ISSN: 0973-7510
E-ISSN: 2581-690X
The accumulation of nitrogen (-N) is a serious problem in aquaculture as it could lead to mass mortality events of the cultivated species. Chemilitotrophic nitrification is the most recognized in nitrogen removal underestimating the role of heterotrophic nitrifiers. In the present study, the heterotrophic nitrification capacity of 8 bacterial strains isolated from mangrove soil, periphyton and biofilters was evaluated. The strains were grown in heterotrophic nitrification base medium (HNM medium) with three different nitrogen sources, ammonium, nitrite or nitrate at a final concentration of 8 mg L-1, 5 mg L-1 and 80 mg L-1 respectively. The concentration of nitrogen (-N) and OD (600 nm) were determined periodically. Only in 4 strains belonging to the Bacillus genus was the capacity for heterotrophic nitrification and aerobic denitrification observed. Among these strains, SM4 strain presented a good removal profile of ammonium, nitrite and nitrate, achieving an average nitrification efficiency of 98.33 ± 2.89%, 83.67 ± 7.51% and 98.00 ± 0.01% respectively, and a nitrification rate (mg L-1 h-1) of 1.71 ± 0.70, 0.13 ± 0.07 and 0.21 ± 0.06 respectively. The nxrB, nirS, nirK genes in the selected strains were identified by PCR. Additionally, several proteins (enzymes) involved in the nitrogen cycle were identified by proteomic analysis, reporting for the first time the presence of the enzymes ammonia monoxygenase (AMO) and nitrite oxide reductase (NXR) in the genus Bacillus. These findings suggest that the strains studied would have a potential use in the biological removal of nitrogen in aquaculture systems.
Heterotrophic nitrification, Bacillus spp, proteomics, aquaculture.
Aquaculture is the cultivation of aquatic organisms of commercial interest, it implies either the intervention to improve productivity as well as the ownership of cultivated stock. This activity is positioned as the key sector for the development of cities, generating positive impacts at an economic and social level.
The health and productivity of cultivated species is influenced by the physical, chemical and biological conditions of the water. In intensive aquaculture systems it is common to find high levels of organic matter and compounds such as hydrogen sulfide (H2S), ammonia (NH3), ammonium (NH4+), inorganic phosphorus (Pi) and carbon dioxide (CO2), which cause stress in aquatic organisms making them susceptible to diseases1. Nitrogen (-N) is an important nutrient for cultivated species, however, only 27% is assimilated and 73% of food nitrogen ends up in the water as unconsumed food and excretion products2.
The total amount of ammoniacal nitrogen (TAN) includes NH3 (ammonia) and NH4+ (ammonium). Although both NH3 and NH4+ can be toxic to aquatic organisms, NH3 is the most toxic form due to the fact that it is discharged (non-ionized) so it is more soluble in lipids and, consequently, crosses phospholipid membranes more easily than the charged NH4+ form3. The high concentration of organic matter in intensive cultivation leads to environmental hypercapnia (high levels of CO2) which favors the cellular assimilation of nitrites, triggering its toxicity in aquatic animals4. In vertebrates, nitrite combines with hemoglobin to form methemoglobin, which reduces the ability of blood to transport oxygen to body parts5, in the case of invertebrates the effects would be similar.
The transformation of TAN is mainly carried out by chemilitotrophic bacteria through nitrification and denitrification processes6. The biotransformation of ammonia to nitrite is a key step of nitrification and can be carried out by ammonia oxidizing bacteria (AOB) of the genus Nitrosomonas and Nitrosococcus or ammonia oxidizing archaeas (AOA) such as Nitrosopumilus maritimus since these microorganisms possess the amoA gene that encodes an enzyme monoxygenase ammonia that oxidizes ammonia in hydroxylamine7. Hydroxylamine is oxidized to nitrite by the action of a hydroxylamine oxidase encoded by the hao gene that is also considered a nitrification marker gene. The oxidation of ammonia to nitrite is followed by the oxidation of nitrite to nitrate carried out by nitrite oxidizing bacteria (NOB) of the genus Nitrobacter8 thanks to the fact that they possess the nxrB gene that encodes the enzyme nitrite oxido- reductase9.
Denitrification is the most important process for the nitrogen cycle, since it returns nitrogen to the atmosphere through microbial respiration processes by reducing the assimilation of nitrates (NO3–) and nitrites (NO2–) to oxide nitrous (N2O) and dinitrogen (N2), respectively. Gao J et al., 2018 point out that although it is traditionally known that the denitrification process occurs in anoxic environments, it is currently known that denitrification can occur at oxygen conditions. In recent years, various aerobic denitrifying microorganisms have been selected. Some members of the genus Alcaligenes and Thiobacillus possess key genes such as nirS, nirK and nosZ that encode enzymes; nitrite reductase containing cytochrome cd1, nitrite reductase containing copper (Cu) and the enzyme nitrous oxide reductase, respectively10.
Although the chemolithotrophic nitrification is the most recognized in the N-removal processes, the role of heterotrophic bacteria that are detritivorous par excellence should not be underestimated. An example is the enzyme hydroxylamine oxido-reductase (participates in the oxidation of ammonia to nitrite), which was believed to be present only in AOB or AOA, has been isolated from a strain of Bacillus sp strain N31 highly efficient in removal – N 11. Therefore, the study of the interaction between the chemolithotrophic and heterotrophic communities in culture systems is of vital importance.
In this context, a great interest arises for the isolation of native heterotrophic bacteria that participate in the nitrogen cycle to eliminate the excess of these compounds in the ponds, therefore, the present study aimed to evaluate the capacity of heterotrophic nitrification in native strains isolated from mangrove soil, periphyton and biofilters.
Sampling and media
Mangrove sediment samples were collected in Puerto Pizarro (Tumbes) at 20, 40 and 80 m from the coast. One kilogram of sample was collected from a depth of 20 cm at each sampling point, then these samples were homogenized until a pool was obtained, the samples were placed in 50 ml conical tubes, then transported and stored at 4°C for microbiological analyzes.
For the isolation of the strains the mineral medium AMO12 which consisted of the following components (g L-1): NH4Cl2 0.15, CaCO3 1.00, KH2PO4 1.00, MgSO4.7H2O 0.30, NaCl 20, FeSO4 0.03 and agar 15.00 (optional).
To study the nitrogen removal capacity, the heterotrophic nitrification base medium (NMH) described by Huang et al., 2017, modified in this investigation, which consisted of the following components (g L-1): K2HPO4 1.00, KH2PO4 0.30, MgSO4.7H2O 0.05, FeSO4.7H2O 0.01, MnSO4.4H2O 0.01 and NaCl 30.00. Saccharose was used as a carbon source (-C) and added to the HNM medium at a final concentration of 0.1g L-1 in each assay. The nitrogen source (-N) was added to the HNM medium from sterile 0.10 g L-1 and 0.50 g L-1 (w / v) stocks, reaching a final concentration of 8.00 mg L-1 NH4SO4 (HNM- 1), 5.00 mg L-1 NaNO2 (HNM-2) and 80.00 mg L-1 NaNO3 (HNM-3).
