ISSN: 0973-7510
E-ISSN: 2581-690X
Hepatitis B virus (HBV) is a significant public health concern, particularly in low-income countries. The host immune response plays an essential role in hepatitis B virus outcome, mainly orchestrated by cytokines and immune modulators molecules such as vitamin D. This study aimed to determine the levels of cytokines (IL-10), interferon-gamma (IFN-gamma), interferon-lambada (IFN-lambada), granzyme B, and vitamin D in Khartoum, Sudan, and to assess the association between them and variable HBV viral load as well as liver enzyme levels. A total of 174 participants were enrolled in the trial, with 100 HBV-infected patients (who were HIV and HCV negative) and 74 healthy volunteers. Patients’ HBV viral load, vitamin D levels, liver enzymes, and circulatory cytokines were measured. This study revealed a remarkable decrease in vitamin D levels in HBV infected patients with an elevation in levels of liver biomarkers. The level of IL-10 was significantly higher in patients than in apparently healthy controls (p-value <0.001). There was a positive correlation between IFN-ϒ, Granzyme B, and viral load. According to the findings of this study, there is a marked hypovitaminosis D among hepatitis B infected patients with a significant increase in IL-10, which may implicate the persistence of HBV infection. Moreover, HBV DNA levels were significantly associated with IFN- g and granzyme B levels.
Hepatitis B Virus, Cytokines, Vitamin D
Hepatitis B virus is a partially double-stranded DNA enveloped virus belonging to the family Hepadnaviridae. It is a highly contagious virus that causes more than 240 million chronic hepatitis cases worldwide.1 Infection with HBV is a significant health concern that can be fatal due to frequent adverse outcomes, including severe clinical acute hepatitis, liver failure, and hepatocellular carcinoma (HCC). Chronic HBV infection is responsible for more than 50% of cirrhosis and hepatocellular carcinoma, with a 0.5-1 million mortality rate.2-4 In Sudan, asymptomatic HBV infection is common in middle-aged adults and has been linked with higher cases of liver cancer.2 In humans, HBV is among the most prominent etiological causes of HCC. It can cause HCC either directly by triggering cellular oncogenes or indirectly by causing persistent liver damage, allowing the mutation to occur. According to the emerging data, inflammation appears to have a critical role in advancing liver disease. Therefore, early diagnosis of infected patients and viral load determination will provide guidelines for a better treatment option.5
Interactions between host defense and HBV determine the manifestations of HBV infection. Innate immune mechanisms play a significant role in HBV clearance by producing inflammatory cytokines. Cytokines represent a large group of low molecular weight glycoproteins that play a crucial role in orchestrating the host immune responses against hepatitis B virus infection.6 It includes cytokines associated with Type 1 (Th1) T cell responses interleukin-2, interferon, which play a vital role in host defense; type 2 (Th2) T cell responses (IL-4,6 and 10), that contribute to humoral immune responses; and regulatory T cell-associated cytokines. There is an imbalance in cytokine secretion in chronic HBV-infected patients, showing that IFNs are weakly induced, and IL-10 is markedly elevated in patients’ serum.7 These may directly affect viral replication and liver function biomarkers.8-10
Vitamin D is a fat-soluble vitamin that aids bone formation and hemostasis. Vitamin D deficiency has already been related to carcinogenesis and the progression of several infectious diseases, including liver infections. Recently, it has been shown to regulate innate and adaptive immune responses against viral infections.11 Low levels of vitamin D concentration were linked with decreased host immune response and increased HBV DNA load.12 Therefore, the host immune response status is critical for determining the outcome of HBV infections such as liver injury, persistent infections, and/or viral clearance.13 The present study sought to determine the association between vitamin D, cytokines, and viral loads in Sudanese patients infected with HBV.
Study Design
A cross-sectional analytical case-control study was designed to include 174 participants. This study was carried out at the Research Center, Ibnsina University, Khartoum, Sudan, from December 2019 to December 2020. The Ethical Approval Committee approved the study, the Medical and Health Campus, University of Khartoum, and all participants provided prior informed consent. Before participating in the study, all volunteers were given a clear explanation of the study’s aim.
Sample Size
This study comprised 174 participants, including 100 patients, and 74 healthy control. The sample size was calculated using an online sample size calculator, OpenEpi software, with the HBV prevalence rate of 6.8% in Sudan.14 The sample size was 98 with 95% CI. A random sampling method was used to collect samples from Fedail Private Hospital in Khartoum, Sudan.
