ISSN: 0973-7510
E-ISSN: 2581-690X
Salmonella continues to be a significant zoonotic pathogen, with poultry products serving as a major vehicle for its transmission to humans. The intensification of poultry farming in India, along with the widespread and often unregulated use of antibiotics, has led to the rise and spread of antimicrobial-resistant (AMR) strains of Salmonella. This review systematically compiles and analyses published data on the prevalence and antibiotic resistance patterns of Salmonella in Indian poultry from 2000-2025, utilizing a bibliometric approach to place Indian research in the global context. Data from 1,780 original studies, including 35 conducted in India, revealed a striking regional variation in Salmonella positivity, ranging from as low as 0.5% to over 80%, and highlight a worrying trend of multidrug-resistance, particularly to commonly used antibiotics such as tetracycline, ampicillin, and third-generation cephalosporin. Bibliometric mapping further demonstrates that India, despite being a leading poultry producer, lags behind many countries in research output on this topic. The review also critically assesses the potential of antibiotic alternatives, including phytogenic feed additives and probiotics, as sustainable strategies for AMR mitigation in Indian poultry production. The article discusses about ongoing problems like the misuse of antibiotics, farmers not knowing enough, and holes in surveillance systems. It also gives practical suggestions for better regulation, integrated One Health approaches, and the use of proven non-antibiotic treatments. Strengthening research and surveillance, alongside capacity building and policy reform, will be essential to ensure food safety, protect public health, and sustain the growth of India’s poultry industry.
Salmonella, Poultry Industry, India, Bibliometric Mapping, Antimicrobial Resistance, One Health
Salmonella continues to pose a major public health challenge around the world, especially as a cause of foodborne illness. Salmonella are generally divided into two groups: typhoidal and non-typhoidal Salmonella (NTS). In poultry, infections can occur either through specific serovars such as S. Pullorum and S. Gallinarum, which cause a systemic, typhoid-like disease, or through a broader range of non-typhoidal Salmonella strains.1 Among the many sources of Salmonella, poultry and poultry products stand out as key reservoirs, linking animal health directly to human disease. Cross-contamination between meat, workers, and equipment-especially when cleaning and disinfection are inadequate-has been identified as a major risk factor for Salmonella contamination, particularly through chopping boards, knives, and tables.2 In addition, the microbial load found in raw meat is often linked to poor hygiene practices in slaughterhouses and during transportation.3,4 NTS is a concern in poultry because birds can carry the bacteria without showing symptoms, making them hidden sources of contamination through meat, eggs, and the farm environment. Among the many serotypes, S. enterica serovars Enteritidis and Typhimurium are the most significant, being commonly transmitted from animals to humans worldwide.5 In the United States, NTS causes an estimated 1.35 million illnesses, 26,500 hospitalizations, and 420 deaths annually.6 According to the World Health Organization, NTS is one of the four leading global causes of diarrheal disease, contributing to millions of cases each year. In Europe, it remains the second most reported zoonosis, with 91,857 confirmed cases of salmonellosis recorded in 2018 by European Food Safety Authority (EFSA) and European Centre for Disease Prevention and Control (ECDC).7
In India, where poultry farming has expanded rapidly over recent decades, controlling Salmonella is not just a matter of animal health but also of food safety and economic security. Antimicrobial resistance (AMR) has added another layer of complexity to this problem. In poultry farms, antibiotics have long been used not only to treat infections but also to promote faster growth and prevent disease outbreaks. However, frequent and often unregulated use of these drugs has accelerated the emergence of Salmonella strains that are resistant to multiple antibiotics.8,9 This trend is worrying, as resistant bacteria can spread from poultry to humans, limiting treatment options for serious infections and putting both animal and public health at risk. International agencies, including the World Health Organization (WHO) and Food and Agriculture Organization, have identified AMR in food animals as an urgent issue that demands stronger surveillance and coordinated action. The Indian poultry sector deserves special attention in this context. India is currently among the leading global producers of eggs and broiler meat, characterised by a multifaceted business that encompasses extensive commercial farms as well as small-scale backyard enterprises. While this growth has brought affordable protein to millions, it has also exposed gaps in regulation, farm biosecurity, and awareness about responsible antibiotic use. Several Indian studies have reported high rates of Salmonella contamination in poultry products and worrying levels of resistance to important antibiotics. Nevertheless, compared to countries like the United States, China, or Brazil, India’s published research on this topic remains limited, making it harder to design effective national responses.
The selection of non-typhoidal Salmonella for this review is driven by its dual importance as both a leading cause of foodborne disease in humans and a major constraint to poultry production systems. In low- and middle-income countries, including India, NTS infections are frequently underreported, yet they contribute substantially to morbidity, productivity losses, and healthcare costs. Poultry-associated salmonellosis results in economic losses through reduced growth rates, increased mortality, trade restrictions, and costs related to biosecurity and disease control.8,9 Moreover, the public health relevance of this pathogen has been formally recognized at the global level. In 2024, the World Health Organization included non-typhoidal Salmonella in its Bacterial Priority Pathogens List, highlighting its high burden and the growing challenges related to antimicrobial resistance, limited treatment options, and prevention in both human and animal health sectors.10 This designation underscores the urgency of strengthening surveillance and control strategies for Salmonella across the food chain, particularly in countries with rapidly expanding poultry industries such as India.
This review aims to address these gaps by synthesizing current evidence on the prevalence and AMR patterns of Salmonella in Indian poultry, placing these findings within a global context through bibliometric analysis, and evaluating promising alternatives to antibiotics that may reduce drug dependence in the poultry sector. The specific objectives are to determine the regional prevalence trends and resistance profiles of Salmonella in Indian poultry, assess of bibliometric patterns and identify gaps in India’s research output compared with global contributions. Bibliometric analysis offers a quantitative and systematic approach to evaluating research trends, authorship patterns, collaborations, and citation networks, which traditional narrative reviews cannot provide. Unlike conventional reviews that qualitatively summarize findings, bibliometric analysis identifies hotspots, emerging topics, and research gaps across a large body of literature, providing an objective, data-driven overview. This is especially useful for fields where publications are growing quickly, as it lets researchers keep track of how knowledge is changing and decide what areas of research to focus on next. Therefore, the use of bibliometric analysis in this study is novel because it goes beyond summarizing content to reveal structural and temporal patterns in Salmonella research that traditional reviews may overlook. And lastly, evaluation of the potential of non-antibiotic strategies such as probiotics, phytogenics, and other emerging alternatives in poultry production. By addressing these objectives, the review highlights knowledge gaps, provides practical recommendations, and supports the development of evidence-based policies for controlling Salmonella and AMR in India’s rapidly expanding poultry sector.
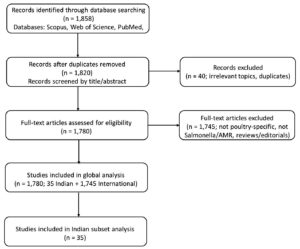
A comprehensive literature search was conducted to gather relevant data on the prevalence and AMR patterns of Salmonella in poultry across India and globally. Scholarly articles were retrieved from search engines Scopus, Google Scholar, and PubMed using primary keywords such as Salmonella, prevalence, poultry, India, broiler chicken, and eggs. The search covered studies published between January 2000 and March 2025, with the final search completed on March 31, 2025. In total, 1,858 records were identified. After removal of 38 duplicates and 40 irrelevant studies, 1,780 peer-reviewed original research articles were retained for analysis. Of these, 35 studies were conducted in India, which were analyzed separately to compare India-specific findings with global trends. To ensure quality, only peer-reviewed original research published in English was included. Eligible studies had to report either (i) prevalence of Salmonella in poultry or poultry products, (ii) AMR patterns in Salmonella isolates, or (iii) data relevant to bibliometric comparisons. Exclusion criteria were: studies not focused on poultry or Salmonella, reviews, editorials, conference abstracts, theses, and articles without original laboratory or field data.
A PRISMA flow diagram (Figure 1) illustrates the study selection process. Given the wide variability in study design, laboratory protocols, and reporting standards, a formal risk-of-bias assessment was not applied. Instead, quality assurance was ensured by restricting inclusion to peer-reviewed original research, and data extraction focused on reported prevalence and resistance outcomes.
To derive deeper insights and identify research trends, a bibliometric analysis was conducted using VOS viewer a powerful text-mining and visualization software developed by Van Eck and Waltman in 2010.11 VOS viewer enables the mapping and clustering of relationships between research objects, such as keywords, authors, publications, citations, and geographic locations. In the visualizations generated, each node represents a unique term or item, while nodal distance signifies the degree of similarity or co-occurrence. The thickness of connecting lines reflects the strength of the linkage between nodes. This analytical approach allowed for the identification of key research themes, geographic trends, and collaborative networks in Salmonella research within India, offering a foundation for meaningful interpretations and future directions. The keyword co-occurrence was analyzed using full counting to identify research themes and trends. In the global dataset (1,780 documents), a threshold of at least 10 keyword occurrences was applied, while in the smaller India-specific dataset (35 documents), the threshold was reduced to 5 to capture relevant terms. For bibliographic coupling, organizations were compared based on citation impact, requiring at least two documents and 10 citations globally, and at least one document and 10 citations for the Indian dataset. At the country level, bibliographic coupling was performed using a stricter threshold of 30 documents and 500 citations, with results visualized according to the number of publications.