Isolation of bacterial strains
Ten grams of biofilm from biofilter or 10g of mangrove soil was placed in 90 ml of AMO mineral medium and incubated at 25°C for 1 week with constant oxygenation. After this time, 100µl aliquots were seeded by spreading on the surface of AMO mineral agar. The isolation of each strain was performed by streak plate method on the surface of TSA (MERCK, New Jersey, USA) supplemented with 2.00% NaCl. Bacterial isolates were identified by analyzing the color, morphology and staining of Gram by optical microscopy (׳ 1000). The strains were stored at -20°C using 15.00% glycerol as a cryoprotectant.
A strain BM6 isolated from periphyton was granted by the Crustaceans Group of Incabiotec SAC.
Molecular identification
DNA extraction was performed using the Stool DNA Extraction kit (GENEAID BIOTECH LTD, Taipei, Taiwan) according to the manufacturer’s instructions. From the bacterial DNA samples, PCR amplification of the 16S rRNA gene was performed using 27F Forward primers (AGAGTTTGATCMTGGCTCAG) and Reverse 1492R (TACGGYTACCTTGTTACGACTT). The amplification was performed under the following conditions; 5 min at 94°C, followed by 35 cycles at 30 s at 94°C, 40 s at 54°C, 1 min at 72°C and 6 min at 72°C.
The sequencing of the amplicons was performed by the Sanger method in the company MACROGEN, USA (https://www.macrogenusa.com/) , the sequences of each isolate were identified by sequence homology using the BLASTN bioinformatic platforms (https://blast.ncbi.nlm.nih.gov/Blast.cgi? PROGRAM= blastn&PAGE_TYPE=BlastSearch&LINK_LOC= blasthome) and EZBioCloud (https://www.ezbiocloud.net/).
Assessment of heterotrophic nitrification capacity
The assessment of the efficiency and rate of heterotrophic nitrification was carried out according to the methodology described by Huang et al., 2017 with some modifications. After 36 h of culture in TSB medium (MERCK, New Jersey, USA), 6 ml of bacterial preculture was harvested by centrifugation at 4000 g (4°C, 15 min) in a refrigerated centrifuge.
The pellet was washed with 6 ml of sterile saline solution 2.00% NaCl (pH 7.40) three times to purify the cell suspension. Then 0.4 ml of bacterial suspension (108 cfu ml-1) was inoculated into 40 ml of heterotrophic nitrification base medium with ammonium (HNM-1), nitrite (HNM-2) and nitrate (HNM-3) respectively. They were then incubated at 28°C in constant motion at 160 rpm using a shaker.
In order to rule out the death of bacteria by refrigerated centrifugation, a positive control consisting of 50µl of bacterial suspension inoculated in 5 ml of TSB 2.00% NaCl was performed. The negative control was formed by sterile HNM-1, HNM-2, HNM-3 and TSB 2% NaCl media. All tests were performed in triplicate.
Aliquots from the tests were taken periodically to determine cell density (OD600), ammonium, nitrite and nitrate respectively. The OD600 was measured by a biophotometer (EPPENDORF BIOPHOTOMETER, Hamburg, Germany) at 600 nm. The concentrations of NH4+ -N, NO2 -N and NO3 -N were measured using the commercial API kit (AQUARIUM PHARMACEUTICALS, USA) following manufacturer’s instructions.
The efficiency and heterotrophic nitrification rate of each strain was determined using the following formulas:
- Nitrification efficiency (%) = (C1 – Cn) x 100% / C1; C1 = Initial concentration, Cn = final concentration.
- Nitrification rate (mg L-1 h-1) = (C1 -Cn) / t; t = incubation period.
Molecular characterization
The functional genes involved in the nitrification and denitrification processes were amplified by conventional PCR from bacterial DNA of the selected strains. The sequences of the specific primers and their respective hybridization temperatures are shown in Table 1. The amplification was performed under the following conditions: 5 min at 94°C, followed by 35 cycles at 30 s at 94°C, 40 s at 54°C, 1 min at 72°C and 6 min at 72°C.
Table (1):
List of primers used for the identification of genes related to nitrification and denitrification.
| Genes | Primersname | Sequence (5´-3´) | Hybridization temperature (°C) | Size of the product (bp) | References |
|---|---|---|---|---|---|
|
amoA |
amoA-1F | GGGGTTTCTACTGGTGGT | 50.5 | 482 | 7 |
| amoA-2R | CCCCTCKGSAAAGCCTTCTTC | ||||
|
nxrB |
nxrB 1F | ACGTGGAGACCAAGCCGGG | 54.6 | 418 | 7 |
| nxrB 1R | CCGTGCTGTTGAYCTCGTTGA | ||||
|
nirS |
Nir S1F | CCTAYTGGCCGCCRCARA | 54.3 | 890 | 35 |
| Nir S6R | CGTTGAACTTRCCGGT | ||||
|
nirK |
nirK1F | GGMATGGTKCCSTGGCA | 50.4 | 439-515 | 35 |
| nirK5R | GCCTCGATCAGRTTRTGG | ||||
| nosZ | nosZ-F | CGYTGTTCMTCGACAGCCAG | 58.3 | 454 | 7 |
| nosZ-1622R | CGSACCTTSTTGCCSTYGCG |
Proteomic analysis
A proteomic analysis was performed in order to identify possible proteins (enzymes) related to the nitrification and denitrification processes in the selected strains.
A volume of 0.4 ml of bacterial suspension (108 cfu ml-1) was inoculated into 40 ml of heterotrophic nitrification base medium with ammonium (HNM-1), nitrite (HNM-2) and nitrate (HNM-3) respectively, including control tubes positive (strains in TSB) and an additional treatment (HNM base medium supplemented with saccharose), it was also considered a general negative control tube (sterile HNM). They were then incubated at 28°C in constant motion at 160 rpm using a shaker.
The samples were centrifuged at 4000 g for 20 min at 4°C, then the supernatant was removed and the pellet was resuspended with 100-500µl of rehydration buffer (8M urea, 20mM DTT, 4% CHAPS) and 1µl PMSF (for every 100µl of rehydration buffer). Then the samples were sonicated for 10 min at 28°C. They were subsequently centrifuged at 10 000 rpm for 10 min at 4°C, 80µl of supernatant (containing the soluble protein fraction) was removed and stored at -20°C.
The preparation of the protein samples for migration was performed by mixing 30µl of supernatant (of the 80µl stored) and 20µl of loading buffer (125 mM Tris pH 6.80, 4% SDS, 20% glycerol, 0.02% Blue Bromophenol, 200 mM DTT, double distilled water). The mixture was incubated in a dry bath at 95°C for 5 min before starting the migration.
