ChAdOx1 nCoV-19 (AZD1222) is a replication-deficient chimpanzee adenovirus vectored vaccine developed by Oxford and AstraZeneca for a disease we all know as Coronavirus, or COVID-19. Ongoing clinical studies reveal that the ChAdOx1 nCoV-19 vaccine has a tolerable safety profile and is effective against symptomatic COVID-19. This vaccine may prove crucial in boosting herd immunity, averting life threatening illness, and relieving the current pandemic. In this mini review, we performed a thorough literature search through PubMed and Google Scholar and reported various case reports associated with complications of the adenovirus-vectored COVID-19 vaccine. Various adverse effects of the ChAdOx1 nCoV-19 vaccine were reported around the globe, which were often serious but rare and developed into life-threatening pathologies such as GBS, thrombocytopenia, demyelinating neuropathies, progressive dementia, cerebral infarction, IgA vasculitis, hemophagocytic lymphohistiocytosis, herpes zoster, cutaneous reactions, and vein thrombosis. These worldwide reported complications, which are usually rare and severe, will aid clinicians in understanding and managing unforeseen situations. There is a need for more research to find out more about these complications and their etiopathogenesis. However, the benefits of these vaccinations for stopping the spread of the outbreak and lowering the fatality rate outweigh the potential risk of the uncommon complications.
SARS-CoV-2, ChAdOx1 nCoV-19, Health, Complications, Thrombocytopenia
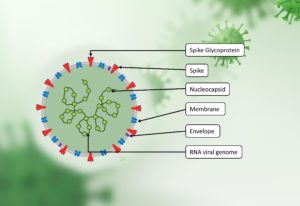
In December of 2019, a disease called Coronavirus was first observed in the province of Wuhan, China, caused by a novel coronavirus variant known as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) belonging to the family of Coronaviridae.1 Coronaviruses are large in size (diameter = 0.1 micrometers), spherical shaped (as shown in Figure 1)., enclosed in an envelope, and have positive strand RNA that acts as messenger RNA in the host body. Scientists from all over the world worked tirelessly to develop vaccines, and several were successful.2 The genomic knowledge of SARS-CoV-2 as well as its mutations urgently requires novel and distinctive techniques, which is why a number of novel medications have been again proposed and also discovered from various natural sources.3 Numerous products of natural origin were tried out for the treatment and prevention of COVID-19. Some carotenoids, such as crocin, which has the highest binding affinity for the SARS-CoV-2 replication complex, have been reported to be possibly beneficial for patients of COVID-19 because of their powerful antioxidant capabilities and also have the potential to be used as alternative treatments and dietary supplements for the prevention and treatment of COVID-19.4 The structural and molecular design of the SARS-CoV-2 genome has been accurately understood using bioinformatics techniques and various software programs. The primary three methods used in in silico research are computer aided drug design (CADD), next-generation sequencing, and genome-wide association (GWA)studies.3,5 One of the vaccines is known as ChAdOx1 nCoV-19, which is a replication-deficient chimpanzee adenovirus vectored vaccine developed by Oxford and AstraZeneca. The spike protein present on the surface of the SARS-CoV-2 is encoded by a gene that is expressed by the vaccine, instructing host cells to manufacture S-antigen protein, which is specific to SARS-CoV-2. This causes the body to produce an immunological response and encodes the knowledge in memory immune cells.6 Data from the trial’s initial analyses in the US indicate that the vaccination has a 76% effectiveness rate against symptomatic cases of SARS-CoV-2 exposure. The COVID-19 vaccine from AstraZeneca offers the public a safe and reliable defence against the virus’s potential risks, such as life-threatening illness, hospitalization, and fatality. 7 All of the evidence evaluated supports the finding that the known & possible benefits of the ChAdOx1-nCoV-19 vaccine exceed the known and potential threats. Only when the benefits of vaccination for the expectant mother outweigh any potential risks does the WHO recommend that pregnant women receive the AstraZeneca vaccine.7 Chinese biopharmaceutical manufacturer CanSino Biologics Inc. has developed an aerosol form of an adenovirus vaccine that is being marketed as Convidecia Air (Ad5-nCoV-IH) that also utilises a replication-deficient adenoviral vector.8 The Indian pharmaceutical company Bharat Biotech recently developed the nasal vaccine iNCOVACC (ChAd36-SARS-CoV-2-S), which is intended for active vaccination against SARS-CoV-2 viral infection.9 The CDSCO authority in India has authorised the commercialization of the iNCOVACC vaccine to fight coronavirus infection in individuals aged 18 years or older for specific usage in the interest of the public and emergency situations.9 After a thorough literature search, various complications associated with vaccination with ChAdOx1 nCoV-19 were compiled, and these are: acute demyelinating polyradiculoneuropathy with bilateral facial palsy, encephalitis; acute disseminated encephalomyelitis (ADEM), bilateral superior ophthalmic vein thrombosis (SOVT), ischemic stroke, and immune thrombocytopenia. Delayed cutaneous refection around the injection site of the vaccine (as shown in Figure 2), hemophagocytic lymphohistiocytosis, herpes zoster (shingles), IgA vasculitis in adults, malignant cerebral infarction, and rapidly progressive dementia with asymmetric rigidity.