Inclusion and Exclusion Criteria
This study excluded all subjects who had undergone the Vitamin D treatment protocol. Patients with Hepatitis B virus infection were chosen using a stringent selection criterion; the included patients did not have HIV, HCV infection, or other systemic diseases. The study included age-matched control participants without having HBV, HIV, HCV, infection, or another systemic disease.
Sampling
Blood samples were taken in a simple container, centrifuged, and the extracted sera were stored at -20°C unless required.
Estimation of Cytokines
To estimate the level of cytokines enzyme-linked immunosorbent assay was utilized. Commercially available kits were purchased, and samples were processed in accordance with the manufacturer’s recommendations. For interferon lambda (IFN- l) and granzyme B kits from R& D system, bio-techne brand, Minneapolis, USA (IFN- l Cat No. DY1598B-05; granzyme B Cat No.DY2906-05) were used. For IL-10 and interferon-gamma (IFN-g) kits of BioLegend, San Diego, USA, (IFN-g Cat No.430104) (IL-10 Cat No.430604) were used.
Serum Biochemistry
Routine automated methods (Cobas, Roche Diagnostics, Germany) were used to determine the level of biomarkers like serum aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphates (ALP), albumin (ALB), total protein (TP,), g-glutamyltransferase (GGT), bilirubin total (BILT) and bilirubin direct measurements.
Based on chemiluminescent immunoassay technology, serum vitamin D level was measured by Elecsys Vitamin D total (Cobas, Roche Diagnostics, Germany, ms_05894913190V8.0). Kits manufacturers provided control materials to optimize the protocols for all measurements.
Detection of HBV DNA and Viral Load Estimation
Serum HBV DNA level was quantified using AccuPower® HBV Quantitative PCR Kit V4.2 (BIONEER, South Korea). The lower limit of quantification was 20 IU/ml. Patients are categorized into three categories based on viral load, and later for easy calculations, viral load is converted into log10 values. The categorization of viral load is based on WHO guidelines for the chronic HBV infection management, HBV viral load > 20000 evidence of high-level HBV replication, HBV DNA levels that fluctuate between 2000 and 20,000 IU/mL were considered clinically significant, and a value <2000 IU/mL evidence of low levels of HBV DNA replication.
Statistical Analysis
SPSS version 24.0 and GraphPad Prism were used for data analysis. To determine the association of variables with viral load, the data were expressed as mean with standard deviation, and the Mann-Whitney test was used. Statistical significance was considered when the P-value was equal to or less than 0.05. Linear regression analysis examined the correlations between cytokines, biochemical variables, and viral load.
Demographic and Clinical Findings of the Study Population
This study included 100 HBV cases and 74 healthy controls (Table 1). In the study population, no statistical difference was observed in the gender of patients with the frequency of HBV infection; however, mean age was higher in cases (38.98±13.89) when compared with healthy controls (29.95±8.10), and it was statistically significant with a p-value <0.001.
Table (1):
Study participants’ demographic fcharateristics (n = 174).
| Characteristics | Cases (n=100) | Control (n=74) | P-value | |
|---|---|---|---|---|
| Age (years) | Mean ± s.d. | 38.93±13.89 | 29.95±8.10 | <0.001 |
| Gender | Male | 63 | 52 | 0.37 |
| Female | 37 | 22 | ||
The clinical findings of the 100 HBV-infected patients indicated that serum liver biomarkers’ mean level was highly elevated. In contrast, there was a marked decrease in serum 25 (OH) D in the patients (Table 2).
Table (2):
Laboratory findings of the 100 HBV patients investigated.
Biomarkers |
Mean ± s.d. n (%) |
|---|---|
Total Protein (g/dL) |
7.92±0.50 |
Albumin (g/dL) |
4.03±0.41 |
Total Bilirubin (mg/dL) |
0.77±1.02 |
Direct Bilirubin (mg/dL) |
0.38±0.94 |
ALP (U/L) |
96.51±32.50 |
ALT (U/L) |
68.85±297.80 |
AST (U/L) |
44.15±91.67 |
GGT (U/L) |
34.72±32.36 |
Serum 25(OH)D (ng/mL) |
13.54±6.17 |
Viral Load Log10 (IU/L) |
3.87±1.81 |
Viral Load Log10
| 47 (47%) |
Viral Load Log10 3.3- 4.3 IU/ml |
20 (20%) |
Viral Load Log10 >4.3 IU/ml |
33 (33%) |
Cytokines Level in HBV-infected Patients
There was no statistical difference in the levels of IFN-g and IFN-l among HBV-infected patients. However, a statistically significant difference was observed in the level of IL-10 in HBV-infected patients (12.61±10.43) when compared with apparently healthy controls (7.84±6.98) with a p-value of 0.001 (Table 3).
