ISSN: 0973-7510
E-ISSN: 2581-690X
In this investigation, growth promoting role of indole-3-acetic acid (IAA) was assessed on yeasts representing three genera and four species viz Wickerhamomyces anomalus, Saccharomyces cerevisiae, Candida tropicalis and Candida glabrata isolated from traditional fermentative starter materials of Ahom, Rabha, Bodo, Karbi, Kachari and Mishing communities of Assam, India. Isolates were first assessed for their ability to synthesize IAA in presence and absence of external tryptophan and was examined for stimulatory effect of growth on the tested isolates. Tryptophan dependent IAA synthesis was observed in 92% of isolates while 72% of isolates could synthesize IAA in absence of exogenous tryptophan. Candida glabrata KC3X could synthesize maximum IAA while Saccharomyces cerevisiae KR5.6 did not synthesize IAA in presence of external tryptophan. 3 out of 14 Wickerhamomyces isolates and the sole Saccharomyces isolate KR4.10 were found to synthesize significant amount of IAA in absence of tryptophan. Treatment of exogenous IAA on the growth of tested yeasts revealed that Saccharomyces isolates were more pronounced than others. Wickerhamomyces anomalus, Candida tropicalis and Candida glabrata did not show any significant response of growth in presence of exogenous IAA. This study concludes that plant condiments present in starter materials may aid in accelerated growth of Saccharomyces yeasts compared to non-Saccharomyces ones. Yeasts capable of synthesizing IAA but unable to show appreciable growth in presence of IAA may presumably facilitate sustenance of Saccharomyces spp. in fermentative consortia.
Ethnic community, traditional fermentation, yeasts, indole-3-acetic acid, growth.
Yeasts are a major group of microorganisms along with mucoraceous fungi and bacteria, widely used in formulating inoculum or starter materials for preparation of fermented beverages worldwide1. The starter materials are composed of dried substances along with small amounts of plant parts as additives and shaped in the form of flattened cakes, small spherical balls or white powder1. Preparation of such household fermented liquor is one of unique traditional practices among the ethnic communities of northeast India. Assam is home to a number of indigenous ethnic communities, is unique in terms of rich biodiversity and ethnicity of its diversified people2. These ethnic communities commonly use rice or millet as sources of starter cultures for perpetuation of yeasts along with addition of various plant condiments. Yeasts inherent in those starter materials preserved as concoction and used during traditional fermentation is somewhat different and unique for each and every community2. The microbial inoculum responsible for fermentation has been perpetuated generation after generation through the starter materials whereby the starchy materials along with the plant condiments are believed to play a major role in resuscitation of microbes as well as in maintaining the quality of final fermented product.
Among the various plant metabolites, Indole-3-acetic acid (IAA) is known to promote rapid and long-term responses in plants3. Yeasts have been previously reported to produce IAA4,5,6 that promote plant growth4 which suggest the role of IAA producing micro-organisms as a potential source of biofertilizers7,8. Published reports suggest that these plant growth promoting factors may be applied in the form of microbial inoculum6,9 implying the importance these IAA producing yeasts as an excellent alternative source for synthetic IAA. Considering the essential inclusion of plant condiments in starter material preparation, it is presumed that IAA probably play active role in the growth of yeasts whilst the addition of plant parts contributes to the perpetuation of yeast species. However, for industrial applications, yeasts from traditional starter materials are desired to be able to synthesize IAA, where tryptophan (trp) plays a decisive role since the amino acid is reported as a precursor for IAA biosynthesis10. Studies on IAA biosynthesis in yeasts from leaf phylloplane11, from phyllosphere of Drosera indica L12 and endophyes13 suggest that these microbes are capable of producing IAA in presence of external L-Tryptophan. However, Rao et al.14 reported the existence of yeasts which were capable of producing IAA in absence of external tryptophan indicating the presence of Trp independent pathway in yeast as well. A bacterial tryptophan-independent pathway was demonstrated by Prinsen et al.15 in Azospirillum brasilense where 90% of the IAA is synthesized via the tryptophan-independent pathway even when no tryptophan is supplied to the medium. Such detailed study on tryptophan independent mechanism of IAA synthesis has been not been fully deciphered in yeasts.
The vast diversity of traditional knowledge is an excellent source to generate goods of industrial importance with a scientific outlook for countries like India which promises rapid growth of economic development in recent decades. India’s rich heritage of traditional knowhow culminated with experiences gained over thousands of years can act as a source of treasure house to identify micro-organisms and molecules that can be exploited for production of substances with economic potential to fulfil the enormous demand of its huge population. Therefore, it becomes important to exploit any probable industrial trait inherent of these traditionally preserved and maintained stock of microorganisms. Keeping this in view, the present study is focused on assessing the ability of a few selected indigenous yeast isolates to produce IAA in presence and absence of external tryptophan as well as to circumscribe the effect of external IAA on growth of these isolates.
Yeast isolates
For the purpose of current investigation, yeasts belonging to four different species viz Wickerhamomyces anomalus, Saccharomyces cerevisiae, Candida tropicalis and Candida glabrata were analyzed. These isolates were originally isolated from fermentative starter materials of six indigenous communities collected by Parasar et al.16 from various locations of Assam. The details of the isolates are provided in Table 1. Purity of isolates were checked by streaking them onto YPD (yeast extract 1%, mycological peptone 2%, dextrose 2%) agar plates and allowed to incubate for 30 ± 2°C for 24 ± 2 hours. Individual colonies were picked and observed under Leica DM750 phase contrast microscope followed by another 24 ± 2 hours incubation in YPD broth at 30 ± 2°C in a rotary shaking incubator maintained at 100 rpm. The pure cultures were preserved in cryovials and stored at 4°C for further analyses.
