ISSN: 0973-7510
E-ISSN: 2581-690X
The growing interest in the key and less explored organic compounds from the diverse mycobiota has been the domain of scientific interest due to their varied properties and potential applications in diverse sectors. The heterogeneity of fungal isolates from different geo-climatic habitats obtrudes every organism as unique and a potential source, awaiting exploration for novel biomolecules. In the current study, we isolated and characterized Aspergillus flavus from the soil of pine tree vegetation. The isolated was further explored for its metabolites using Gas chromatography-mass spectrometry (GC-MS) and Liquid chromatography-mass spectrometry (LC-MS). The dried extract was analyzed to be rich consortia of the diverse organic metabolites, Indole-3-acetic acid, Tryptophan, flavonoids (TFC), and others, that may confer catalogs of therapeutic activities including antioxidants, antimicrobials, and anti-cancerous properties. Further Ultra-performance liquid chromatography (UPLC) analysis showed 0.41 mg/mL of Indole-3-acetic acid (IAA) and 1.2 mg/mL of Tryptophan in the dried extract. The dried broth also showed antioxidant activity in the range of 70%-80% ascorbic acid equivalent, obtruding the isolate as a potential source of contribution to plant growth therapeutic and commercially important metabolites.
Indole-3-acetic Acid, Bioactive Molecule, GC-MS, LC-MS, UPLC, Tryptophan, Antioxidant, ITS Sequencing
The growing demand for safe natural alternatives to conventional chemical and synthetic drugs, which are often associated with potentially hazardous side effects and global concern due to increasing pathogen resistance, has provided a paradigm shift toward the exploration of novel natural compounds from diverse microbial sources.1 Some recent studies have reported catalogs of such metabolites being explored in fungi from diverse habitats.2-5 The studies on the therapeutic potential of fungal-derived pigments have also been explored.6 The studies in the last few decades have been focused on the fungal endophytes from plants, followed by the characterization of their metabolites, and an exploration of the associated therapeutic properties.2,4,7 Plant endophytes are attracting scientific interest as the source of novel natural products due to their association with plants. A crucial study from Murali et al.8 have suggested that fungal endophytes account for 7% of the total fungal diversity. The non-disruptive changes conferred by endophytes within the host tissues impact plant physiology through the production of various metabolites. These metabolites may modulate plant growth, and metabolic pathways, and regulate genetic expression. This modulates plant growth during biotic and abiotic stress.9 The review from Riberio et al.,7 explored the chemical diversity in the natural products such as secondary metabolites of Brazilian endophytic fungi, in terms of the natures and types of phenolic, terpenes, and nitrogen-containing compounds produced by Agrawal and Bhatt10 reviewed the bioprospecting of diverse metabolites from endophytes in terms of plant-derived metabolites.10 Such studies also signify the importance of the habitat in the metabolite profiling of fungal isolates due to adaptive changes and acclimatization. Further studies by Awad et al.11 also characterized Trichoderma viridae, isolated from cucumber soil (rhizosphere) for antimicrobial, antioxidant, and anticancer activities. Fungi are renowned for bolstering the growth and yield of host plants through the production of bioactive compounds, which foster interactions between plants and endophytes.11 various biotic and abiotic factors regulate plant growth and the association of plants and soil microbes that have ranged from parasitic to symbiotic is well known. Microbial support in plant growth through the biosynthesis of phytohormones further fosters the pivotal roles of microbes in agriculture. The current study aimed at genetic identification, metabolite profiling, and antioxidant potential from the soil-isolated Aspergillus flavus. fungal was isolated from the agricultural field of pine trees (Pinus roxburghii) rhizosphere at Waknaghat, Himachal Pradesh, India. The isolated was unique due to diversities in the types of metabolites and bioactivities observed in the fermentation broth, which differentiates it from other recent studies.12 The isolated fungi were explored to produce IAA and tryptophan. The unique nature of the metabolites observed in the current study may be an outcome of the adaptive changes and acclimatization of the isolate to the dynamic habitat.
Isolation and characterization of fungal strain
In this investigation, the fungal strain was isolated from the Soil of Pine Tree Vegetation Site in District Solan, Himachal Pradesh, India. The isolation of fungal strain was performed by serial dilution of soil suspension in autoclaved distilled water, followed by spreading the liquid on Yeast Extract Peptone Dextrose (YEPD) growth agar media (HiMedia Pvt. Ltd., India).
Morphological identification
The isolated fungal was cultured on a YEPD growth agar media (HiMedia, Germany) plate at 30 °C for 5 days. The isolated fungus was morphologically identified with the help of Indian Type Culture Collection (ITCC), Pusa Road, New Delhi.
Genomic DNA extraction
The genomic DNA extraction from the fungal isolate was performed using the assay method of Heintz and Gong13 with moderation. Briefly, the isolated fungal biomass was obtained after overnight growth in YEPD media (HiMedia, Germany) at 30 °C in an orbital shaker. The biomass was resuspended in a 2 mL Sorbitol buffer supplemented with 20 µl Lyticase (2.5 mg/mL) (Sigma Aldrich) and this mixture was incubated overnight at 37 °C. Post incubation, the solution (volume 1 mL) at 10000 rpm was centrifuged at a temperature of 4 °C for 60 seconds followed by resuspension of the pellet in 0.5 mL yeast suspension buffer. The solution was further supplemented with 50 µl Sodium lauryl sulfate (SLS) solution (10% w/v) and it was incubated at a temperature of 80 °C for 1 hour. The debris from the pellet was further removed by adding 0.2 mL of Potassium acetate (5M) and the tube was incubated on ice for 1 hour. To remove the debris, at 10000 rpm the tube was centrifuged at 4 °C for 1 minute. The collected supernatant was blended with isopropanol with equilibrium of supernatant and incubated for 5 minutes at 37 °C, and then at 10000 rpm centrifuged for 1 min. The pellet collected was dried at room temperature to dry isopropanol. The pellet was resuspended in 300 µl of autoclaved distilled water with 10 µl of RNase (10 mg/mL), followed by incubation or room temperature for 30 minutes. Furthermore, 30 µl Sodium Acetate (3M) was added. The DNA was again precipitated using 0.2 mL Isopropanol, and centrifugation at 10000 rpm for 60 seconds. The extracted DNA was analysed by the 8% agarose gel electrophoresis and the buffer was Tris-borate-EDTA.