Prevalence of Salmonella spp. in Indian Poultry
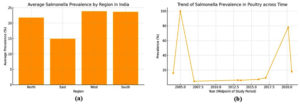
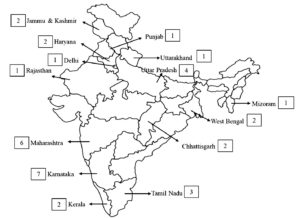
The prevalence of Salmonella spp. in Indian poultry (Table 1, Figures 2a, 2b) demonstrates considerable regional variation, influenced by diverse epidemiological patterns, sources of contamination, and risk factors. In North India, studies have reported a wide range of prevalence rates. For instance, a 4.8% positivity rate was documented in chicken egg samples across states like Uttar Pradesh, Haryana, Punjab, Delhi, and Uttarakhand,12 while a higher prevalence of 9.43% in meat swabs, poultry feces, and environmental samples was observed from the same region.13 In Rajasthan, the prevalence reached 6.3%,14 but an alarming 78% was reported from intestinal content samples in a recent study.15 Chhattisgarh showed particularly high positivity rates in both egg samples (15.5%)16 and swabs/tissues (30%),17 indicating a growing concern in Central India.
Table (1):
Prevalence of Salmonella spp. in Poultry and Associated Sources Across Different Regions of India
| Region | Period of Study (State) | No. of Samples | Sample type | No. of Salmonella Positive Samples | % of Salmonella Positive Samples | Ref. |
|---|---|---|---|---|---|---|
| North India | April 2006 to July 2007 (Uttar Pradesh, Haryana, Punjab, Delhi and Uttarakhand) | 560 | Chicken eggs | 27 | 4.8% | [12] |
| January to August 2017 (Uttar Pradesh, Uttarakhand) | 742 | meat swabs, poultry faeces, environment | 70 | 9.43 | [13] | |
| March 2013 to August 2014 (Rajasthan) | 507 | caecal contents & faecal sample | 32 | 6.3% | [14] | |
| July 2019 to Dec 2020 (Rajasthan) | 151 | intestinal content | 118 | 78% | [15] | |
| August 2020 to July 2021 (Chhattisgarh) | 180 | Eggs | 28 | 15.5% | [16] | |
| Not Mentioned (Chhattisgarh) | 10 | Swab of heart and intestinal contents; tissue pieces of ovary and liver | 03 | 30% | [17] | |
| Not Mentioned (Bareilly) | 720 | Egg, Feed, Drinking water, Faeces, Cloaca | 24 | 3.3% | [31] | |
| Not Mentioned (Haryana) | – | heart blood, liver and bile | 150 | – | [32] | |
| Not Mentioned (Jammu & Kashmir) | 101 | Poultry droppings | 30 | 16.67% | [33] | |
| 2019 to 2022 (Uttar Pradesh, Uttarakhand, Jammu and Kashmir) | 26 | – | – | – | [34] | |
| East India | July to September 2013 (West Bengal) | 360 | cloacal swabs, feed, drinking water, eggs | 22 | 6.1% | [18] |
| Mizoram | 50 | cloacal swabs | 6 | 12% | [19] | |
| Not mentioned (Howrah, Hooghly, Jalpaiguri, Darjeeling, Kolkata) | 150 | cloacal swabs | 40 | 26.7% | [20] | |
| West India | Not Mentioned (Mumbai) |
48 39 |
Chilled Ready-to-cook meat poultry samples Frozen Ready-to-cook meat poultry samples |
25 01 |
52% 2.6% |
[21] |
| December 2015-2017 (Mumbai) | 956 | poultry farm origin, poultry and product processing stages wise and environmental samples | 71 | 7.4% | [22] | |
| August to December 2020 (Shirwal, Maharashtra) | 100 | Faecal Sample Food and water samples |
18 03 |
18% 6% |
[23] | |
| Not Mentioned (Udgir, Maharashtra) | 400 | faecal and cloacal samples, environmental samples, chicken meat, and restaurant meat products | 02 | 0.5% | [24] | |
| Not Mentioned
(Across Mumbai) |
17 | Surface swab from chilled broiler chicken meat from retail poultry shop | 14 | 82.35% | [25] | |
| Oct 2023-Mar 2024 (Marathwada, Maharashtra) | 364 | Cloacal swab, shell waste, feed, water, etc. | 15 | 4.1% | [35] | |
| South India | Not mentioned Salem, Tamil Nadu) |
120 | litter | 10 | 8.3% | [26] |
| Not Mentioned (Kottayam, Kerala) | 600 | Eggs | 02/150 | 1.33% in commercial layer hen | [27] | |
| 77/150 | 51.33% in duck eggs | |||||
| 06/150 | 4% in Backyard raised Layer Hen | |||||
| 31/150 | 20.66% in Commercial Layer Hen | |||||
| January 2016- December 2017 (Manipal) | 396 | poultry meat, intestinal contents and faecal samples | 58 | 14.64% | [28] | |
| Not Mentioned (Bengaluru) | 182 | Swab from different sources | 47 | 25.82 % | [29] | |
| Not Mentioned (Karnataka) | 503 | Environmental sources of poultry wet markets | 09 | 1.8% | [30] | |
| June 2003 to May 2005 (Namakkal, Tamil Nadu) |
578 | chicken meat | 92 | 15.91% | [36] | |
| September-October 2005 (Kerala) | 3 case study | organ samples from liver, spleen, gall bladder and heart blood | 03 | 100% | [37] | |
| Not Mentioned (Mangalore) | 50 | Meat from retail stores | 06 | 12% | [38] | |
| Not Mentioned (Tamil Nadu) |
1215 | Tissue samples Poultry products Environmental samples |
12/392 | 3.06% | [39] | |
| 7/460 | 1.52% | |||||
| 2/362 | 0.53% | |||||
| Not Mentioned (Kolar, Karnataka) |
30 | Tissue samples (livers and ovaries) | 10/30 | 33.3% | [40] | |
| January 2010 to December 2018 (Vellore, Tamil Nadu) | – | – | 826 | – | [41] | |
| Not Mentioned (Karnataka) | 655 | chicken breast meat, eggs, cloacal | 22 | 3.35% | [42] | |
| Not Mentioned (Kanchipuram, Tamil Nadu) | 60 | meat samples | 25 | 42% | [43] |
Figure 2. a) Geographical prevalence of Salmonella in Poultry Birds Across India; b) its trend over the time
In East India, moderate to high prevalence was reported. A study from West Bengal reported a 6.1% positivity rate in cloacal swabs and environmental samples,18 while Mizoram reported 12%.19 Notably, prevalence of 26.7% in cloacal swabs was observed from various districts in West Bengal, suggesting localized hotspots of infection.20
West India revealed some of the highest and lowest prevalence rates. In Mumbai, a 52% prevalence was noted in chilled ready-to-cook meat samples, whereas frozen samples showed only 2.6%.21 A prevalence of 7.4% was reported across various poultry farm and processing stages.22 In Shirwal, 18% positivity was observed in fecal samples and 6% in food and water samples.23 However, only 0.5% prevalence reported in Udgir,24 while an extremely high prevalence of 82.35% was observed in surface swabs from broiler meat in Mumbai’s retail shops, emphasizing contamination risks in urban wet markets.25
In South India, prevalence rates also varied widely. In Tamil Nadu’s Salem region, 8.3% occurrence was reported in litter samples,26 while Kerala’s duck eggs exhibited a 51.33% positivity rate, indicating species-level differences in contamination.27 Manipal showed a significant 14.64% prevalence in poultry meat and fecal samples,28 and Bengaluru recorded 25.82% from various sources.29 Conversely, some studies noted lower prevalence, such as 1.8% in environmental samples from poultry wet markets in Karnataka.30
Across all regions, contamination sources include poultry feed, drinking water, feces, cloacal matter, meat products, and environmental elements such as litter and swabs from processing and retail environments. These findings suggest that Salmonella can persist at multiple points in the poultry production and distribution chain, increasing the risk of cross-contamination. Risk factors influencing Salmonella prevalence include poor biosecurity measures, unhygienic handling of poultry products, inadequate sanitation in live bird markets and processing units, improper storage conditions, and the use of contaminated water and feed. Backyard poultry and duck farming also pose higher risks due to lack of regulated health management. The varied prevalence across India reflects the need for region-specific surveillance, better hygiene practices, and stringent monitoring of poultry production to mitigate public health risks associated with Salmonella infections.