Table (3):
Concentrations of cytokines in HBV infected patients and healthy controls.
Cytokines |
Cases (n=100) Mean s.d. |
Controls (n=74) Mean s.d. |
P-value* |
|---|---|---|---|
IFN-γ (pg/mL) |
33.99±41.30 |
26.57±31.25 |
0.19 |
IL-10 (pg/mL) |
12.61±10.43 |
7.84±6.98 |
0.001 |
Granzyme B (pg/mL) |
196.68±194.35 |
196.94±170.85 |
0.99 |
IFN-λ (pg/mL) |
122.67±43.28 |
116.30±64.40 |
0.42 |
s.d=standard deviation; *A nonparametric Mann-Whitney test was utilized.
Association of Serum Cytokines and Biochemical Indexes with HB Viral Load
To investigate whether there is an association between the viral load and cytokines level and liver biomarkers, we categorized the HBV-infected patients based on the viral load into three groups: group 1: patients with viral load log10 < 3.3 IU/ml, group 2 patients have viral load log10 3.3- 4.3 IU/ml, and group 3 with log10 > 4.3 IU/ml. Compared to the other groups, group 3 significantly increased IFN-g and granzyme B concentrations. In comparison, there were no statistically significant differences in IL-10 and Granzyme levels between the groups (Table 3).
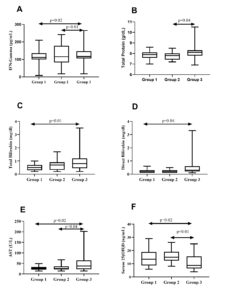
There was a significant difference in IFN-g total protein, bilirubin, and AST levels between the three groups of HBV patients. These biochemical indexes were more significantly increased in HBV-infected individuals with a viremia than log10 > 4.3 IU/ml, as presented in Figure. Serum 25(OH) D levels in group 3 participants were substantially lower than in groups 1 and 2 (Table 4).
Table (4):
Comparative analysis of cytokines and biochemical indexes among HBV patients with different viral load range.
| Variable | Viral Load (HBV-DNA) | Group 1 vs. 2 | Group 2 vs. 3 | Group 1 vs. 3 | ||
|---|---|---|---|---|---|---|
| Log10 < 3.3 IU/ml (n=47) | Log10 3.3-4.3 IU/ml (n=20) | Log10 > 4.3 IU/ml (n=33) | ||||
| IFN-γ (pg/mL) | 26.86 ± 18.14 | 35.34 ± 55.93 | 43.31 ± 52.74 | 0.55 | 0.01 | 0.02 |
| IL-10 (pg/mL) | 12.28 ± 11.08 | 12.16 ± 9.31 | 13.35 ± 10.39 | 0.96 | 0.67 | 0.66 |
| Granzyme B (pg/mL) | 196.95 ± 202.95 | 131.36 ± 58.42 | 235.88 ± 226.66 | 0.16 | 0.06 | 0.07 |
| IFN-Lambda (pg/mL) | 117.12 ± 38.00 | 124.80 ± 53.52 | 129.26 ± 43.88 | 0.78 | 0.65 | 0.25 |
| Total Protein (g/dL) | 7.88 ± 0.39 | 7.73 ± 0.36 | 8.08 ± 0.65 | 0.14 | 0.04 | 0.15 |
| Albumin (g/dL) | 4.04 ± 0.38 | 3.98 ± 0.44 | 4.04 ± 0.46 | 0.52 | 0.61 | 0.97 |
| Total Bilirubin (mg/dL) | 0.52 ± 0.22 | 0.67 ± 0.37 | 1.20 ± 1.68 | 0.14 | 0.20 | 0.01 |
| Direct Bilirubin (mg/dL) | 0.21 ± 0.13 | 0.23 ± 0.13 | 0.01 ± 0.03 | 0.70 | 0.07 | 0.04 |
| ALP (U/L) |
89.12 ± 27.35 | 99.30 ± 32.62 | 105.33 ± 37.35 | 0.19 | 0.55 | 0.08 |
| ALT
(U/L) |
29.93 ± 12.52 | 33.30 ± 21.54 | 145.81 ± 514.54 | 0.42 | 0.34 | 0.12 |
| AST (U/L) |
27.95 ± 9.48 | 29.75 ± 13.71 | 75.93 ± 155.57 | 0.54 | 0.04 | 0.02 |
| GGT (U/L) |
31.12 ± 16.04 | 30.90 ± 14.93 | 42.15 ± 51.50 | 0.95 | 0.60 | 0.40 |
| 25 (OH)D (ng/mL) | 14.27 ± 6.39 | 15.74 ± 5.22 | 11.18 ± 5.78 | 0.25 | 0.02 | 0.01 |
Figure. Association of cytokines and biochemical indexes with viral load in HBV patients: Group 1 indicates patients with viral load log10 < 3.3 IU/ml. Group 2 indicates patients with log10 3.3-4.3 IU/ml, and Group 3 is for the patients’ log10 >4.3 IU/ml. P-values are adjusted for multiple correction analysis.