PCR amplification and Sequence analysis
The fungal identification was carried out by the PCR amplification and sequencing of the Inter Transcribing Space (ITS) gene sequence, as reported previously by Patil et al.2 With modifications. The primer sequences for the forward (ITS1) and reverse (ITS4) were the same as reported previously by Riaz et al.14 The synthetic primers were provided by Barcode Bioscience, India (Table 1). The PCR reaction mixtures were prepared in a total volume of 50 µl with 5 µl 2 mM dNTPs, 5 µl of 10x Buffer, 2.5 µl of Taq Polymerase, 2.5 µl of 20 pmol of both primers with 1 µl gDNA were mixed with deionized water. The following conditions were set for polymerase chain reaction amplification of the ITS sequence condition as initial denaturation was set at a temperature of 94 °C for 2 minutes, The cycle number was 30 at a temperature of 94 °C and the time was 30 seconds, 40 °C for 30 seconds, at temperature 72 °C for 60 seconds, and the period of last extension step was at 5 minutes with temperature 72 °C. The polymerase chain reaction amplified obtained product was analyzed for sequencing at Central Instrument Facility, South Delhi campus, New Delhi, India. The Seq Studio 8 Flex Genetic Analyser was used for Sanger sequencing of the PCR amplified fragment.
Table (1):
Details on the forward and reverse primers used for ITS-1 gene amplification using genomic DNA from fungal isolate, as a template
No. |
Primer Name |
Primer Length |
Sequence |
GC Content |
Melting temperature (as per manufacturer’s specification) |
|---|---|---|---|---|---|
1 |
Forward Primer (ITS-1) |
22 |
CTTGGTCATTTAGAGGAAGTAA |
36.4 |
54.7 °C |
2 |
Reverse Primer (ITS-4) |
20 |
TCCTCCGCTTATTGATATGC |
55.25 |
55.25 °C |
Phylogenetic analysis
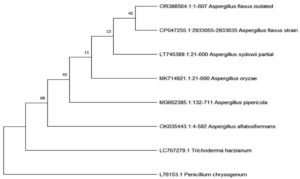
The sequence analysis was performed at the Central Instrument Facility, University of Delhi, South Campus, Delhi, India. The sequence received was aligned using the Basic Local Alignment Search Tool (BLAST) and the identification was done with a sequence showing more than 98.45% similarity. The neighbor-joining technique was employed for the construction of the phylogenetic tree with bootstrap 1000 replications by the use of software Molecular Evolutionary Genetics Analysis (MEGA) X. The received sequence was submitted to the National Center for Biotechnology Information (NCBI) USA GenBank with the accession number OR388564.
Fungal fermentation
The isolated fungal was grown for 5 days on a YEPD growth agar plate at 30 °C and 2.37 × 108 spores/ml was taken from the plate and transferred to 20 mL YEPD incubated for 2 days at 30 °C and 150 rpm using an orbital shaker (Khuner). After the incubation, 1 mL broth was transferred into 12 different flasks containing 50 mL YEPD growth media. The culture studies were performed for 6 days and the flasks were harvested after every 24 hours. The broth was separated from mycelia by centrifugation. Mycelia was discarded and supernatants were dried at room temperature. Dried extracts were resuspended in autoclaved distilled water with a concentration of 20 mg/mL. For all the studies, dried uninoculated media was used as the control.
Analysis of volatile bioactive constituents by gas chromatography-mass spectrometry (GC-MS)
Gas chromatography-mass spectrometry (GC-MS) technique was employed for profiling the organic constituents in the fermentation broth, as reported previously by Farhat et al.15 The fungal dried extract in methanol was imposed to GC-MS with fused silica 60 m x 0.25 mM id x 0.25 µm of DB-5ms Ultra Inert column. Briefly, the extract in methanol of 1 µl was injected and the carrier gas used such as helium. The oven temperature was held for 1 minute at 30 °C then 10 °C/min to 40 °C held for 5 minutes then 40 °C/min to 250 °C held for 5 minutes then 40 °C/min to 275 °C held for 10 minutes. The flow rate of helium was 1 mL/min. The MSD transfer line was 275 °C. The electron energy was conserved (70 eV). The bioactive compounds of the extract were recognized by spectrum obtained from GC-MS using the National Institute of Standards and Technology (NIST) MS data Library at the Indian Institute of Technology Delhi, India.