Geographic Heat Map of Salmonella Prevalence in Poultry Birds Across India: Trends and Overview Regional Prevalence of Salmonella in Poultry
Haryana
Haryana has reported a high prevalence of Salmonella in poultry, particularly in broiler flocks. Salmonella Gallinarum was the most common serovar isolated, accounting for 183 out of 253 isolates, followed by Salmonella Enteritidis (41 isolates) and Salmonella Typhimurium (29 isolates).44,45 The prevalence of Salmonella in Haryana has shown a consistent trend over the years, with outbreaks peaking in the fourth quarter (October-December).44
Chhattisgarh
A study examined Salmonella in chicken and chevon meat and discovered an overall 7% incidence in chicken samples in Chhattisgarh, where a significant portion of birds are raised in backyards and small-scale systems with little regular antibiotic supplementation.46 Interestingly, a large number of isolates exhibited resistance to oxytetracycline and erythromycin, which are not commonly found in commercial broiler diets. This suggests that the isolates may have been cross-contaminated from mixed-species holdings or environmental reservoirs of resistance genes. Of the 32 Salmonella isolates, 14 had multiple-drug-resistance indices ≥0.2, highlighting the peculiar and worrisome AMR dynamics in the mixed-production environment of Chhattisgarh.
Patna
Patna has reported a higher prevalence of Salmonella in chicken meat (23.7%) compared to cattle milk (7.7%). Serotyping of isolates revealed the presence of Salmonella Typhimurium, Salmonella Newport, and Salmonella Gallinarum. The study also noted the presence of multiple drug-resistant Salmonella isolates, emphasizing the need for improved hygienic practices in the production and handling of poultry products.47
Other regions
Other regions in India have also reported Salmonella prevalence in poultry, although data is limited. Studies have consistently shown the presence of Salmonella Enteritidis, Salmonella Typhimurium, and Salmonella Gallinarum as the most common serovars across the country.14,45
Serovar distribution and trends
Salmonella enteritidis
Salmonella Enteritidis is one of the most prevalent serovars in Indian poultry (Table 2). It has been isolated from both chicken and chevon meat, with a prevalence of 9% in chicken meat and 7% in chevon meat.14,46 The serovar is known for its ability to cause severe gastroenteritis in humans, making it a significant public health concern.
Table (2):
Regional Prevalence and Serovar Distribution of Salmonella in Poultry Across India
Region |
Prevalence of Salmonella |
Common Serovars |
Ref. |
|---|---|---|---|
Haryana |
High prevalence |
Salmonella Gallinarum, Salmonella Enteritidis, Salmonella Typhimurium |
[44, 45] |
Chhattisgarh |
7% in chicken meat |
Salmonella Enteritidis, Salmonella Typhimurium |
[46] |
Patna |
23.7% in chicken meat |
Salmonella Typhimurium, Salmonella Newport, Salmonella Gallinarum |
[47] |
Other Regions |
Varies by region |
Salmonella Gallinarum, Salmonella Enteritidis, Salmonella Typhimurium |
[14, 39] |
Salmonella typhimurium
Salmonella Typhimurium is another common serovar in Indian poultry. It has been isolated from broiler flocks in Haryana and Patna, with a prevalence of 5.8% in chicken meat and 2.1% in cattle milk.44,47 The serovar is also known for its multidrug-resistance, with isolates showing resistance to ampicillin, tetracycline, and colistin.14,46
Salmonella gallinarum
Salmonella Gallinarum is a serovar specifically adapted to poultry and is a major cause of fowl typhoid. It has been isolated from broiler flocks in Haryana, with a prevalence of 9.12:183 isolates.44,45 The serovar is known for its high morbidity and mortality rates in poultry, with a case-fatality rate of 71.55% in broiler chicks.44
Antibiotic resistance trends
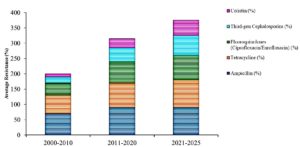
Antibiotic resistance is a significant concern in Salmonella isolates from Indian poultry (Figure 3). Studies have reported high resistance to antibiotics such as ampicillin, tetracycline, and colistin.14,45,46 Multidrug-resistance has also been reported, with isolates showing resistance to multiple antibiotics such as erythromycin, oxytetracycline, and cefotaxime.46,47
Figure 3. Decade-wise illustrative trends in antimicrobial resistance (AMR) of Salmonella enterica in poultry (2000-2024). Values represent approximate resistance levels to tetracyclines, beta-lactams, fluoroquinolones, and cephalosporins, derived qualitatively from narrative descriptions in the manuscript. Percentages are not pooled or statistically calculated but are presented to visualize reported trends: 2000-2010 (moderate resistance, ~40% tetracycline, ~25% beta-lactams, ~15% fluoroquinolones, ~10% cephalosporins); 2011-2020 (increased resistance, ~55%, 45%, 35%, 30% respectively); 2021-2025 (alarming resistance including XDR strains, ~65%, 60%, 55%, 50% respectively)
Public health implications
The prevalence of Salmonella in poultry and poultry products poses a significant risk to public health. Salmonella Enteritidis and Salmonella Typhimurium are zoonotic pathogens that can cause severe gastroenteritis in humans. The presence of multidrug-resistant isolates further complicates treatment, making it essential to implement effective surveillance and control measures.14,46,47 In India, Salmonella enterica serovar Typhimurium is the dominant cause of NTS infections, accounting for 26.7% of non-invasive cases and over 52% of invasive disease. The second most common serovar is S. Worthington (18.1%). Other frequently isolated serovars include S. Weltevreden (5.8%) and S. Enteritidis (5.9%). AMR) shows concerning trends: resistance to ciprofloxacin (9.8%), ceftriaxone (9.9%), and azithromycin (16.1%) is reported in non-invasive isolates. Multidrug-resistance (MDR) remains a significant issue, detected in about 30.2% of non-invasive and 22.3% of invasive isolates. Overall, the data highlight that India faces a substantial burden of serovar diversity and MDR, with S. Typhimurium and S. Worthington emerging as key public health concerns.48 However, direct evidence of zoonotic Salmonella transmission to humans from contaminated meat or contact with birds remains limited in India. In April 2008, ten cases of gastroenteritis caused by Salmonella Wien were reported at a tertiary hospital in Mangalore. Biochemically similar to S. Paratyphi B or S. Typhimurium, the serovar was confirmed at the Central Research Institute, Kasauli. The likely source was poultry. This appears to be the first reported occurrence of S. Wien in humans in India.49 Another report in January 2010, a foodborne gastroenteritis outbreak occurred among 150 students in a hostel in Pune, India, following consumption of a meal including milk, rice, sprouted pulses, and curd. Eight isolates were identified as multidrug-resistant Salmonella enterica serovar Weltevreden.50 A foodborne outbreak of gastroenteritis occurred among staff and students of a tertiary care hospital in southern India after consuming chicken shawarma. A case-control study among 348 students found a strong association between illness and consumption of chicken shawarma from a specific outlet (OR 121.8; P < 0.001). Stool samples and the implicated food tested positive for Salmonella Enteritidis. The study highlights meat-based shawarma as a potential source of non-typhoidal Salmonella infection and underscores the need for strict food safety practices.51 A 52 year-old male from rural Mysuru, India, developed acute gastroenteritis after contact with cattle and poultry. He was diagnosed with Salmonella enterica serovar Hvittingfoss, marking the first documented case in India.52 The pathogenicity of Salmonella is increasing rapidly, and the true burden of non-typhoidal Salmonella (NTS) infections in India is likely underestimated due to the lack of a comprehensive national surveillance system.
The geographic heat map of Salmonella prevalence in poultry birds across India highlights the regional variations in serovar distribution and antibiotic resistance trends. Haryana, Chhattisgarh, and Patna have reported high prevalence rates, with Salmonella Enteritidis, Salmonella Typhimurium, and Salmonella Gallinarum being the most common serovars. The presence of multidrug-resistant isolates underscores the need for improved hygienic practices, judicious use of antibiotics, and effective surveillance to mitigate the risk of Salmonella transmission to humans.
Salmonella load is significantly reduced by commercial pelleting procedures (usually 80-90 °C for 60-90 sec), although numerous Indian studies still demonstrate its recovery from final feed. For instance, a 7.4% positivity rate was observed in feed and ambient samples from Maharashtra,22 while Salmonella was detected in 6.1% of feed samples combined with cloacal swabs in West Bengal.18 These findings most likely represent post-pelleting recontamination-through contaminated dust, equipment, or storage bins-and, in certain backyard or small-scale mills, the sporadic addition of meat-bone meal (MBM). Following restrictions imposed by the Food Safety and Standards Authority of India (FSSAI), the use of MBM in commercial poultry rations has drastically decreased. However, residual use in unregulated sectors may still be a factor in feed-borne Salmonella.