Table (1):
Details of the yeast isolates selected for the current investigation. Data reproduced from Parasar et al (2017)16
Sl. No |
Isolate |
Molecular identity |
Community |
Colony texture in YPD media |
Accession Number (NCBI and DDBJ) |
|---|---|---|---|---|---|
1 |
RAA1X |
Wickerhamomyces anomalus |
Rabha |
Smooth, flat, semi opaque |
KM603613 (NCBI) |
2 |
RAB1X |
Wickerhamomyces anomalus |
Rabha |
Rough, Opaque, flat |
KM603604 (NCBI) |
3 |
RAB1Y |
Wickerhamomyces anomalus |
Rabha |
Smooth, opaque, flat |
KM603609 (NCBI) |
4 |
RAB1.1Y |
Wickerhamomyces anomalus |
Rabha |
Smooth, flat, semi opaque |
KM603618 (NCBI) |
5 |
RAB3X |
Wickerhamomyces anomalus |
Rabha |
Smooth, opaque, elevated |
KM603601 (NCBI) |
6 |
BOA1X |
Wickerhamomyces anomalus |
Bodo |
Smooth, flat, semi opaque |
KM603612 (NCBI) |
7 |
BOA2Y |
Wickerhamomyces anomalus |
Bodo |
Smooth, flat, semi opaque |
KM603614 (NCBI) |
8 |
BOB1X |
Wickerhamomyces anomalus |
Bodo |
Smooth, flat, semi transparent |
KM603616 (NCBI) |
9 |
BOB3X |
Wickerhamomyces anomalus |
Bodo |
Rough, elevated, semi transparent |
LC011409 (DDBJ) |
10 |
BOB3Y |
Wickerhamomyces anomalus |
Bodo |
Smooth, flat, semi transparent |
KM603608 (NCBI) |
11 |
BOC2X |
Wickerhamomyces anomalus |
Bodo |
Smooth, flat, opaque |
KM603617 (NCBI) |
12 |
KR2X |
Wickerhamomyces anomalus |
Karbi |
Smooth, elevated, opaque |
LC011408 (DDBJ) |
13 |
KC6X |
Wickerhamomyces anomalus |
Kachari |
Smooth, flat, semi transparent |
LC011410 (DDBJ) |
14 |
MIB4X |
Wickerhamomyces anomalus |
Mishing |
Smooth, elevated, opaque |
KM603602 (NCBI) |
15 |
KR1.4 |
Saccharomyces cerevisiae |
Karbi |
Smooth, flat, opaque |
KM603621 (NCBI) |
16 |
KR2.11 |
Saccharomyces cerevisiae |
Karbi |
Smooth, flat, opaque |
KM603622 (NCBI) |
17 |
KR4.10 |
Saccharomyces cerevisiae |
Karbi |
Smooth, flat, opaque |
KM603623 (NCBI) |
18 |
KR5.6 |
Saccharomyces cerevisiae |
Karbi |
Rough, opaque, elevated |
KM603620 (NCBI) |
19 |
KR2Y |
Saccharomyces cerevisiae |
Karbi |
Smooth, elevated, opaque |
KM603619 (NCBI) |
20 |
KC4X |
Saccharomyces cerevisiae |
Kachari |
Smooth, opaque, elevated |
KM603607 (NCBI) |
21 |
BOC1X |
Saccharomyces cerevisiae |
Bodo |
Smooth, opaque, elevated |
KM603603 (NCBI) |
22 |
AH3SA |
Candida tropicalis |
Ahom |
Smooth, flat, semi opaque |
KM603605 (NCBI) |
23 |
2D |
Candida tropicalis |
Ahom |
Smooth, flat, semi opaque |
KM603615 (NCBI) |
24 |
AH2SA |
Candida glabrata |
Ahom |
Smooth, elevated, opaque |
KM603625 (NCBI) |
25 |
KC3X |
Candida glabrata |
Kachari |
Smooth, opaque, elevated |
LC011411 (DDBJ) |
Determination of evolutionary relationship
In order to determine phylogenetic relationship, the sequences representing D1/D2 domain of yeast genomic DNA was obtained through polymerase chain reaction (PCR) and deposited to Genebank of NCBI database as per method already described in Parasar et al.16.
Genomic DNA extraction
Genomic DNA from the yeast isolates were extracted following the methodology of Xiao et al.17 with minor modification. Briefly, 1 ml of overnight grown yeast cells in YPD broth were centrifuged at 15000g for 5 minutes in a refrigerated centrifuge (Eppendorf 5430R) at 4°C. Pellets were resuspended in 230µL of DNA lysis buffer (Himedia R075) and vortexed for 3 minutes with 0.4gm of acid washed glass beads (0.2-0.6µm). Washed cells were centrifuged and resuspended in 200µl of phenol: chloroform: isoamyl alcohol (1:1:1). After centrifugation for 5 minutes, the aqueous phase (top was transferred to new tube where 600µL of cold 95% ethanol was added and kept at -20°C for 30 minutes. DNA was pelleted by centrifugation at 15000g for 15 minutes and ethanol was discarded. The tubes were dried under vacuum for 5 minutes. The DNA pellets were resuspended in 200µl of TE to which 5µl of RNase A was added and allowed to incubate at 37°C for 10 minutes. 8µl of 5 M NaCl and 400µl of cold 95% ethanol was added in each sample and kept at -20°C for 30 minutes. DNA was finally pelleted by centrifugation at 15000g for 15 min, while the supernatant containing ethanol was discarded. Finally, DNA pellets were air dried and resuspended in 200µl of TE buffer.
PCR amplification
The ITS1 and ITS4 regions of yeast DNA were subjected to PCR amplification using the primers 52 -TCCGTAGGTGAACCTGCG-32 and 52 -TCCTCCGCTTATTGATATGC-32 respectively for D1/D2 domain of 5.8S fragment of ITS region18. The reactions were performed in a thermal cycler (Master cycler Nexus Gradient, Eppendorf) in a final volume of 25µl. The final concentration of each ingredients was optimized at 1X of the standard buffer that included 1.5 mM of MgCl2, 0.2µM of each primer, 0.2 mM of dNTPs, 0.25 U of Taq DNA polymerase and 25 ng of template DNA. Thermal cycling was performed with initial denaturation at 94°C for 3 minutes, followed by 30 cycles of denaturation at 94°C for 1 minute, annealing at 55°C for 1 minute and extension at 72°C for 3 minutes followed by a final extension at 72°C for 10 minutes18. Amplicons were analyzed on a 1.2% agarose gel and visualized under Bio Doc-It Imaging System (UVP, USA). The amplified products were eluted and purified through Himedia Agarose Gel DNA extraction kit (MB503) and were sequenced in a 96-capillary sequencer (Shimadzu).
Phylogenetic analysis
To examine the evolutionary relationships between the selected isolates, the obtained sequences were analysed through a BLAST search against the non-redundant nucleotide database, and were submitted to NCBI Genbank and DDBJ (Table 1). Mmultiple sequence analysis was done using Clustal X program version 2.019. Pphylogenetic dendrogram was deducted using UPGMA (Unweighted Pair Group Method with Arithmetic Mean) method20 considering bootstrap value from 1000 re-samplings to evaluate branching tree patterns21. MEGA4 software was used to evaluate phylogenetic analysis and to portray tree structure20.