Complications of ChAdOx1 nCoV-19
Acute demyelinating polyradiculoneuropathy with bilateral facial palsy
Nasuelli et al. presented a case of a 59-year-old Caucasian man who reportedly had postural instability and four limb distal paresthesia 10 days after receiving ChAdOx1 nCoV-19 (ABW12777).10 On day 15, patient had neurological evaluation. Patient’s past medical history was positive for hypertension and hyperuricemia. Physical examination revealed presence of conditions like global areflexia, gait ataxia and paresthesia of lower and upper limbs and pallesthesia or vibratory sensations was normal. Following Electromyography (EMG), it was discovered that the patient had motor polyradiculoneuropathy, absence of F reflex in any of the districts and temporal dispersion of the tibial nerve cMAP bilaterally. There was no sensory involvement, specifically no bilateral temporal sural nerve SAP. Electrophysiological results supported the diagnosis of demyelinating motor polyneuropathy. Routine lab tests were clinically insignificant and serological tests for known viral infections were conducted and results were negative.
After 24 hours, patient’s condition deteriorated as a result of bilateral facial palsy (House-Brackmann grade 5). LP was performed which revealed elevated proteins (140 mg/dl; normal – 20-40 mg/dl) and glycorrhachia in the CSF.10
Treatment IVIg at 0.4 mg/kg for 5 days improved the patients’ condition. His gait, forehead motility, and moderate movement improved slowly. With full effort, the eyes closed completely and the mouth was weak (House-Brackmann grade 4).
This case, presented by Nasuelli et al., reported a complication of Guillain-Barre syndrome (GBS) in a patient who got vaccinated with the ChAdOx1 nCoV-19 vaccine recently. Guillain-Barré is defined as an acute generalised inflammatory polyradiculoneuropathy condition that is generally associated with infections caused by viruses. But the aetiology and pathogenesis in this case seem to be immune-mediated. According to the postulated mechanism, the spike protein of SARS-CoV-2 or adenoviral vector components causes the production of cross-reactive antibodies to peripheral myelin proteins or to peripheral nerves and roots, thus causing an autoimmune process that destroys the neurons. This could involve T-cell-mediated immunological reactions. About two-thirds of individuals with GBS have previous infections, therefore, one of the proposed pathophysiologic mechanisms is the molecular similarity involving the nerve cell surface and microbial proteins. Earlier neurological manifestations were a well-known complication of SARS-Cov-2, and now there are reports of neurological side effects like GBS and CIDP (chronic inflammatory demyelinating polyneuropathy) after vaccination. A well-described GBS phenotype that occurs concurrently with SARS-CoV-2 immunisation is bilateral facial palsy with paresthesias. This case reported a possible correlation between the ChAdOx1 nCoV-19 vaccine and Guillain-Barre syndrome (GBS) in a patient without any predisposing factors or history of autoimmune conditions. 10 Several case reports have demonstrated that many GBS variations can manifest themselves following the injection of the ChAdOx1 nCoV-19 vaccination. The bilateral form of facial palsy and paresthesia appears to be the most common version. Literature data unequivocally demonstrates the need for careful observation of individuals who develop GBS following vaccination for COVID-19 just after the acute stage. Neurologists should be aware of this possibility even though a direct causal link between various COVID-19 vaccine types and autoimmune neuropathies has not yet been established. Prompt diagnosis and treatment are essential for improving outcomes in patients with GBS, CIDP, and other serious potential neurologic problems.10
ChAdOx1 nCoV-19 vaccine-associated encephalitis