Antimicrobial resistance patterns
The AMR pattern (Table 3) observed across different regions of North India reveals substantial variations in resistance profiles among Salmonella isolates derived from poultry products, environmental samples, and clinical specimens. A striking finding is the high resistance to commonly used antibiotics, notably colistin (COL) and polymyxin B (PMB), with some isolates-particularly from egg samples, showing 100% resistance.12 Similarly, erythromycin (ERY) demonstrated complete resistance in certain isolates obtained from chicken meat and poultry feces.13 Resistance to penicillin (PEN) and oxacillin (OXA) was also universally reported in multiple studies,14,21 underscoring the diminished efficacy of several frontline antibiotics.
Table (3):
Prevalence, Serotype Distribution, and Antimicrobial Resistance Patterns of Salmonella Isolates from Poultry and Related Sources Across Different Regions of India (2000-2025)
| Zone | Region | No. of Samples | Sample type (positive/total isolate) | Salmonella serotypes (n/N, %) | Antimicrobial Resistance (n/N, %) | Ref. |
|---|---|---|---|---|---|---|
| North India | Uttar Pradesh, Haryana, Punjab, Delhi and Uttarakhand | 560 | Eggs from poultry form (10/260) Eggs from wholesale market (6/150) Eggs from retail market (27/560) |
S. Typhimurium (9/260, 3.46%), S. Africana (1/260, 0.38%) S. Typhimurium (6/150, 4%) S. Lagos (6/560, 1.07%) Rough (4/560, 0.7%) S. II. (1/560, 0.185) |
CEP (4/27, 14.8%), OXY (11/27, 40.7%), AMP (7/27, 26%), PEF (1/27, 3.7%), KAN (2/27, 7.4%), BAC (17/27, 62.96%), NEO (1/27, 3.7%), CIP (0/27, 0%), TET (0/27, 0%), PMB (27/27, 100%), GEN (5/27, 18.5%), SPZ (21/27, 77.8%), STR (0/27, 0%), ERY (22/27, 81.5%), ENR (0/27, 0%), TRI (5/27, 18.5%), AMO (10/27, 37%), COL (27/27, 100%), CHL (4/27, 14.8%), DOC (17/27, 63%), SMZ (18/27, 66.7%) | [12] |
| Uttar Pradesh, Uttarakhand | 742 | Chicken meat (28/188) Poultry faeces (15/214) Environment (27/340) | S. Kentucky (52/70, 74.29%) S. Virchow (12/70, 17.14%) S. Typhimurium (5/70, 7.14%) |
STR (19/70, 27.14%), NA (69/70, 98.57%), CIP (58/70, 82.56%), LEV (35/70, 50%), GAT (57/70, 81.43%), CEF (53/70, 75.71%), CXN (11/70, 15.71%), CTX (36/70, 51.43%), ERY (70/70, 100%), AMP (67/70, 95.71%), TET (70/70, 100%), SFX (33/70, 47.14%) | [13] | |
| Rajasthan | 507 | caecal contents (18/202) faecal sample (14/305) |
S. Enteritidis (9/32, 28%), Untypeable (6/32, 18.75%), S. Typhimurium (5/32, 15.6%) S. Virchow (4/32, 12.5%), S. Gallinarum (3/32, 9.4%) S. Reading (2/32, 6.25%) Rough (2/32, 6.25%) S. Altona (1/32, 3%) |
OXA (32/32, 100%), CTZ (6/32, 18.75), PEN (32/32, 100%), CHL (0/32, 0%), GEM (10/32, 31.25%), LEV (3/32, 9.37%), COL (15/32, 46.87%), NAL (18/32, 56.25%), AMP (22/32, 68.75%), TRI (8/32, 25%), CEF (9/32, 28%), CLI (32/32, 100%), CIP (5/32, 15,6%), CTX (6/32, 18.75%), TET (21/32, 65.6%), KAN (13/32, 40.6%), TIC (12/32, 37.5%), MER (3/32, 9.37%), CTN-SAL (3/32, 9.37%), AZT (4/32, 12.5%), AMK (12/32, 37.5%), PIP (18/32, 56.25%), GEN (6/32, 18.75%), CPM (4/32, 12.5%) | [14] | |
| Rajasthan | 151 | Intestinal content (118/151) | S. Typhimurium (51/118, 43.2%) S. Weltevreden (57/118, 48.3%) Untypable (10/118, 8.5%) |
AMP (82/118, 70%), TET (90/118, 76.27%), CTZ (94/118, 79.66%), CTN (75/118, 63.35%), CIP (86/118, 73.33%), AMK (51/118, 43.33%), GEN (59/118, 50%); AZI (71/118, 60%) | [15] | |
| Chhattisgarh | 180 | Commercial chicken eggs (20/60) Backyard chicken eggs (11/60) Duck eggs (5/60) |
–
|
AMP (11/28, 39.28%), CTX (14/28, 50%), CEP (6/28,21.42%), CIP (1/28, 3.57%), GEN (0/28, 0%), OXY (7/28, 25%) | [16] | |
| Chhattisgarh | 10 | Swab of heart and intestinal contents; tissue pieces of ovary and liver | S. Gallinarum (3/10, 30%) | CHL, AMP, CAZ, CXM, CPM, AZI, NA, TET, OXY, STR | [17] | |
| Bareilly | 720 | Chicken Egg (8/180) Feed (3/120) Drinking water (4/120) Faeces (3/120) Cloacal swab (8/180) |
S. Typhimurium (6/180, 3.3%), S. Senftenberg (1/180, 0.56%) and S. Kottbus (1/180, 0.56%) S. Kottbus (1/120, 0.8%), S. Typhimurium (1/120, 0.8%) and S. II (1/120, 0.8%) S. Kottbus (2/120, 1.7%) and S. Typhimurium (2/120, 1.7%) S. Kottbus (2/120, 1.7%) and S. II (1/120, 0.8%) S. Typhimurium (5/180, 2.8%), S. Kottbus (2/180, 1.1%) and S. II (1/180, 0.56%) |
CLI (26/26, 100%), CHL (6/26 6, 23.08%), CIP (3/26, 11.5%), OXA (26/26, 100%), GEN (2/26, 7.69%), PEN (26/26, 100%), AMP (0/26, 0%), NIT (6/26, 23.08%), CTZ (6/26, 23.08%), STR (3/26, 11.5%), TET (6/26 6, 23.08%), VAN (26/26, 100%) | [31] | |
| Haryana | 150 | Heart blood Liver Bile |
Salmonella Gallinarum (126/150, 84%) Salmonella Enteritidis (15/150, 10%) Salmonella Typhimurium (9/150, 6%) |
AMK (27/150, 18%), AMP (27/150, 18%), AMP–SAL (20/150, 13%), CTZ (26/150, 17%), CIP (26/150, 17%), CHL (15/150, 10%), CTX (30/150, 20%), CTN (32/150, 21%), ENR (21/150, 14%), CAR (84/150, 56%), NA (102/150, 68%), NOR (78/150, 52%), TET (23/150, 15%), SFZ (21/150, 14%), KAN (21/150, 14%), GEN (21/150, 14%), CPZ (29/150, 19%), AMC (27/150, 18%) | [32] | |
| Jammu & Kashmir | 101 | Poultry droppings (19/30, 16.67%) | S. Kentucky (3/19, 15.8%) S. Infantis (5/19, 26.3%) S. Agona (4/19, 21%) S. Typhimurium (7/19, 37%) |
AMP (19/19, 100%), CPZ (2/19, 10.53%), CTX (3/19, 15.79%), CTN (3/19, 15.79%), CHL (5/19, 26.31%), CIP (7/19, 36.84%), DOX (18/19, 94.74%), GEN (5/19, 26.31%), NA (14/19, 73.68%), TET (19/19, 100%) | [33] | |
| Uttar Pradesh, Uttarakhand, Jammu and Kashmir | 26 | – | S. Typhimurium (10/26, 38.5%) S. Infantis (5/26, 19.2%) S. Virchow (3/26, 11.5%) S. Kentucky (4/26, 15.4%) S. Agona (4/26, 15.4 %) |
LEV (7/26, 26.9%), CIP (6/26, 23.1%), NIT (6/26, 23.1%), TET (17/26, 65.4%), NA (16/26, 61.5%), KAN (8/26, 21%), AMC (3/26, 11.5%), COT (3/26, 11.5%), TRI (9/26, 34.6%) | [34] | |
| East India | West Bengal | 360 | cloacal swabs (6/40) feed (4/40) drinking water (8/40) eggs (4/40) |
Salmonella sp. (22/360, 6.1%) S. Enteritidis (6/22, 27%) S. Typhimurium (2/22, 9%) |
CHL (100%), CIP (100%), GEN (100%), LEV (100%), NOR (100%), OXY (100%), TET (100%), CTZ (75%), ERY (75%) | [18] |
| Mizoram | 50 | cloacal swabs (6/50) | S. Virchow (2/6, 33.3%) S. Typhimurium (4/6, 66.7%) |
CAZ (5/6, 80%), CTN (5/6, 80%), CXM (4/6, 70%), CTX (4/6, 70%), GEN (4/6, 70%), CTZ (4/6, 60%), AMP (4/6, 60%) | [19] | |
| Howrah, Hooghly, Jalpaiguri, Darjeeling, Kolkata | 150 | Cloacal swabs Backyard Broiler (27/100) Contract Farm Broiler (13/50) |
Salmonella sp. (40/150, 26.7%) | TET (100%), CHL (100%), DOX (91.3%), CTZ (91.3%), AMP (88.1%), CEC (83.8%), CIP (82%), NOR (82.7%), MOX (81%), ACN (81.7%). | [20] | |
| West India | Mumbai | 48 39 |
Chilled Ready-to-cook meat poultry samples (25/48, 52.1%) Frozen Ready-to-cook meat poultry samples (1/39, 2.56%) |
S. Typhimurium (81) S. Enteritidis (24) S. Weltevreden (2) S. Typhimurium (4) S. Enteritidis (2) |
AMP (38/113, 35.5%), CEP (25/113, 22.1%), NA (94/113, 83.1%), SMZ (73/113, 64.6%), CTC (91/113, 80.5%), KAN (96/113, 85%), OXY (101/113, 89.4%), TRI (61/113, 54%), TET (100/113, 88.5%) | [21] |
