ISSN: 0973-7510
E-ISSN: 2581-690X
Urinary tract infections are gaining much importance among community acquired infections. Poor personal hygiene may act as a contributory factor in such frequent infections in developing countries. The condition becomes serious with antimicrobial resistance among uropathogens. Specifically, surge of Extended Spectrum beta Lactamase (ESBL) producers poses much concern in the available treatment options. Present work was aimed to observe the bacteriological profile and antimicrobial susceptibility pattern of uropathogens with special reference to ESBL producing strains. This study was carried out in the microbiology department, SGT University, Gurugram, Haryana. Total 600 samples were processed as per standard bacteriological procedures i.e, microscopy, culture and biochemical reactions followed by antibiotic susceptibility testing and interpreted as per CLSI guidelines. ESBL producer strains were spotted by double disc synergy testing (DDST). Out of 600 specimens, 128( 21.3%) showed culture positivity. Females contributed more compared to males. 21-40 years age group showed highest isolation rate. E.coli was predominant organism. Highest resistance towards amikacin was shown by gram negative organisms. Gram positive organisms demonstrated high resistance towards gentamicin, cotrimoxazole, ampicillin and ciprofloxacin. 34.4% ESBL producing E.coli strains were observed. Alarming rate of antibiotic resistance as well as beta lactamase production by strains and increasing urinary tract infections should be considered as potential threat to the community. Routine investigation should be done to see the burden in order to implicate advance treatment policies in order to treat such infections.
Urinary Tract Infection (UTI), Enterobacteriaceae, Community-Acquired Infections, Rural, ESBL
Urinary tract infection (UTI) is considered as a very commonly encountered clinical problem among all community acquired infections. Lack of sanitation and unhygienic personal habits in developing countries contribute a major part for such infections.1
Pathogenic microorganisms present in the kidney, bladder, urethra or urine can cause UTI with or without symptoms. In addition, UTI may be considered as a contributory factor for morbidity among rural population and a second most common cause for frequent hospital visits of the population.2
Bacterial spectrum causing complicated UTI is much broader compared to those causing uncomplicated UTI. Complicated UTI may be given a thought in situations with treatment failure and repeat infections and they will lead to significant morbidity and mortality with a poor outcome.3 However uncomplicated UTI may be defined among healthy nonpregnant women, premenopausal having no abnormal urinary tract anomaly previously.
The most commonly encountered microorganisms associated with complicated UTI are Gram negative (GN) bacteria i.e, Escherichia coli (E.coli), Pseudomonas aeruginosa, Citrobacter spp, Proteus vulgaris and Enterobacter aerogenes whereas Coagulase Negative Staphylococcus (CoNS), Acinetobacter spp. and Salmonella spp. are observed less commonly.4
E.coli, a commonly isolated bacteria, is causative agent of more than 80% of all the community associated UTI. The organism is responsible for range of UTI’s, including symptomatic cystitis, acute prostatitis, prostatic abscess, uncomplicated urethritis and urosepsis. Primarily sexually active females who are colonized by uropathogenic strains of Escherichia coli complaining of uncomplicated cystitis.5,6 Extensive injudicious use of antimicrobials may result into emergence of multidrug resistance (MDR) in different pathogens. In addition many healthcare systems have initial outbreaks of infections which are majorly contributed by surge of ESBL in pathogenic microorganisms which in turn result into high mortality rate as antibiotics are found inactive in response towards ESBL producing strains. Hiked multidrug resistance (MDR) in bacterial strains responsible for uropathies are an important and emerging public health concern these days.7
Most common antibiotics in the cure of bacterial ailments belong to β-lactam group however β-lactamases production makes bacterial strains resistant to this group. β-lactamases are extracellular enzymes produced by bacteria breaking the amide bond of β-lactam ring of Penicillin and capable of inactivating Oxyimino- cephalosporins group and Aztreonam group but are inactive against Cephamycin and Carbapenem.8
Extended Spectrum Beta Lactamases
(ESBL) are majorly plasmid or chromosomal mediated. It has been observed that the pre- existing broad spectrum β-lactamases (TEM-1, TEM-2, SHV-1) had been mutated to result in ESBL as a sequel for use of Aztreonam and 3rd generation Cephalosporins.9 These enzymes are coded by plasmids and their ability to spread to other bacteria led to dramatic increase in their prevalence worldwide in a very short span of life.10
ESBL Production in Bacteria Causing
UTI leave physicians with very limited options of antibiotics for treating such patients. ESBL enzymes are found predominantly in Escherichia coli and Klebsiella spp. Therefore the study was designed and conducted to find out the profile of bacteria causing UTI and the antibiotic sensitivity patterns with special emphasis on the presence of ESBLs.