Junko et al. covered a case of a 22-year-old woman with a 3-week history of recurrent headache in the frontal region and exhaustion following vaccination with the second dose of the ChAdOx1 nCoV-19 vaccine.11 Her symptoms didn’t resolve on administration of acetaminophen and advanced to tactile and visual hallucinations and confusion. The patient described seeing “disco balls” and feeling touches on her skin. She had a condition called “non-syndromic retinitis pigmentosa” and no other conditions. Her family and personal histories of psychiatric illness were unremarkable. The patient was awake but agitated by labile touch and disoriented by TPP. The patient reported auditory and visual hallucinations, such as believing the room was on fire, as well as frequently hyper-religious delusions. She was also seen gesturing in the air as if responding to unseen stimuli. On examination, apart from very restricted visual fields, there was no focal neurology, meningism, or photophobia, and the cardio-respiratory and abdominal examinations were also normal. The results of the PCR for SARS-CoV-2 and other respiratory viruses were negative. Based on her symptoms, she was started on IV Ceftriaxone 2g BD and Acyclovir 800 mg TDS for potential meningoencephalitis. To control her extreme agitation, she was administered lorazepam and haloperidol. Her antivirals and antibiotics were stopped as her fever disappeared. Her psychotic symptoms worsened, which required a change of drug, and haloperidol was replaced with olanzapine and promethazine as given for dystonia. Her brain MRI and EEG were unremarkable, and a sign of encephalopathy was seen. Her LP revealed an increased WCC count in CSF, which was evaluated as autoimmune encephalitis owing to the COVID-19 vaccine as described in Graus criteria. However, given the likelihood of infection, a repeat LP was ordered, and the administration of steroids and immunomodulatory medications was avoided as her clinical condition improved. As her condition improved, she was weaned off olanzapine, and no new episodes of hallucinations occurred. After some time, the patient had a mild relapse in her delirium along with other psychotic symptoms, visual and auditory hallucinations like seeing “green lights” and hearing “the sound of coins pouring into a dish,”, highly religiousness, non-stop prayers, and a labile mood. She was restarted on Olanzapine 5mg BD and was discharged with continuous follow-up appointments with neurology and psychiatry. The COVID-19 vaccine adverse event reporting programme was notified of the case. One month after being released, the patient is still taking a low dose of olanzapine and is capable of doing independent daily activities. Her family reports that she has not yet made a full recovery from her pre-morbid condition. She has become quieter and more isolated, although she still exhibits some psychotic symptoms, such as seeing green lights sometimes, praying more frequently than previously.11
The patient’s suffering during her condition and the diagnostic challenge exhibited in this case posed serious difficulties to the treating medical team. Limbic encephalitis was the presentation. Although neuroborreliosis was taken into consideration, due to negative serum serology, the absence of epidemiological risk, such as travel to endemic regions within the UK or overseas, resolution of the CSF pleocytosis, and normal brain imaging, it was felt that it was very unlikely and that very extensive workup for commonly known viruses is required. There are isolated cases of encephalitis associated with anti-NMDA receptors following vaccines for diseases such as polio, diphtheria, pertussis, Japanese encephalitis, and tetanus, as well as the H1N1 virus. It is well-known that COVID-19 can be worsened by psychiatric disorders, encephalopathy, encephalitis, and delirium, and that these diagnoses may be supported by possible biomarkers as well as imaging alterations. Although an asymptomatic COVID-19 infection at the time of immunisation cannot be ruled out, this diagnosis was made less plausible in this patient due to several negative SARS-CoV-2 PCR tests.11
ADEM after ChAdOx1 nCoV-19 vaccine
ADEM, or acute disseminated encephalomyelitis, is an inflammatory demyelinating CNS disease. It is characterised by an acute or chronic polyfocal neurological syndrome, usually with monophasic progression, which can possibly transform into a chronic inflammatory disease of the CNS. It generally occurs due to infections, but about 10% of cases are due to vaccinations. Numerous cases of ADEM have been reported in patients infected with SARS-CoV-2 since December 2019, while no case of ADEM has been reported after administration of mRNA or an adenovirus-vectored COVID-19 vaccine.