Quantification of indole-3-acetic acid (IAA) isolates in presence and absence of exogenous tryptophan
Growth assay
To quantify the IAA production, yeast isolates were grown in liquid synthetic minimal medium (SD, pH 4.3) without any amino acids as described by Chen et al.22 at 30 ± 2°C for 24 ± 2 hours in shaker incubator at 120 rpm. After completion of incubation, absorbance was noted at 600 nm and OD600 of all isolates were adjusted to ~1.0 with fresh SD medium. Cells were centrifuged at 15000g for 5 minutes, washed twice with distilled water and finally re suspended in 20mL SD media formulated with and without addition of L-tryptophan to find out the ability of the isolates to produce IAA both in presence and in absence of exogenous tryptophan. Cultures were allowed to incubate at 30 ± 2°C for 48 ± 2 hours at rotary shaker incubator at 120 rpm.
Detection and quantification of IAA
Detection and quantitative estimation of produced IAA was done following the methodology of Sun et al.12 with minor modifications. Briefly, 2mL of the previously grown cell culture was pelleted by centrifuging at 15000g for 5 minutes at 4°C. 0.5 mL of the cell free supernatant from each sample was mixed with 0.5 mL of Salkowaski reagent {2 mL of 0.5M iron(III) chloride and 98 mL of 35% perchloric acid23}, vortexed for 1 minute and allowed to stand at room temperature for 30 min. Color development (red) was quantified at 530 nm after 30 minutes. Standard IAA was used to prepare calibration curve for quantitative IAA estimation in each isolate.
Evaluating growth of isolates in presence and absence of exogenous IAA
In order to determine if IAA has any effect on cell growth rate, the isolates were subjected to growth both in presence and absence exogenous IAA. Liquid synthetic minimal medium (SD, pH 4.3) was used for the assay. Isolates were incubated in SD medium overnight and OD600 of each isolate were adjusted to ~0.1. Cells were pelleted at 15000g for 5 minutes and resuspended in 20ml fresh SD medium supplemented with (10µg.ml-1, 50µg.ml-1 and 100µg.ml-1) and without (blank) exogenous IAA (Merck, India). Cultures were allowed to incubate at 30 ± 2°C for 24 ± 2 hours at rotary shaker incubator at 120 rpm. After completion of incubation period, 1ml of each culture was palleted at 15000g, washed twice and resuspended in 1ml distilled water prior to measuring the OD600 for estimating relative growth of isolates.
Statistical analysis
Statistical analysis was performed in GraphPad Prism software version 7.04 and data were reported as means ± SEM of measurements from three replicates of experiments. Two-way ANOVA was performed using Tukey’s multiple range test (Post Hoc) to compare the effect of exogenous tryptophan on IAA production and Dunnett’s multiple comparison to asses growth of isolates in presence of exogenous IAA compared to blank (without IAA). The significance was determined at three levels (*p<0.05, **p<0.005 and ***p<0.0001).
Yeast isolates and their phylogenetic relationship