| Mumbai | 956 | Poultry farm origin (432) Poultry product processing stages wise (324) Environmental samples (154) | Salmonella spp. (71/956, 7.4%) S. Virchow (20/42, 50%), S. Newport (6/42, 14.3%) S. Typhimurium (5/42, 11.9%) Untypable (11/42, 26.2%) |
GEN (7.14%), AZT (21.43%), CZX (35.71%), AMK (4.76%), AC (4.29%), NOR (26.19%), OXY (97.62%), ENR (14.29%), CIP (19.05%), STR (16.67%), COL (16.67%), CHL (4.76%), CTX (14.29%), AMP (21.43%), NEO (88.10%), ERY (83.33%), TET (78.57%), DOC (100%) | [22] | |
| Shirwal, Maharashtra | 100 | Faecal Sample (18/100, 18%) Food and water samples (3/50, 6%) | NA | NA | [23] | |
| Udgir, Maharashtra | 400 | faecal and cloacal samples, environmental samples, chicken meat, and restaurant meat products | S. Typhimurium (1/400, 0.25%) S. Enteritidis (1/400, 0.25%) |
TET (100%), NAL (100%), AMP (100%), SDZ (100%) | [24] | |
| Mumbai | 17 | Surface swab from chilled broiler chicken meat from retail poultry shop | Salmonella spp. (14/17, 82.35%) | GEN (2/14, 14.28%), NA (9/14, 64.28%), TRI (11/14, 78.57%), TET (13/14, 92.85%), CIP (3/14, 21.42%), CHL (1/14, 7.14%), ERY (8/14, 57.14%), AMP (14/14, 100%), OFL (2/14, 14.28%) | [25] | |
| Marathwada, Maharashtra | 364 | Cloacal swab, shell waste, feed, water, etc. | Salmonella spp. (15/364, 4.1%) | ERY (100%), CAZ (40%) | [35] | |
| South India | Salem, Tamil Nadu | 120 | litter | Salmonella spp. (10/120, 8.3%) | NA | [26] |
| Kottayam, Kerala | 600 | Eggs | S. Weltevreden, S. Worthington, S. Dublin, S. Bareilly, S. Typhimurium, S. Infantis | VAN (100%), NOV (100%), TET (40%), STR (35%), OXY (30%), NA (10%), KAN (10%), AMP (10%), CIP (5%), AMK (1%) | [27] | |
| Manipal, Karnataka | 396 | poultry meat, intestinal contents and faecal samples | S. Infantis (25/58, 43.1%) S. Kentucky (13/58, 22.41%) S. Poona (2/58, 3.45%) S. Kouka (1/58, 1.72%) |
AMP (19/58, 32.8%), COT (17/58, 29.31%), GEN (10/58, 17.24%), AMC (9/58, 15.38%. CIP (42/58, 72.41%) | [28] | |
| Bengaluru, Karnataka | 182 | Swab from different sources | Salmonella (47/182, 25.82%) | DOX (44/47, 94.34%), CPD (40/47, 84.91%), CIP (34/47, 72.64%), GEN (31/47, 65.09%), ENR (29/47, 61.32%), COL (19/47, 40.42%), AMK (16/47, 34.91%), AMP (16/47, 33.96%), NEO (16/47, 33.02%), CTX (14/47, 30.19%), CAZ (14/47, 29.25%), COT (11/47, 23.58%), AMC (10/47, 21.70%), CHL (6/47, 12.26%); ESBL producers (8/47, 16.98%) | [29] | |
| Karnataka | 503 | Environmental sources of poultry wet markets | S. Typhimurium | KAN, PMB, PIP, IPM, Cephalosporins | [30] | |
| Namakkal, TamilNadu | 578 | chicken meat | S. Enteritidis (92/578, 15.91%) | AMP (92/92, 100%), ERY (92/92, 100%), KAN (81/92, 88%), CEP (82/92, 89.1%), NA (53/92, 57.6%), TET (82/92, 89.1%), AMC (40/92, 43.5%), CTZ (37/92, 40.2%), CIP (7/92, 7.6%), GEN (11/92, 12%), CHL (64/92, 70%), AMK (6/92, 6.5%) | [36] | |
| Kerala | 3 case study | organ samples from liver, spleen, gall bladder and heart blood | S. Gallinarum (3/3, 100%) | PEN, ERY, STR, CTZ, CX, AMP | [37] | |
| Mangalore, Karnataka | 50 | Meat from retail stores | Salmonella sp. (6/50, 12%) | TET (6/6, 100%), NA (4/6, 66.7%), NIT (6/6, 100%), MRP (3/6, 50%), CT (3/6, 50%), CIP (1/6, 16.7%), PIT (1/6, 16.7%), GEN (1/6, 16.7%), CTX (2/6, 33.3%), AMP (2/6, 33.3%) | [38] | |
| Tamil Nadu | 1215 | Tissue samples, Poultry products, Environmental samples | Salmonella sp. (21/1215, 1.7%) | OXY (21/21, 100%), ENR (2/21, 10%), NOR (4/21, 19%), CHL (8/21, 38%), AMO (8/21, 38%), CTZ (13/21, 62%) | [39] | |
| Kolar, Karnataka | 30 | Tissue samples (livers and ovaries) | Salmonella sp. (10/33, 10%) | CIP (10/10, 100%), ENR (10/10, 100%), AMO (10/10, 100%), COL (7/10, 70%), DOX (2/10, 20%), NOR (3/10, 30%), KAN (6/10, 60%) | [40] | |
| Vellore, Tamil Nadu | – | – | 826 | CTN (42/826, 5.1%) | [41] | |
| Karnataka | 655 | Breast meat (13/269, 4.83%) Eggs (4/186, 2.15%) Cloacal swab (5/200, 2.5%) | S. Enteritidis (10/269, 3.71%); S. Typhimurium (3/269, 1.11%) S. Enteritidis (2/186, 1.07%); S. Typhimurium (2/186, 1.07%) S. Enteritidis (3/200, 1.5%); S. Typhimurium (2/200, 1%) |
AMP (10/22, 45.5%), AMP-SAL (9/22, 40.5%), CAZ (1/22, 4.5%), CTZ (3/22, 13.6%), PMB (18/22, 82%), COL (13/22, 59.1%), CHL (1/22, 4.5%), CFR (4/22, 18.2%), NA (16/22, 72.7%), STR (2/22, 9.1%), TET (10/22, 45.5%), CIT/Tz (1/22, 4.5%) | [42] | |
| Kanchipuram, Tamil Nadu | 60 | meat samples | Salmonella (25/60, 42%) | Not retrievable of chicken meat sample only | [43] |
STR, Streptomycin; NA, Nalidixic acid; CIP, Ciprofloxacin; LEV, Levofloxacin; GAT, Gatifloxacin; CEF, Cefazolin; CXN, Cefoxitin; CTX, Cefotaxime; ERY, Erythromycin; AMP, Ampicillin; TET, Tetracycline; SFX, Sulfisoxazole; GEN, Gentamicin; OXY, Oxytetracycline; CEP, Cephalexin; CHL, chloramphenicol; CAZ, ceftazidime; CXM, Cefexime; CPM, Cefepime; AZI, Azithromycin; AMK, Amikacin; AMP-SAL, Ampicillin–Salbactum; CTZ, Cotrimoxazole; CTN, Ceftriaxone; ENR, Enrofloxacin; CAR, Carbenicillin; NOR, Norfloxacin; STM, Spectinomycin; SFZ, Sulphafurazole; KAN, Kanamycin; CPZ, Cefoperazone; AMC, amoxicillin/clavulanic acid; CLI, Clindamycin; OXA, Oxacillin; PEN, Penicillin; NIT, Nitrofurantoin; VAN, Vancomycin; CEP, Cephalexin; PEF, Pefloxacin; BAC, Bacitracin; NEO, Neomycin; PMB, Polymyxin-B; SPZ, Sulphaphenazole; TRI, Trimethoprim; AMO, Amoxycillin; COL, Colistin; DOC, Doxycycline; SMZ, Sulphamethizole; CFX, Cefuroxime; GEM, Gemifloxacin; TIC, Ticarcillin; MER, Meropenem; CTN-SAL, Ceftriaxone/Sulbactam; AZT, Aztreonam; PIP, Piperacillin; DOR, Doriprem; COT, Trimethoprim-sulphathoxazole/co-trimoxazole; CEC, Cefotaxime/clavulanic acid; MOX, Moxifloxacin; ACN, ampicillin/cloxacillin; CZX, Ceftizoxime; AXC, Amoxyclav; OFL, Ofloxacin; CEP, Cephalothin; CTC, Chlortetracycline; SDZ, Sulphadiazine; CFR, Cefadroxil; CIT/Tz, Cefriaxone/Tazobactum; CX, cloxacillin; CT, Cotrimazole; IPM, imepenem; MRP, meropanem; PIT, piperacillin/tazobactin; DOX. Doxycylcline; CPD, cefpodoxime; extended spectrum β-lactamase (ESBL); NOV, Novobiocin
Moreover, multidrug-resistance (MDR) and extensive drug resistance (XDR) were frequently observed in specific Salmonella serovars such as Salmonella Typhimurium, Salmonella Kentucky, and Salmonella Weltevreden, which exhibited resistance to multiple antibiotic classes. Several studies highlighted high resistance to β-lactam antibiotics, including ampicillin (AMP), amoxicillin-clavulanic acid (AMC), ceftazidime (CTZ), and cefotaxime (CTX).15,22 Notably, Salmonella Kentucky demonstrated significant resistance to fluoroquinolones such as ciprofloxacin (CIP), levofloxacin (LEV), gatifloxacin (GAT), and nalidixic acid (NA), raising concerns about its role in persistent poultry-related infections.13
Distinct regional variations in resistance patterns were also evident. For instance, isolates from Rajasthan showed elevated resistance to third-generation cephalosporins like ceftazidime (CAZ), cefotaxime (CTX), and ceftriaxone (CEF). In contrast, isolates from Chhattisgarh exhibited a higher prevalence of resistance to macrolides like erythromycin (ERY), azithromycin (AZI) and tetracyclines (TET).16,17 Haryana reported significant resistance to carbapenems in Salmonella Gallinarum and Salmonella Enteritidis strains.32 Emerging resistance to critical antibiotics was especially concerning. Fluoroquinolone resistance was prominently noted in Salmonella Kentucky and Salmonella Typhimurium,13,34 while carbapenem resistance in certain regions points to the rise of extensively drug-resistant Salmonella strains.14,32