A 45-year-old man, who was previously free from any neurological conditions, presented with numbness in his hands 12 days after the administration of the first dose of the ChAdOx1 nCov-19 vaccine. 12 After a few days, his condition deteriorated, and the numbness from his hands extended to his legs, trunk, and all the upper limbs. He gradually began to have urge incontinence, dysphagia, dysarthria, and clumsy right-hand movements. On admission, the patient reported numbness in the abdomen, lateral calves, thighs, chest, and both upper extremities. Neurological examination revealed bilateral nystagmus on lateral gaze and right arm pronator drift. Brain MRI revealed large, poorly marginated T2-weighted hyperintensities in the pons, right thalamus, right cerebellar peduncle, and multiple spinal cord segments. Anti-SARS-CoV-2 IgG was positive, whereas IgM was not detected. Blood work and infectious workup tests were unremarkable. CSF analysis revealed mild lymphocytosis. A CSF scan of the entire body revealed no abnormalities.12
Treatment
The patient was started on a 5-day course of high-dose IV methylprednisolone, followed by orally administered Prednisone. Patients’ clinical symptoms improved in a few days. Numbness in the trunk and legs had gone away, but numbness in the hands and fingers remained. Right arm pronator drift and nystagmus were extensively resolved. After 10 days, a brain MRI revealed significantly reduced sizes of T2-weighted hyperintensities in previously discovered areas of the brain. With a gradual volume reduction of the spinal cord and brainstem regions and the ablation of the thalamic lesion, MRI follow-up at 1 and 2 months exhibited marked improvement. The MRI scan at the 4-month follow-up showed complete clinical remission and no relapses.12
The diagnosis done by the medical team led them to the conclusion and diagnosis of acute disseminated encephalomyelitis (ADEM). The clinical symptoms of the patient, including the presence of polyfocal neurological syndrome, which improves quickly on administration of corticosteroids, were all supportive of the diagnosis made. Following a brain MRI, it was discovered that there were large active lesions involving deep grey matter areas, excluding the periventricular and corpus callosum areas, which are commonly involved in multiple sclerosis (MS). The pathophysiology underlying this case was thought to be a cell-mediated mechanism; in this case, the triggering factor was the ChAdOx1 nCoV-19 vaccine, which elicited a T-cell immune response in addition to humoral immunity.12
Bilateral superior ophthalmic vein thrombosis (SOVT), ischemic stroke, and immune thrombocytopenia after ChAdOx1 nCoV-19 vaccination
A 55-year-old woman was admitted 10 days after her first dose vaccination with the ChAdOx1 nCoV-19 vaccine with clinical symptoms of retro-orbital pain, congestion in the conjunctiva, and diplopia. 13 She started having pyrexia and flu-like symptoms on the night of the vaccination, and similar symptoms reappeared after 7 days. Her past medical history was devoid of any visual difficulties, stroke, thrombocytopenia, risk factors for arterial disease, autoimmune disorders, thrombosis, or neurological disorders. On examination, it was revealed that the patient had bilateral visual acuity of 0.85 and binocular diplopia at right lateral and vertical gaze. An MRI scan revealed SOVT. Antiplatelet antibodies IgG and borderline IgM were found in lab tests, indicating significant ischaemic thrombocytopenia. To confirm the diagnosis of immune thrombocytopenia (ITP), a monoclonal antibody-specific immobilisation of platelet antigens assay and a PSIFT test were done and found to be positive. 13
Treatment
ITP was treated by daily administering IV dexamethasone 40 mg for 4 days, which improved the platelet count. Despite receiving therapeutic heparinization, the patient experienced a temporary, mild right-sided hemiparesis and aphasia 8 days after being admitted. An MRI revealed that the patient had an ischemic stroke in the left parietal lobe and territory of the cerebral artery. Following the onset of right-sided focal seizures that were managed by levetiracetam and lacosamide, the patient’s anticoagulation drug was changed to phenprocoumon, and 26 days after being admitted, she was released to go home.13
The patient in this case, who had previously been perfectly healthy and fine prior to receiving the ChAdOx1 nCoV-19 vaccine, developed symptoms of two rare conditions known as ITP and bilateral SVOT, as well as an event ischemic stroke and the flu, eight, ten, and eighteen days after vaccination.
On March 18, the European Medicines Agency released a review that aimed at alerting HCPs to the possibility of thromboembolisms such as pulmonary embolism, DVT, and cerebral venous sinus thrombosis after receiving the ChAdOx1 nCoV-19 vaccine.13
Delayed cutaneous reaction to the ChAdOx1 nCoV-19 vaccine
Kim et al. reported four cases of delayed cutaneous reactions after vaccination with ChAdOx1 nCoV-19 at the site of injection. The reaction occurred at least 3 days after the administration of the first dose. All patients were female and of Korean origin, and they worked as healthcare workers. None of them had any previous medical history of hypersensitivity reactions to any vaccine or medication except one, who had a history of allergic rhinitis but does not require continuous treatment for her condition. All of the patients had a severe delayed skin reaction that started with erythematous swelling, chills with fever, and myalgia on the day of the first dose vaccination and resolved before the delayed skin reactions appeared.A skin biopsy was done, which revealed perifollicular and superficial perivascular lymphocytic infiltration with scattered eosinophils.14
Treatment
The delayed cutaneous lesions were successfully ablated within 4–18 days on treatment with antihistamines given orally or corticosteroids given orally or topically.
