ISSN: 0973-7510
E-ISSN: 2581-690X
Fungi can cause airborne diseases that need to consider in health plans due to its risk and threats of many communities worldwide. This study aims to assess indoor air quality of laboratory room environment for health industry sector. Open plate method was used to collect and culture of airborne fungi from 12 rooms of laboratories in faculty and hospital buildings. Identification of filamentous fungi grown on culture plates was based on standard mycological texts and manual. The results showed that the occurrence frequency and relative abundance were used to show the risk of airborne fungi. The colony forming unit of fungi species isolated from air samples of faculty and hospital laboratories were 304 and 83 respectively. Thirteen fungi genera including twenty-one species were identified overall air samples. The predominant fungi were Aspergillus, Penicillium and Alternaria among fungi. Among these species the most prevalence fungi was Penicillium chrysogenum at the highest at species level. Therefore, it is essential to set urgent steps to enhance the indoor air quality in laboratory environment such as providing air purification system in these labs.
Airborne fungi, Aspergillus, Penicillium, indoor environment.
Airborne microorganisms scattered everywhere in both outdoor and indoor environments. Many common infections in both human and animals can spread by airborne transmission (Shams-Ghahfarokhi et al., 2014). Airborne transmission increases the opportunity of human exposure to pathogenic microorganisms during breathing the polluted air or contamination of food and water (Durugbo et al., 2012; Fernstrom & Goldblatt, 2013; Martםnez-Herrera et al., 2016). Airborne fungi is the most important part of human pathogens which cause several human health problems such as allergic, contagious, infectious, and respiratory diseases (Gots et al., 2003).
Airborne microorganisms scattered everywhere in both outdoor and indoor environments. Many common infections in both human and animals can spread by airborne transmission (Shams-Ghahfarokhi et. al., 2014). Airborne transmission increases the opportunity of human exposure to pathogenic microorganisms during breathing the polluted air or contamination of food and water (Durugbo et. al., 2012; Fernstrom & Goldblatt, 2013; Martínez-Herrera et al., 2016). Airborne fungi is the most important part of human pathogens which cause several human health problems such as allergic, contagious, infectious, and respiratory diseases (Gots et. al., 2003).
Fungi spread in building via ventilation, air outdoor intakes, human movement, building materials and others. Fungal growth and sporulation occur with increasing of humidity of air (Shelton et. al., 2002). Many studies have been reported the elevated concentrations of fungi in air samples of some critical indoor environments such as health care facilities. Particularly, high frequency levels of Aspergillus sp., Cladosporium sp. , Penicillium sp. and Alternaria sp. were recorded by Pavan and Manjunath (2014); Tong et al. (2017); Gonçalves et al. (2017). These fungi produce large quantities of spores that easily spread over a wide area. Spores also remain dominant for a long period of time during unfavorable conditions. Although, the abundance of some fungi species showed a link with different seasons of the year (Mebi et. al., 2012). Iraq is one of the west Asia countries that characterize with the sunny weather. Average temperatures in Iraq range from 48 °C (118.4 ° F) in July and August to zero occasionally in January. Winds accompanied by dust sometimes comes at intervals of the year because low rainfall in winter over the past few years. Thus it is expected that increase number of airborne diseases during this conditions. Therefore, this study aims to assess indoor air quality of various laboratories from Faculty and hospital buildings located in Thi-Qar Governorate southern of Iraq.
The open plate method (Kumari et al., 2016) was adopted to collect and analysis air samples from indoor environment of two distinct sites. The selected sites included room laboratories of biology department in Faculty of Science, University of Thi-Qar and pathogenic analysis laboratories in Suq Alshuyukh public hospital. A total of 36 plates of Sabouraud dextrose agar (SDA) containing chloramphenicol (Oxoid, France) were prepared and exposed to air for the experiment. The plates were distributed in 12 groups, three plates for each room. Five rooms of hospital laboratories were tested to assess the indoor environments of this facility and namely: Microbiology unit, Serology unit, Viruses unit, Parasitology unit and Samples Collection unit. At the faculty laboratories, twenty one plates were exposed in seven rooms. The opened plates for air were left at least for 5 hours during working hours, when students, workers and patients were present in these locations. The culture plates were incubated at 30°C for 3 to 5 days. Then, the isolates were identified based on morphological and microscopic characteristics using standard mycological texts and manuals (Kidd et al., 2016).
