci Univ. 2020;15(2):261-265.
Crossref - Sepsis. https://www.cdc.gov/nchs/data/icd/SEPSIS_tabular_final_dp.pdf
- Sangita K, Tomar R, Saha N. Bacteriological Profile and Antibiogram of Blood Culture Isolates From a Tertiary Care Hospital. IJMSIR. 2019;4(6):187-192.
- WHO. Global report on the epidemiology and burden of sepsis: current evidence, identifying gaps and future directions. https://apps.who.int/iris/handle/10665/334216
- Jeganathan N. Burden of Sepsis in India. Chest. 2022;161(6):1438-1439.
Crossref - Katyal A, Singh D, Sharma M, Chaudhary U. Bacteriological Profile and Antibiogram of Aerobic Blood Culture Isolates from Intensive Care Units in a Teaching Tertiary Care Hospital. J Health Sci Res. 2018;9(1):6-10.
Crossref - Pandey S, Raza S, Bhatta C. The Aetiology of the Bloodstream Infections in the Patients Who Presented to a Tertiary Care Teaching Hospital in Kathmandu, Nepal. JCDR. 2013;7(4):638-641.
Crossref - Dash M, Panda R, Mohapatra D, Paty B, Sarangi G, Chayani N. Bacteriological profile and antimicrobial resistance patterns of bloodstream infections in a tertiary care hospital, Eastern India. Int J Health Allied Sci. 2016;5(4):210-214.
Crossref - Prabhu K, Bhat S, Rao S. Bacteriologic Profile and Antibiogram of Blood Culture Isolates in a Pediatric Care Unit. J Lab Physicians. 2010;2(02):85-88.
Crossref - BACT/ALERT® 3D: Healthcare https://www.biomerieux-usa.com/bactalert3d/automatedsystem. Accessed on 2022 Feb 7
- VITEK® 2. https://www.biomerieux-diagnostics.com/vitek-2. Accessed on 2022 Feb 7.
- Banik A, Bhat S, Kumar A, Palit A, Snehaa K. Bloodstream infections and trends of antimicrobial sensitivity patterns at Port Blair. J Lab Physicians. 2018;10(03):332-337.
Crossref - Khara R, Sucheta JL. Bacteriological Profile of Blood Culture from Adult Sepsis Patients from a Rural Based Tertiary Care and Teaching Hospital, Piparia, Vadodara, India. Int J Curr Microbiol App Sci. 2018;7(05):3173-3182.
Crossref - Hall KK, Lyman JA. Updated review of blood culture contamination. Clin Microbiol Rev. 2006;19(4):788-802.
Crossref - Raheja P, Antarikshdeep, Chaudhary U. Microbiological profile of hospital-acquired bloodstream infections in seriously ill medical patients admitted in tertiary care hospital. Int J Res Med Sci. 2016;4(5): 1636-1640.
Crossref - Vasudev N, Nirwan PS, Shrivastava P. Blood stream infections and antimicrobial sensitivity patterns in a tertiary care hospital of India. Ther Adv Infectious Dis. 2016;3(5):119-127.
Crossref - Jena PP, Gur R, Duggal SD, Kumar A, Rongpharpi SR. Microbiological Profile and Antibiogram of Blood Stream Isolates at a Referral Hospital in North Delhi: A One Year Study. IJBR. 2015;6(10):819-824.
Crossref - Abrahamsen SK, Haugen CN, Rupali P et al. Fever in tropics: aetiology and case fatality – a prospective observational study in a tertiary care teaching hospital in South India. BMC Infect Dis.2013;13:355.
Crossref - Sudaramano P, Aman A, Arif M, et al. Causes and outcomes of sepsis in southeast Asia: a multinational multicentre cross-sectional study. The Lancet Global Health. 2017;5(2):e157-e167.
Crossref - ICMR. Annual Report Antimicrobial Resistance Research and Surveillance Network January 2020 to December 2020. https://main.icmr.nic.in/sites/default/files/guidelines/AMRSN_annual_report_2020.pdf. Accssed on 09 February 2022.
- Mamtora D, Saseedharan S, Bhalekar P, Katakdhonal S. Microbiological profile and antibiotic susceptibility pattern of Gram-positive isolates at a tertiary care hospital. J Lab Physicians. 2019;11(02):144-148.
Crossref - Stein G, Craig W. Tigecycline: A Critical Analysis. Clin Infect Dis. 2006;43(4):518-524.
Crossref - Binkhamis K, Forward K. Effect of the Initial Specimen Diversion Technique on Blood Culture Contamination Rates. J Clin Microbiol. 2014;52(3):980-981.
Crossref
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.