The above findings suggest that hypersensitivity mechanisms may be responsible for the diagnosed condition, but the exact pathophysiological mechanism is still not clear. Contrary to PEG, which was used as an excipient in Pfizer-BioNTech’s BNT162b2 vaccine and produced hypersensitivity reactions15,16, polysorbate 80 was previously used in other vaccinations; however, it is still unknown which antigen in the ChAdOx1nCoV-19 vaccine caused these events. Thus, it concludes that delayed skin reactions to ChAdOx1nCoV-19 vaccine are not specifically due to the vaccine and may be caused by the excipient used since both the viral-vector vaccine (ChAdOx1nCoV-19 vaccine) and the mRNA vaccine (BNT162b2 vaccine) showed such hypersensitivity reactions.14
Hemophagocytic lymphohistiocytosis after vaccination with the ChAdOx1 nCoV-19 vaccine
Hemophagocytic lymphohistiocytosis, or HLH, is an erratic and rare disease that causes a lethal dysregulated hyper immune response in the body, similar to sepsis. Secondary HLH (sHLH) is caused by autoimmune disease, malignancy, or long-term infection and is also triggered by vaccination. The pathogenesis behind sHLH is not clear, but it may result from interference with the homeostasis of the immune system, which results in anomalous activation of immune cells such as T-cells, NK cells, and macrophages, which leads to excess production of inflammatory cytokines such as TNF-alpha, TNF-gamma, IL-1, IL-2, IL-6, and hemophagocytosis.17
A 68-year-old man presented with complaints of pyrexia associated with lethargy, rigors, and night sweats since the last seven days. His symptoms started about 10 days after vaccination with the first dose of the ChAdOx1 nCoV-19 vaccine. He was started on the antibiotics gentamicin and flucloxacillin for treating pyrexia of an obscure cause. Patient had previously travelled to a rural area two months prior. Physical examination disclosed splenomegaly. The patient had gout, hypertension, and Bowen’s disease and was being treated with diltiazem, hydrochlorothiazide, allopurinol, quinapril, prazosin, and colchicine. Biochemical investigations revealed hyponatremia, thrombocytopenia, and elevated levels of ferritin, LDH, D-dimer, AST, and ALT. Tests done for the detection of common and supposed viral infections were negative. A biopsy done on a sample taken from the bone marrow revealed a possible case of hemophagocytosis. His fever of 103.28 oF was resolved on the fifth day, along with relief from malaise and lethargy. After 4 weeks, his ferritin levels dropped from 11801 mcg/L to 710 mcg/L, platelets increased to 198 x 109/L, and a CT scan of the abdomen region showed improvement in his splenomegaly.17
The H-score is a biologic, clinical, and cytologic-based diagnostic HLH grading system created for adults. The H-score of our patient is 250, which indicates a greater than 99% likelihood that they have HLH. This case report supports the well-described connection between the ChAdOx1 nCoV-19 vaccine and HLH, as there was no evidence of infection or other precipitating factors that could have caused HLH in this patient. Conservative management was successful in treating the condition. Clinicians should take HLH into account when treating individuals who experience recurrent fevers after immunization with the ChAdOx1 nCoV-19 vaccine.17
Herpes zoster after vaccination with the ChAdOx1-nCoV-19 vaccine
This report contains three cases that show an association between the ChAdOx1 nCoV-19 vaccine and recurrence of the herpes zoster virus. The first case is about a 76-year-old female who presented with tight vesicular lesions on an erythematous ground on her right breast. Her symptoms appeared seven days after getting vaccinated with the ChAdOx1 nCoV-19 vaccine. The second case is about a 79-year-old male who presented with vesicular lesions over the right thigh six days after getting vaccinated. The third case is about a 70-year-old male who presented with the same vesicular lesions located on the left side of his neck and which appeared ten days after getting vaccinated. On examination of the skin, she excoriated tense vesicular lesions that were occasionally excoriated and associated with itching and burning. Based on the dermatological examination and clinical history, herpes zoster was diagnosed, and systemic antiviral medication was administered to all patients in accordance with guidelines, which led to the remission of the symptoms.18
Varicella-Zoster virus is responsible for causing chickenpox, which then remains in a latent state in dorsal root ganglia and cranial nerves and causes herpes zoster in the event of immunosuppression. SARS-CoV-2 infection undoubtedly causes immunosuppression, which then results in reactivation of the virus, subsequently leading to herpes zoster.19 Likewise, the ChAdOx1 nCoV-19 vaccine, upon administration, causes immunomodulation in the body, which may possibly lead to reactivation of the varicella virus, leading to herpes zoster. 20 Hence, these findings needed to be recognized.