The 12% SDS PAGE (Sodium dodecyl sulfate) gel was prepared, and 25µl of each previously prepared protein sample was migrated. Protein migration was performed at 90V and 120A for 3 h.
The polyacrylamide gel containing the migrated proteins was disassembled to fix the proteins using a fixation solution (50.00% methanol; 10.00% acetic acid in double distilled water) for 20 min, then the dyeing of the gel was performed using a staining solution (50.00 % methanol, 10.00% acetic acid, 0.10% Coomasii blue in double distilled water) for 12 h. Subsequently, the gel was discolored using a wash solution (45.00% methanol, 10.00% acetic acid, diluted in double distilled water) for 40 min.
The bands present were cut with a sterile scalpel and deposited in 0.2 ml tubes individually, and were discolored according to the protocol described by Shevchenko et al., 2007, by incubation and homogenization in ammonium bicarbonate and acetonitrile (1: 1) for 30 min. Followed by incubation in 100µl of acetonitrile until complete discoloration of the bands.
Saturation and digestion of the proteins was performed by incubation in 25µl of trypsin buffer (13 ngµl-1) at -4°C for 2 h, followed by incubation at 55°C for 30 min. For the extraction of peptides, extraction buffer (5% formic acid / acetonitrile 1: 2 (V / V)) was used, followed by incubation steps (37°C for 15 min) and sonication. Subsequently, a 10µL aliquot was removed and placed in a vacuum drying oven for 3 h.
The samples were resuspended in 0.1% TFA, mixed with 10 mg ml-1 CHCA matrix (“-cyano-4-hydroxycinmic acid). The matrix was previously resuspended in 0.1% TFA in HPLC grade water and 0.1% TFA in acetonitrile. Aliquots of the resuspended samples were splashed on an opti-TOF 1-1 V / V plate.
The plate containing the peptide samples was analyzed in a MALDI-TOF / TOF mass spectrophotometer (5800 AB SCIEX SYSTEM, Framingham, USA), which was calibrated using a Mass Standards AB SCIEX TOF / TOFTM Instruments kit (AB SCIEX SYSTEM, Framingham MA, USA). The analysis was carried out with the equipment in positive reflective ion mode, with a 349 nm Nd: YAG laser at an intensity of 3800 and a speed of 600µm / second, the team made 750 shots per spectrum which were analyzed within a mass range between 800 to 4000 m / z, and processed using ProteinPilot ™ software. Sequences not identified by this software were analyzed using Protein BLAST (https://blast.ncbi.nlm.nih.gov/Blast.cgi?PAGE=Proteins&).
Statistical analysis
All statistical analyzes were performed using a one-way ANOVA with Tukey’s HSD test (P <0.05) in the SPSS statistics 21.0 program, the graphics were generated in the Graph Pad program.
Isolation, selection and identification of bacterial strains
In total, 9 bacterial strains were isolated using the selective AMO culture medium for nitrifying bacteria. In addition, a strain BM6 isolated from periphyton was granted by the Crustaceans Group of Incabiotec SAC (Table 2).
Table (2):
Molecular identification of native bacterial strains isolated from biofilter, periphyton and mangrove soil by sequence homology.
Strain Code |
Bacteria |
Identity level (%) |
Genbank access number |
Morphology |
Isolation site |
|---|---|---|---|---|---|
SM4 |
Bacillus megaterium |
100 |
JJMH01000000 |
Large gram-positive bacilli |
Mangrove soil |
SM5 |
Bacillus haynesii |
100 |
MRBL01000000 |
gram-positive bacilli |
Mangrove soil |
BM6 |
Bacillus wiedmannii |
100 |
LOBC01000000 |
Gram-positive streptobacilli |
Periphyton |
EBA-P |
Bacillus tequilensis |
100 |
AYTO01000000 |
Gram-positive streptobacilli |
Biofilter |
SM1 |
Bacillus altitudinis |
100 |
MH017401 |
Large gram-positive bacilli |
Mangrove soil |
SB1 |
Bacillus sp |
99 |
MF321840 |
Gram positive bacilli |
Biofilter |
ESM-P1 |
Bacillus aryabhattai |
100 |
MH045946 |
Gram positive bacilli |
Mangrove soil |
ESM-P2 |
Bacillus infantis |
100 |
MF993020 |
Gram positive bacilli |
Mangrove soil |
EBB-P |
Vibrio alginolyticus |
98 |
MF680287 |
Short gram negative bacilli |
Biofilter |
SM3 |
Vibrio parahaemolyticus |
99 |
KY229843 |
Gram negative bacilli |
Mangrove soil |
The isolated strains correspond to those that managed to overcome the culture conditions in the AMO medium, that is; 30 ppb salinity, inorganic carbon source and a high concentration of ammonium (NH4Cl 0.15 g L-1). Due to these conditions, the AMO medium has been reported for the isolation of nitrifying bacteria such as Nitrosomonas sp and Nitrobacter sp.12.
However, in the present study we obtained bacteria of the Bacillus and Vibrio genera, which allows us to hypothesize that these strains would have the ability to use ammonia as a source of nitrogen, surviving the culture conditions in AMO medium. In addition, many heterotrophic microorganisms contribute to nitrification processes, including ammonium oxidation and nitrite oxidation 14.
Isolates of the Vibrio genus were not taken into account for the tests because they are considered deadly pathogens in the aquaculture. So heterotrophic nitrification capacity was evaluated in eight strains of the genus Bacillus.
Assessment of heterotrophic nitrification capacity in strains of the genus Bacillus
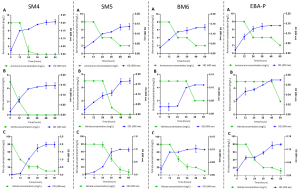
It was observed that only 4 strains were able to perform heterotrophic nitrification. Nitrification characteristics demonstrate that there is a relationship between the increase in OD600 biomass and the decrease in the source of supplemented nitrogen.
The strains obtained their maximum growth between 36 and 48 h reaching their stationary phase after 48h. The OD 600 of SM4 strain was increased from 0.03 to 0.16, 0.13 and 1.04 to 24 hours of culture in HNM-1, HNM-2 and HNM-3 medium respectively, reaching a 95% efficiency of ammonium removal with a rate of nitrification of 0.32 mg L-1 h-1.
At 48 hours it reached 100% removal efficiency of ammonium. This strain also presented a good nitrite removal profile reaching a removal efficiency of 98% with a nitrification rate of 0.21 mg L-1 h-1. The OD 600 of SM5 strain was increased from 0.03 to 0.10, 0.09 and 0.31 to 24 hours of culture in medium HNM-1, HNM-2 and HNM-3 respectively. This strain was able to remove the nitrite with a nitrification efficiency of 80% and a nitrification rate of 0.08 mg L-1 h-1 (at 48 h).