In eastern India, particularly West Bengal and Mizoram, several studies have highlighted the increasing role of backyard poultry in the persistence of Salmonella. A 6.1% prevalence was recorded among 360 diverse poultry samples, with universal resistance to ciprofloxacin, tetracycline, and chloramphenicol, indicating long-standing drug pressure.18 In Mizoram, Salmonella Typhimurium and Salmonella Virchow was detected in 50 cloacal swabs from Mizoram, with 80% of isolates resistant to third-generation cephalosporins, pointing towards severely limited treatment options in these semi-urban settings.19 A comprehensive study in multiple districts of West Bengal found Salmonella in 26.7% of 150 cloacal swabs from backyard and contract broilers.20 Nearly all isolates showed resistance to tetracycline and chloramphenicol, with over 80% also resistant to fluoroquinolones and cephalosporins. These findings highlight the potential risk of community-acquired Salmonella infections emanating from unregulated backyard poultry systems.
The western region, notably Maharashtra, has also shown a high burden of MDR Salmonella. Gautam et al. reported that chilled and frozen poultry samples from Mumbai were heavily contaminated with S. Typhimurium and S. Enteritidis, with nearly 90% resistance to tetracyclines and nalidixic acid, posing a significant food safety hazard in urban centers.21
Similarly, Salmonella was found in 7.4% of nearly 1,000 samples across the poultry production chain, with 83.3% resistant to erythromycin and 78.5% to tetracycline.22 These trends underscore the risk of MDR Salmonella infiltration into consumer supply chains. Data from Shirwal,23 Udgir,24 and Marathwada35 reinforce this threat, with resistance rates approaching 100% for ampicillin, tetracycline, and sulfadiazine. Alarmingly, it was also noted the presence of S. Enteritidis and S. Typhimurium in retail settings, indicating poor post-harvest sanitation.24
The recent study from Mumbai further confirmed these trends, with 82.35% of surface swab samples testing positive for Salmonella, and 100% resistance to ampicillin, alongside 92.85% resistance to tetracycline.25 These findings signal a dire need for enhanced regulatory oversight and farm-to-fork interventions.
The southern states of India, notably Tamil Nadu, Kerala, and Karnataka, have demonstrated varied patterns of Salmonella contamination across poultry production and retail sectors, accompanied by alarming trends in AMR. In early surveillance from Salem, Tamil Nadu, an 8.3% prevalence of Salmonella was reported from poultry litter, emphasizing the environmental persistence of the pathogen.25 More concerning data came from Namakkal, a major poultry hub, where S. Enteritidis was identified in 15.91% of 578 chicken meat samples.36 These isolates exhibited 100% resistance to ampicillin and erythromycin, and over 88% resistance to kanamycin, tetracycline, and cephalexin, underscoring the endemicity of multidrug-resistance in commercial broiler chains.
In Kerala, the situation was no better, diverse serotypes was found including S. Weltevreden, S. Worthington, and S. Dublin in eggs, with all isolates showing complete resistance to vancomycin and novobiocin. A significant proportion (40%) were also resistant to tetracycline, suggesting widespread antimicrobial usage in layer farms.27 S. Gallinarum was reported in organ samples from diseased poultry, again noting resistance to core veterinary drugs like penicillin and ceftazidime.37 These findings highlight the pathogenic burden of host-specific serotypes in Kerala’s poultry systems.
Karnataka too reflects concerning trends. From meat retail stores in Mangalore, Salmonella was found in 12% of samples, with complete resistance to tetracycline and nitrofurantoin.38 In Kolar, Salmonella was found in 33% of liver and ovary samples from poultry, with 100% resistance to fluoroquinolones like ciprofloxacin and enrofloxacin, both of which are considered critically important in human medicine.40 The widespread presence of Salmonella in tissue and environmental samples was also reported in Tamil Nadu (1.7% prevalence from over 1,200 samples), reflects the complexity of transmission routes and contamination hotspots.39
A broader examination of serotypes was conducted in Manipal, where S. Infantis (43.1%), S. Kentucky (22.41%), and rarer types like S. Poona and S. Kouka were isolated. Fluoroquinolone resistance was again significant, with 72.4% of isolates resistant to ciprofloxacin.28 with Surveillance in Vellore added another dimension, where S. Typhimurium and S. Enteritidis were commonly found in poultry environments, although specific AMR data were limited.41 In environmental samples from Karnataka’s wet markets, identified S. Typhimurium with resistance to a wide range of cephalosporins and carbapenems, pointing towards the probable circulation of extended-spectrum beta-lactamase (ESBL) or carbapenemase-producing strains.30
Further studies confirm that the pathogen is not restricted to high-density farms. In Karnataka, S. Enteritidis and S. Typhimurium was found in breast meat (4.83%), eggs (2.15%), and cloacal swabs (2.5%). These isolates showed high resistance to polymyxin B (82%) and colistin (59.1%), both of which are considered last-resort antibiotics.42 In Bengaluru, recorded a Salmonella prevalence of 25.82% from swabs of different sources, with high resistance to doxycycline (94.3%), cephradine (84.9%), and ciprofloxacin (72.6%). Importantly, 17% of isolates were identified as ESBL producers, signifying the presence of strains with advanced resistance mechanisms.29
The most recent report from Kanchipuram (Tamil Nadu) showed an extremely high prevalence (42%) of Salmonella in meat samples, although detailed AMR profiles were not retrievable. Still, the high detection rate itself indicates severe lapses in meat hygiene and slaughterhouse practices.43
The aggregated data from 2009-2024 paints a concerning picture of Salmonella prevalence and AMR in India’s poultry sector. While serotype distribution varies by region, S. Typhimurium and S. Enteritidis remain dominant across most zones. The emergence of MDR strains such as S. Kentucky and S. Weltevreden in certain pockets warrants close surveillance. The most common antibiotics showing high resistance rates include tetracycline, ampicillin, nalidixic acid, erythromycin, and even third-generation cephalosporins such as cefotaxime and ceftriaxone. These results indicate not only the excessive utilization of antimicrobials in veterinary medicine but also the possibility of zoonotic transmission and therapeutic failure in human healthcare. Coordinated national surveillance, stringent antimicrobial stewardship, and public awareness campaigns are urgently needed to mitigate this escalating public health threat.