IgA vasculitis in adult patients after vaccination with the ChAdoX1-nCov-19 vaccine
Immunoglobulin A-associated vasculitis (IgAV) is a rare immune cell disease in which IgA-containing immune complexes get deposited in the skin and other areas’ small blood vessels. The occurrence of IgAV is generally associated with various vaccines and infections.
A 72-year-old male presented with complaints of skin haemorrhages or vascular purpura. He had a medical history of T2DM, hypertension, asthma, and obesity and was on drugs to control them. 2 weeks after receiving the first dose of the ChAdOx1 nCoV-19 vaccine, he complained of joint inflammation and pain, or inflammatory arthralgia of the shoulders, ankles, and knees, as well as skin haemorrhage or vascular purpura. Biochemical investigations revealed increased levels of CRP, and other parameters were unremarkable. Tests were done to rule out SARS-CoV-2 infection, HIV-1, HBV, and HCV, which were all negative. A skin biopsy revealed evidence of vasculitis of small capillaries as well as excessive RBCs in the superficial dermal layer and the presence of neutrophils and eosinophils in the perivascular region with signs of leukocytosis. IgA deposits were discovered by immunofluorescence in the vessel wall, which led to the diagnosis of immunoglobulin A-associated vasculitis (IgAV).21
Treatment
This case of IgAV was successfully treated with corticosteroid drugs, specifically prednisone 20 mg/day.
The diagnosis of IgAV was confirmed, but its actual cause was unknown, which was supposed to be because of malignancies, viral or bacterial infections, drugs, or mostly vaccines. The mechanism behind this was supposed to be the possible imitation of an infectious state by the vaccine, which leads to an immune response similar to an immune response due to pathogens. To date, Vigibase® has received reports of nine (9) cases of IgAV.21
Malignant cerebral infarction after ChAdOx1-nCoV-19 vaccination
The ChAdOx1 nCoV-19 vaccine is associated with various complications, one of which is thrombotic thrombocytopenia induced by the vaccine (VITT). Recently reported cases give insight about several unusual cases of thrombosis associated with elevated PF-4 and thrombocytopenia after vaccination with the ChAdOx1 nCoV-19 vaccine.
A 57-year-old female presented with left hemiplegia, dysarthria, right gaze deviation, and left neglect caused by the occlusion of the middle cerebral artery (MCA) on the right side. She had previously received the ChAdOx1 nCoV-19 vaccine nine days prior. Biochemical investigations revealed a severe case of thrombocytopenia, severe progressive normochromic normocytic anemia, and a decreased level of hemoglobin. She underwent mechanical thrombectomy twice to reduce the risk of possible recurrence of occlusion of the right MCA, but unfortunately suffered from re-occlusion of the right MCA and malignant infarction, leading to rapid deterioration of her neurological state. A CT scan revealed thrombosis of the portal vein and pulmonary artery. The thrombotic clot collected from thrombectomy revealed that clot 1 was mainly composed of thrombocytes and neutrophils, while the second clot was made up of RBCs, fibrin, and platelets.22
Treatment
Patient was started on high-dose IVIG, which increased thrombocyte counts for a short period of time. Later, plasma exchange therapy was done, which gave an unremarkable result. The drug fondaparinux 2.5 mg OD was started via the subcutaneous route, which caused a marked increase in platelet count. ABG analysis revealed a worsening condition in the patient. A CT scan revealed a severe state of ARDS.22
This report presents a case of venous thrombosis due to VITT associated with malignant cerebral infarction along with thrombocytopenia after vaccination with the ChAdOx1 nCoV-19 vaccine.22,23
Rapidly progressive dementia with asymmetric rigidity after ChAdOx1 nCoV-19 infection
A female patient, previously healthy and in her 60s, was admitted a day after her 2nd dose of the ChAdOx1 nCoV-19 vaccine with complaints of behavioural anomaly and social seclusion in the form of movement without any purpose, rejection of food, and unrelated verbal replies. The patient’s condition deteriorated rapidly. She developed echolalia, or the repetition of words, and walking difficulty over 2 days, and over the next 5 days she developed confusion, hallucinations, and amnesia. She was introduced to various drugs such as amantadine, clonazepam, and trihexyphenidyl to stabilise her, but her condition worsened, and she developed irritability, increased amnesia, visual and auditory hallucinations, jumbled speech, and atypical limbs and jaw movement, along with dystonia of the neck. She was started on quetiapine after all previous drugs were withdrawn. On examination, the patient was immobile and in delirium. The patient had asymmetric rigidity on the left side as well as bilateral plantars that went up. Her biochemical tests, CSF analysis, and tests for autoimmune and viral encephalitis were also negative. An MRI scan revealed FLAIR hyperintensities in caudate heads bilaterally, which showed diffusion restriction, as well as patchy diffusion restriction in the left posterior parietal and occipital gyri. A repeat MRI scan was also performed 2 weeks later, which revealed worse results.23