A total of 25 yeast isolates were procured for current investigations consisting fourteen Wickerhamomyces anomalus, seven Saccharomyces cerevisiae, two Candida tropicalis and two Candida glabrata. The details of these isolates are listed in Table 1. Evolutionary relationship is shown in Fig. 1. The tree dendrogram establishes close relationship among all the yeast isolates despite their different source of perpetuation with an exception of two Saccharomyces isolates viz KC4X and BOC1X (Fig. 1) which are grouped with Wickerhamomyces spp. ascertaining their genetic resemblances with the genus rather than the other Saccharomyces isolates. Moreover, Candida glabrata was found to be in close evolutionary relationship with Saccharomyces cerevisiae.
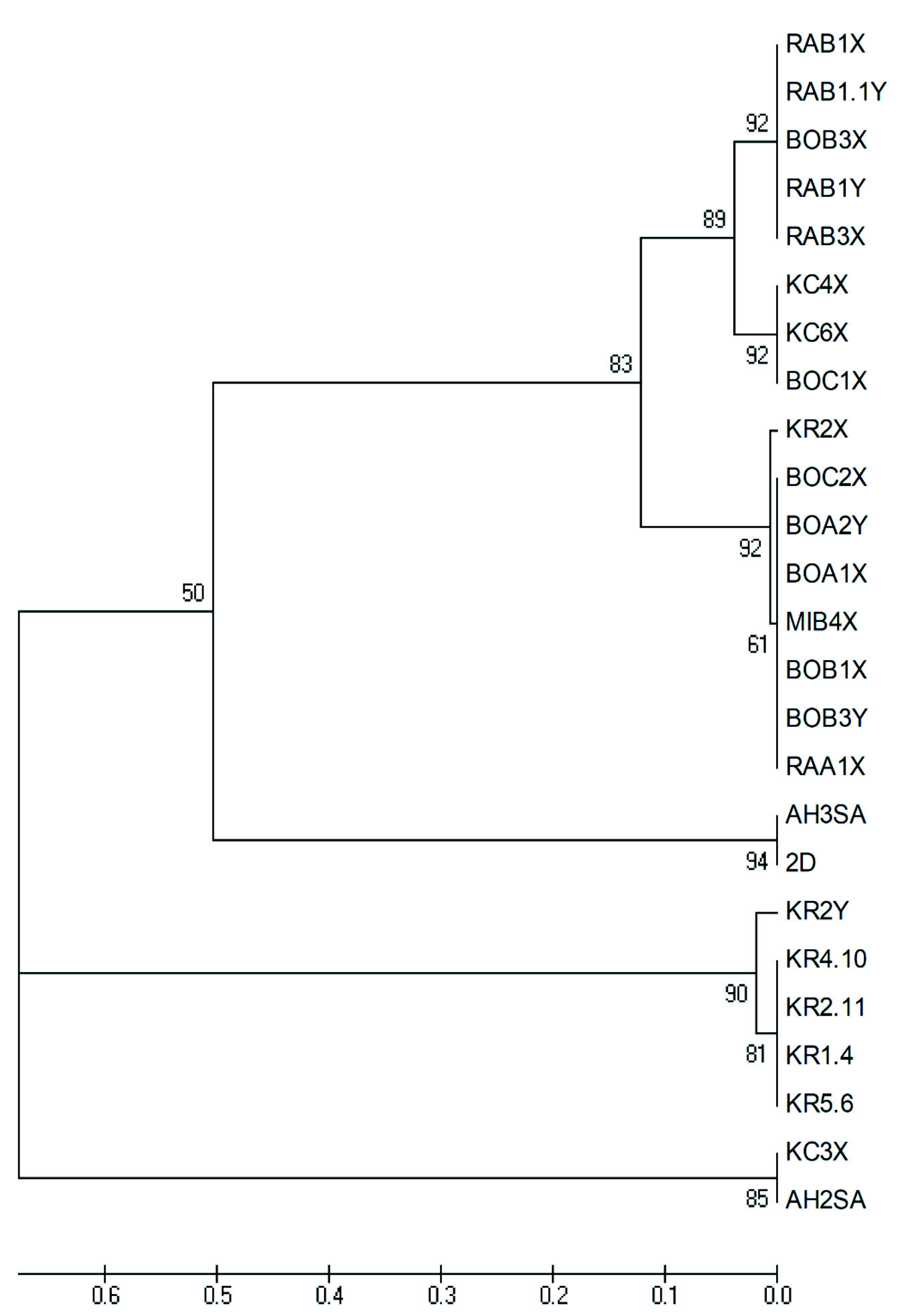 Fig. 1.Bootstrap analysis of UPGMA (Unweighted Pair Group Method with Arithmetic Mean) tree showing the phylogenetic relationship amongst the tested isolates viz Wickerhamomyces anomalus (RAB1X, RAB1.1Y, BOB3X, RAB1Y, RAB3X, KC6X, KR2X, BOC2X, BOA2Y, BOA1X, MIB4X, BOB1X, BOB3Y, RAA1X), Saccharomyces cerevisiae (KC4X, BOC1X, KR2Y, KR4.10, KR2.11, KR1.4, KR5.6), Candida tropicalis (AH3SA, 2D) and Candida glabrata (KC3X, AH2SA)
Fig. 1.Bootstrap analysis of UPGMA (Unweighted Pair Group Method with Arithmetic Mean) tree showing the phylogenetic relationship amongst the tested isolates viz Wickerhamomyces anomalus (RAB1X, RAB1.1Y, BOB3X, RAB1Y, RAB3X, KC6X, KR2X, BOC2X, BOA2Y, BOA1X, MIB4X, BOB1X, BOB3Y, RAA1X), Saccharomyces cerevisiae (KC4X, BOC1X, KR2Y, KR4.10, KR2.11, KR1.4, KR5.6), Candida tropicalis (AH3SA, 2D) and Candida glabrata (KC3X, AH2SA)Quantification of indole-3-acetic acid produced by isolates
The isolates were grown in presence and absence of external tryptophan. The isolates exhibited variation in production of IAA. The presence of exogenous Tryptophan could imply its effect in some isolates, whereas for some isolates tryptophan could merely affect the production rate (Fig. 2). Synthesis of IAA in presence of tryptophan was found in Wickerhamomyces isolates (RAB3x, RAB1.1Y), Saccharomyces isolates (KR1.4, KR2.11, KR2Y), Candida glabrata isolates (KC3x, AH2SA), but were unable to produce any in absence of external tryptophan. Exogenous tryptophan was found to enhance the production of IAA in five Wickerhamomyces isolates (~1.8 fold for RAB1X, ~1.1 fold for RAA1X, ~1.3 fold for BOB3X, ~1.8 folds for BOB1X and ~1.12 fold for KC6X), two Saccharomyces isolates (~1.87 fold for BOC1X and ~2.56 fold for KC4X) and one Candida tropicalis (~4.7 fold for AH3SA) isolate. On the contrary, IAA was found to be produced more in absence of external tryptophan in six Wickerhamomyces isolates (~1.85 fold for MIB4X, ~2.44 fold for BOB3Y, ~2.45 fold for RAB1Y, ~1.24 fold for BOA1X, ~1.44 fold for BOA2Y and ~1.15 fold for BOC2X), one Saccharomyces isolate (~5.13 fold for KR4.10) and one Candida tropicalis isolate (~1.1 fold for 2D). Among these tested isolates, Candida glabrata isolates were found to produce highest amount of IAA (16.71 ± 1.45µg.ml-1 for KC3X and 8.66 ± 1.1µg.ml-1 for AH2SA) in presence of tryptophan.