ESBL detection has gained high attention among uropathogens from the suspected cases of UTI as urine specimen may be considered as a tool for consideration of epidemiological markers in the assessment of community spread of pathogenic drug resistant organisms.11
In recent storyline, antibiotic resistance is increasing alarmingly with emergence of new multidrug resistant bacteria which in turn results into enhanced mortality and morbidity rate.
The present work gives an insight to the burden of urinary pathogens and their antimicrobial resistance rate and enhanced ability to produce ESBL enzymes.
Study Area
The current research was accomplished in the Microbiology department, SGT University, Gurugram, Haryana. Total 600 urine samples received in the microbiology laboratory were taken into consideration for the study after approval from Institutional ethics committee.
Sample Collection and Processing
Appropriate sterile collection of mid-stream urine samples was ensured from all patients. All the specimens were received in sterile, disposable universal containers within one hour of collection.
Processing
All the collected specimens were processed during sixty minutes of receiving.
Direct Microscopy
Uri n e samples were examined microscopically for significant pyuria, RBCs and crystals.
Standard Loop Method (Semi Quantitative Method)
Significant bacteriuria was considered as a marker of positive urine culture bacteriuria i.e, isolation of 1 or 2 pathogens with >105 CFU (colony forming units)/ ml, for pyuria detection of >5 pus cells/ high power field (HPF).
Culture
Urine samples were inoculated on to CLED medium (Cystine Lactose Electrolyte Deficient medium) and the Culture plates were subjected to incubation at 37°C for overnight or 18-24 hrs. The isolates were identified on the basis of colony morphology, gram’s reaction, motility testing, oxidase test and catalase test. These primary testing methods were further followed by chain of biochemical reactions for further identification of organisms as per standard guidelines.12
Antimicrobial Sensitivity Testing
Antimicrobial sensitivity of bacterial strains was conducted by disk diffusion method on MHA (muller-hinton agar). The susceptibility result will be interpreted as per the CLSI (Clinical Laboratory Standard Institute).13
Antibiotics used Gram Negative Bacilli
Amikacin (30μg), Amoxyclav (30μg), Levofloxacin (5μg),Ampicillin (10μg), Cefotaxime (30μg), Azithromycin (15μg), Ceftriaxone (30μg), Ciprofloxacin (5μg), Imipenem (10μg), Cefaclor (30μg),Pipracilin /Tazobactum (100/10μg ), Gentamycin (10μg).
For Gram Positive Cocci
Amikacin (30μg), Cefotaxime (30μg), Amoxyclav (30μg), Ampicillin (10μg), Azithromycin (15μg), Cefoxitin (30μg), Cephalexin (30μg), Clindamycin (2μg), Cotrimoxazole (25μg), Erythromycin (15μg), Gentamycin (10μg), Levofloxacin (5μg), Linezolid (30μg), Ofloxacin (5μg), Ciprofloxacin (5μg), Vancomycin (30μg).
ESBL Detection
DDST (Double-Disc Synergy Test)
Strains of E. coli were checked for ESBL production by placing a ceftazidime disc at 20mm distance centre to centre from the combined disc (clavulanic acid +ceftazidime). An increase in zone of inhibition more than 5mm towards ceftazidime disc was considered as the ESBL production.
A total of 600 mid-stream urine specimens were analysed in the laboratory throughout the study duration. Out of 600 samples, 128 (21.3%) showed culture positivity.
Isolation of uropathogens vary significantly between samples from male and female population.