Statistical analysis
The occurrence frequency and relative abundance were computed depending on colony forming unit (CFU) of fungal species, genera or a fungi group to show the exposure risk of collected fungi as described by (Chan et al., 2008).
The results revealed that all a total of 36 plates were positive to at least one colony of fungi species. The colony forming unit (CFU) of fungi species was 304 colonies grown on 21 plates distributed in biology laboratories of faculty and 83 colonies grown on 15 plates distributed in Hospital laboratories. Over all air samples in both faculty and hospital fungi populations, thirteen fungi genera including twenty-one species were identified (Table 1).
Table (1):
General statistics of airborne fungi populations from faculty and hospital laboratories.
| Parameters | Faculty (21 plates) | Hospital (15 plates) |
|---|---|---|
| Number of observed individuals (Colony Forming Unit CFU) | 304 | 83 |
| Number of observed genera | 8 | 8 |
| Number of observed shared genera | 3 | |
| Total fungi genera | 13 | |
| Number of observed species (Species Richness and diversity) | 11 | 16 |
| Number of observed shared species | 6 | |
| Total fungi species | 21 | |
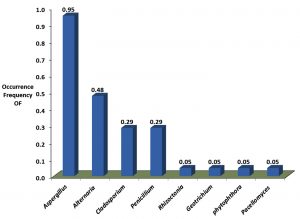
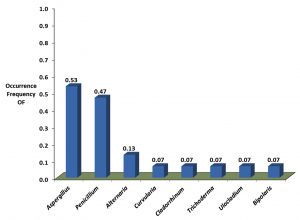
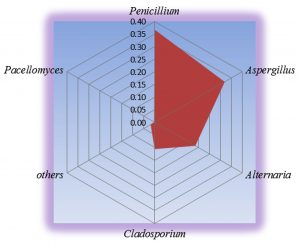
Both populations shared three fungi genera Aspergillus, Penicillium and Alternaria. Aspergillus was isolated with highest occurrence frequency in both populations (Faculty 95% and hospital 53%) (Fig. 1, 2). Penicillium followed by Aspergillus was observed with highest Relative abundance from both populations. Alternaria was isolated with high occurrence frequency of Faculty population; on other hand it was isolated with high relative abundance of Hospital population (Figure 3). Therefore, we found that high diversity within populations as is the case between populations.
The species richness and diversity were significantly lower in faculty fungi population (11 fungi species) than hospital fungi population (16 fungi species) (Table 1). The most abundant species in faculty fungi population were P. chrysogenum (22%), A. alternata (16%), A. niger and Cladosporium cladosporioides (13%), A. flavus (12%) and A. fumigatus (11%). In contrast, the most abundant in hospital fungi population was P. aurantiogriseum (64%) followed by P. chrysogenum (12%) according to relative abundance (Table 4). Other fungi species were isolated with sporadic relative abundance and occurrence frequency in both populations (Table 2, Fig. 1, 2).
Table (2):
Relative abundance depending on colony forming unit CFU for both fungi populations from Faculty and Hospital laboratories.