The OD 600 of BM6 strain was increased from 0.03 to 0.11 and 0.09 to 24 h of culture in HNM-1 and HNM-3 medium respectively, no growth or nitrite removal was observed in HNM-2. There was no significant increase in the OD600 of EBA-P strain, however at 24 h it was able to remove the nitrite by 50% (Fig. 1).
Fig. 1. Heterotrophic nitrification characteristics of the selected Bacillus strains with A) ammonium (NH4+ -N), B) nitrite (NO2- -N) y C) nitrate (NO3- -N) as the only source of nitrogen (-N) in relation to OD 600 nm, in aerobic conditions.
The SM4 strain had a good removal profile of ammonium, nitrite and nitrate, achieving a nitrification efficiency of 95.00%, 98.00% and 75.00% respectively at 24 h. These values were high compared to those found by Huang et al., 2017, who report that Bacillus litoralis N31 has an nitrification efficiency of ammonium, nitrite and nitrate of 86.30%, 89.30% and 89.40% respectively after 48 h of cultivation.
Song, An, Fu, & Yang, 2011, report that the strain Bacillus sp. YX-6 removed 10 mg L-1 of nitrite at zero in 14 h. In this investigation, the SM5 strain removed the nitrite with an efficiency of 80.00% at 48 h. On the other hand, BM6 and EBA-P strains removed ammonia with an efficiency of 75.00% (at 60 h) and nitrate with an efficiency of 48.00% and 50.00% respectively at 24 h. The difference in efficiency vs. time could be attributed to the various degrees of oxidation of these compounds by some strains of Bacillus spp, which would be influenced by the initial concentration of ammonium, nitrite or nitrate, for example, the strain Bacillus sp. P7 that achieved a removal efficiency of ammonium of 92.00-95.00% only when the initial levels were 1-2 mg L-1 16. In our case the initial concentrations were 8.00 mg L-1, 5.00 mg L-1 and 80.00 mg L-1 for ammonium, nitrite and nitrate respectively, taking as criteria the highest levels found in L. vannamei culture systems. The removal of ammonium, nitrite or nitrate was not observed in the SM1, SB1, ESM-P1 y ESM-P2 strains (zero values were obtained), so these strains would not have the nitrification capacity.
According to the results, the strains selected were; SM4, SM5, BM6 and EBA-P. Table 3 shows the mean of the efficiency and rate nitrification observed at 24, 48 and 60 hours, it is observed that the nitrification efficiency of ammonium and nitrite in SM4 was higher than the rest of the strains (p <0.05). Regarding nitrate removal, no significant difference was observed between the strains.
Table (3):
Heterotrophic nitrification capacity of selected strains with ammonium (NH4+ -N), nitrite (NO2- -N) and nitrate (NO3- -N) as the sole source of nitrogen under aerobic conditions.
| Source of nitrogen | Strain Code | Nitrification efficiency (%) | Nitrification rate (mg L-1h-1) |
|---|---|---|---|
| Ammonium (NH4+ -N) | SM4 | 98.33 ± 2.89a | 0.21 ± 0.06a |
| SM5 | 66.67 ± 14.43b | 0.13 ± 0.02a | |
| BM6 | 58.33 ± 14.43b | 0.12 ± 0.03a | |
| EBA-P | 58.33 ± 14.43b | 0.12 ± 0.03a | |
| Nitrite (NO2– -N) | SM4 | 98.00 ± 0.01a | 0.13 ± 0.07a |
| SM5 | 58.33 ± 51.07a,b | 0.05 ± 0.05b | |
| BM6 | 20.00 ± 34.64b | 0.02 ± 0.03b | |
| EBA-P | 20.00 ± 34.64b | 0.02 ± 0.03b | |
| Nitrate (NO3– -N) | SM4 | 83.67 ± 7.51a | 1.71 ± 0.70a |
| SM5 | 59.00 ± 49.57a | 0.89 ± 0.72a | |
| BM6 | 70.33 ± 20.40a | 1.34 ± 0.23a | |
| EBA-P | 71.00 ± 19.31a | 1.36 ± 0.27a |
Values are means ± SD for three replicates. Different letters indicate statistically significant differences (p <0.05).
Identification of genes involved in nitrification and denitrification processes
The amoA, nxrB, nirS, nirK and nosZ genes in the selected strains were evaluated by conventional PCR (Table 4).
Table (4):
Identification of specific genes related to nitrification and denitrification processes in the strains evaluated.
| Sample | Nitrification genes | Denitrification genes | |||
|---|---|---|---|---|---|
| amoA | nxrB | nirS | nirK | nosZ | |
| SM4 | – | – | – | + | – |
| SM5 | – | + | + | – | – |
| BM6 | – | – | – | – | – |
| EBA-P | – | + | – | + | – |
| Positive Control | + | + | + | + | + |
| Negative Control | – | – | – | – | – |
The amoA gene encodes the subunit A of the enzyme ammonia monoxygenase (catalyzes the oxidation of ammonia in hydroxylamine) that together with amoC and amoB are part of the AMO operon present in all the chemilitotrophic AOBs studied (Nitrosomonas europaea, Nitrosaspira multiformis, Nitrosococcus oceanus and Nitosospira sp). In the genus Bacillus, the amoA gene has been poorly studied, finding only a report of the presence of the amoA gene in the strain Bacillus sp LY where they also determined that the amoB gene was absent unlike what was found in AOB where the amoA and amoB gene are co-transcribed which suggests that amoB would not be part of the same transcriptional unit and, therefore, is not a member of the master operon in Bacillus sp LY 17. In the present study, the amoA gene was absent in the four strains that have the ability to remove ammonia, which raises doubts, since it could be due to the primers used in both studies, which differ in the sequences.
The nxrB gene encodes the subunit B of the nitrite oxide reductase enzyme that has a key role in the nitrification process, is used as a functional and phylogenetic marker of Nitrospira species of the NOB group18. In nitrifying heterotrophic bacteria, the nxrB gene has been poorly studied, finding only a report of the presence of the nxrA and nxrB gene in the Pseudomonas denitrificans strain which explains its ability to transform nitrite to nitrate19. In this study, the presence of the nxrB gene in SM5 and EBA-P strains is reported for the first time. It should be noted that the primers used in this research were pre-designed for the nxrB gene of Nitrobacter sp 7, which suggests that the gene found in the Bacillus strains evaluated could be closely related to the nxrB gene of Nitrobacter.
Recently, a study on the genome of Bacillus subtilis DM2 revealed the presence of several horizontal transfer genes from phagos, which play critical roles in the acquisition of resistance genes and adaptation to hostile environments20 as the stress conditions caused by high concentrations of nitrite which is toxic to many microorganisms21. The findings in the present study, together with the coexistence of the Nitrobacter genus and heterotrophic bacteria in natural environments14, allow us to hypothesize the horizontal transfer of Nitrobacter genes to Bacillus either directly or phage-mediated.