Correlations of Viral Load with 25(OH)D and Cytokines Level
Serum 25(OH)D was negatively correlated with the viral load in HBV-infected patients (R=-0.31, P<0.01). No significant correlation was observed between the levels of cytokines and viral load. Intercorrelation analysis of different cytokines suggests a substantial correlation between IFN-g and IL-10 (R=0.25, P=0.01) (table 5).
Table (5):
Correlations of viral load with cytokines and serum 25(OH)D.
| Variables | Statistics | IFN-Gamma | IL10 | IFN-Lambda | Granzyme B | Serum 25(OH)D |
|---|---|---|---|---|---|---|
| Viral Load | PC | 0.19 | 0.03 | 0.10 | 0.007 | -0.31 |
| P-value | 0.05 | 0.71 | 0.27 | 0.94 | <0.01 | |
| IL-10 | PC | 0.25 | ||||
| P-value | 0.01 | |||||
| Granzyme B | PC | 0.001 | 0.01 | -0.08 | ||
| P-value | 0.99 | 0.85 | 0.40 | |||
| IFN–λ | PC | 0.003 | 0.26 | -0.5 | ||
| P-value | 0.97 | 0.008 | 0.61 | |||
| Serum 25(OH)D | PC | -0.05 | 0.09 | -0.05 | -0.01 | |
| P-value | 0.62 | 0.33 | 0.611 | 0.85 |
PC: Pearson correlation; significant p value ≤ 0.05.
Currently, HBV infection is distributed worldwide, particularly in low-income countries. Most HBV infections are asymptomatic, and the virus remains persistent within the liver cells for years. The mean age of hepatitis B infection is 20-39 years old, with socioeconomic effects representing the active workforce age.16 Similarly, hepatitis B infected patients included in this study were 38.93±13.8 who had been born before the nationwide hepatitis B immunization program for newborns.
The non-cytopathogenic replication behavior of the virus in the liver cells and the immune response mediated through cytokines play a crucial role in the different outcomes. Therefore, the circulating levels of immune mediators, including IFN-g and IFN-l, IL-10, and granzyme B, were compared between HBV-infected patients and healthy controls. The present study showed a substantial increase of IL-10 in HBV infected patients compared with healthy individuals. Interleukin 10 is a cytokine with anti-inflammatory properties; it can suppress the antiviral immune response and sustain the host cells’ viral.17 The elevated IL-10 levels in HBV-infected patients have been reported in several studies.18,19 Previously, it was shown that the low levels of IL-10 were associated with severe exacerbation of disease.20 The high detected levels of IL-10 match with the clinical conditions of apparently asymptomatic patients.
IFN-g and IFN-l are indispensable for the innate and adaptive host defense against viral infection through retarding viral replication.21,22 The present study showed that type II and type III IFN levels were comparable to healthy controls. Consistently, the chronic inactive HBV infection was not associated with increased IFN-g and IFN-l.23,24 However, our study significantly correlated high levels of IFN-g and granzyme B with increased viral load. Granzyme B is one of the NK cell weapons, which can mediate liver cell damage and increase HBV load.25
The significant viral load was found to be correlated to impaired liver function tests.26 The present study illustrates a significant association between increased AST activity and high viral load. Similar findings were reported by Esmaeelzadeh et al. in Iranian HBV patients.27 Taken together, AST might be used as a biomarker to predict viral load in chronic inactive HBV patients.