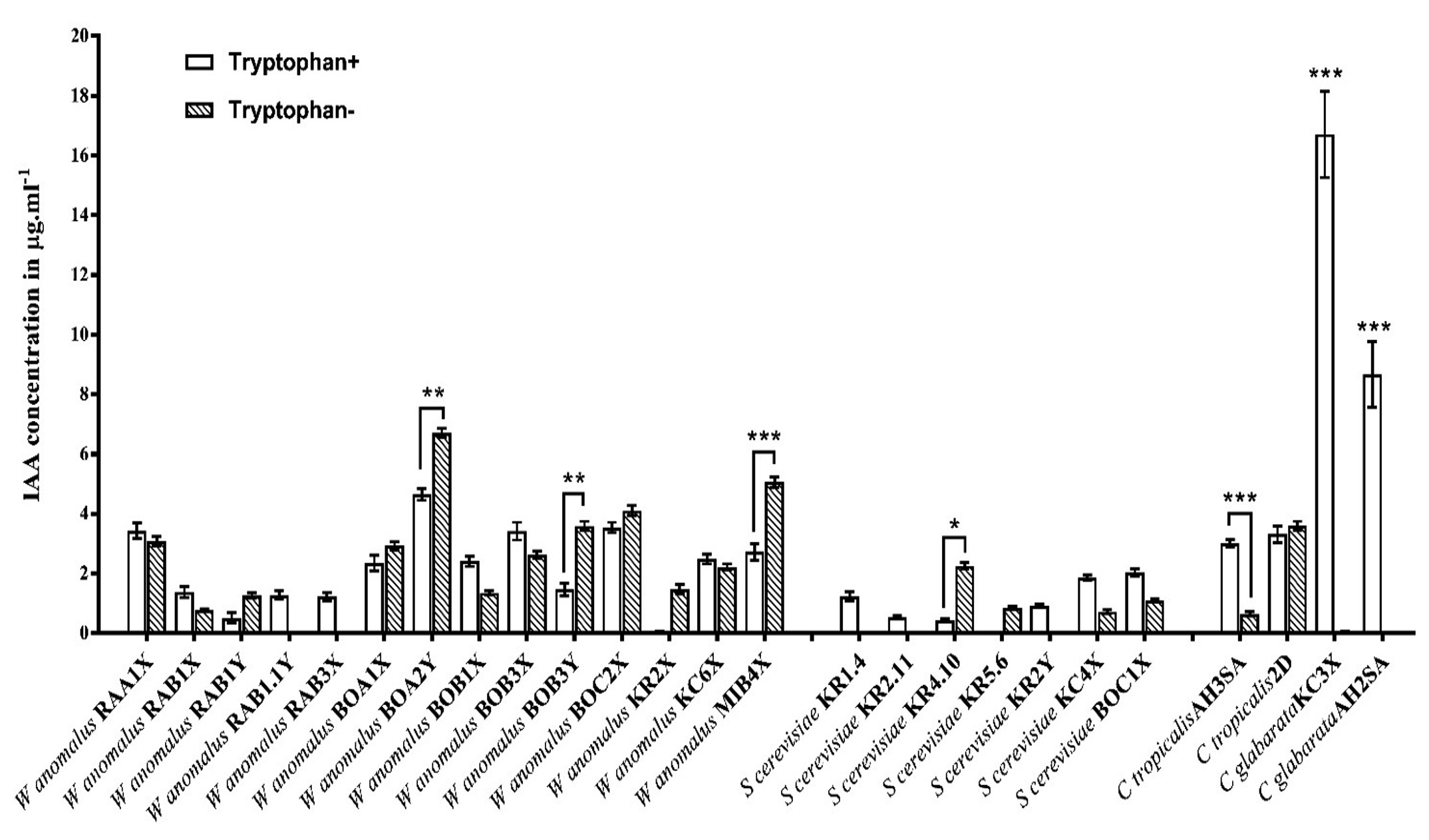 Fig. 2. Fig. showing Indole-3-acetic acid (IAA) production capacity of yeast isolates in presence and absence of external tryptophan (*p<0.05, **p<0.005 and ***p<0.0001).
Fig. 2. Fig. showing Indole-3-acetic acid (IAA) production capacity of yeast isolates in presence and absence of external tryptophan (*p<0.05, **p<0.005 and ***p<0.0001). Growth of isolates in presence and absence of external IAA
The role of exogenous IAA during growth was evaluated on 25 isolates and is shown in Fig. 3. In case of Wickerhamomyces isolates, no significant enhancement of growth was observed in presence of IAA. Minor increase or decrease of growth was noticed which was found to be non-uniform among these isolates with an exception to isolates BOB3X which showed ~1.1 fold increase in growth in presence of 50µg.ml-1 exogenous IAA. Noticeable enhancement of growth was observed in Saccharomyces isolates. Isolate KR1.4 exhibited ~1.8 fold increase (p<0.05) in presence of 100µg.ml-1 IAA, Isolate KR2.11 showed ~1.14 fold increase in 10µg.ml-1 IAA, ~1.3 fold increase (p<0.005) in 50µg.ml-1 IAA and ~1.4 fold increase (p<0.0001) in 100µg.ml-1 IAA. Similarly, isolate KR4.1 bared ~1.2 fold increase in 10µg.ml-1 IAA, ~1.3 fold increase (p<0.005) in 50µg.ml-1 IAA and ~1.4 fold increase (p<0.0001) in 100µg.ml-1 IAA. Likewise, ~1.2 fold increase (p<0.05) in 10µg.ml-1 IAA, ~1.3 fold increase (p<0.005) in 50µg.ml-1 IAA and ~1.5 fold increase (p<0.0001) in 100µg.ml-1 IAA was seen in case of isolate KR5.6. Noticeable increase in growth was observed in 100µg.ml-1 IAA only in case of isolate KR2Y (~1.16 fold), KC4X (~1.23 fold) and BOC1X (~1.3 fold). On the contrary, Candida tropicalis and Candida glabrata could not exhibit noticeable increase in growth in presence of IAA.
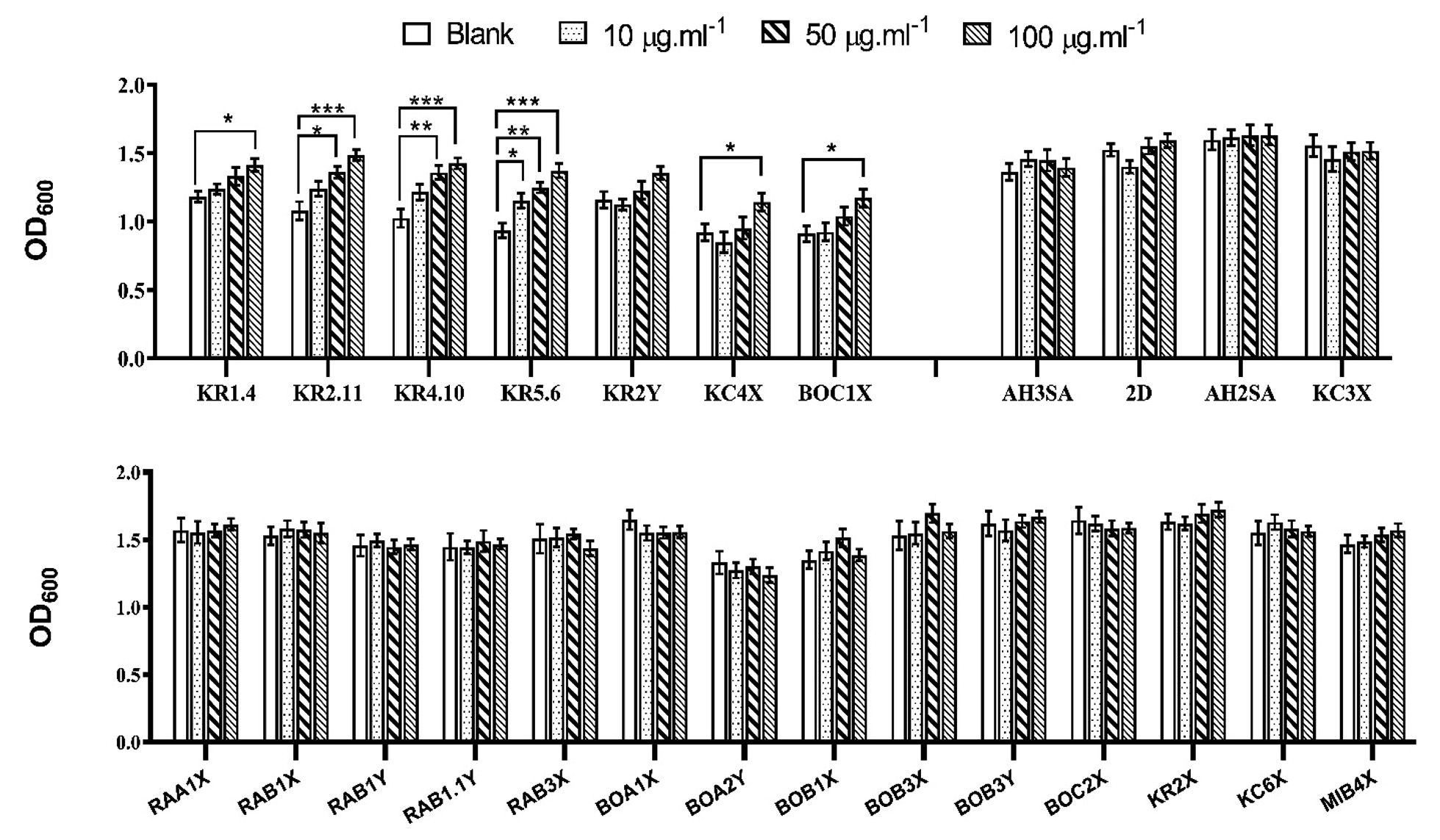 Fig. 3. Fig. showing relative growth of yeasts viz Saccharomyces cerevisiae (KC4X, BOC1X, KR4.10, KR2.11, KR1.4, KR5.6), Candida tropicalis (AH3SA, 2D), Candida glabrata (KC3X, AH2SA) and Wickerhamomyces anomalus (RAA1X, RAB1X, RAB1Y, RAB1.1Y, RAB3X, BOA1X, BOA2Y, BOB1X, BOB3X, BOB3Y, BOC2X, KR2X, KC6X, MIB4X,) in absence and presence(10, 50 and 100µg.ml-1) exogenous IAA (*p<0.05, **p<0.005 and ***p<0.0001)
Fig. 3. Fig. showing relative growth of yeasts viz Saccharomyces cerevisiae (KC4X, BOC1X, KR4.10, KR2.11, KR1.4, KR5.6), Candida tropicalis (AH3SA, 2D), Candida glabrata (KC3X, AH2SA) and Wickerhamomyces anomalus (RAA1X, RAB1X, RAB1Y, RAB1.1Y, RAB3X, BOA1X, BOA2Y, BOB1X, BOB3X, BOB3Y, BOC2X, KR2X, KC6X, MIB4X,) in absence and presence(10, 50 and 100µg.ml-1) exogenous IAA (*p<0.05, **p<0.005 and ***p<0.0001)Table (2):
Data of Two-Way Analysis of Variance (ANOVA) analysed for screening total interactions among individual isolates. Tukey’s multiple comparison test was done to evaluate interactions among isolates for production of IAA and Dunnett’s multiple comparison test was done to screen relative growth of individual isolates in presence and absence of IAA. Three levels of significance were observed (*p<0.05, **p<0.005 and ***p<0.0001).