The presence of nirS and nirK genes that encode two functionally equivalent but structurally distinct forms, enzymes, was also evaluated; nitrite reductase containing a cytochrome cd1 and nitrite reductase containing Cu, which have a primary role in the denitrification process22. Both nirS and nirK are present in the periplasm of Gram-negative bacteria or between the cell membrane and the cell wall of some Archaeas. At the phylogenetic level nirK and nirS are prevalent in a- and b-Proteo-bacteria, respectively23, where some of their members are involved in aerobic heterotrophic denitrification.
The nirK gene was found in Alcaligenes faecalis JCM20522 and Hyphomicrobium sp NL23 while the nirS gene was amplified in Bacillus sp YX-6 and Pseudomonas stutzeri X3124. In the present study the nirK gene was identified in SM4 and EBA-P while the nirS gene was absent in both strains, however, it was present in SM5. One possible explanation would be that the nirS and nirK genes do not coexist in the same bacteria25. Likewise, the results coincide with those found by Gao et al., 2018, who first reported the presence of the nirK gene in Bacillus firmus.
Nitrous oxide reduction is the last step in the denitrification pathway and is catalyzed by the enzyme nitrous oxide reductase encoded by the nosZ gene27. This gene has been identified in several strains of Bacillus spp.28 using degenerate primers designed based on heterotrophic bacteria, which differ from those used in the present study. This could be an explanation of the absence of the nosZ gene in the strains evaluated.
Some aerobic denitrifiers are also capable of performing heterotrophic nitrification 24 which would explain the presence of nxrB (nitrification) and nirS or nirK (denitrification) genes in the SM5 and EBA-P strains.
Identification of proteins involved in nitrification and denitrification processes
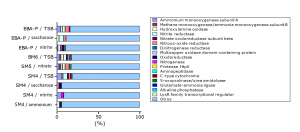
The presence of genes in microorganisms is not indicative enough to affirm their metabolic activity, therefore, in the present investigation by MALDI TOF TOF mass spectometry the presence of enzymes (proteins) related to nitrification and denitrification processes in the selected strains grown in HNM-1, HNM-2, HNM-3 medium, in HNM medium with saccharose and in TSB medium.
The number of different peptide sequences identified in each test was; 69 in SM4 / ammonium, 35 in SM4 / nitrite, 35 in SM4 / saccharose, 158 in SM4 / TSB, 146 in SM5 / nitrate, 181 in BM6 / TSB, 66 in EBA-P / nitrite, 36 in EBA-P / saccharose and 181 in EBA-P / TSB. From these amounts, the percentage of proteins (not absolute) involved in the nitrification and denitrification processes for each test was estimated, resulting; 5.80% in SM4 / ammonium, 8.57% in SM4 / nitrite, 5.71% in SM4 / saccharose, 18.99% in SM4 / TSB, 17.81% in SM5 / nitrate, 17.68% in BM6 / TSB, 10.61% in EBA-P / nitrite, 11.1% in EBA-P / saccharose, 16.02% in EBA-P / TSB. Proteins belonging to other metabolic pathways / cell structure were grouped in the category of others (Fig. 2).
Fig. 2. Diversity of proteins (not absolute) expressed during the evaluation of heterotrophic nitrification capacity of the selected Bacillus strains.
Knowledge about the activity and structure of the enzyme ammonium monoxygenase (AMO) has been obtained mainly from studies in Nitrosomonas europaea 29, however, it is known that the purified AMO enzyme of the heterotrophic bacterium Paracoccus denitrificans is a quinol oxidase that has similar properties to AMO enzyme from Nitrosomonas europaea and methane monoxygenase (MMO) enzyme from Methylococcus capsulatus. The amoA genes of the nitrifying heterotrophic bacteria appear to share similarities with the AOB amoA gene sequences14. In the present study the AMO enzyme was first identified in SM4 and EBA-P, while in SM5 and BM6 both AMO and MMO were identified.
Both enzymes, AMO and MMO, are integral membrane proteins30 which would explain their presence frequently when the strains were cultured in TSB medium since in this medium cell growth (pellet bacterial) was higher compared to the other tests. These enzymes would be responsible for the capacity exhibited by the strains selected in the removal of ammonium.
The hydroxylamine oxidase / hydroxylamine oxidoreductase (HAO) enzyme is a periplasmic enzyme that catalyzes the oxidation of hydroxylamine to nitrite29 where several type C cytochromes are involved in electron transport. HAO is a key enzyme within this chain, since the electrons generated by its reaction are used by AMO who in turn generates hydroxylamine during the electron transport chain for power generation31. In autotrophic nitrifiers it is a trimeric enzyme whose subunits contain 8 heme groups. The HAO of the nitrifying heterotrophic bacteria, such as Pseudomonas denitrificans and Pseudomonas strain PB16, are soluble and do not contain heme groups14.
Yang, 2017, reports for the first time the purification of HAO from Bacillus sp K5. In the present study, both HAO and cytochrome C protein were present in SM4, SM5, BM6 and EBA-P which reinforces the evidence of ammonium removal in these strains.
The nitrite oxide reductase enzyme (NOR or NXR) contains multiple subunits, iron and sulfur centers and a molybdenum cofactor. It is attached to the internal cytoplasmic surface of the NOB bacterial membrane and catalyzes the oxidation of nitrite to nitrate using cytochrome c as an electron acceptor32. No reports of NXR have been found in heterophic bacteria. In the present study, the alpha subunit of the nitrite oxidoreductase enzyme in EBA-P has been identified for the first time.
The nitrite reductase enzyme is a periplasmic protein and is key in denitrification since it produces the first gaseous product, nitric oxide. There are two types of dissimilatory nitrite reducing enzymes that catalyze precisely the same reaction but differ in their cofactors. One type, encoded by nirS genes, contains a cytochrome cd1, while the second type, encoded by nirK genes, contains copper33. Nitrite reductase encoded by nirK was present in all strains while nitrite reductase encoded by nirS was only present in EBA-P.
Nitrous oxide reductase (N2OR) is a periplasmic enzyme encoded by the nosZ gene, which belongs to a group of nosRZDFYL genes, which encode several auxiliary proteins required for expression, maturation and maintenance. Typically, the N2OR enzyme has long been considered the only enzyme involved in the mitigation of nitrous oxide (N2O), however, a closely related enzymatic variant called atypical nosZ has been identified in various microbial taxa34. In our study, although it was not possible to identify the nosZ gene in the evaluated strains, the N2OR protein was identified in all selected Bacillus strains. This suggests that the selected strains would play an important role in the loss of gaseous nitrogen and the regulation of nitrous oxide emissions in various environments28. Table 5 shows a summary of the proteins involved in the nitrification and denitrification processes identified in the selected Bacillus strains.