Fungi species |
Faculty-CFU |
Relative Abundance (%) |
Hospital-CFU |
Relative Abundance (%) |
|---|---|---|---|---|
Aspergillus niger |
38 |
13 |
1 |
0.01 |
Aspergillus fumigatus |
34 |
11 |
3 |
0.04 |
Aspergillus flavus |
36 |
12 |
1 |
0.01 |
Aspergillus parasiticus |
4 |
1 |
0 |
0 |
Aspergillus terreus |
4 |
0.01 |
0 |
0 |
Aspergillus flocculosus |
0 |
0 |
2 |
0.02 |
Aspergillus pseudoelegans |
0 |
0 |
1 |
0.01 |
Cladosporium cladosporioides |
41 |
13 |
0 |
0 |
Penicillium citrinum |
0 |
0 |
2 |
0.02 |
Penicillium glaucum |
10 |
0.03 |
0 |
0 |
Penicillium chrysogenum |
66 |
22 |
10 |
0.12 |
Penicillium aurantiogriseum |
0 |
0 |
53 |
0.64 |
Penicillium lanosumtetra |
0 |
0 |
1 |
0.01 |
Alternaria alternata |
50 |
16 |
1 |
0.01 |
Alternaria solani |
20 |
0.07 |
1 |
0.01 |
Pacellomyces varotii |
1 |
0.003 |
0 |
0 |
Trichoderma |
0 |
0 |
1 |
0.01 |
Curvularia lunata |
0 |
0 |
1 |
0.01 |
Ulocladium |
0 |
0 |
2 |
0.02 |
Bipolaris |
0 |
0 |
2 |
0.02 |
Cladorrhinum samala |
0 |
0 |
1 |
0.01 |
Airborne fungi usually spread their spores in the ambient air. Qualitative knowledge of spread of these fungi in our regions is of great importance to avoid several respiratory diseases such as asthma and rhinitis that can be occurred in human (Menezs et al., 2004). This study revealed that a great abundance of fungal species isolated from air samples in two distinct sites (faculty and hospital laboratories).
The most dominate fungal genera in our findings were Aspergillus and Penicillium due to the ability of their spores to easily spread in various aerobic environments (Asan et al., 2003). In particular, Aspergillus is characterized by production of small conidial spores which are a widespread in air environment of tropical regions (Rosas et al., 1992). However, previous study suggested that fungal spores spread from outdoor to indoor environment through staff, visitors, patients and ventilation systems (Kiasat et al., 2017).
High occurrence frequency of Penicillium and Alternaria were also recoded in hospital fungi population. This indicates that potential infections of respiratory allergy may be occurred due to inhalation spores of Penicillium and Alternaria (Bush & Prochnau, 2004; Menezs et al., 2004).
However, Aspergillus, Penicillium and Alternaria, which were called allergenic fungi, can colonize in indoor environments and thereby increase fungi exposure levels. Human exposure to fungal aeroallergens due to increase asthma severity (Gabriel et al., 2016).
The existence of some fungi species in hospital associated with specific location, such as Penicillium had strong relevance with serology unit, while the existence of Aspergillus and Cladosporium linked with microbiology unit in both hospital and faculty. This is in agreement with previous study (Sepahvand et al., 2013).
The results of our study are comparable with previous studies (Asan et al., 2003; Kumari et al., 2016; Shelton et al., 2002) which isolated similar fungi in indoor environment. In other word, fungi species isolated here endemic to indoor environments.
The density of airborne fungi species in indoor environments of educational and health institutions increase the exposure to opportunistic fungi, which may have a potential negative impact on public health. Indoor environment likely contaminate by opportunistic fungi from outdoor environment.
ACKNOWLEDGMENTS
The authors acknowledge the staff of laboratories of Suq Alshuyukh public hospital to facilitate access into the laboratory.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
MHA and RA carried out the experiment. AKA wrote the manuscript with support from MHA who conceived the original idea. SE supervised the project. All authors read and approved the manuscript for publication.
FUNDING
None.
ETHICS STATEMENT
This article does not contain any studies with human participants or animals performed by any of the authors.
AVAILABILITY OF DATA
All datasets generated or analyzed during this study are included in the manuscript.
- Asan Ahmet, Kirgiz Timur , Sen Burhan, Camur-Elipekbelgin, G.U. and G.H. Isolation, identification and seasonal distribution of airborne and waterborne fungi on terkos lake (Istanbul0Turkey). Journal of Basic Microbiology, 2003; 43(2): 83–95.
Crossref - Bush, R.K., & Prochnau, J.J. Alternaria-induced asthma. Journal of Allergy and Clinical Immunology, 2004; 113(2): 227–234.
Crossref - Chan, W.Y., Mui, K.W., & Wong, L.T. Fungal exposure risk in indoor environment. Environmental Health, 2008; 17–22.
- Durugbo, E.U., Kajero, A.O., Omoregie, E.I., & Oyejide, N.E. A survey of outdoor and indoor airborne fungal spora in the Redemption City, Ogun State, south-western Nigeria. Aerobiologia, 2012; 29: 201–216.
Crossref - Fernstrom, A., & Goldblatt, M. Aerobiology and Its Role in the Transmission of Infectious Diseases [Research article], 2013.
