ISSN: 0973-7510
E-ISSN: 2581-690X
Bacteriological evaluation of contact surfaces (counters, chairs/benches, railings, poles, tables and door handles of rest rooms) at selected bus terminals in Uyo metropolis was carried out using standard procedures. Counters and poles revealed highest (log10 6.3 ± 0.7CFU/cm2) and least (log10 1.5±0.3 CFU/cm2 ), total heterotrophic bacterial counts respectively. Bacterial isolates associated with the contact surfaces included Staphylococcus aureus, S. epidermidis, Escherichia coli and species of the genera Bacillus, Micrococcus, Streptococcus, Proteus, Pseudomonas, Klebsiella and Serratia. Bacillus sp (16.6%) and Serratia sp (2.8%) revealed highest and least frequency distribution among the isolates. The Gram positive bacteria associated with the contact surfaces showed highest susceptibility (93%) to erythromycin and least susceptibility (12.2%) to augmentin while the Gram negative bacteria revealed highest (92.3%) and least (13.8%) susceptibility to ofloxacin and augmentin respectively. This study has revealed that contact surfaces at bus terminals can harbor potential pathogenic microorganisms hence adequate and regular sanitation practices should be embraced by staff and travelers at these places to avoid possible health risk.
Bacteriological, Contact surfaces, antibiotics, Pathogenic, Bus terminals
Bus terminals are places where commuters board and/or transport goods to different destinations which could be intra city or intercity. It may comprise both indoor and outdoor spaces or exist singly to form public spaces used by a large number of people that commute in public transport systems. Microorganisms constitute a major part of the ecosystem; soil, air, water and food as well as environmental surfaces or objects (Neely and Maley, 2002). Studies have implicated environmental surfaces in the transmission of microorganisms with the hands acting as chief organ for physical manipulation of the environment. Human hands usually habour microbes as part of the body normal flora as well as transient microorganisms contracted from the environment (Dodrill et al., 2011; Li et al., 2014; Koroglu et al., 2015; Fraser et al., 2015; Ayalew et al., 2015). Several organisms possess the potentials to survive on dry surfaces. These organisms have developed complicated separate physiologic resting stages which accord them the surviving advantage or potentials to hibernate effectively due to low water activity (Grant, 2004). Highly virulent spore forming pathogenic bacteria in the genus Bacillus have reportedly survived on dry surfaces by some studies (Singh et al., 2002; Toba et al., 2007)
Terminals in the transport systems serve as centres that aid the movement of people as well as goods daily thus making it the beehive of activities especially in a growing population such as Uyo metropolis. Several bus terminals in Uyo metropolis cater for people of different works of life, tribe and beliefs and their activities at this important centres is such that can involve possible interactions between different inanimate objects which could habour microorganisms. Adegoke and Okoh (2011) showed that inanimate materials like currency notes can serve as vehicles for transmission of plasmid bearing vancomycin resistant Staphylococcus aureus. The study showed the need for more surveillance on the antibiotic profiles of the bacterial contaminants to which large numbers of individuals are exposed.
Irrespective of where isolated, bacteria showing resistance to amoxicillin are likely Studies have revealed the microbiological status of contact surfaces at public areas in some towns/locality but there is paucity of such report about the contact surfaces at bus terminals in Uyo metropolis. This study aimed at assessing the level of microbial contamination on hand contact surfaces at selected bus terminals in Uyo metropolis.
Sample collection
Samples were collected from the contact surfaces using surface swab technique as adopted by (Cetin et al., 2006). The surface swabs from counters, railings, poles, door handles of rest rooms and chairs/benches were collected aseptically using sterile cotton swabs moistened with normal saline. This was done by rubbing the moistened cotton swabs firmly over the predetermined surface area using parallel stroke line with slow rotation with respectively chosen template surface area to be swabbed. The moistened sterile swabs were used to swab 20cm2 of the facilities. The swabs were replaced into their packs, sealed, labeled and transported in ice packs to University of Uyo Microbiology Laboratory for microbiological analysis
Microbiological quality assessment
Inoculation by direct streaking was done according to Cheesbrough (2006). This involved direct streaking of the swabs on the surface of the sterile molten culture media (Nutrient agar, MacConkey agar and blood agar) The surface swabs were processed using the swab-rinse method for enumeration of microorganisms associated with the contact surfaces. The swab sticks were agitated up and down in the tubes containing normal saline to aid on rinsing of the swab sticks. Serial dilution of the swab-rinse was made to provide appropriate dilutions from which Aliquots for inoculation unto sterile media were obtained.