Table (5):
Summary of the identified proteins related to nitrification and denitrification processes in selected Bacillus strains during culture in HNM-1, HNM-2, HNM-3 medium and in TSB.
Test |
Prec MW |
Prec m/z |
Sequence |
Predicted protein |
Identified by |
Identity (%) |
Accession |
|---|---|---|---|---|---|---|---|
SM4 / HNM-1 |
1791.74 |
1792.75 |
DRDYIAAIDWRAAEK |
TPA: Sec-dependent nitrous-oxide reductase [Acidobacteria bacterium]* |
Protein BLAST |
100.00 |
HBL27222.1 |
SM4 / HNM-1 |
1702.86 |
1703.86 |
PPMVPPAIERTRPAR |
Nitrite reductase, copper-containing [Candidatus Rokubacteria bacterium GWA2_73_35] |
Protein BLAST |
100.00 |
OGK85672.1 |
SM4 / HNM-2 |
2002.89 |
2003.89 |
YPPPTLPPHMTPIEPQVG |
Ammonia monooxygenase [Uncultured archaeon] |
Protein BLAST |
100.00 |
ADW19711.1 |
SM4 / HNM-2 |
2002.89 |
2003.89 |
ENRAQEIYIVMSGEMMA |
Nitrogenase [uncultured organism]* |
Protein BLAST |
100.00 |
ADV92516.1 |
SM4 / TSB |
1805.80 |
1806.81 |
VATSMLLGLSTGAQAAGGAN |
TPA: Nitrite reductase, copper-containing [Oxalobacteraceae bacterium] |
Protein BLAST |
100.00 |
HCE07025.1 |
SM4 / TSB |
2005.92 |
2006.93 |
EADGQWLLSLNEFSKGR |
Nitrous oxide reductase [uncultured forest soil bacterium]* |
Protein BLAST |
100.00 |
|
SM4 / TSB |
2005.92 |
2006.93 |
WWSHYPINFVLPSTMI |
Ammonia monooxygenase subunit A [uncultured Nitrosomonadales bacterium]* |
Protein BLAST |
100.00 |
|
SM4 / TSB |
1702.84 |
1703.85 |
SPETYLIKNKDGLPL |
Hydroxylamine oxidase [candidate division Zixibacteria bacterium RBG_16_48_11] |
Protein BLAST |
100.00 |
OGC87529.1 |
SM4 / TSB |
1523.69 |
1524.70 |
RPEDGIVLIDQNR |
Nitrate oxidoreductase subunit beta [Candidatus Nitrospira nitrificans]* |
Protein BLAST |
100.00 |
WP_090900834.1 |
SM5 / HNM-3 |
1399.67 |
1400.67 |
LLTQQQALSLER |
Methane monooxygenase/ammonia monooxygenase subunit A [Candidatus Methylomirabilis limnetica] |
Protein BLAST |
100.00 |
WP_107560874.1 |
SM5 / HNM-3 |
1399.67 |
1400.67 |
LVEQGSLRTFGGH |
Ammonia monooxygenase subunit A [uncultured ammonia-oxidizing beta proteobacterium]* |
Protein BLAST |
100.00 |
ABN12990.1 |
SM5 / HNM-3 |
2225.96 |
2226.96 |
HTEMNFTNTPWKIGEDFK |
Hydroxylamine oxidase [Thermodesulfobacterium geofontis] |
Protein BLAST |
100.00 |
PMP67185.1 |
SM5 / HNM-3 |
1462.72 |
1463.72 |
DNPLLAKVGDKVR |
Nitrite reductase [Bacillus oryziterrae] |
Protein BLAST |
100.00 |
WP_017754331.1 |
SM5 / HNM-3 |
2225.96 |
2226.96 |
FNVGETVLMIHNQANRDSR |
Nitrite reductase, copper-containing [Sulfitobacter sp. SK012] |
Protein BLAST |
100.00 |
WP_114884555.1 |
SM5 / HNM-3 |
1462.72 |
1463.72 |
SQDTLAALAAMDQK |
Dinitrogenase reductase [uncultured forest soil bacterium] |
Protein BLAST |
100.00 |
AAX10405.1 |
SM5 / HNM-3 |
1399.67 |
1400.67 |
KTEGGTLNLGTKR |
C-type cytochrome [Pseudomonas stutzeri] |
Protein BLAST |
100.00 |
WP_011914555.1 |
SM5 / HNM-3 |
1462.72 |
1463.72 |
DNPLLAKVGDKVR |
Multicopper oxidase domain-containing protein [Bacillus oryziterrae] |
Protein BLAST |
100.00 |
WP_017754331.1 |
SM5 / HNM-3 |
2665.31 |
2666.31 |
GGDYMNGDLHHPHMSFTDGAYDGR |
TAT-dependent Nitrous-oxide reductase [Kiloniella laminariae]* |
Protein BLAST |
100.00 |
WP_020590667.1 |
BM6 / TSB |
1702.77 |
1703.78 |
ISMKNDATAFGEEGFA |
Ammonia monooxygenase subunit A [uncultured bacterium] |
Protein BLAST |
100.00 |
AFF59116.1 |
BM6 / TSB |
1858.83 |
1859.84 |
EIKMEAKDALWEIQR |
Methane monooxygenase/ammonia monooxygenase subunit B [Nitrosomonas sp. Nm132]* |
Protein BLAST |
100.00 |
WP_090544350.1 |
BM6 / TSB |
1858.83 |
1859.84 |
ELKMEAKDALWEIQR |
Ammonia monooxygenase subunit B [uncultured bacterium]* |
Protein BLAST |
100.00 |
ASM48262.1 |
BM6 / TSB |
2001.88 |
2002.89 |
GLVYAHPHPQSPDTTLIR |
TPA: Hydroxylamine oxidase [Desulfobacteraceae bacterium] |
Protein BLAST |
100.00 |
HBF43703.1 |
BM6 / TSB |
1858.83 |
1859.84 |
IGAAPFLHDGGWDATKR |
Dissimilatory nitrite reductase [uncultured bacterium]* |
Protein BLAST |
100.00 |
CCA94885.1 |
BM6 / TSB |
1858.83 |
1859.84 |
ISAAPFLHDGGWDASKR |
Nitrite reductase [uncultured bacterium]* |
Protein BLAST |
100.00 |
ADR10915.1 |
BM6 / TSB |
1702.77 |
1703.78 |
AEADGINLTSDSKIIR |
Nitrous-oxide reductase [Pseudomonas bauzanensis] |
Protein BLAST |
100.00 |
WP_036989209.1 |
BM6 / TSB |
2001.88 |
2002.89 |
LILHAKAQTTVLHMAAER |
Dinitrogenase reductase [uncultured bacterium]* |
Protein BLAST |
100.00 |
AAV30937.2 |
BM6 / TSB |
1523.72 |
1524.73 |
RPEDGIVLIDQNR |
Nitrate oxidoreductase subunit beta [Candidatus Nitrospira nitrificans]* |
Protein BLAST |
100.00 |
WP_090900834.1 |
EBA-P / HNM-2 |
1688.69 |
1689.69 |
ASDGMRHSFMFTFR |
Cytochrome c nitrite reductase small subunit [Ignavibacteriales bacterium] |
Protein BLAST |
100.00 |
RPI61568.1 |
EBA-P / HNM-2 |
1704.73 |
1705.74 |
GMTYDTQEYHPEPR |
Nitrite reductase [Bradyrhizobiaceae bacterium] |
Protein BLAST |
100.00 |
WP_116457134.1 |
EBA-P / HNM-2 |
1704.73 |
1705.74 |
VPGIVYLMPCGKSPR |
Nitrous oxide reductase [uncultured bacterium]* |