Vitamin D plays a decisive role in augmenting innate host defense against viral infection by regulating multiple genes encoding immune mediators, including cytokines, chemokines, and pro-apoptotic factors.28 This study illustrates that serum 25(OH) D was severely deficient in HBV patients. Moreover, serum 25-hydroxyvitamin D was negatively correlated with the viral load in HBV-infected patients. Vitamin D can suppress transcription and progeny HBV production, but HBV can evade the Vitamin D signaling pathway by downregulating the Vitamin D receptor.29 Meanwhile, Vitamin D deficiency associated with high viral load was previously reported.12,30,31 These findings recommend that Vitamin D therapy combined with antiviral chemotherapy can hasten HBV eradication.
This study has some limitations, including the study’s design, a cross-sectional, that can not precisely show the temporal relationship of the studied variables to be observed. In addition, the sample size was relatively small, and all patients enrolled in this study were from Khartoum State, which may not represent the whole country. Moreover, a significant difference in age between cases and control is another limitation of this study. The mean age of the HBV patients was higher than control, and the immune system gets affected in elder age. Further experiments such as molecular characterization of HBV and longitudinally measurement of cytokines profiles and vitamin D will provide a better option for the treatment intervention.
In conclusion, there was a marked hypovitaminosis D among hepatitis B infected patients with a significant increase in IL-10, which may involve the persistent infection of HBV. In addition, IFN-g and granzyme B levels were significantly correlated with HBV DNA levels.
In HBV infection, these serum biomarkers show promising predictive biomarkers for the progression and management of the disease. Thus, there is a need for an inclusive understanding of the effect of cytokines and vitamins in chronic HBV infection.
ACKNOWLEDGMENTS
None.
CONFLICT OF INTEREST
The author declares that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This article does not contain any studies with human participants or animals performed by any of the authors.
- Jefferies M, Rauff B, Rashid H, Lam T, Rafiq S. Update on global epidemiology of viral hepatitis and preventive strategies. World J Clin Cases. 2018;6(13):589-599.
Crossref - Ibrahim SAE, Mohamed SB, Kambal S, et al. Molecular Detection of Occult Hepatitis B virus in plasma and urine of renal transplant patients in Khartoum state Sudan. Int J Infect Dis. 2020;97:126-30.
Crossref - Teng CF, Li TC, Wang T, et al. Increased infiltration of regulatory T cells in hepatocellular carcinoma of patients with hepatitis B virus pre-S2 mutant. Sci Rep. 2021;11(1):1136.
Crossref - Wang WT, Zhao XQ, Li GP, et al. Immune response pattern varies with the natural history of chronic hepatitis B. World J Gastroenterol. 2019;25(16):1950-1963.
Crossref - Li H, Yan L, Shi Y, et al. Hepatitis B Virus Infection: Overview. Adv Exp Med Biol. 2020;1179:1-16.
Crossref - Xia Y, Protzer U. Control of Hepatitis B Virus by Cytokines. Viruses. 2017;9(1):18.
Crossref - Rybicka M, Woziwodzka A, Sznarkowska A, et al. Genetic variation in IL-10 influences the progression of hepatitis B infection. Int J Infect Dis. 2020;96:260-265.
Crossref - Wang M, Hou Y, Meng SH, et al. Abnormal IL-10 levels were related to alanine aminotransferase abnormalities during postpartum in HBeAg positive women with chronic hepatitis B. Medicine (Baltimore). 2019;98(46):e17969.
Crossref - Li MH, Lu Y, Zhang L, et al. Association of Cytokines with Alanine Aminotransferase, Hepatitis B Virus Surface Antigen and Hepatitis B Envelope Antigen Levels in Chronic Hepatitis B. Chin Med J (Engl). 2018;131(15):1813-1818.
Crossref - Vinhaes CL, Cruz LAB, Menezes RC, et al. Chronic Hepatitis B Infection Is Associated with Increased Molecular Degree of Inflammatory Perturbation in Peripheral Blood. Viruses. 2020;12(8):864.
Crossref - Siddiqui M, Manansala JS, Abdulrahman HA, et al. Immune Modulatory Effects of Vitamin D on Viral Infections. Nutrients. 2020;12(9):2879.
Crossref - Ko WS, Yang YP, Shen FP, et al. The Study of Correlation Between Serum Vitamin D(3) Concentrations and HBV DNA Levels and Immune Response in Chronic Hepatitis Patients. Nutrients. 2020;12(4):1114.