| Two-way ANOVA | SS | df | MS | F (DFn, DFd) | P value | Significance | |
|---|---|---|---|---|---|---|---|
| Tukey’s multiple comparison test | Total interaction (Row vs Column) | 546.5 | 24 | 22.77 | F (24, 100) = 88.7 | p<0.0001 | *** |
| Interaction of W anomalus, S cerevisiae, C tropicalis and C glabrata isolates (Row factor) | 530.8 | 24 | 22.11 | F (24, 100) = 86.13 | p<0.0001 | *** | |
| Relative comparison of produced IAA in presence and absence of tryptophan (Column factor) | 38.39 | 1 | 38.39 | F (1, 100) = 149.5 | p<0.0001 | *** | |
| Residual | 25.67 | 100 | 0.2567 | ||||
| Dunnett’s multiple comparison test | Total interaction (Row vs Column) | 1.206 | 72 | 0.01675 | F (72, 200) = 1.414 | p=0.0317 | * |
| Interaction of W anomalus, S cerevisiae, C tropicalis and C glabrata isolates (Row factor) | 10.58 | 24 | 0.4408 | F (24, 200) = 37.21 | p<0.0001 | *** | |
| Relative growth of isolates in 10, 50 and 100µg.ml-1 IAA compared to blank (No IAA) (Column factor) | 0.45 | 3 | 0.15 | F (3, 200) = 12.66 | p<0.0001 | *** | |
| Residual | 2.369 | 200 | 0.01185 | ||||
This is the first reported investigation on the effect of IAA on the survivability of fermentative yeasts from Assam perpetuated on starchy materials and assessing the ability of those isolates to produce necessary IAA in presence and absence of external tryptophan. Several studies have been constructed on the IAA synthesizing ability of yeasts from natural sources11,12,13, but none of the study share reports of such activities on yeasts actively involved in household fermentation processes including the traditional fermentation of this region. The predominance of selective species of yeasts from the selected regions of northeast India has already been discussed in our previous report16. From phylogenetic analysis it was observed that similar isolates from different communities showed resemblances such as Wickerhamomyces anomalus from Bodo, Rabha, Kachari and Mishing communities. Such similarity could also be observed in Candida tropicalis and Candida glabrata as well. However, exception could be seen in Saccharomyces cerevisiae isolates, where all Saccharomyces from Karbi community showed similarity amongst themselves, isolates KC4X from Kachari community and BOC1X from Bodo community showed more resemblance with Wickerhamomyces isolates than to Saccharomyces isolates (Fig. 1). Such similarity could be explained by probable genomic interchanges since yeasts are known to exhibit various mechanisms of adaptive evolution such as gene duplication, polyploidy, chromosomal rear-rangements, interspecies hybridization and introgression during fermentation processes24. Such introgression types from S. cerevisiae into S. paradoxus has already been reported by Liti et al.25. Additionally, Novo et al.26 reported that sequence of tested wine strain Saccharomyces cerevisiae EC1118 contained several genome segments from outside S. cerevisiae, but within the Saccharomyces superfamily.