Protein BLAST |
100.00 |
ASF90315.1 |
EBA-P / TSB |
2225.95 |
2226.96 |
KNKHSLILMGGVLCGMSMSM |
Ammonia monoxygenase [uncultured archaeon]* |
Protein BLAST |
100.00 |
ACZ06628.1 |
EBA-P / TSB |
2225.95 |
2226.96 |
HTEMNFTNTPWKIGEDFK |
Hydroxylamine oxidase [Thermodesulfobacterium geofontis] |
Protein BLAST |
100.00 |
PMP67185.1 |
EBA-P / TSB |
1805.76 |
1806.77 |
NCHAAVAMDFTKQTR |
Cytochrome C3 family protein [Paracoccus denitrificans] |
Protein BLAST |
100.00 |
WP_011750943.1 |
EBA-P / TSB |
1523.70 |
1524.71 |
RPEDGIVLIDQNR |
Nitrate oxidoreductase subunit beta [Candidatus Nitrospira nitrificans]* |
Protein BLAST |
100.00 |
WP_090900834.1 |
EBA-P / TSB |
1523.70 |
1524.71 |
VGETVLMVHAQANR |
MULTISPECIES: Nitrite reductase, copper-containing [unclassified Rhizobiales (miscellaneous)] |
Protein BLAST |
100.00 |
WP_137859800.1 |
EBA-P / TSB |
2068.92 |
2069.92 |
VVGAPSMRELIRIPVFNR |
TAT-dependent Nitrous-oxide reductase [Paracoccus chinensis]* |
Protein BLAST |
100.00 |
WP_090753257.1 |
EBA-P / TSB |
1523.70 |
1524.71 |
ARYYNGTPRGVVR |
Nitrite oxidoreductase/nitrate reductase alpha subunit [uncultured Acetothermia bacterium] |
Protein BLAST |
100.00 |
BAL57377.1 |
* Sequences with conserved domains found.
Extreme conditions in shrimp farming could inhibit the activity of nitrifying chemilitotrophic bacteria. in these cases, heterotrophic nitrifying microorganisms contribute greatly to nitrification processes in aquaculture systems. In the present work it was possible to isolate, select and characterize at the molecular level 4 strains of the genus Bacillus capable of performing heterotrophic nitrification and aerobic denitrification. The nxrB, nirS and nirK genes considered markers of the nitrification and denitrification processes were identified by PCR. In addition, by MALDI TOF TOF mass spectometry, several enzymes involved in the nitrogen cycle were identified, the presence of the enzymes ammonia monoxygenase (AMO) and nitrite oxide reductase (NXR) in the genus Bacillus being reported for the first time. The strains studied would have a potential use in the biological removal of nitrogen in intensive aquaculture systems.
ACKNOWLEDGMENTS
A special acknowledgments to the Postgraduate Scholarship program provided by FONDECYT in coordination with the National University of Tumbes and the INCABIOTEC SAC research center.
CONFLICT OF INTEREST
The authors declares that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
LFDM, JGQM, JMRC, VACE and EMM involved in the design of the research and the experiment. LFDM, GWAL, JJIA and VISD involved in performing the designed experimental procedure. LFDM and GWAL involved in analysis the data. All authors wrote the manuscript and agreed with the final version of the manuscript.
FUNDING
The research was funded by a master’s degree grant from FONDECYT-CONCYTEC through management agreement N°000190-2015-FONDE-CYT of the National University of Tumbes in collaboration with the Incabiotec SAC research center.
ETHICS STATEMENT
The biological material and the research process did not require specific permits.
AVAILABILITY OF DATA
The datasets are available from the corresponding author on reasonable request.
- Antony SP, Philip R. Bioremediation in Shrimp Culture Systems. NAGA, World Fish Center Q., 2006; 29(3): 62–6.
- Crab R, Avnimelech Y, Defoirdt T, Bossier P, Verstraete W. Nitrogen removal techniques in aquaculture for a sustainable production. Aquaculture, 2007; 270(1–4): 1–14.
Crossref - Korner S, Das SK, Veenstra S, Vermaat JE. The effect of pH variation at the ammonium/ammonia equilibrium in wastewater and its toxicity to Lemna gibba. Aquat. Bot., 2001; 71(1): 71–8.
Crossref - Gam LTH, Jensen FB, Huong DTT, Phuong NT, Bayley M. The effects of elevated environmental CO2 on nitrite uptake in the air-breathing clown knifefish, Chitala ornata. Aquat. Toxicol., 2018; 196(January): 124–31.
Crossref - Yang H, Cheng H. Controlling nitrite level in drinking water by chlorination and chloramination. Sep Purif Technol., 2007; 56(3): 392–6.
Crossref - Ferrera I, Sanchez O. Insights into microbial diversity in wastewater treatment systems: How far have we come? Biotechnol Adv. [Internet]. 2016; 34(5): 790–802.
Crossref - Geets J, De Cooman M, Wittebolle L, Heylen K, Vanparys B, De Vos P, et al. Real-time PCR assay for the simultaneous quantification of nitrifying and denitrifying bacteria in activated sludge. Appl. Microbiol. Biotechnol., 2007; 75(1): 211–21.
Crossref - Klotz MG, Stein LY. Nitrifier genomics and evolution of the nitrogen cycle. FEMS Microbiol Lett., 2008; 278(2): 146–56.
Crossref - Li J, Hao H, Cheng G, Liu C, Ahmed S, Shabbir MAB, et al. Enhancing nitrate removal from freshwater pond by regulating carbon/nitrogen ratio. Front Microbiol., 2017; 8(SEP): 1–9.
Crossref - Simon J. Enzymology and bioenergetics of respiratory nitrite ammonification. 2002; 26.