Crossref - Gabriel, M.F., Postigo, I., Tomaz, C.T., & Martinez, J. Alternaria alternata allergens: Markers of exposure, phylogeny and risk of fungi-induced respiratory allergy. Environment International. Pergamon, 2016.
Crossref - Goncalves, C.L., Mota, F.V, Ferreira, G.F., Mendes, J.F., Pereira, E.C., Freitas, C.H., … Nascente, P.S. Airborne fungi in an intensive care unit. Brazilian Journal of Biology, 2017; 78(2): 265.
Crossref - Gots, R.E., Layton, N.J. & Pirages, S.W. Indoor health: Background levels of fungi. American Industrial Hygiene Association Journal, 2003; 64(4): 427–438.
Crossref - Kiasat, N., Fatahinia, M., Mahmoudabadi, A.Z. & Shokri, H. Qualitative and quantitative assessment of airborne fungal spores in the hospitals environment of Ahvaz city (2016). Jundishapur Journal of Microbiology, 2017; 10(10): 1–6.
Crossref - Kidd, S., Halliday, C., Alexiou, H. & Ellis, D. Description of Medical Fungi, 2016; Retrieved from www.mycology.adelaide.edu.au
- Kumari, N., Kumar, A. & Khare, P. Fungal pollution in indoor environments. IOSR Journal of Pharmacy and Biological Sciences, 2016; 11(1): 5–7.
- Martinez-Herrera, E.O., Frias De-Leon, M.G., Duarte-Escalante, E., Calderon-Ezquerro, M. del C., Jiminez-Martinez, M. del C., Acosta-Altamirano, G., … Reyes Montes, M. del R. Fungal diversity and Aspergillus species in hospital environments. Annals of Agricultural and Environmental Medicine, 2016; 23(2), 264–269.
Crossref - Mebi, G., Samuel, W. & Ojogba, M. Relationship Between Fungal Contamination of Indoor Air and Health Problems of Some Residents in Jos. Air Quality – Monitoring and Modeling, 2012; 1–19.
Crossref - Menezs, E.A., Carvalho, P.G., Trindade, E., Sobrinho, G., Cunha, F. & Casrto, F. Airborne fungi causing respiratory allergy in patients from Fortaleza, Ceara, Brazil. Bras. Patol. Med. Lab, 2004; 40(2), 79–84.
Crossref - Pavan, R., & Manjunath, K. Qualitative Analysis of Indoor and Outdoor Airborne Fungi in Cowshed. Journal of Mycology, 2014; 1–8.
Crossref - Rosas, I., C.C. & ESCAMILLA, B., ULLOA, M. Seasonal distribution of Aspergillus in the air of an urban area: Mexico City Grana Seasonal distribution of Aspergillus in the air of an urban area/ : Mexico City. Grana, 1992; 31: 315–319.
Crossref - Sepahvand, A., Shams-Ghahfarokhi, M., Allameh, A. & Razzaghi-Abyaneh, M. Diversity and distribution patterns of airborne microfungi in indoor and outdoor hospital environments in Khorramabad, Southwest Iran. Jundishapur Journal of Microbiology, 2013; 6(2): 186–192.
Crossref - Shams-Ghahfarokhi, M., Aghaei-Gharehbolagh, S., Aslani, N. & Razzaghi-Abyaneh, M. Investigation on distribution of airborne fungi in Outdoor environment in Tehran, Iran. Journal of Environmental Health Science and Engineering, 2014; 12(1): 1–7.
Crossref - Shelton, B.G., Kirkland, K.H., Flanders, W.D. & Morris, G.K. Profiles of Airborne Fungi in Buildings and Outdoor Environments in the United States Profiles of Airborne Fungi in Buildings and Outdoor Environments in the United States. Applied and Environmental Microbiology, 2002; 68(4): 1743–1753.
Crossref - Tong, X., Xu, H., Zou, L., Cai, M., Xu, X., Zhao, Z., … Li, Y. High diversity of airborne fungi in the hospital environment as revealed by meta-sequencing-based microbiome analysis. Scientific Reports, 2017; 7: 39606.
Crossref
© The Author(s) 2019. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.