Bibliometric perspective of the current status of research in India compared to world
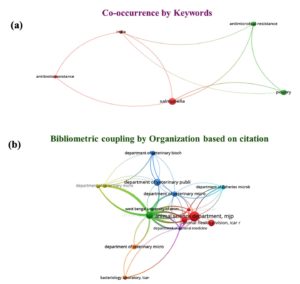
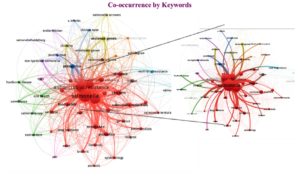
Bibliometric analysis is an effective way to obtain a clear view of the worldwide status of any research area.53 Figure 4 illustrate the bibliometric mapping of several reported publications in the past two decades in India (2000-2025) focussing on co-occurrence and bibliographic coupling by organization where studies were conducted. The mapping reveals interconnected keywords between reported articles, revealing the similarity in searches for articles under the roof that bring Salmonella, antibiotic resistance and poultry for the strong surveillance system to certain food safety. Figure 5 shows the distribution of the total number of collected original studies on occurrence of Salmonella and antibiotic resistance from the poultry sector in India, 2000-2025.
Figure 4. (a) Bibliometric mapping of reports on prevalence of Salmonella in Poultry in India. (b) Bibliometric coupling of organizations working of surveillance of poultry sector in India
Figure 5. Spatial distribution of the number of studies on occurrence of Salmonella and antibiotic resistance from the poultry sector in India, 2000-2025
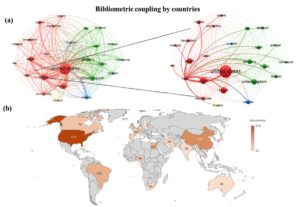
Country-wide bibliometry analysis connecting the coauthorships revealed the names of the leading nations with the maximum reported literature on occurrence of Salmonella in poultry and its antibiotic resistance pattern. Thirteen developed nations contribute about 1142 articles and 12 developing countries contribute 780 articles (Table 4). Among developed countries USA topped the list with 373 documents and developing nations China published maximum 194 documents, followed by Brazil. India lags behind at position 9 with only 35 published documents (Table 4 and Figures 5 & 6). Figures 7 and 8 illustrate the bibliometric mapping of several reported publications in the past two decades in the whole world focussing on co-occurrence and bibliographic coupling by organization where studies were conducted, the mapping was much more complex and intricate due to large of studies as compared to Indian data only.
Table (4):
Distribution of Publications on Salmonella and Antibiotic Resistance in Poultry by Country Classification and Region (2000-2025)
| Country | Documents | Region | |
|---|---|---|---|
| Developed Nations | United States | 373 | North America |
| United Kingdom | 104 | Europe | |
| Canada | 79 | North America | |
| Germany | 75 | Europe | |
| France | 64 | Europe | |
| South Korea | 63 | East Asia | |
| Spain | 58 | Europe | |
| Italy | 53 | Europe | |
| Denmark | 37 | Europe | |
| Belgium | 37 | Europe | |
| Japan | 36 | East Asia | |
| Netherlands | 33 | Europe | |
| Total | 12 | 1142 | |
| Developing Nations | China | 194 | East Asia |
| Brazil | 122 | Latin America | |
| Egypt | 107 | North Africa | |
| Nigeria | 60 | Sub-Saharan Africa | |
| Iran | 49 | Middle East | |
| Poland | 41 | Eastern Europe* | |
| South Africa | 39 | Sub-Saharan Africa | |
| Bangladesh | 39 | South Asia | |
| India | 35 | South Asia | |
| Thailand | 32 | Southeast Asia | |
| Malaysia | 32 | Southeast Asia | |
| Portugal | 30 | Europe* (Emerging) | |
| Total | 12 | 780 |
Figure 6. (a) Bibliometric coupling of reports on prevalence of Salmonella in Poultry by countries; (b) Spatial distribution of the number of studies on occurrence of Salmonella and antibiotic resistance from the poultry sector in India, 2000-2025
Figure 7. Bibliometric mapping of reports on prevalence of Salmonella and its antibiotic resistance pattern in Poultry (World)
Alternative Solutions to tackle AMR challenge
The discovery of the growth-promoting effects of antibiotics led to their incorporation into animal feed as antibiotic feed additives (AFAs) at sub-therapeutic levels. Poultry farming has widely used AFAs for nearly eight decades to enhance growth rates, feed efficiency, and overall productivity. According to estimates by Van Boeckel et al., global antimicrobial consumption in food animal production is projected to reach approximately 105,596 (±3605) tons by 2030.54 While the use of AFAs has brought significant benefits in terms of animal health and performance, it has also posed serious challenges. The continuous and indiscriminate use of these additives has contributed to the emergence and spread of antibiotic-resistant strains of pathogens, notably Salmonella, Campylobacter, and Escherichia coli in poultry. This escalation in AMR, alongside the release of antibiotic residues into the environment, has become a matter of global concern. The unregulated and prolonged use of antimicrobials in animal agriculture has accelerated the selection of MDR pathogens. For instance, a well-documented MDR strain of Salmonella typhimurium, a major cause of salmonellosis in humans, was initially detected in livestock.55 Increasing global pressure from the WHO and EU to limit antibiotic use in poultry production has highlighted the urgent need for sustainable alternatives to AFAs. As AMR continues to rise, the poultry industry faces a critical challenge: to develop effective, long-term solutions that support animal health and productivity without exacerbating resistance. Several promising substitutes have been explored, including phytogenic feed additives, probiotics, prebiotics, antimicrobial peptides, and feed acidifiers, bacteriophages, vaccines and CRISPR-based methods. These alternatives offer various mechanisms to support animal health, modulate gut microbiota, and enhance immunity. A summary of their potential effects and mechanisms of action in poultry is presented in Table 5.56
Table (5):
Comparison of Antibiotic Feed Additives and Proposed Alternatives to AFAs in Poultry
Parameter |
Antibiotic Feed Additives |
Phytogenic Feed Additives |
Probiotics |
Prebiotics |
Feed Acidifiers |
AMPs |
|---|---|---|---|---|---|---|
Direct Antibacterial Activity |
Yes |
Yes |
No |
No |
Yes |
Yes |
Immunomodulation |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
Proliferation of Beneficial Bacteria |
No |
Yes |
Yes |
Yes |
Yes |
No |
Nutrient Absorption |
Increased due to thinning of epithelial wall |
Increased due to induction of digestive enzymes |
Increased due to induction of digestive enzymes |
Unknown |
Increased due to gut acidification and protease activity |
Thought to increase |
Development of Resistance |
Very common |
Could be difficult due to multiple targets of action |
No (indirect antimicrobial activity) |
No (indirect antimicrobial activity) |
Not very common |
Not very common (not energy efficient for cell) |
Stability |
Stable |
Stable |
Stable |
Stable |
Stable |
Vulnerable to proteolysis |
Effect in Poultry |
(i) Enhancement in body weight and feed efficiency (ii) Reduction in pathogenic microbial load (iii) Improved overall productivity and yield |
(i) Enhanced body weight gain (ii) Better feed conversion ratio (FCR) and higher carcass yield (iii) Reduction in pathogenic microorganisms (iv) Improved fatty acid composition in egg yolk (v) Elevated serum protein levels and antioxidant status |
(i) Improvement in body weight and FCR (ii) Reduction in pathogenic bacterial load (iii) Enhancement of beneficial intestinal microbiota |
(i) Enhanced resistance to diseases and improved nutrient utilization (ii) Greater weight gain and enrichment of beneficial gut microbiota (iii) Lower incidence of pathogenic bacteria (iv) Healing or mitigation of coccidial lesions |
(i) Decrease in pathogenic load (ii) Enhanced weight gain and FCR (iii) Better utilization of phytate-bound phosphorus (iv) Lower mortality and feed expenses, with increased dressing percentage and liver mass |
(i) Reduction in pathogenic microorganisms (ii) Enhancement of beneficial microbial populations, nutrient uptake, body weight, and feed efficiency |
Several studies carried out in India have explored the use of Phytogenic feed additives and probiotics as an alternative to antibiotics to be used as growth enhancers. A collaborative study was conducted between the College of Veterinary Science and Animal Husbandry, R.K. Nagar, Agartala, Tripura, and the R&D team of Ayurvet Ltd., Baddi, Himachal Pradesh.57 The study evaluated the efficacy of a herbal growth promoter developed by Ayurvet Ltd. as an alternative to conventional antibiotic growth promoters. The formulation, comprising herbal oils from Eruca sativa (arugula), Trigonella foenum-graecum (fenugreek), Zingiber officinale (ginger), and Allium sativum (garlic), was administered at a dose of 500 g/ton of basal feed to 120 day-old chicks. Results showed that birds receiving the herbal supplement exhibited significantly higher growth performance compared to those fed with the antibiotic growth promoter (Vetclin 112). A recently published report investigates the combined use of Andrographolide (from Andrographis paniculata) and Ajwain (Trachyspermum ammi) as a phytoremediation-based alternative to antibiotics for treating Salmonella gallinarum infections in poultry. Through in vitro and in vivo experiments, the combination showed antimicrobial, immunomodulatory, and anti-inflammatory effects, significantly reducing mortality, clinical symptoms, and pathological damage in infected chickens. The approach highlights a sustainable and effective strategy for managing Salmonella infections in poultry production.58