Analysis of bioactive constituents by liquid chromatography-mass spectrometry (LC-MS)
The extract of the fungal isolate was analyzed by Dionex Ultimate 3000, Thermo Scientific. For the separation of bioactive compounds, the column Hypersil Gold C18 (2.1 mM x 100 mM, 3.0 µm) was used with a temperature of 40 °C. The flow amount was employed at 0.300 mL/min (300 µl/min). The method duration was 28 minutes. The mobile phase carried the buffer A: 1 mM Ammonium formate in 1% MeOH in Water (pH: 8) and Buffer B: 100% Acetonitrile. The mobile phase contains Buffer (A): 1 mM Ammonium formate in 1% MeOH in Water (pH: 8) and Buffer B: 100% Acetonitrile. Q Exactive, Thermo Scientific was used for Mass spectrometry (MS) detection, and ESI mode was used for Ionization with Probe Heater Temp: 200 °C, Capillary Temperature: 275 °C, Capillary voltage: (+) 4.0 kV, Aux Gas flow rate (arbitrary unit): 10, S-Lens RF Level: 50, Sheath Gas flow rate (arbitrary unit): 30, Sweep Gas flow rate: 0, Scan Range: 80-1100 m/z, Resolution: 70,000. Thermo Fisher Scientific Compound Discoverer 3.3 was used for the identification and investigation of the bioactive compounds.16
Determination of Total Flavonoid Content (TFC)
An aluminium chloride colorimetric assay was performed for the determination of TFC as reported by Shraim et al.,17 with modifications. The quercetin (1 mg/mL) was prepared in methanol. Reagents used such as sodium nitrate (5% w/v), aluminium chloride (2% w/v), and 1M Sodium hydroxide. The 100 µl of the different concentrations of quercetin solution (200 to 1000 µg/mL) were assorted with 30 µl sodium nitrate (5% w/v). The solution was incubated for 5 min followed by the addition of 30 µl aluminium chloride (2% w/v). After 6 min incubation, 200 µl Sodium hydroxide with a concentration of 1 Molar was mixed, and distilled water was blended to attain its final volume of 1 mL. Absorbance was measured at 510 nm by the ELISA (Thermo Scientific), using distilled water as blank. The TFC in the fungal broth extract was measured using a standard plot of quercetin concentration versus absorbance (A510 nm). Every analysis was carried out twice.
Quantification of tryptophan in dried Extract by UPLC
The Ultra High-Pressure Liquid Chromatography (UPLC) imposed quantify of Tryptophan from dried extract was achieved using Waters Acquity UPLC, in Central Instrument Facility, South Delhi campus, New Delhi, India. BEH C18 Column with dimension 1.7 µm, 2.1 mM x 100 mM was used. The sample was filtered through a 0.2 µm filter and a 5 µl sample was injected into the column. The solvent system used 55:45 (5 mM Ammonium Formate in water: Methanol). The flow rate of the mobile phase was 0.3 mL/min. PDA was the detector and the wavelength was 276 nm. Tryptophan (Himedia Pvt. Ltd., India) was used as a standard.1
Determination of IAA by UPLC
UPLC analysis of dried extract Isolated Aspergillus flavus, was conducted using a C18 column model. The dried extract was dissolved in 1 milliliter of HPLC-grade methanol and subjected to chromatographic separation at room temperature. In this method, Waters Acquity UPLC was used with BEH C18 (100 mM × 2.1 mM), a mobile phase consisting of Solvent A:B:- 40:60 (0.01% Glacial acetic acid in water: Acetonitrile), particle size 1.7 µm, Column Temp.: 35. Detector Used: PDA el Detector, Wavelength: 265 nm, Run time: 5 mins. was employed. The flow rate of the mobile phase was maintained at 0.3 mL/min, with an injection volume of 1 µl of the respective sample. The generated peak was compared with the retention time of authentic IAA. Indole-3-acetic acid (Central Drug House (P) Ltd.) was used as a standard.18
Determination of antioxidants activity
The dried extract of isolated fungi was further used for its antioxidant activity and it was quantified by using the 2,2-diphenyl-2-picrylhydrazyl (DPPH) as reported previously, by Zhang et al.,5 with modifications. Briefly, the DPPH solution was prepared in methanol (900 µl) and mixed with a volume of 100 µl of extract. The solutions were mixed by vortexing and incubated for 30 minutes at room temperature. The absorbances were measured at 517 nm. The extract’s antioxidant activity was measured using a standard plot of ascorbic acid concentration versus DPPH scavenging activity.
Percentage inhibition of free radical DPPH was calculated using equation 1, where AC is the absorbance of the control and AS is the absorbance of the sample.5
Drug-likeness analysis of bioactive compounds
The bioactive compounds from LC-MS were the predictive study of pharmacokinetics especially Absorption, Distribution, Metabolism, and Excretion (ADME) drug-likenesses, bioavailability, and medicinal chemistry of ligands were performed with the help of the Swiss ADME tool developed by Daina et al.19 Identified compounds from LC-MS their canonical simplified molecular-input line-entry system (SMILES) string was taken from PubChem database (http://pubchem.ncbi.nlm.nih.gov/) it was subjected to tools for computational simulation. Swiss ADME tool predicts bioavailability radar works on six physicochemical properties including lipophilicity, flexibility, size, saturation, solubility, and polarity, to detect drug-likeness. For further information refer to Daina et al.19
Statistical analysis
In this investigation, experiments were conducted twice, and the obtained outcome illustrates the average of the observations. The statistical analysis was performed for hypothesis testing using a T-test, wherever necessary.
The isolated fungus was morphologically characterized by the Indian Type Culture Collection (ITCC), New Delhi, India. It was found as Aspergillus flavus. Further molecular identification of the isolate was performed using Polymerase chain reaction amplification of the ITS region from the genomic DNA followed by sequencing further validating the isolated fungus as Aspergillus flavus (Accession No. OR388564). The homology analysis was carried out by employing the BLAST (https://blast.ncbi.nlm.nih.gov/Blast.cgi) revealed around 98.45% similarity with the Aspergillus flavus. The phylogenetic tree was constructed through the neighbour-joining method from a sequence achieved. It confirmed the species-level identification with Aspergillus flavus (Figure 1).
Gas chromatography-mass spectrometer (GC-MS) Profiling of Volatile Constituents
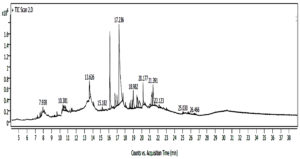
Gas chromatography-mass spectrometry (GC-MS) is a robust technique for profiling of the volatile constituents using the resolution peak profile of the same and analysis using mass fragmentations, peak area, retention times, and molecular weights. The GC-MS analysis (Figure 2) showed the existence of bioactive compounds with their retention time, molecular formula, and biological activity Table 2. The diversity of compounds, detected using GC-MS analysis, revealed organic compounds that may be explored for their potential therapeutic or other associated bioactivities.