Crossref - Iannacone M, Guidotti LG. Immunobiology and pathogenesis of hepatitis B virus infection. Nat Rev Immunol. 2022;22(1):19-32.
Crossref - Mudawi HM. Epidemiology of viral hepatitis in Sudan. Clin Exp Gastroenterol. 2008;1:9-13.
Crossref - WHO. Guidelines Approved by the Guidelines Review Committee. Guidelines for the Prevention, Care and Treatment of Persons with Chronic Hepatitis B Infection. Geneva: World Health Organization. 2015. https://www.ncbi.nlm.nih.gov/books/NBK305553/
- Kolou M, Katawa G, Salou M, et al. High Prevalence of Hepatitis B Virus Infection in the Age Range of 20-39 Years Old Individuals in Lome. Open Virol J. 2017;11:1-7.
Crossref - Wilson EB, Brooks DG. The role of IL-10 in regulating immunity to persistent viral infections. Curr Top Microbiol Immunol. 2011;350:39-65.
Crossref - Arababadi MK, Pourfathollah AA, Jafarzadeh A, Hassanshahi G. Serum Levels of IL-10 and IL-17A in Occult HBV-Infected South-East Iranian Patients. Hepat Mon. 2010;10(1):31-5. https://pubmed.ncbi.nlm.nih.gov/22308123/
- Ozguler M, Akbulut HH, Akbulut A. Evaluation of Interleukin-10 Levels in Patients Diagnosed with Chronic Hepatitis. West Indian Med J. 2015;64(2):71-75.
Crossref - Wang K, Wu ZB, Ye YN, et al. Plasma Interleukin-10: A Likely Predictive Marker for Hepatitis B Virus-Related Acute-on-Chronic Liver Failure. Hepat Mon. 2014;14(7):e19370.
Crossref - Phillips S, Mistry S, Riva A, et al. Peg-Interferon Lambda Treatment Induces Robust Innate and Adaptive Immunity in Chronic Hepatitis B Patients. Front Immunol. 2017;8:621.
Crossref - Kang S, Brown HM, Hwang S. Direct Antiviral Mechanisms of Interferon-Gamma. Immune Netw. 2018;18(5):e33.
Crossref - de Groen RA, McPhee F, Friborg J, Janssen HL, Boonstra A. Endogenous IFN-l in viral hepatitis patients. J Interferon Cytokine Res. 2014;34(7):552-556.
Crossref - Ribeiro CRA, de Almeida NAA, Martinelli KG, et al. Cytokine profile during occult hepatitis B virus infection in chronic hepatitis C patients. Virol J. 2021;18(1):15.
Crossref - Li X, Zhou L, Gu L, et al. Veritable antiviral capacity of natural killer cells in chronic HBV infection: an argument for an earlier anti-virus treatment. J Transl Med. 2017;15(1):220.
Crossref - Tufon KA, Anong DN, Meriki HD, et al. Characterization and assessment of HBV chronically infected patients: Identification of those eligible for treatment in the South West region of Cameroon. PLoS One. 2018;13(9):e0203312.
Crossref - Esmaeelzadeh A, Saadatnia H, Memar B, et al. Evaluation of serum HBV viral load, transaminases and histological features in chronic HBeAg-negative hepatitis B patients. Gastroenterol Hepatol Bed Bench. 2017;10(1):39-43.
- Ghosh J. Potential role of Vitamin D as an antiviral agent. Medical Journal of Dr DY Patil Vidyapeeth. 2021;14(1):3-12.
Crossref - Gotlieb N, Tachlytski I, Lapidot Y, Sultan M, Safran M, Ben-Ari Z. Hepatitis B virus downregulates vitamin D receptor levels in hepatoma cell lines, thereby preventing vitamin D-dependent inhibition of viral transcription and production. Mol Med. 2018;24(1):53.
Crossref - Hoan NX, Khuyen N, Binh MT, et al. Association of vitamin D deficiency with hepatitis B virus – related liver diseases. BMC Infect Dis. 2016;16(1):507.
Crossref - Hu YC, Wang WW, Jiang WY, Li CQ, Guo JC, Xun YH. Low vitamin D levels are associated with high viral loads in patients with chronic hepatitis B: a systematic review and meta-analysis. BMC Gastroenterol. 2019;19(1):84.
Crossref
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.