Our current investigation revealed that all Wickerhamomyces isolates were able to produce IAA in presence and absence of external tryptophan with an exception of isolate KR2X being unable to synthesize IAA in presence of tryptophan while isolates RAB1.1Y and RAB3X were unable to do so in absence of tryptophan which contradict a previous report where Wickerhamomyces isolates (W. edaphicaus LM02811, W siamensis DMKU-RK35927) were unable to produce detectable IAA. Saccharomyces isolates did not show significant production of IAA except isolate KR4.10 which produced IAA in absence of tryptophan significantly (p<0.05). Candida tropicalis isolate AH3SA exhibited significant (p<0.0001) IAA production in presence of tryptophan and finally both Candida glabrata isolates were able to synthesize IAA significantly (p<0.0001) in presence of tryptophan that resemble with the previous studies where Candida tropicalis was found to produce IAA27 along with other Candida isolates12,27. There is no clear indication describing the ability Candida glabrata to synthesize IAA, but investigation done by Mayser et al.28 showed the ability to produce other indole compounds by the isolate. Current study demonstrates that the two C glabrata isolates have the ability to produce significant amount of IAA.
Earlier reports and current study suggest that yeasts are capable of producing IAA both in presence and absence of tryptophan. In-spite of being a major precursor, tryptophan may not be available in sufficient quantity or not at all to the yeasts for production of IAA29. In such case, presence of trp independent pathway to synthesize IAA proves to be of great importance to yeasts, but details about independent pathway in yeats is not properly deciphered. Trp-dependant and trp-independent pathways of IAA biosynthesis has been reported in plants30,31,32 and in microbes15. Zhang et al.33 reported the existence of indole synthase (INS) gene, a homolog of TSA1 in Arabidosis that was reported to take part in Trp-independent pathway of auxin biosynthesis. Rao et al.14 suggested that a Trp-independent pathway for IAA synthesis does exist in yeasts. These collective evidences support the finding of this study where significant amount of increase in IAA production was observed in Wickerhamomyces isolates viz BOA2Y(p<0.005), BOB3Y (p<0.005), MIB4X (p<0.005) and Saccharomyces isolate KR4.10 (p<0.05) in absence of external tryptophan. The presence of such isolates capable of producing IAA in absence of external tryptophan prove to be potent candidates in bio fertilizer industry34.
To evaluate the possible effect of IAA on yeast growth, isolates were subjected to incubation in presence and absence of exogenous IAA. Significant increase in growth was observed in presence of IAA only in Saccharomyces isolates (Fig. 3). Previous study has also reported that IAA does not only stimulate growth, but also promote filamentous morphogenesis in Saccharomyces35 which support the fact that IAA act as a primary signaling molecule to regulate yeast growth. Growth promoting nature of IAA was also reported in previous works on Fusarium delphinoides36 where 50µM exogenous IAA stimulated 80% growth. The present study showed similar increase in growth as has been described in other fungal species. Notably Wicker-hamomyces, Candida tropicalis and Candida glabrata did not show any significant increase in growth in presence of IAA suggesting the absence of any stimulatory effect of IAA on these isolates.
From the study it may be concluded that yeast isolates perpetuated in starchy starter materials rich in plant condiments are adapted to growth in presence of IAA obtained from plant materials. Although Wickerhamomyces, C tropicalis and C glabrata did not respond to IAA, yet the ability of some of the isolates to synthesize IAA in absence as well as in presence of external tryptophan prove to be advantageous since it could presumably affect the other fermentative yeasts in consortium, such as Saccharomyces, which positively reacts to the presence of external IAA. This commensalism could prove to be beneficial in co-culture fermentation, where slow growing yeasts like Saccharomyces gain advantage from such IAA producing isolates in consortial growth.
Conflicts Of Interest
The author declare that there are no conflicts of interest.
Author Contributions
BN and DP conducted entire sets of experiments, generated and analyzed data, performed statistical analysis and drafted the manuscript. EV performed PCR reactions and generated genomic data. HS formulated the initial concept, supervised the work, collated and interpreted data, provided intellectual input and finalized the manuscript. AM guided the experiments, provided intellectual input and is a collaborator of the work.
Funding
This investigation was supported by DBT Twining project (BT/303/NE/TBP/2012 dt 04/01/2013) sanctioned to H.K.S. and A.K.M. by the Department of Biotechnology, Govt. of India. B.J.N. received DBT fellowship (2013-2016) and now a recipient of NFOBC-UGC fellowship for PhD under H.K.S. D.P.P. was a recipient of BSR-UGC fellowship (2012-2017) for PhD under HKS.
Data Availability
The dataset generated during the investigation are included in the manuscript and supplementary files.
Ethics
The study does not contain any studies with Human participants or animals performed by any of the authors.
- Hesseltine C.W., Rogers R., Winarno F.G. Microbiological studies on amylolytic oriental fermentation starters. Mycopathologia, 1988; 101: 141–155.
- Sarma H.K. Isolation and Characterization of Yeast Strains from Indigenous Starter Culture for Alcoholic Fermentation, 2002, pp. 85-97. In Tiwari S.C., Sharma G.D. (eds.), Microbial Diversity: Status and Potential Applications, Scientific book publishers, Guwahati, Assam.
- Cleland R.E. Auxin and Cell Elongation, 2010, pp. 204-220. In Davies PJ (eds.), Plant Hormones, Springer, Dordrecht.
- El-Tarabily K.A. Suppression of Rhizoctonia solani diseases of sugar beet by antagonistic and plant growth-promoting yeasts. J. Appl. Microbiol., 2004; 96: 69–75.
- Nakamura T., Murakami T., Saotome M., Tomita K., Kitsuwa T., Meyers S. Identification of Indole-3-Acetic Acid in Pichia spartinae, an Ascosporogenous Yeast from Spartina alterniflora Marshland Environments. Mycologia, 1991; 83: 662–664.
- Nassar A.H., El-Tarabily K.A., Sivasithamparam K. Promotion of plant growth by an auxin-producing isolate of the yeast Williopsis saturnus endophytic in maize (Zea mays L.) roots. Biol. Fertil. Soils, 2005; 42: 97–108.
- Sasikala C., Ramana C.V. Biodegradation and Metabolism of Unusual Carbon Compounds by Anoxygenic Phototrophic Bacteria. Advances in Microbial Physiology, 1997; 39: 339-377.
- Ahmad F., Ahmad I., Khan M.S. Screening of free-living rhizospheric bacteria for their multiple plant growth promoting activities. Microbiol. Res., 2008; 163: 173–181.