Table (2):
Characteristic properties of the compound found in broth GC-MS analysis
No. |
PubChem ID |
Name of Compounds |
Retention time (Minutes) |
Area % |
Molecular Formula |
Activity (based on cited literature) |
Ref. |
|---|---|---|---|---|---|---|---|
1 |
548318 |
Ethanethiol, 2-(diethylboryloxy) |
7.938 |
2.43 |
C6H15BOS |
Antioxidants |
20 |
2 |
571719 |
1,3,2- Dioxaphosphorinane-2- methanol, 2-oxo-alpha.-phenyl |
10.381 |
3.63 |
C10H13O4P |
– |
|
3 |
546203 |
2-Azido-2,4,4,6,6- pentamethylheptane |
13.626 |
11.21 |
C12H25N3 |
Anti- inflammatory Antimicrobial |
21,22 |
4 |
543892 |
trans-2-methyl-4-n-pentylthiane, S,S-dioxide |
17.236 |
100 |
C10H20O2S |
Antimicrobial, Antioxidants, Anticancer |
23 |
5 |
5375934 |
Iron, tricarbonyl- 1,4,5,6-.eta.-4-3- methylhex-4-ene-1,6- diyl-(exo)-carboxylic acid, methyl ester |
21.391 |
18.22 |
C12H14FeO5 |
– |
|
6 |
584098 |
1-[4-Chlorophenyl]-N-[3-[1-pyrolidinyl]propyl]-1H-tetrazol-5-amine |
22.123 |
3.01 |
C14H19ClN6 |
– |
|
7 |
584098 |
Phosphonic acid, 2-methylenecyclopropyl-, diethyl ester |
25.03 |
1.91 |
C8H15O3P |
– |
|
8 |
5306184 |
Piperidine, 1-(1-phenyl-1H-1,2,3,4-tetrazol-5-yl) |
26.44 |
1.67 |
C12H15N5 |
– |
|
9 |
250036013 |
2-Ethylcyclohexylamine, N-(2-chloropropylidene)-, N-oxide |
18.892 |
4.91 |
– |
Figure 2. Chromatogram of Gas chromatography-mass spectrometer of extract of isolated fungi Aspergillus flavus
Liquid chromatography-mass spectrometry (LC-MS) profiling of bioactive constituents
The extract derived solely from Aspergillus flavus possesses a significant amount of flavonoids and exhibits good antioxidant activity. Analysis of chemical constituents of the dried extract of Aspergillus flavus using LC-MS revealed catalogues of organic metabolites. It is important to discuss that the control (uninoculated broth) used in the current study has not shown any match with the metabolite profile. The LC-MS results tabulated in Table 3 showed the molecular formula, retention time, and molecular weight of bioactive compounds, and Figure 3, shows the chromatogram of dried extract. The identified compounds in LC-MS exhibited diverse bioactivities including their role as scavengers of hydroxyl radicals, antibacterial activity, for the treatment of asthma and treatment of canine tegumentary leishmaniasis, along with treatment of total joint arthroplasty (TJA). The role of fungal metabolites, as therapeutic and commercial natural metabolites have gained significant relevance in the last few decades. However, the uniqueness of some of the proposed metabolites, as detected using LC-MS analysis further obtrudes the quest for their characterization and purification. Furthermore, the in silico pharmacokinetic analysis, performed using Swiss ADME revealed that most of the detected compounds follow the Lipinski rule, suggesting their possible exploration as drug targets for diseases.
Table (3):
LC–MS analysis of the compounds found in the dried culture broth of Aspergillus flavus
No |
Groups name |
Identified compound |
PubChem ID |
RT |
Area Max |
Molecular Formula |
Activity |
References |
|---|---|---|---|---|---|---|---|---|
1 |
– |
1,1-Dichloroethylene-d2 |
12233278 |
0.847 |
1657511641 |
C2Cl2D2 |
– |
|
2 |
Organofluorine |
5-Fluoropentyl thiocyanate |
69577 |
0.876 |
1468506233 |
C6H10FNS |
– |
|
3 |
– |
NP-016455 |
86178349 |
1.405 |
1343268575 |
C11H18N2O4 |
– |
|
4 |
Amino Acids and Derivatives |
D-(+)-Tryptophan |
9060 |
1.464 |
627167020.3 |
C11H12N2O2 |
Precursor of IAA |
24 |
5 |
Monosaccharides |
D-(-)-Mannitol and Derivatives |
6251 |
1.007 |
1277650586 |
C6H14O6 |
• Apply in medicine for • helps in brain oedema and reduces dye osmotic diuretic • Hypertonic Mannitol is used for life-threatening brain diseases • Inhaled the mannitol helps to increase hydration and surface properties of sputum of patients have cystic fibrosis • Mannitol hexanitrate used for cure of Hypertention • Mannitol used for scavenger of hydroxyl radicals |
25 |
6 |
– |
1-Pentofuranosyl-2,4(1H,3H) -pyrimidinedione |
312160 |
1.167 |
919433823.1 |
C9H12N2O6 |
– |
|
7 |
Organofluorine |
Diethyl(ethynyl)fluorosilane |
71398188 |
1.372 |
832424230.2 |
C6H11FSi |
– |
|
8 |
Organofluorine compounds |
3-Fluoropropyl thiocyanate |
67893 |
0.968 |
671403636.2 |
C4H6FNS |
– |
|
9 |
Methylamino group |
1-Deoxy-1-(methylamino) hexitol |
4049 |
1.464 |
627167020.3 |
C7H17NO5 |
• Treatment of Canine tegumentary leishmaniasis |
26 |
10 |
Amino Alcohol |
Pirbuterol |
4845 |
6.007 |
382457346.7 |
C12H20N2O3 |
• Treatment of asthma |
27 |
Figure 3. Liquid chromatography-mass spectrometry chromatogram of dried extract of isolated fungi Aspergillus flavus
Total Flavonoid content (TFC)
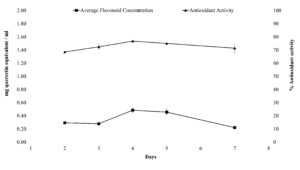
The Total Flavonoid Content (TFC) in the dried broth extract from the fungi showed a consistent yield of around 0.36 mg/mL. The quantification of total flavonoid content in the dried material with the help of the standard graph of an aluminum chloride colorimetric assay. The analysis was performed for 7 days and showed the yield to range between 0.2-0.6 mg Quercetin equivalent/mL. Time-dependent analysis of total flavonoid content and antioxidant activity in the dried culture broth over 7 days in yeast extract peptone dextrose (YEPD) medium (Figure 4).