Aliquot (1 mL) of swab-rinsed dilutions were inoculated unto appropriate culture media using pour plate according to Etok et al. (2004). Inoculated plates were incubated at 37oC for 18-24hours for the enumeration of Total Heterotrophic Bacterial Counts. Pure isolates were preserved in MacCartney bottle (Agar Slants) as stock cultures in the refrigerator at 4oC for further analysis. The bacterial isolates were characterized based on their colonial morphology, microscope appearance and biochemical characteristics using standard identification procedures as described by Holt et al., (1994).
Antibiotic sensitivity test
The antibiotic test was carried out to detect organisms that were susceptible or resistant to standard antibiotics. Each inoculum of the bacterial isolate was suspended in 2ml of sterile water and subsequently diluted to the turbidity of the McFarland standard. Susceptibility testing was carried out according to Clinical and Laboratory Standard Institute, CLSI (CLSI M100-S20-2010; M100-2017) using the commercially prepared antibiotic discs (Abtek Biologicals Ltd) with the following antibiotics; Amoxicillin (25µg), Gentamicin (10µg), Cotrimoxazole (25µg), Nitrofurantoin (20µg), Nalidixic acid (30µ), Ofloxacin (5µg), Augmentin (30µg), Tetracycline (10µg), Cloxacillin (5µg), Erythromycin (5µg), Streptomycin (10µg) and Chloramphenicol (10µg).
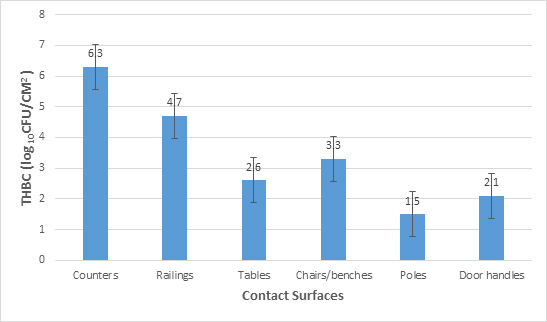
Large counts per cm2 were estimated in various sampling points for this study. Figure 1 shows the microbial load of the assessed hand contact surfaces at the bus terminals in Uyo metropolis. Highest (log10 6.3 ± 0.7CFU/cm2) and least (log10 1.5±0.3 CFU/cm2 ) microbial loads were shown by counters and poles respectively.
Fig. 1. Mean microbial load of hand contact surfaces at bus terminals. THBC means total heterotrophic bacterial count
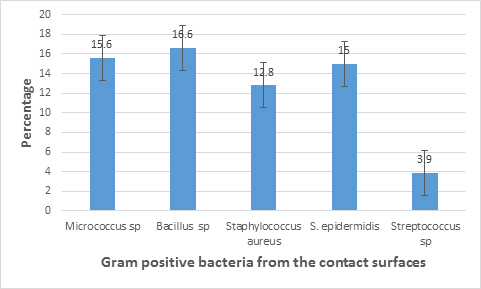
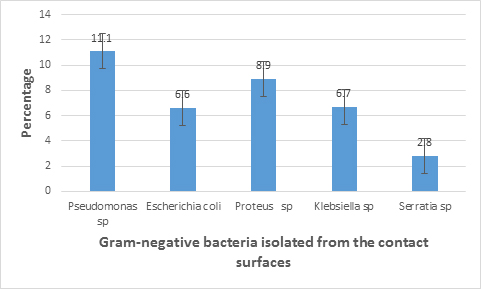
The results from characterization showed that both Gram positive and Gram negative bacteria were present in the sampling site. Figures 2 and 3 show the bacterial isolates and their frequency distribution on Hand contact surfaces at bus terminals in Uyo metropolis to include Staphylococcus aureus(12.8%), S.epidermidis(15%), Escherichia coli(6.6%) and species of Bacillus(16.6%), Micrococcus(15.6%), Streptococcus(3.9%), Proteus(8.9%), Pseudomonas(11.1%), Klebsiella (6.7%) and Serratia(2.8%).
Varying resistance profiles to various antibiotics used in this study were observed among both Gram-positive and Gram-negative bacteria. Tables 1 and 2 show the antimicrobial resistance for Gram-positive and Gram-negative isolates respectively. While Gentamicin was the least resisted antibiotic (7%) by Gram-positive bacterial isolates, Augmentin was the most (87.8%) resisted antibiotic against the Gram-positive isolates.