Uropathies were observed in high frequency among females compared to males. Major number of patients were observed from the age group of 21-40 years, as demonstrated by highest isolation frequency in them as compared to other age groups. (Table 1) Of the total 128 bacterial isolates, Gram negative bacteria accounted for 67%, maximum being members of Enterobacterales (62.5%). E. coli was most frequently isolated (72.5%) uropathogen among total Enterobacterales. Gram positive bacteria contributed well as 32.8% Gram positive cocci isolated from culture positive samples from clinically suspected population. (Table 2)
Table (1):
Age group distribution of all the study subjects.
| Age group | Samples | Number of positive samples | |||
|---|---|---|---|---|---|
| Total | Males | Females | Statistical analysis | ||
| <20 | 120 (20%) | 20(6.6) | 6 (30%) | 14(70%) | |
| 21-40 | 306 (51%) | 75(24.5) | 16 (21.3%) | 58(77.3%) | |
| 41-60 | 142 (23.6%) | 28 (19.7%) | 12(42.8%) | 16(57.1%) | |
| 61-80 | 32 (5.3%) | 5 (16.1%) | 1 (3.2%) | 4 (12.9%) | NC |
| Total | 600 (100%) | 128 (21.3%) | 35(27.3%) | 92 (71.8%) | |
Overall E. coli was isolated as the most frequent uropathogen compared to Klebsiella Spp, S.aureus, Enterococcus spp, Proteus spp, Micrococcus, Acinetobacter and Pseudomonas (Table 2)
Table (2):
Distribution of Uropathogens.
| No. | Isolates | Total number of isolates |
|---|---|---|
| 1 | E.Coli | 58 |
| 2 | S.aureus | 23 |
| 3 | Klebsiella.spp | 21 |
| 4 | Enterococcusspp | 11 |
| 5 | Micrococcus | 8 |
| 6 | Acinetobacter | 4 |
| 7 | Pseudomonas | 2 |
| 8 | Proteus | 1 |
| Total | 128 | |
E.coli and Klebsiella showed higher resistance towards amikacin compared to other classes of antibiotics. 3 (75%) of Acinetobacter isolates were not found susceptible towards carbapenem. (Table 3) 100% Pseudomonas strains were found resistant towards ampicillin & cefaclor. All Proteus isolates showed resistance towards amikacin and gentamycin in the current research work. S. aureus strains were not found sensitive towards gentamycin and cotrimoxazole however all the isolates found sensitive towards vancomycin. Enterococcus spp. showed a high resistance towards ampicillin and ciprofloxacin. (Table 3)
Table (3):
Antimicrobial sensitivity profile of uropathogens.
Organisms (128) |
Amikacin (30µg m) |
Co- trimoxazole (25µgm) |
Cefotaxime (30µgm) |
Ampicill in (30µg m |
Cefaclor (30 µgm ) |
Ciprofloxacin (5µgm) |
Gentamycin (10µgm) |
Imipenam (10 µgm) |
Vacomycin |
Ofloxacin |
|---|---|---|---|---|---|---|---|---|---|---|
E.coli (58) |
50 (86.2%) |
14 (24.1%) |
19 (32.7%) |
8 (13.7%) |
12 (20.6%) |
22 (37.9%) |
33 (56.8%) |
23 (39.6%) |
NA* |
NA |
Klebsiella (21) |
15 (71.4%) |
6 (28.5%) |
10 (47.6%) |
3 (14.2%) |
9 (42.9%) |
10 (47.6%) |
10 (47.6%) |
11 (52.3%) |
NA |
NA |
Acineto- bacter (4) |
2 (50.0%) |
2 (50.0%) |
2 (50.0%) |
1 (25.0%) |
1 (25.0%) |
3 (75.0%) |
2 (50%) |
3 (75%) |
NA |
NA |
Pseudo- monas (2) |
2 (100%) |
2 (100%) |
2 (100%) |
– |
– |
2 (100%) |
2 (100%) |
2 (100%) |
NA |
NA |
Proteus (1) |
– |
1 (100%) |
1 (100%) |
1 (100%) |
1 (100%) |
1 (100%) |
– |
1 (100%) |
NA |
NA |
S. aureus (23) |
13 (56.5%) |
3 (13.0%) |
11 (47.8%) |
6 (26.0%) |
NA |
13 (56.5%) |
2 (8.6%) |
NA |
23 (100%) |
15 (65.2%) |
Enterococcus (11) |
6 (54.5%) |
8 (72.7%) |
4 (36.3%) |
4 (36.3%) |
NA |
3 (27.2%) |
6 (54.5%) |
NA |
5 (45.4%) |
7 (63.6%) |
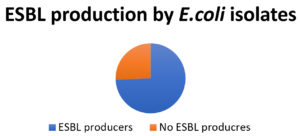
Out of total 58 E.coli isolates, 20(34.4%) isolates were observed as ESBL producers (Figure).