Figure 4. Time-dependent analysis of total flavonoid and antioxidant activities in the dried culture broth, performed for 7 days in Yeast extract peptone dextrose growth media
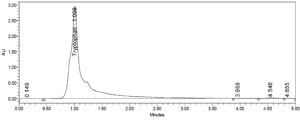
UPLC analysis of tryptophan
The dried broth extract of Aspergillus flavus yielded approximately 1.2 mg/mL of tryptophan. Tryptophan quantification in the dried broth material was conducted using a standard graph method. The calibration curve was constructed using five different concentrations of standard solution, ranging from 50 µg/mL to 250 µg/mL. Figure 5 and tryptophan in dried culture.
UPLC analysis of Indole-3-acetic acid
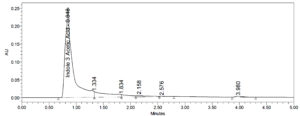
Indole-3-Acetic Acid from the dried extract of isolated Aspergillus flavus was identified and quantified by the UPLC and the yield has exhibited a yield of approximately 0.41 mg/mL of 1AA. IAA quantification was conducted using a standard graph method. The calibration curve was constructed using five different concentrations of standard solution, ranging from 50 µg/mL to 250 µg/mL. The Figure 6 represents the standard of IAA and IAA in dried culture respectively.
Antioxidant activity
The extract of isolated fungi Aspergillus flavus was analyzed for DPPH free radical scavenging potential. The analysis is to augment the possible role of flavonoids as potential antioxidant molecules. In the assay, the DPPH was reduced in occupancy of hydrogen-donating antioxidants by the development of a nonradical formation of DPPH-H. The time-dependent study showed the antioxidant potential in the extracts from the culture. It was further observed that on the fourth day of the fermenter study, the extract had the highest antioxidant potential (76.5%).
Drug-likeness analysis of bioactive compounds
The bioactive compounds of LC-MS were further analysed by the Swiss ADME tool. Swiss ADME tool suggested data on the different parameters such as pharmacokinetics, physicochemical characteristics, solubility, drug lipophilicity, medicinal chemistry, and likeness of compounds. Table 4 shows Blood-Brain Barrier Permanent, GI Absorption, Permeability glycoprotein substrate, cytochromes P450 (CYP1A2) inhibitor, cytochromes P450 (CYP2C19) inhibitor, cytochromes P450 (CYP2D6) inhibitor, Log Kp (skin permeability coefficient), and cytochromes P450 (CYP3A4) inhibitor of compounds detected in LC-MS. Table 5 shows the Lipinski rules and Bioavailability Score of the compounds detected through LC-MS analysis.
Table (4):
Pharmacokinetic properties of bioactive compounds predicted using SwissADME
S. No |
Groups Name |
Compound Name |
PubChem ID |
GI Absorption |
Blood-Brain Barrier (BBB) Permeant |
Permeability glycoprotein (P-gp) substrate |
CYP1A2 Inhibitor |
CYP2C19 Inhibitor |
CYP2D6 Inhibitor |
CYP3A4 Inhibitor |
Log Kp |
|---|---|---|---|---|---|---|---|---|---|---|---|
1 |
1,1-Dichloroethylene-d2 |
12233278 |
Low |
No |
No |
No |
No |
No |
No |
-5.24 cm/s |
|
2 |
Organofluorine |
5-Fluoropentyl thiocyanate |
69577 |
High |
Yes |
No |
No |
No |
No |
No |
-5.64 cm/s |
3 |
NP-016455 |
86178349 |
High |
No |
No |
No |
No |
No |
No |
-7.56 cm/s |
|
4 |
Monosaccharides and Derivatives |
D-(-)-Mannitol |
6251 |
Low |
No |
No |
No |
No |
No |
No |
-9.61 cm/s |
5 |
1-Pentofuranosyl-2, 4(1H,3H)-pyrimidinedione |
312160 |
High |
No |
No |
No |
No |
No |
No |
-9.06 cm/s |
|
6 |
Organofluorine |
Diethyl(ethynyl)fluorosilane |
71398188 |
Low |
Yes |
No |
No |
No |
No |
No |
-4.99 cm/s |
7 |
Organofluorine compounds |
3-Fluoropropyl thiocyanate |
67893 |
High |
Yes |
No |
No |
No |
No |
No |
-5.87 cm/s |
8 |
methylamino group |
1-Deoxy-1-(methylamino) hexitol |
4049 |
Low |
No |
No |
No |
No |
No |
No |
-9.51 cm/s |
9 |
Amino Acids and Derivatives |
D-(+)-Tryptophan |
9060 |
High |
No |
No |
No |
No |
No |
No |
-8.30 cm/s |
10 |
Amino Alcohol |
Pirbuterol |
4845 |
High |
No |
Yes |
No |
No |
No |
No |
-7.81 cm/s |
Table (5):
Drug-likeness properties of bioactive compounds predicted using SwissADME
S. No. |
Groups Name |
Compound Name |
Lipinski’s Rule of Five |
Bioavailability |
|---|---|---|---|---|
1 |
– |
1,1-Dichloroethylene-d2 |
Yes |
0.55 |
2 |
Organofluorine |
5-Fluoropentyl thiocyanate |
Yes |
0.55 |
3 |
– |
NP-016455 |
Yes |
0.56 |
4 |
Monosaccharides and Derivatives |
D-(-)-Mannitol |
No |
0.55 |
5 |
1-Pentofuranosyl-2,4(1H,3H)- pyrimidinedione |
Yes |
0.55 |
|
6 |
Organofluorine |
Diethyl(ethynyl)fluorosilane |
Yes |
0.55 |
7 |
Organofluorine compounds |
3-Fluoropropyl thiocyanate |
Yes |
0.55 |
8 |
methylamino group |
1-Deoxy-1-(methylamino)hexitol |
No |
0.55 |
9 |
Amino Acids and Derivatives |
D-(+)-Tryptophan |
Yes |
0.55 |
10 |
Amino Alcohol |
Pirbuterol |
Yes |
0.55 |
The quest to explore novel biomolecules, whether natural or nature-derived, has led to a paradigm shift towards an exploration of a diverse catalog of natural microflora, as a potential contributor of metabolites that may be characterized as a potential lead molecule or drug associated with diverse human needs. The recent decade has witnessed growing research, focused on the isolation and characterization of different fungi from assorted habitats and the exploration of their metabolome profile is the quest towards the isolation and characterization of new and efficacious biomolecules. The diversity observed among fungal isolates from different habitats is significant due to concomitant changes in the pool of metabolites produced, as reported previously.2,28,29 In the current study, one such agricultural field isolate, characterized by morphological and molecular identification techniques, Aspergillus flavus, was screened for potential metabolites, their characterization, and bioactivity assessment for therapeutic, its