Table (1):
Antibiotic resistance of the isolated of gram positive bacteria from the contact surfaces (n=115).
| Antibiotics | Percentage resistance | ||||
|---|---|---|---|---|---|
| Micrococus sp | Bacillus sp | S. aureus | S. epidermidis | Streptococus sp | |
| Erythromycin | 70 | 35 | 65 | 100 | 55 |
| Gentamycin | 0 | 35 | 0 | 0 | 5 |
| Cloxacillin | 65 | 30 | 75 | 100 | 52 |
| Cotrimoxazole | 12 | 95 | 72 | 100 | 50 |
| Streptomycin | 0 | 37 | 30 | 5 | 15 |
| Tetracycline | 100 | 75 | 60 | 80 | 50 |
| Chloramphenicol | 100 | 36 | 55 | 58 | 45 |
| Augmentin | 100 | 92 | 85 | 100 | 70 |
Table (2):
Antibiotic resistance of the isolated gram-negative bacteria from the contact surfaces.
| Antibiotics | Percentage resistance per isolates | ||||
|---|---|---|---|---|---|
| Pseudomonas sp. | E. coli | Proteus sp | Klebsiella sp | Serattia sp | |
| Ofloxacin | 10 | 22 | 0 | 0 | 0 |
| Amoxicillin | 100 | 84 | 58 | 69 | 75 |
| Gentamycin | 8 | 20 | 13 | 15 | 0 |
| Nitrofuratoin | 47 | 68 | 30 | 30 | 80 |
| Nalidixic acid | 100 | 79 | 42 | 30 | 0 |
| Tetracycline | 100 | 72 | 37 | 20 | 50 |
| Augmentin | 100 | 87 | 79 | 83 | 55 |
| Cotrimoxazole | 65 | 60 | 5 | 25 | 75 |
Generally, the antimicrobial susceptibility for Gram negative bacterial isolates showed that ofloxacin was the most effective antibiotics for the Gram negative bacteria with a susceptibility rate of 92.3%, while Augmentin with a susceptibility rate of 13.8% was least effective on the Gram negative bacteria.
The results of this study revealed the microbial load of the assessed hand contact surfaces at the bus terminals to be in the order counter > railings > benches /chairs > tables > door handles of rest rooms > poles. The contamination level was proportional to the activities in the surface. Counters highest microbial load could be attributed to it being a surface that exist where intense activities such as buying of tickets, enquiry and complaint by passengers, drivers and all who visit these bus terminal interact. The least microbial load of the poles could be attributed to less interaction of people with this facility since it is a fixture that demarcates between units at the stations. The microbial load range of log10 1.5±0.3 CFU/cm2 – log10 6.3 ± 0.7CFU/cm2 on the contact surfaces at the bus terminals is indicative of poor sanitary status of facilities and could pose a health risk to commuters and staff at these station.
Surface bio contamination is a problem that has been shown to aid outbreaks of community-acquired and nosocomial infections through fomite transmission (Nwankiti et al.,2012) This study revealed the presence of both Gram positive and Gram negative bacteria on the contact surfaces. The bacterial isolates associated with the surfaces include Staphylococcus aureus ,S. epidermidis ,Escherichia coli ,and species of Micrococcus, Bacillus, Streptococcus, Pseudomonas, Proteus, Klebsiella and Serratia This results corroborates with the reports of Ikeh and Isamede (2011), Nwankiti et al. (2012), Orannusi et al. (2013), Fraser et al. (2014), Jaykus et al. (2014), Ayelew et al. (2015). The predominance of Bacillus on the surfaces could be attributed to its spore forming ability which probably cause it to be dispersed into the air and thus be able to settle on the surface of fomites, though some studies have reported the persistence of non-spore formers on dry surfaces (Adegoke and Okoh, 2011). It has, however, been shown to be a transient. Microflora of hands and can adapt to varying environmental conditions (Willey et al.,2008). The isolation of Staphylococcus aureus and S. epidermidis could be attributed to the ubiquitous nature of the organisms being found as part of the normal flora of the human skin and hands which often make contact with objects in the environment. Micrococcus spp. often present in fine dust particles may colonize the skin or mucus membrane of human. Pseudomonas spp, opportunistic pathogens are also found in soil. The presence of Streptocococcus sp. and Staphylococcus sp. indicate the possibility of mouth or nasal contamination (aerosol discharge from mouth and nose), i.e. body flora might have been shed to those surfaces by the passengers (Adegoke and Komolafe, 2009; Komolafe and Adegoke, 2008). The isolation of Escherichia coli from the contact surfaces is indicative of fecal contamination probably from the hands of people who do not practice proper hand washing after using the rest rooms or changing baby diapers at the bus terminals.