Urinary tract infections are the commonly occurring infections having potential to produce ailments among all age groups throughout the world.14 Urinary tract infections (UTI) impose a high load on hospitals due to high occurrence of infection in both community and nosocomial settings. Variety of pathogens including P. aeruginosa, E.coli and K. pneumonia are found responsible for UTI. Surveillance programs for antimicrobial resistance of urinary pathogens is not helping with this surge in antimicrobial resistance rate, in turn provides an in sight for management and empirical treatment of patients suffering from UTIs.15
Out of 600 symptomatic patients, included in the present study, urine culture was positive in 128 subjects (21.3%). The positivity was found to be lower than other reports in the year 201415 and 2015 (41.18% and 43.07% respectively). Female population contributed 22.5% of the total study group, which is a concordant finding with another study (24.5%).16
High isolation rate of uropathies among 21-40 yrs age group may be justified due to that females being more susceptible to develop UTI than males due to certain anatomical and physiological factors such as absence of prostatic secretions, easy contamination of the urinary tract with faecal normal flora and short urethra and it has been well documented in some studies also.17- 19 These facts also supported by clinical studies and findings of present study are in total agreement.20 About 40-50% of females belonging to reproductive age group had gone through at least a single episode of UTI in their lifetime.21 Uropathies are most commonly occurring among sexually active females. Among males, high incidence of urinary tract infections was observed in 41 to 60 years age group which could be explained due to co-morbid conditions like prostate hypertrophy and propensity to have high blood sugar levels in the male population of this age group.
Several types of microorganisms may be the causative agents of UTI, including, viruses, fungi and protozoan parasites and among all the categories bacterial pathogens are considered as the major category of microorganisms accounting for much than 90% of all the UTI cases.17
The most commonly identified uropathogen was E. coli followed by Klebsiella pneumoniae in the present study. E.coli has been documented as a cause of UTI in over 80% of cases.22,23 Many other studies have also demonstrated E.coli as their majorly encountered microorganism in UTI accounting for 66.9%, 61.02%, 39% and 37.95% respectively.5,6,24,25 However the pathogenesis by uropathogenic E. coli (UPEC) include a range of factors i.e, vaginal, periurethral and urethral colonization, growth of planktonic cells in urine, interference with the bladder epithelium which is the primary defense system and biofilm formation. UPEC colonization in bladder and presence of various virulence factors i.e, pili, outer membrane proteins (OMP), lipopolysaccharide capsule, secretory toxins, also play condemnatory characters in urinary infections.
In the present study showed a countable involvement in the total uropathies. Besides Enterobacteriaceae, gram-positive organisms are also exerting countable impact in uropathies specifically among old age population, pregnant females or those who are immunocompromised. Gram positive bacteria include S. aureus, Enterococcus and Micrococcus specifically found associated with uropathies.
Our study supports this fact as 32.8% GPC were identified S. aureus as the predominant species over Enterococcus spp. and Micrococcus. (Table 2) Gram-positive organisms are associated with uncomplicated uropathies quite commonly seen in nonpregnant females, premenopausal and sexually active females. However, there’s no doubt in the percentage of gram negative/gram positive association in uncomplicated uropathies, as gram negatives account for a major portion (70-75%) of the organisms associated with uncomplicated uropathies but the remaining proportion (25-30 %) is contributed by gram positives only. Similar order of isolation was also observed in another study.26 Symptoms for uncomplicated uropathies resembles as that for complicated uropathies which include urinary frequency, dysuria, flank pain, nausea suggestive of upper UTI.27
In the present study, most frequent isolation of S. aureus is in contrast to findings of many studies where either Enterococcus spp or S. saprophyticus is most commonly isolated gram-positive bacteria from the samples of clinically suspected cases of UTI.28,29
Bacteremia due to S. aureus is strongly associated with bacteriuria supports the fact that bacteriuria is invariably preceded by bacteremia.30 In patients with S. aureus bacteriuria (UTI), importance of history related to concomitant or preceding bacteremia cannot be overlooked.