role in plant growth, and commercial benefits. The metabolites produced by fungi have been segregated into volatile and non-volatile organic metabolites. The volatile metabolites produced by fungal isolates are significantly important components of the fungal metabolome.12 The fungi are well known for enhancing host plant growth and yield by producing bioactive compounds that facilitate interactions between plants and endophytes.30 Studies have also explored the isolated organisms as potential sources of amino acids.12 The diverse pool of amino acids has been isolated and purified from fungal isolates, including glutamic, threonine, methionine, and tryptophan.31 Most likely, all fungi rely heavily on amino acids as a primary nutritional source.32 Tryptophan is well-known as the precursor of Indole-3-acetic acid.33 Tryptophan is one of the important amino acids used in human beings and animals. Tryptophan also plays a crucial role in protein synthesis and regulates various physiological mechanisms. It serves as a precursor for important compounds such as the neurotransmitter serotonin and the vitamin niacin. Tryptophan is generally considered safe for consumption, and it offers potential health benefits.34 It is crucial for human and animal health, and serves as a valuable supplement in the food, feed, and pharmaceutical industries, promoting better sleep quality and mood enhancement. Being an essential aromatic amino acid, it has found extensive use in medicine, food, and animal feed applications.35 As a result, research attention has intensified on developing efficient production technologies. Among various methods such as chemical synthesis, enzyme conversion, and microbial fermentation, microbial fermentation stands out due to its utilization of cost-effective raw materials, generation of high-purity products, and straightforward extraction processes, making it the preferred method for industrial tryptophan production.35 Tryptophan was found in dried culture broth 1.2 mg/mL and previous studies reported the production of tryptophan from E. coli.35 To the best of our literature review, limited fungi are reported for the production of tryptophan.31 The diversity of secondary metabolites found in endophytic fungi boosts plant growth by secondary metabolites such as ammonia and plant hormones, with a particular emphasis on Indole-3-acetic acid.36 Plants and microorganisms have the potential role of developing the auxin which promotes plant growth and development. Various physiological processes including, tissue differentiation, responses to gravity and light, and cell division have been reported from primary auxin such as Indole-3-acetic acid (IAA).37 In this investigation, isolated fungi underwent Indole-3-acetic acid analysis and it was found that 0.41 mg/mL of 1AA in the dried culture broth. The yield of IAA was comparable to most of the previous reports on IAA production using fungal isolates.38 The GC-MS examination of the fungal dried extract indicated the existence of variation bioactive composites in the extract. Most of the compounds have already been explored and reported to exhibit bioactivities composites including antimicrobial, antioxidant, anticancer, and anti-inflammatory activities along with others. The results showed variations from the GC-MS profile of Aspergillus flavus, reported in other studies.12 Further analysis using LC-MS also suggested the catalogs of metabolites to be distinct from previous studies. This maybe attributed to adaptive and acclimatization changes in the organisms. The antioxidant activity of the extract was further characterized. Finding natural antioxidants for use in preventative medicine that have few or no side effects has lately regained global interest. Reactive oxygen species (ROS), are the key factor in the expansion of numerous non-infectious diseases including coronary heart disease, hypertension, Alzheimer’s, atherosclerosis, and diabetes. This disease can be prevented by antioxidant chemicals.39,40 Diabetes Mellitus can be prevented by consuming natural antioxidants because utilizing antioxidant compounds removes the reactive oxygen species. The objective is to obtain drugs with both antioxidant and antidiabetic properties.39 The study provided an impetus to an exploration of the role of soil-isolated Aspergillus flavus, as a potential source of naturally derived organic compounds that may be a suitable replacement for synthetic compounds for alleviating diseases, conditions, and symptoms. In a recent study by Bora and Devi29 some of the endophytes, including Aspergillus sp., were compared for the total phenol and total flavonoid contents in different organic and aqueous solvents. Further studies by Patil et al.2 reported a yield of 158.33 mg quercetin/mL from the Aspergillus flavus. The result of this investigation had shown yield of total flavonoid content in the 7-day study to range between 0.2-0.6 mg quercetin equivalent/mL. The diverse abiotic parameters and culture conditions may be pivotal in regulating the flavonoid yield. Further, the analysis of antioxidant activity was observed to be in the range of 65%-75%. Previous reports from Bora and Devi29 had reported changes in the DPPH radical scavenging activity with the change in the extraction solvent. Based on the GC-MS, LC-MS, flavonoid estimation, and antioxidant activity, it may be anticipated that the fungal-derived metabolites may be the potential source of therapeutic activities that may be explored. The studies may be further extended to purify the bioactive compounds and evaluate their bioactivities. Further studies with compound purification and characterization may be pivotal in confirming their potential role.