Treatment
The patient was managed on a trial of broad-spectrum antimicrobials, dexamethasone, and antiepileptics and was later started on bromocriptine, which showed improvement in her condition. Still, the next 2 days were not in her favour as her neurological decline progressed inevitably, and the patient developed shock and respiratory distress before finally succumbing after 1 month of admission.23
The EEG and radiological findings in our patient point to a pathology that resembles prion disease, but there is a chance that a subsequent infection could cause sepsis and shock. Despite a few studies that have stated that COVID-19 was followed by prion disease, prion-like neurodegenerative diseases after COVID-19 immunisation have not been documented. In this case, two plausible mechanisms behind this complication of ChAdOx1 nCoV-19 may be considered. The first being the direct toxicity of S protein, which causes a neurodegenerative disease that mimics the pathophysiology of a prion disease. The second plausible mechanism could be that anti-S protein antibodies interact with host brain tissue antigens to trigger a type of autoimmune reaction. Such a mechanism is responsible for causing conditions like Guillain-Barré syndrome, stroke, VITT, and demyelinating diseases. Thus, long-term postmarketing surveillance of ChAdOx1 nCoV-19 is needed to prevent the occurrence of this rare event.23
COVID-19 was declared a public health emergency of international concern by the World Health Organization (WHO) on January 1, 2020. Coronavirus disease is an infectious illness brought on by the SARS-CoV-2 virus. Most virus-infected individuals will develop mild to severe respiratory distress and may recover without the need for special care. Despite that, some people will have severe symptoms and may require urgent care. To prevent the spread of infection, several vaccines were developed and given accelerated approval for use in the interest of public health and in emergency situations. These vaccines had a great impact on decreasing the spread of SARS-CoV-2 infection but also produced serious side effects, some of which got better without any intervention. The adenoviral vectored vaccine developed by AstraZeneca-Oxford was used in mass vaccinations against COVID-19. Global reports of the ChAdOx1 nCoV-19 vaccine’s side effects include serious but rare complications that may develop into life-threatening pathologies such as GBS, thrombocytopenia, demyelinating neuropathies, progressive dementia, cerebral infarction, IgA vasculitis, hemophagocytic lymphohistiocytosis, herpes zoster, cutaneous reaction, and vein thrombosis. Immunology and epidemiology, vaccine demand, and fair vaccine distribution must all be carefully taken into account when deciding when and who is eligible for COVID-19 immunisation boosters. There is a need for sufficient long-term follow-up with the clinician in these situations since, even with good treatment; a chronic version of the disease may develop. Numerous problems about the long-term reactogenicity of these immunisations remain unresolved since the virus is so unique. More clinical trial data is currently being awaited, which may aid in learning more about the safety of vaccines during pregnancy and lactation. To increase public confidence in COVID-19 vaccinations and to pinpoint all possible risk factors connected to each vaccine’s adverse reactions, more independent research on the efficacy and safety of all these vaccines is absolutely required.
ACKNOWLEDGMENTS
The authors would like to thank Chitkara College of Pharmacy, Chitkara University, Rajpura, Patiala, Punjab, India, for providing the necessary facilities to carry out the work.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
RS conceived and designed the manuscript. RS, HC and TBE performed visualization. RS, PK and RK critically reviewed the manuscript. RK, PK, OS and KD edited the manuscript. RS, HC and KD perform supervision. MAA, HK wrote the manuscript. All authors read and approved the final manuscript for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
Not applicable.
- Sharma A, Tiwari S, Deb MK, Marty JL. Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2): a global pandemic and treatment strategies. Int. J. Antimicrob. Agents 2020;56(2):106054.
Crossref - Pal M, Berhanu G, Desalegn C, Kandi V. Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2): an update. Cureus. 2020;12(3).
Crossref - Thapa K, Verma N, Singh TG, Grewal AK, Kanojia N, Rani L. COVID-19-Associated acute respiratory distress syndrome (CARDS): Mechanistic insights on therapeutic intervention and emerging trends. Int. Immunopharmacol., 2021; 101:.108328.
Crossref - Kciuk M, Mujwar S, Rani I, et al. Computational Bioprospecting Guggulsterone against ADP Ribose Phosphatase of SARS-CoV-2. Molecules, 2022;27(23), 8287.