One recent observation showed the synergy of bacterial infections in urine with the mortality rate which demonstrated twice increased complication of death among the patients having S. aureus bacteremia as well as bacteriuria.31 Enterococci are the normal flora of mouth, vagina and human gastrointestinal tract.32
E. faecalis may be isolated from 80% of human ailments as concluded by a survey.33 In the same direction it could be the major leading cause of urinary infections around the world.34 E. faecalis was reported to have tropism for the kidneys in female C57BL/6J mice. If this finding can be extended to human beings, possibility of lower tract UTI caused by seeding from kidneys in earlier asymptomatic/ apparently symptomatic episodes, cannot be denied.35
Proteus spp. and Pseudomonas spp. are majorly encountered from health care associated UTIs compared to community associated UTIs. Few non fermenter bacteria are also isolated as Pseudomonas and Acinetobacter spp accounting 4.7% of total bacterial isolates. Similar finding have been observed in other studies.22,23
Antimicrobial resistance possess a worldwide threat to the community as well as healthcare system. Intense and overuse of antimicrobials is being the foremost cause for surge of antimicrobial resistance together. In turn the hike is responsible for the transmission of antimicrobial resistant strains among community and healthcare systems.
However there are some principles of antibiotic prescription also available, that must be followed in order to minimize the burden of AMR. Those principles include microbiological therapy guidelines wherever possible, Indications which should be evidence based, Narrowest spectrum requirement, appropriateness of the drug dosage towards site & type of infections and minimization of the therapy duration and to maintain monotherapy in most of the cases. Beside the above principal’s antimicrobial surveillance, teamwork and open pragmatic approach may also adopt to overcome the problem of AMR. Studies are required to monitor local resistance and surveillance for this emerging threat of resistance by means of antimicrobial susceptibility testing of the pathogens responsible for UTI, specifically for E.coli .36 Enterococci are intrinsically resistant to a number of antimicrobial agents and can further easily acquire resistances which increase their potential to cause disease.37 In the present study, increased resistance for 2nd and 3rd generation Cephalosporins like Cefaclor and Cefotaxime is seen. The resistance rates were 79.3%, 67.2% respectively. So the increasing resistance to Cephalosporins prompted us to search for ESBL producers. The incidence of ESBL strains among clinical isolates have steadily increased over the past few years resulting in major problem for clinical therapeutics.
Detection of ESBL isolates is a huge burden for microbiological setups because the ESBL producer GNB may be seen susceptible to some β-lactam antimicrobial agents during In vitro studies and in turn may result into treatment failure. So proper identification is necessary. In this study, the isolation rate of ESBL producers gram-negative bacterial strains were found as 20 (34.4%). Concordant results of ESBL producer microorganisms were observed in 2012-2015 (27.67%).5,6,25,26
There are some organisms which may carry genes coding for resistance towards different classes of antimicrobials and many of those termed as MDR (multi-drug resistant) organisms. Many of isolates were observed to be multidrug resistant. Hence this present study provides an insight towards the emergence of increased antimicrobial resistance among urinary pathogens in the rural community which is majorly a result of indiscriminate and overdose of antibiotics. In addition, the present data may give some assistance to the physician’s towards treatment policies for urinary tract infections by avoiding misprescription of antimicrobial drugs.
High incidence of UTIs among females in 21-40 years age group is a great concern as in the reproductive age treating the infections with various drug resistant bacteria is very challenging. An increasing trend in production of ESBLs among UTI pathogens noted which is more prevalent in E.coli. ESBL producing bacteria make the treatment policies much complicated, In this regard, proper antimicrobial surveillance is needed in every healthcare system.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
AVAILABILITY OF DATA
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
ETHICS STATEMENT
This study was approved by the Institutional Ethics Committee, Shree Guru Gobind Singh Tricentenary University, Gurgaon, India.