The bacterial isolates associated with the contact surfaces showed varying susceptibility pattern to commercial standard antibiotics. The most predominant Gram positive isolates (Bacillus spp.) showed highest (75%) and least (5%) susceptibility to Gentamicin and Cotrimoxazole respectively. The Gram positive isolates with least occurrence (Streptococcus spp.) however showed l00% resistance to Erythromycin, Cloxacilin, Cotrimoxazole and Augmentin and highest sensitivity (95%) to Gentamicin. Pseudomonas the predominant Gram negative isolate showed highest(90%) sensitivity to Ofloxacin and 100% resistance to Amoxacilin, Nalidixic acid, Tetracycline and Augmentin. Serratia which showed least occurrence among the Gram negative bacteria revealed 100% resistance to Nalidixic acid and 100% sensitivity to Ofloxacin and Gentamicin respectively. The Gram positive and Gram negative bacteria associated with the contact surfaces showed highest susceptibility to Gentamicin (93%) and Ofloxacin (92.3%) respectively while both Gram positive (12.2%) and Gram negative (13.8%) bacteria showed least susceptibility to Augmentin. The resistance pattern of bacteria associated with contact surfaces has been reported (Boma and Olieme, 2011; Akubuenyi et al., 2011; Ezeonu and Ugwu, 2011 and David et al., 2011). This present study however, contrasts with the report of Jombo et al. (2010) that indicated commonly isolated organisms from contact surfaces was sensitive to Augmentin.
This study is of further importance due to the recent calls by World Health Organization (WHO, 2017) for more research on some bacterial pathogens like Pseudomonas aeruginosa, Escherichia coli, Staphyloccocus aureus, and some others as isolated. The potential public health impacts of these bacteria when individuals become infected with them is of high significance, especially with increase in the populations of the immunocompromised individuals.
Potential pathogenic bacteria from large counts per surface areas were isolated and identified from contact surfaces in a bus terminal. The bacterial isolates showed resistance to some conventional antibiotics. The distribution of these bacteria were proportional to the frequency of contact of the surfaces. This present study has contributed to the paucity of information on the hygienic status of hand contact surfaces at the assessed bus terminals in Uyo metropolis. The isolation of pathogenic organisms from these surfaces indicates they can be vehicles for disease transmission at these important public settings. There is therefore the need to embrace adequate sanitary practices that can aid prevent contamination and interrupt disease spread through these surfaces at the bus terminals. It is also important to encourage good personal hygienic habits such as hand washing to prevent / control contamination or spread of diseases via these fomites.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
- Adegoke, A.A., and Okoh, A.I. The in vitro effect of vancomycin on multidrug resistant Staphylococcus aureus from hospital currency notes. African Journal of Microbiology Research, 2011; 5(14): 1881-1887.
- Adegoke, A.A. and Komolafe, A.O. Nasal colonization of school children in Ile-Ife by multiple antibiotic resistant Staphylococcus aureus. International Journal of Biotechnology and Allied Sciences, 2008; 3(1): 317-322
- Akubuenyi, F.C., Arikpo, G.E., Ogugbe, C.J., Mfongeh, J.F. and Akpanumun, E.V. Antibiotics resistance profile of waste water isolates obtained from University of Calabar Teaching Hospital and General Hospital ,Calabar,Nigeria. Nigerian Journal ofMicrobiology, 2011; 25: 2243-2250
- Ayalew, H., Berhanu, A., Sibhat, B. and Serda, B. Microbiological assessment of meat contact surfaces at abattoir and retail houses in Jigjiga town, Somali, National Regional State of Ethiopia. ISABB-Journal of Food and Agricultural Science, 2015; 5(3):21-26.
- Boma, A. and Oliemen, P. Sensitivity pattern among bacterial isolates neonatal Septicaemia in Port Harcourt. Annals of Clinical Microbiology and Antimicrobials, 2012; 11:7
- Cetin, O., Kahraman, T. and Buyukunal, S.K. Microbiological evaluation of food contact surfaces at red meat processing plants in Istanbul, Turkey. Italian Journal of Animal Science 2006; 5(3): 277 – 283
- Cheesbrough, M. District Laboratory Practice in Tropical Countries. (Part 2) Cambridge University Press, United Kingdom 2006.
- Clinical Laboratory Standards Institute (CLSI) Perfomance Standards for antimicrobial susceptibility testing,20th informational supplement (M100-S20).Clinical and Laboratory Standards Institute,Wayne,PA,USA 2010.