The diverse demographic conditions, concomitant with the plethora of associated biotic microscopic organisms are awaiting to be explored for their associated benefits. The current study was unique, as the isolated fungi, Aspergillus flavus, was analyzed to be a source of diverse bioactive metabolites using GC-MS and LC-MS analysis. Further, antioxidant analysis revealed significant bioactivity, which is more than some of the previous assessment by other similar isolates. The in-silico analysis obtrude the organic metabolites of fungal isolates as potential drug molecules. The presence of flavonoids opens a new avenue for exploring fungi as an alternative cost-effective source of flavonoids. The study also suggests the fungal broth to be a rich source of tryptophan. The current study will pave the way for exploring fungal metabolites as a suitable alternative to plant metabolites, which are often time and season-dependent.
ACKNOWLEDGMENTS
The authors acknowledge the Jaypee Institute of Information Technology, Noida, for providing all necessary support for the successful execution of the study.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
Both authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This article does not contain any studies on human participants or animals performed by any of the authors.
- Li GR, Cao BH, Liu W, Ren RH, Feng J, Lv DJ. Isolation and Identification of Endophytic Fungi in Kernels of Coix lachrymal-jobi L. Cultivars. Curr Microbiol. 2020;77(8):1448-1456.
Crossref - Patil MP, Patil RH, Maheshwari VL. Biological Activities and Identification of Bioactive Metabolite from Endophytic Aspergillus flavus L7 Isolated from Aegle marmelos. Curr Microbiol. 2015;71(1):39-48.
Crossref - Atiphasaworn P, Monggoot S, Gentekaki E, Brooks S, Pripdeevech P. Antibacterial and Antioxidant Constituents of Extracts of Endophytic Fungi Isolated from Ocimum basilicum var. thyrsiflora Leaves. Curr Microbiol. 2017;74(10):1185-1193.
Crossref - Dos Santos GD, Gomes RR, Goncalves R, et al. Molecular Identification and Antimicrobial Activity of Foliar Endophytic Fungi on the Brazilian Pepper Tree (Schinus terebinthifolius) Reveal New Species of Diaporthe. Curr Microbiol. 2021;78(8):3218-3229.
Crossref - Zhang X, Xu Z, Ma J, Zhou D, Xu J. Phylogenetic Diversity, Antimicrobial and Antioxidant Potential and Identification of Bioactive Compounds from Culturable Endophytic Fungi Associated with Mangrove Bruguiera sexangula (Lour.) Poir. Curr Microbiol. 2021;78(2):479-489.
Crossref - Ashitha A, Radhakrishnan EK, Mathew J. Antibacterial Potential and Apoptosis Induction by Pigments from the Endophyte Burkholderia sp. WYAT7. Curr Microbiol. 2020;77(9):2475-2485.
Crossref - Ribeiro BA, da Mata TB, Canuto GAB, Silva EO. Chemical Diversity of Secondary Metabolites Produced by Brazilian Endophytic Fungi. Curr Microbiol. 2021;78(1):33-54.
Crossref - Murali TS, Suryanarayanan TS, Venkatesan G. Fungal endophyte communities in two tropical forests of southern India: diversity and host affiliation. Mycol Progress. 2007;6(3):191-199.
Crossref - Wani ZA, Ashraf N, Mohiuddin T, Riyaz-Ul-Hassan S. Plant-endophyte symbiosis, an ecological perspective. Appl Microbiol Biotechnol. 2015;99(7):2955-2965.
Crossref - Agrawal S, Bhatt A. Microbial Endophytes: Emerging Trends and Biotechnological Applications. Curr Microbiol. 2023;80(8):249.
Crossref - Awad NE, Kassem HA, Hamed MA, El-Feky AM, Elnaggar MAA, Mahmoud K, Ali MA. Isolation and characterization of the bioactive metabolites from the soil-derived fungus Trichoderma viride. Mycology. 2018;9(1):70-80. doi:10.1080/21501203.2017.1423126. Erratum in: Mycology. 2018;9(4):311.
Crossref - Song F, Geng Q, Wang X, et al. Gas Chromatography-Mass Spectrometry Profiling of Volatile Compounds Reveals Metabolic Changes in a Non-Aflatoxigenic Aspergillus flavus Induced by 5-Azacytidine. Toxins. 2020;12(1):57.
Crossref - Heintz N, Gong S. Growth of Saccharomyces cerevisiae and Preparation of DNA. Cold Spring Harb Protoc. 2020;2020(10).
Crossref - Riaz A, Rasib KZ, Aslam H, Raza S. Molecular Identification of Aspergillus flavus Using Inter Transcribed Spacers (ITS). Biotechnol J Int. 2018;20(4):1-8.
Crossref - Farhat H, Urooj F, Irfan M, et al. Biological Control Potential of Endophytic Fungi with Amelioration of Systemic Resistance in Sunflower and GC-MS Metabolic Profiling of Talaromyces assiutensis. Curr Microbiol. 2023;80(2):61.
Crossref - Shaker KH, Zohair MM, Hassan AZ, Sweelam HTM, Ashour WE. LC-MS/MS and GC-MS based phytochemical perspectives and antimicrobial effects of endophytic fungus Chaetomium ovatoascomatis isolated from Euphorbia milii. Arch Microbiol. 2022;204(11):661.