Crossref - Ray M, Sable MN, Sarkar S, Hallur V. Essential interpretations of bioinformatics in COVID-19 pandemic. Meta Gene. 2021;27:100844.
Crossref - Mascellino MT, di Timoteo F, de Angelis M, Oliva A. Overview of the Main Anti-SARS-CoV-2 Vaccines: Mechanism of Action, Efficacy and Safety. Infect Drug Resist. 2021;14:3459-3476.
Crossref - The Oxford/AstraZeneca (ChAdOx1-S [recombinant] vaccine) COVID-19 vaccine: what you need to know. https://www.who.int/news-room/feature-stories/detail/the-oxford-astrazeneca-covid-19-vaccine-what-you-need-to-know?gclid=EAIaIQobChMIrPyF0t6S_QIVEDdgCh2ZFA4lEAAYASAAEgJH4fD_BwE. Accessed February 14, 2023.
- Inhalable vaccine in trials. 2023. https://www.nature.com/articles/d42473-022-00043-y. Accessed February 14.
- COVID-19 Vaccine ChAd36-SARS-CoV-2-S (Recombinant) By Bharat Biotech. Bharat Biotech.
- Nasuelli NA, de Marchi F, Cecchin M, et al. A case of acute demyelinating polyradiculoneuropathy with bilateral facial palsy after ChAdOx1 nCoV-19 vaccine. Neurological Sciences. 2021;42(11):4747-4749.
Crossref - Takata J, Durkin SM, Wong S, Zandi MS, Swanton JK, Corrah TW. A case report of ChAdOx1 nCoV-19 vaccine-associated encephalitis. BMC Neurol. 2021;21:1-5.
Crossref - Rinaldi V, Bellucci G, Romano A, Bozzao A, Salvetti M. ADEM after ChAdOx1 nCoV-19 vaccine: A case report. Multiple Sclerosis Journal. 2022;28(7):1151-1154.
Crossref - Bayas A, Menacher M, Christ M, Behrens L, Rank A, Naumann M. Bilateral superior ophthalmic vein thrombosis, ischaemic stroke, and immune thrombocytopenia after ChAdOx1 nCoV-19 vaccination. Lancet. 2021;397(10285):e11.
Crossref - Kim JE, Lee H, Paik SS, Moon J, Yoon HJ, Kim S. Delayed cutaneous reaction to ChAdOx1 nCoV-19 vaccine: is it an ‘AstraZeneca arm’? J Eur Acad Dermatol Venereol. 2021;35(11):e711.
Crossref - McMahon DE, Amerson E, Rosenbach M, et al. Cutaneous reactions reported after Moderna and Pfizer COVID-19 vaccination: a registry-based study of 414 cases. J Am Acad Dermatol. 2021;85(1):46-55.
Crossref - Farinazzo E, Ponis G, Zelin E, et al. Cutaneous adverse reactions after m-RNA COVID-19 vaccine: early reports from Northeast Italy. J Eur Acad Dermatol Venereol. 2021;35(9):e548.
Crossref - Attwell L, Zaw T, McCormick J, Marks J, McCarthy H. Haemophagocytic lymphohistiocytosis after ChAdOx1 nCoV-19 vaccination. J Clin Pathol. 2022;75(4):282-284.
Crossref - Vastarella M, Picone V, Martora F, Fabbrocini G. Herpes zoster after ChAdOx1 nCoV-19 vaccine: a case series. J Eur Acad Dermatol Venereol. 2021;35(12):e845.
Crossref - Tartari F, Spadotto A, Zengarini C, et al. Herpes zoster in COVID-19-positive patients. Int J Dermatol. 2020;59(8):1028.
Crossref - Eid E, Abdullah L, Kurban M, Abbas O. Herpes zoster emergence following mRNA COVID-19 vaccine. J Med Virol. 2021;93(9):5231.
Crossref - Badier L, Toledano A, Porel T, et al. IgA vasculitis in adult patient following vaccination by ChadOx1 nCoV-19. Autoimmun Rev. 2021;20(11):102951.
Crossref - De Michele M, Iacobucci M, Chistolini A, et al. Malignant cerebral infarction after ChAdOx1 nCov-19 vaccination: a catastrophic variant of vaccine-induced immune thrombotic thrombocytopenia. Nat Commun. 2021;12(1):4663.
Crossref - Chakrabarti SS, Tiwari A, Jaiswal S, et al. Rapidly progressive dementia with asymmetric rigidity following ChAdOx1 nCoV-19 vaccination. Aging Dis. 2022;13(3):633.
Crossref
© The Author(s) 2023. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.