- Oscar S, Saucedo JT, Garcia-Mora A, Dehesa- Davila M, Naber KG. Risk factors and predisposing conditions for urinary tract infection. Ther Adv Urol. 2019;11:19-28.
Crossref - Lekha Tuli, Sujeet Rai, Dilshad Arif, Deepak K. Singh. Bacteriological profile and Antimicrobial Susceptibility pattern ofisolates from Urinary Tract Infections in Eastern Uttar Pradesh, India. Int J Curr Microbiol App Sci. 2016;5(3):428-435.
Crossref - Das RN, Chandrashekhar TS, Joshi HS, Gurung M, Shrestha N, Shivananda PG. Frequency and susceptibility profile of pathogens and causing Urinary tract infection at a teritary care hospital in western Nepal. Singapore Med. 2006;47(4):281-285. PMID: 16572238
- Sabih A, Leslie SW. Complicated Urinary Tract Infections. 2021. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021. https://www.ncbi.nlm.nih.gov/books/NBK436013/
- Razak Sk, Guru SV. Bacteriology of Urinary tract infection and Antibiotic Susceptibility Pattern in a Territory care hospital in south India. Int J Med Sci Public Health. 2012;1(2):109-112.
Crossref - Shah LJ, Vaghela GM, Mahida H. Urinary tract infection: Bacteriological profile and its antibiotic susceptibility in western India.Natl J Med Res. 2015;5(1):71-74.
- Aggarwal R, Chaudhary U, Sikka R. Detection of Extended Spectrum β-lactamase Production Among Uropathogens. J Lab Physicians. 2009;1(1):7-10.
Crossref - Prakash D, Saxena RS. Distribution and antimicrobial susceptibility pattern of bacterial pathogens causing urinary tract infection in Urban Community of Meerut City, India. ISRN Microbiol. 2013;2013:749629.
Crossref - Laghawe A, Tripathi A, Saxena SB. Aetiology of urinary tract infection and antimicrobial susceptibility pattern of urinary isolates in tertiary care hospital in central India: A retrospective analysis. Int J Curr Microbiol Appl Sci. 2015;4:962-970
- Dugal S, Purohit H. Antimicrobial susceptibility profile and detection of Extended spectrum beta – lactamase production by gram negative uropathogens. Int J Pharm Pharm Sci. 2013;5(4):434-438.
- Shilpakar A, Ansari M, Rai KR, Rai G, Rai SK. Prevalence of multidrug resistant and extended spectrum beta-lactamase producing gram negative isolates from clinical samples in a tertiary care hospital of Nepal. Tropical Medicine and Health 2021;49(23):1- 9.
- Collee JG, Miles RS, Watt B. Tests for the identification of bacteria. In:ColleeJG, editor. Mackie and MCCartney Practical Medical Microbiology. 14th edition; New Delhi. 2014:131-150.
- Performance standards for antimicrobial susceptibility: sixteenth informational supplement. Wayne, PA, USA: CLSI; 2006. Clinical and Laboratory Standards Institute; pp. M100-S16
- Maripandi A, Al-Salamah AA, Amuthan M. Prevalence and Antibiotics Susceptibility of Uropathogens in Patients from a Rural Environment, Tamilnadu. Am J Infect Dis. 2010;6(2):29-33.
Crossref - Okonko IO, Ijandipe LA, lusanya I, et al. Incidence of Urinary tract infection among pregnant women in Ibadan, South Western Nigeria. Afr J Biotech. 2009;8(23):6649-657.
- Bano S, Tunio SA, Memon AA,Detho H, Bano R, Kumari K. Evaluation of Antibiotic susceptibility Pattern of Uropathogen circulating in Hyderabad; Pakisthan. Khyber Med University. 2014;6(3):110-115.
- Saha A, Majumdar T, Dasgupta A, Saumandal P. Prevalence of extended spectrum beta-lactamases (ESBLs) among uropathogenes at a tertiary care hospital in Tripura. The Health Agenda. 2015;3(2):55-62.
- Baron EJ, Finegold SM, Eds Microorganism encountered in the urinary tract In Bailey & Scott’s diagnostic microbiology (9th edition), Mosby publishers, St. Louis Missouri; 1994:256.