- Tsavkelova E.A., Klimova S.Y., Cherdyntseva T.A., Netrusov A.I. Microbial producers of plant growth stimulators and their practical use: A review. Appl. Biochem. Microbiol., 2006; 42: 117–126.
- Hazelwood L.A., Daran J.M., van Maris A.J.A., Pronk J.T., Dickinson J.R. The Ehrlich pathway for fusel alcohol production: a century of research on Saccharomyces cerevisiae metabolism. Appl Environ Microbiol, 2008; 74: 2259–2266.
- Limtong S., Koowadjanakul N. Yeasts from phylloplane and their capability to produce indole-3-acetic acid. World J. Microbiol. Biotechnol., 2012; 28: 3323–3335.
- Sun P.F., Fang W.T., Shin L.Y., Wei J.Y., Fu S.F., Chou J.Y. Indole-3-acetic acid-producing yeasts in the phyllosphere of the carnivorous plant Drosera indica L, PLoS One, 2014; 9: 1–22.
- Xin G., Glawe D., Doty S.L. Characterization of three endophytic, indole-3-acetic acid-producing yeasts occurring in Populus trees. Mycol. Res. 2009; 113: 973–980.
- Rao R.P, Hunter A., Kashpur O., Normanly J. Aberrant synthesis of indole-3-acetic acid in Saccharomyces cerevisiae triggers morphogenic transition, a virulence trait of pathogenic fungi. Genetics, 2010; 185: 211–220.
- Prinsen E., Costacurta A., Michiels C., Vanderleyden J., Van Onckelen H. Azospirillum brasilense Indole-3-acetic acid biosynthesis: Evidence for a non-tryptophan dependent pathway. Mol. Plant Microbe Interact, 1993; 6: 609–615.
- Parasar D.P., Sarma H.K, Kotoky J. Exploring the genealogy and phenomic divergences of indigenous domesticated yeasts cultivated by six ethnic communities of Assam, India. J. Biol. Sci., 2017; 17: 91–105.
- Hanna M, Xiao, W. Isolation of Nucleic Acids, 2006, pp 15-20. In Xiao W (eds.), Yeast Protocol. Methods in Molecular Biology, vol 313, Humana press, Totowa, N.J.
- Naumova E.S., Serpova E.V., Naumov G.I. Molecular systematics of Lachancea yeasts. Biochemistry (Mosc.), 2007; 72: 1356–1362.
- Larkin M.A., Blackshields G., Brown N.P., Chenna R., McGettigan P.A., McWilliam H., Valentin F., Wallace I.M., Wilm A., Lopez R., Thompson J.D., Gibson T.J., Higgins D.G. Clustal W and ClustalX version 2. Bioinformatics, 2007; 23: 2947–2948.
- Tamura K., Dudley J., Nei M., Kumar S. MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol. Biol. Evol., 2007; 24: 1596–1599.
- Felsenstein J. Confidence Limits on Phylogenies: An Approach Using the Bootstrap; Confidence Limits on Phylogenies: An Approach Using the Bootstrap. Evolution (N.Y.), 1985; 39: 783–791.
- Chen H., Fujita M., Feng Q., Clardy J., Fink G.R. Tyrosol is a quorum-sensing molecule in Candida albicans. Proc. Natl. Acad. Sci., 2004; 101: 5048–5052.
- Gordon S.A., Weber R.P. Colorimetric estimation of Indoleacetic acid. Plant Physiol., 1951; 26: 192-195.
- Barrio E., Gonzבlez S.S., Arias A., Belloch C., Querol A. Molecular mechanisms involved in the adaptive evolution of industrial yeasts, 2006, pp. 153-174. In Querol A, Fleet G (eds.), Yeasts in Food and Beverages, Springer Berlin Heidelberg.
- Liti G., Barton D.B.H, Louis E.J. Sequence diversity, reproductive isolation and species concepts in Saccharomyces. Genetics, 2006; 174: 839–850.
- Novo M., Bigey F., Beyne E., Galeote V., Gavory F., Mallet S., Cambon B., Legras J.L., Wincker P., Casaregola S., Dequin S. Eukaryote-to-eukaryote gene transfer events revealed by the genome sequence of the wine yeast Saccharomyces cerevisiae EC1118. Proc. Natl. Acad. Sci., 2009; 106: 16333–16338.
- Limtong S., Kaewwichian R., Yongmanitchai W., Kawasaki H. Diversity of culturable yeasts in phylloplane of sugarcane in Thailand and their capability to produce indole-3-acetic acid. World J. Microbiol. Biotechnol., 2014; 30: 1785–1796.
- Mayser P., Wenzel M., Krהmer H.J., Kindler B.L.J, Spiteller P., Haase G. Production of indole pigments by Candida glabrata. Med. Mycol., 2007; 45: 519–524.
- Fu S.F., Wei J.Y., Chen H.W., Liu Y.Y., Lu H.Y., Chou J.Y. Indole-3-acetic acid: A widespread physiological code in interactions of fungi with other organisms. Plant Signal Behav., 2015; 10: e1048052.
- Woodward A.W., Bartel B. Auxin: Regulation, action, and interaction. Ann. Bot., 2005; 95: 707–735.
- Chandler J.W. Local auxin production: A small contribution to a big field. Bioessays, 2009; 31: 60–70.
- Normanly J. Approaching cellular and molecular resolution of auxin biosynthesis and metabolism. Cold Spring Harb. Perspect Biol., 2010; 2: 1–17.
- Zhang R., Wang B., Ouyang J., Li J., Wang Y. Arabidopsis indole synthase, a homolog of tryptophan synthase alpha, is an enzyme involved in the Trp-independent indole-containing metabolite biosynthesis. J. Integr. Plant. Biol., 2008; 50: 1070–1077.
- Nandal M., Hooda R. Plant growth promoting Rhizobacteria: A review article. International Journal of Current Research, 2013; 5: 3863–3871.
- Prusty R., Grisafi P., Fink G.R. The plant hormone indoleacetic acid induces invasive growth in Saccharomyces cerevisiae. Proc. Natl. Acad Sci., 2004; 101: 4153–4157.
- Kulkarni G.B., Sanjeevkumar S., Kirankumar B., Santoshkumar M., Karegoudar T.B. Indole-3-acetic acid biosynthesis in Fusarium delphinoides strain GPK, a causal agent of wilt in chickpea. Appl. Biochem. Biotechnol., 2013; 169: 1292–1305.
© The Author(s) 2019. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