- CLSI. Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing; 27th Edition . M100-S27. Wayne, PA 2017.
- David. O.M., Akinunmi, A., Oluduro, A.O., Egbebi, A.O. and Famurewa,O. Antibiotic resistance and plasmid profile of bacteria pathogens isolated from drinking water in Ado-Ekiti, Nigeria. Nigerian Journal of Microbiology, 2011; 25: 2339-2344.
- Dodrill, L., Scmidt, W.P., Cobb, E., Donachie, P., Curtis, V. and De-Barra, M. The Effect of Hand Washing with water or soap on Bacterial Contamination of Hands. International Journal of Environmental Public Health Research, 2011; 8(1): 97-104.
- Etok, C.A., Udo, S.M. and Eja, M.E. General Microbiology Practical Manual. Abison Printing Press, Calabar, Nigeria 2004.
- Ezeonu, I.M. and Ugwu, K.O. Incidence of diarrhea and antibiotics resistance profiles of enteric bacteria in HIV positive individuals. Nigerian Journal of Microbiology, 2011; 24(2): 2251-2260
- Li, Y., Fraser, A., Chen, X., Cates, S., Wohigenant, K. and Jaykus, L.A. Microbiological analysis of environmental samples collected from child care facilities in North and South Carolina. Amreican Journal of Infectious Disease Control, 2014; 42(10): 1049-1055
- Fraser, A., Wohigenant, K., Cates, S., Chen, X., Jaykus, L.A., Li, Y. and Chapman, B. An Observational Study of Frequency of Provider Hand Contacts in Child Care facilities in North and South Carolina. American Journal of Infectious Disease Control, 2015; 43(2):107- 111.
- Grant, W.D. Life at low water activity. Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences, 2004; 29; 359(1448): 1249–1267.
- Holt, J.G., Krieg, N.R., Sneath, P.H., Stanley, J.T. and Williams, S.T. Bergey’s Manual of Determinative Bacteriology (9th edition) Williams and Wilking, Baltimore Maryland, USA 1994.
- Ikeh, E. and Isamade, E. Bacterial flora of fomites in a Nigerian multi-disciplinary intensive care unit. Laboratory Medicine, 2011; 42: 411-413.
- Jombo, G.T., Akpan, S., Epoke, J. and Etukumana, E.A. Antimicrobial Susceptibility profile of community acquired and nosocomial isolates of Staphylococcus aureus and that of coagulase negative Staphylococci from clinical blood culture specimens at a Nigerian University Teaching Hospital. Journal of Clinical Medicine and Research 2010; 2(6): 83-90.
- Komolafe, A. O. and Adegoke, A. A. Incidence of bacterial septicaemia in Ile-Ife Metropolis, Nigeria. Malaysian Journal of Microbiology, 2008; 4(2): 51- 61
- Koroglu, M., Gunai, S., Yildiz, F., Stravas, M., Ozar, A. and Altindis, M. Comparison of Keyboards and Touch-sreen Mobile Phones/Devices as Potential Risk for Microbial Contamination. The Journal of Infection in Developing Countries, 2015; 9(12): 1308-1314
- Neely, A. and Maley, M. Survival of enterococci and Staphylococci on hospital fabric and plastic. Journal of Clinical Microbiology 2000; 38: 724-726
- Nwankiti,O., Ndako, J., Nwakiti, A., Okeke, O., Uzoechina, A. and Agada, G. Computer keyboards and mouse: etiologic agents for microbial infections. Natural Science, 2012; 10(10):162-166.
- Orannusi I., Dahunsi, S., Owosio,O.and Olatile, T. Microbial profile of hands, food, easy contact surfaces and food surfaces: A case study in a University campus. Novus. International Journal of Biotechnology and Bioscience, 2013; 2(1):30-38.
- Singh, D.V., Thakur, K., Goel, K.A. Microbiological surveillance of currency. Indian Journal of Medical Microbiology, 2002; 20:53
- Toba, O., Loughrey, A., Goldsmith, C.E., Millar, B.C., Rooney, P.J., Moore, J.E. Survival of epidemic strains of nosocomial and community-acquired methicillin-resistant S. aureus on coins. American Journal of Infection Control, 2007; 35(5): 342-346.
- Willey, J.M., Sherwood,L.M. and Woolverton, C.J. Prescott,Harley and Klein’s Microbiology (7th Edition)McGraw –Hill Companies Inc.New York 2008.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.