- Singhal A, Sharma R, Jain M,Vyas L. Hospital and Community Isolates of Uropathogens and their Antibiotic Sensitivity Pattern from a Tertiary Care Hospital in North West India. Ann Med Health Sci Res. 2014;4(1):51-56.
Crossref - Haque R, Akter ML, Salam MA. Prevalence and susceptibility of uropathogens: a recent report from a teaching hospital in Bangladesh. BMC Res Notes. 2015;8:416-420.
Crossref - Biswas R, Rabbani R, Ahmed SH, Sarker MAS, Zafrin N, Rahman M. Antibiotic sensitivity pattern of urinary tract infection at a tertiary care hospital. Bangladesh Crit Care J. 2014;2(1):21-24.
Crossref - Shah SU, Ahmed A, Gul I, Rahman G. Etiology and antibiotic resistance pattern of community- acquired urinary tract infections in children. KJMS. 2015;8(3):428.
- Patel P, Garala RN. Bacteriological profile and antibiotic susceptibility pattern (antibiogram) of urinary tract infections in paediatric patients. Journal of Research in Medical and Dental Science. 2014;2(1):20-23.
Crossref - Eshwarappa M, Dosegowda R, Vrithmani A, Khan MW, Kumar PS, Kempegowda P. Clinico-microbiological profile of urinary tract infection in south India, Indian J Nephrol. 2011;21(1):30-36.
Crossref - Hussein NS. Clinical, Etiology and Antibiotic Susceptibility Profiles of Community- Acquired Urinary Tract Infection in a Baghdad Hospital. Medical & Surgical Urology. 2014;3(2):1-5
- Agarwal P, Maheshwari M, Arora DR. Profile of bacterial uropathogens and their antibiotic susceptibility pattern among pregnant women in a rural setup of North India. Asian J Med sci, 2014;3(2):57-62.
- Kline KA, Lewis AL. Gram-Positive Uropathogens, Polymicrobial Urinary Tract Infection, and the Emerging Microbiota of the Urinary Tract. Microbiol Spectr. 2016;4(2):10.
Crossref - Khattar S, Sharma B, Bajaj A, Gupta K, Kaur IR. Bacteriological profile of uropathogens and their susceptibility pattern at a teaching hospital in north india: A retrospective study. Int J Scien Res. 2017;6(12):89-91.
- Sabharwal ER. Antibiotic susceptibility patterns of Uropathogens in obstetric Patients. N Am J Med Sci 2012;4(7):316-319.
Crossref - Lee BK, Crossley K, Gerding DN: The association between Staphylococcus aureus bacteremia and bacteriuria. Am J Med. 1978;65(2):303-306.
Crossref - Huggan PJ, Murdoch DR, Gallagher K, Chambers ST. Concomitant Staphylococcus aureus bacteriuria is associated with poor clinical outcome in adults with S. aureus bacteraemia. J Hosp Infect. 2008;69(4):345- 349.
Crossref - Goel V, Kumar D, Kumar R, Mathur P, Singh S. Community acquired Enterococcal urinary tract infections and antibiotic resistance profile in North India. J Lab Physicians. 2016;8(1):50-54.
Crossref - Huycke MM, Sahm DF, Gilmore MS. Multiple-drug resistant enterococci: the nature of the problem and an agenda for the future. Emerg Infect Dis. 1998;4(2):239- 249.
Crossref - Flores-Mireles AL, Walker JN, Caparon M, Hultgren SJ. Urinary tract infections: epidemiology, mechanisms of infection and treatment options. Nat Rev Microbiol. 2015;13(5):269-284.
Crossref - Kau AL, Martin SM, Lyon W, Hayes E, Caparon MG, Hultgren SJ. Enterococcus faecalis Tropism for the Kidneys in the Urinary Tract of C57BL/6J Mice. Infect Immun. 2005;73(4):2461-2468.
Crossref - Alos JI. Epidemiology and etiology of urinary tract infection in the community. Antimicrobial susceptibility of the main pathogen and clinical significance of resistance. Enfermedades Infectious y Microbiological Clinical. 2005;23(4):3-8.
- Murray BE. The life and times of the Enterococcus.Clin Microbiol Rev. 1990;3(1):46-65.
Crossref
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.