Crossref - Huang F, Pan L, Lv N, Tang X. Characterization of novel Bacillus strain N31 from mariculture water capable of halophilic heterotrophic nitrification–aerobic denitrification. J. Biosci. Bioeng., 2017; 124(5): 564–71.
Crossref - Rodriguez-Moreno N, Toro-Lozano C, Martinez-Salgado M, Mercado-Reyes M. Estandarizacion de condiciones para la prueba cuantitativa del NMP con bacterias nitrificantes y denitrificantes usando como matriz compost. 2007; 1: 69–81. Retrieved from https://revistas.javeriana.edu.co/index.php/scientarium/article/view/4867.
- Shevchenko A, Tomas H, Havli J, Olsen J V., Mann M. In-gel digestion for mass spectrometric characterization of proteins and proteomes. Nat. Protoc., 2007; 1(6): 2856–60.
- Hayatsu M, Tago K, Saito M. Various players in the nitrogen cycle: Diversity and functions of the microorganisms involved in nitrification and denitrification. Soil Sci. Plant. Nutr., 2008; 54(1): 33–45.
Crossref - Song ZF, An J, Fu GH, Yang XL. Isolation and characterization of an aerobic denitrifying Bacillus sp. YX-6 from shrimp culture ponds. Aquaculture, 2011; 319(1–2): 188–93.
Crossref - Ghosh S, Sasmal D, Abraham TJ. Microcosm evaluation of indigenous microflora of traditional shrimp farming system as bioremediators. Indian J. Fish, 2010; 57(1): 97–101.
- Lin Y, Kong H, Wu D, Li C, Wang R, Tanaka S. Physiological and molecular biological characteristics of heterotrophic ammonia oxidation by Bacillus sp. LY. World J. Microbiol. Biotechnol., 2010; 26(9): 1605–12.
Crossref - Pester M, Maixner F, Berry D, Rattei T, Koch H, L cker S, et al. NxrB encoding the beta subunit of nitrite oxidoreductase as functional and phylogenetic marker for nitrite-oxidizing Nitrospira. Environ. Microbiol., 2014; 16(10): 3055–71. 10.1111/1462-2920.12300.
- Rodrigues Araujo L. Functional genomic characterization of bioremediation genes in the main wastewater treatment bacteria. Univ. Fed. Uberlandia., 2019; 53(9): 1689–99.
- Li S-W, Liu M-Y, Yang R-Q. Comparative Genome Characterization of a Petroleum-Degrading Bacillus subtilis Strain DM2 . Int. J. Genomics, 2019; 2019: 1–16.
Crossref - Chen R, Deng M, He X, Hou J. Enhancing Nitrate Removal from Freshwater Pond by Regulating Carbon/Nitrogen Ratio. Front Microbiol., 2017; 8(SEP): 1–9.
Crossref - Che Y, Liang P, Gong T, Cao X, Zhao Y, Yang C, et al. Elucidation of major contributors involved in nitrogen removal and transcription level of nitrogen-cycling genes in activated sludge from WWTPs. Sci. Rep., [Internet]. 2017; 7(March). Available from:
Crossref - Simon J, Klotz MG. Diversity and evolution of bioenergetic systems involved in microbial nitrogen compound transformations. Biochim. Biophys. Acta-Bioenerg., [Internet]. 2013; 1827(2): 114–35. Available from:
Crossref - Ji B, Yang K, Zhu L, Jiang Y, Wang H, Zhou J, et al. Aerobic denitrification: A review of important advances of the last 30 years. Biotechnol. Bioprocess Eng., 2015; 20(4): 643–51.
Crossref - Ben Taheur F, Fdhila K, Elabed H, Bouguerra A, Kouidhi B, Bakhrouf A, et al. Molecular identification of potential denitrifying bacteria and use of D-optimal mixture experimental design for the optimization of denitrification process. Microb. Pathog., [Internet]. 2016; 93: 158–65.
Crossref - Gao H, Li C, Ramesh B, Hu N. Cloning, purification and characterization of novel Cu-containing nitrite reductase from the Bacillus firmus GY-49. World J. Microbiol. Biotechnol., [Internet]. 2018; 34(1).
Crossref - Kandeler E, Deiglmayr K, Tscherko D, Bru D, Philippot L. Abundance of narG, nirS, nirK, and nosZ genes of denitrifying bacteria during primary successions of a glacier foreland. Appl. Environ. Microbiol., 2006; 72(9): 5957–62.
Crossref - Jones CM, Welsh A, Throback IN, Dorsch P, Bakken LR, Hallin S. Phenotypic and genotypic heterogeneity among closely related soil-borne N2- and N2O-producing Bacillus isolates harboring the nosZ gene. FEMS Microbiol Ecol., 2011; 76(3): 541–52.
Crossref - Bennett K, Sadler NC, Wright AT, Yeager C, Hyman MR. Activity-based protein profiling of ammonia monooxygenase in Nitrosomonas europaea. Appl. Environ. Microbiol., 2016; 82(8): 2270–9.
Crossref - Holmes AJ, Costello A, Lidstrom ME, Murrell JC. Evidence that participate methane monooxygenase and ammonia monooxygenase may be evolutionarily related. FEMS Microbiol Lett., 1995; 132(3): 203–8.
Crossref - Hommes NG, Sayavedra-Soto LA, Arp DJ. Mutagenesis and expression of amo, which codes for ammonia monooxygenase in Nitrosomonas europaea. J. Bacteriol., 1998; 180(13): 3353–9.
- Meincke M, Bock E, Kastrau D, Kroneck PMH. Nitrite oxidoreductase from Nitrobacter hamburgensis: redox centers and their catalytic role. Arch. Microbiol., 1992; 158(2): 127–31.
Crossref - Hooper AB, Vannelli T, Bergmann DJ, Arciero DM. Enzymology of the oxidation of ammonia to nitrate by bacteria. Antonie Van Leeuwenhoek. 1997; 71: 59–67.
Crossref - Torres MJ, Simon J, Rowley G, Bedmar EJ, Richardson DJ, Gates AJ, et al. Nitrous Oxide Metabolism in Nitrate-Reducing Bacteria: Physiology and Regulatory Mechanisms [Internet]. 1st ed., Advances in Microbial Physiology. Elsevier Ltd.; 2016; 68; 353-432 p.
Crossref - Braker G, Fesefeldt A, Witzel K-P. Development of PCR Primer Systems for Amplification of Nitrite Reductase Genes nirK and nirS To Detect Denitrifying Bacteria in Environmental Samples. Appl. Environ. Microbiol. [Internet]. 1998; 64(10): 3769–75.
Crossref - Gao J, Gao D, Liu H, Cai J, Zhang J, Qi Z. Biopotentiality of High Efficient Aerobic Denitrifier Bacillus megaterium S379 for Intensive Aquaculture Water Quality Management. J. Environ. Manage., 2018; 222(November 2017): 104-111.
Crossref
© The Author(s) 2019. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.