Crossref - Shraim AM, Ahmed TA, Rahman MM, Hijji YM. Determination of total flavonoid content by aluminum chloride assay: A critical evaluation. LWT. 2021;150:111932
Crossref - Szkop M, Bielawski W. A simple method for simultaneous RP-HPLC determination of indolic compounds related to bacterial biosynthesis of indole-3-acetic acid. Antonie Van Leeuwenhoek. 2013;103(3):683-691.
Crossref - Daina A, Michielin O, Zoete V. SwissADME: a free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci Rep. 2017;7:42717.
Crossref - Khan MS, Yusufzai SK, Ying LY, Zulnashriq W. GC-MS based chemical profiling and evaluation of antioxidant potential of leaves and stems of alternanthera sessilis red from sabah, Malaysia. Int J Pharm Pharm Sci. 2018;10(7):4.
Crossref - Peng DQ, Yu ZX, Wang CH, Gong B, Liu YY, Wei JH. Chemical Constituents and Anti-Inflammatory Effect of Incense Smoke from Agarwood Determined by GC-MS. Int J Anal Chem. 2020;2020:1-19.
Crossref - Mohiuddin I, Kumar TR, Zargar MI, et al. GC-MS Analysis, Phytochemical Screening, and Antibacterial Activity of Cerana indica Propolis from Kashmir Region. Separations. 2022;9(11):363.
Crossref - Selvakumar JN, Chandrasekaran SD, Vaithilingam M. Bio Prospecting of Marine-derived Streptomyces spectabilis VITJS10 and Exploring its Cytotoxicity Against Human Liver Cancer Cell Lines. Pharmacogn Mag. 2015;11(issue 44):S469-S473.
Crossref - Wary S, Sarma A, Talukdar R, Tayung K. Leaf endophytic fungi of Cymbidium aloifolium L. produces antimicrobials and indole-3-acetic acid. S Afr J Bot. 2022;149:381-388.
Crossref - Saha BC, Racine FM. Biotechnological production of mannitol and its applications. Appl Microbiol Biotechnol. 2011;89(4):879-891.
Crossref - Lago J, Fraga D, Guimarães LH, Lago T, Santos YdJ, Lago E, et al. Efficacy of intralesional meglumine antimoniate in the treatment of canine tegumentary leishmaniasis: a randomized controlled trial. PLoS Negl Trop Dis. 2023;17(2):e0011064. doi:10.1371/journal.pntd.0011064. Erratum in: PLoS Negl Trop Dis. 2023;17(4):e0011261.
Crossref - Rayees S, Din I. Current Asthma Treatments. In: Asthma: Pathophysiology, Herbal and Modern Therapeutic Interventions. SpringerBriefs in Immunology. Springer, Cham. 2021:19-25.
Crossref - Elgorban AM, Bahkali AH, Al Farraj DA, Abdel-Wahab MA. Natural products of Alternaria sp., an endophytic fungus isolated from Salvadora persica from Saudi Arabia. Saudi J Biol Sci. 2019;26(5):1068-1077.
Crossref - Bora P, Devi NN. Exploration of the chemical constituents and its antioxidant, antibacterial activities of endophytic fungi isolated from the medicinal plant Dillenia indica. Arch Microbiol. 2023;205(2):67.
Crossref - Hassan SE, Hijri M, St-Arnaud M. Effect of arbuscular mycorrhizal fungi on trace metal uptake by sunflower plants grown on cadmium contaminated soil. N Biotechnol. 2013;30(6):780-787.
Crossref - Sahasrabudhe NA, Sankpal NV. Production of organic acids and metabolites of fungi for food industry. Appl Mycol Biotechnol. 2001;1:387-425.
Crossref - Gournas C, Prevost M, Krammer EM, Andre B. Function and Regulation of Fungal Amino Acid Transporters: Insights from Predicted Structure. In: Ramos, J., Sychrová, H., Kschischo, M. (eds) Yeast Membrane Transport. Advances in Experimental Medicine and Biology, vol 892. Springer, Cham. doi: 2016;892:69-106.
Crossref - López-Gómez P, Smith EN, Bota P, et al. Tryptophan Levels as a Marker of Auxins and Nitric Oxide Signaling. Plants. 2022;11(10):1304.
Crossref - Friedman M. Analysis, Nutrition, and Health Benefits of Tryptophan. Int J Tryptophan Res. 2018;11:1178646918802282.
Crossref - Liu L, Bilal M, Luo H, Zhao Y, Iqbal HMN. Metabolic Engineering and Fermentation Process Strategies for L-Tryptophan Production by Escherichia coli. Processes. 2019;7(4):213.
Crossref - Khalil AMA, Hassan SE, Alsharif SM, et al. Isolation and Characterization of Fungal Endophytes Isolated from Medicinal Plant Ephedra pachyclada as Plant Growth-Promoting. Biomolecules. 2021;11(2):140.
Crossref - Itkina DL, Suleimanova AD, Sharipova MR. Pantoea brenneri AS3 and Bacillus ginsengihumi M2.11 as Potential Biocontrol and Plant Growth-Promoting Agents. Microbiology. 2021;90(2):210-218.
Crossref - Patel D, Patel A, Vora D, et al. A resourceful methodology to profile indolic auxins produced by rhizo-fungi using spectrophotometry and HPTLC. 3 Biotech. 2018;8(10):413.
Crossref - Kantari SAK, Biswal RP, Kumar P, Dharanikota M, Agraharam A. Antioxidant and Antidiabetic Activities, and UHPLC-ESI-QTOF-MS-Based Metabolite Profiling of an Endophytic Fungus Nigrospora sphaerica BRN 01 Isolated from Bauhinia purpurea L. Appl Biochem Biotechnol. 2023;195(12):7465-7482.
Crossref - Jeong GJ, Khan S, Tabassum N, Khan F, Kim YM. Marine-Bioinspired Nanoparticles as Potential Drugs for Multiple Biological Roles. Mar Drugs. 2022;20(8):527.
Crossref
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.