The potential of Emblica officinalis (amla) fruit powder as a natural growth promoter was investigated in commercial broiler chickens.59 Their findings revealed a significant increase in average body weight over a 6-week period in birds supplemented with amla powder. A recent study reported that combination of grape and olive pomace extracts, administered to male Ross 308AP95 broiler chicks via drinking water (2 L:1000 L) for 1-42 days, improved body weight and feed conversion ratio (FCR), and significantly reduced mycotoxin-induced damage.60
Anand Agricultural University evaluated the effects of dietary inclusion of probiotics, prebiotics, and their combination (synbiotics) on broiler chickens and found synbiotic supplementation significantly improved performance efficiency and economic returns.61 Kemin Industries South Asia Pvt. Ltd. in collaboration with the West Bengal University of Animal and Fishery Sciences, conducted a study comparing the growth-promoting potential of the probiotic Bacillus subtilis PB6 with conventional antibiotic growth promoters in broiler chickens.62 Probeads-EC® is an enteric-coated probiotic bead developed by the Translational Research Platform for Veterinary Biologicals (TRPVB), Tamil Nadu Veterinary and Animal Sciences University, using the unique polymer Cellulose Acetate Phthalate. It contains Bacillus firmus, Bacillus subtilis, Enterococcus faecium, Enterococcus faecalis, and Saccharomyces cerevisiae. This study assessed the effects of Probeads-EC® supplementation on the growth performance of Aseel cross chicks, revealing significant improvements in body weight, feed efficiency, and livability without compromising gut health.63 Another study conducted in collaboration with Cargill and West Bengal University of Animal and Fishery Sciences, this study assessed the effects of Bacillus subtilis (probiotic) and Saccharomyces cerevisiae fermentation product (postbiotic) on broiler chickens. These findings demonstrated enhanced growth performance, improved vaccine efficacy, and reduced prevalence of antimicrobial-resistant bacteria.64 Bhagwat et al. evaluated HimFlora, a synbiotic formulation with phytoactives (SFP), containing probiotic Bacillus strains and herbal actives, in broiler chicken diets. Nine hundred Cobb 430y chicks were divided into five groups, including controls, antibiotic (Enramycin), SFP (100 g and 150 g/ton feed), and a competitor probiotic product (CPP). Supplementation with SFP at 150 g/ton improved growth performance, ready-to-cook weight, gut health, nutrient digestibility, and litter quality, performing comparably to Enramycin and CPP. Additionally, SFP reduced fecal water-soluble phosphorus by up to 23.08%, suggesting its potential as an effective antibiotic alternative in broiler production.65 Another study showed supplementation with 1% citric acid, alone or with antibiotics over 45 days, significantly improved body weight gain, feed intake, and FCR compared to controls and antibiotic-only groups. The study confirmed that 1% citric acid can effectively replace chlortetracycline as a growth promoter without compromising performance.66
There are quite a few reports from India regarding the use of bacteriophages, vaccines and antimicrobial peptides. Bacteriophages as an alternative to antibiotics was evaluated for controlling Salmonella in poultry. Salmonella isolates from poultry sources that showed high resistance to nalidixic acid (71%), tetracycline (71%), nitrofurantoin (50%), and ampicillin (43%) targeted with Salmonella-specific lytic phage PSE5 isolated from poultry wastewater. The lytic activity of PSE5 significantly reduced bacterial counts at a multiplicity of infection (MOI) of 0.01 within 2 hours. In an egg-surface contamination model, PSE5 treatment reduced S. Enteritidis levels by approximately 2 × 106 CFU/ml compared to untreated controls. These findings highlight the potential of bacteriophage therapy as an effective strategy for controlling Salmonella in poultry production and reducing pathogen transmission.67 The role of vaccination in managing Salmonella in poultry was evaluated by Maiti et al. The research group has developed a novel trivalent outer membrane vesicle (OMV) based vaccine targeting invasive non-typhoidal Salmonella (iNTS) in poultry. OMVs were isolated from Salmonella Typhimurium, S. Enteritidis, and S. Gallinarum and combined in a 1:1:1 ratio. were orally immunized thrice at two-week intervals. One-day-old vaccinated chicks showed reduced fecal shedding and organ invasion following challenge with homologous and heterologous (S. Infantis) serovars, with protective effects maintained up to one year. These results indicate that the trivalent OMV formulation is a promising broad-spectrum vaccine candidate for preventing iNTS infections in both broiler and layer poultry.68 The use of chicken intestinal-derived antimicrobial peptides (AMPs) as an alternative to antibiotic growth promoters in broilers was evaluated by Vaani et al. AMP supplementation significantly modulated cytokine expression, downregulating pro-inflammatory cytokines (IL-17A, IFN-α, IFN-γ) and upregulating the anti-inflammatory cytokine IL-10, indicating an enhanced cell-mediated immune response.69
The advancement in the CRISPR-based method of genome editing is also explored in eliminating Salmonella infection. CRISPR-Cas systems offer a powerful tool for selectively targeting pathogenic bacteria, such as S. enterica, while sparing beneficial or commensal species. Programmable CRISPR RNAs can be designed to target specific genetic sequences, such as the mviM virulence gene in S. enterica, enabling precise pathogen elimination with strain-level discrimination and quantitative control of microbial populations-capabilities not achievable with traditional antibiotics or bacteriophages.70 Building on this concept, CRISPR/Cas9 was used to delete the SpvB gene from the virulence plasmid of S. Gallinarum (SG18). The resulting ִSpvB_SG18 strain was avirulent in broiler chickens, causing no disease or mortality, did not affect bird weight, and was rapidly cleared from the liver, although it persisted in the intestine for 4-5 days.71 These findings demonstrate the essential role of SpvB in systemic infection and underscore the dual potential of CRISPR technology both as a “smart antibiotic” platform to target foodborne pathogens and as a tool for generating live attenuated vaccine strains against fowl typhoid.
Alternative therapies for antibiotic resistance hold significant promise but face multiple challenges that hinder their widespread adoption. Scientifically, many remain in early research stages with limited clinical evidence, making their effectiveness less certain compared to conventional antibiotics. Some therapies also struggle with specificity, potentially harming beneficial microbiota, while issues of standardization and quality control complicate consistent safety and efficacy. Economically, these treatments are less attractive to pharmaceutical companies due to low profit potential, given that antibiotics are prescribed for short durations, unlike chronic disease drugs. High development and production costs further limit accessibility, especially in resource-limited settings, and unclear regulatory pathways create delays in approval. On a systemic level, lack of awareness among healthcare professionals, inadequate infrastructure for delivery, and persistent misuse of antibiotics by the public exacerbate resistance challenges. Overcoming these barriers requires not only scientific advancements but also stronger regulatory support, economic incentives, public education, and collaborative efforts between researchers, clinicians, policymakers, and industry.
Economic impact and cost-benefit insights
The economic ramifications of AMR in Indian poultry production are substantial and multifaceted. Annual losses due to poultry diseases are estimated at ₹ 25,000 crore (approximately $3 billion), encompassing mortality, reduced productivity, treatment costs, and trade restrictions. Small-scale farmers, who constitute approximately 80% of India’s poultry producers, are particularly vulnerable to these economic shocks due to limited resources for biosecurity measures and veterinary care. Undercooked poultry products contaminated with Salmonella and Campylobacter are estimated to cause approximately 175,000 reported cases of illness annually in India, although the true incidence is likely significantly higher due to underreporting. According to the National Institute of Nutrition, poultry accounts for around 43% of all Salmonella infections in humans. These bacteria are capable of surviving for extended periods in processed meat products, particularly when cold chain systems are poorly maintained. Additionally, cross-contamination during food preparation increases the risk of infection, extending exposure beyond direct consumers of poultry.72 Cost-benefit analyses of antibiotic restrictions reveal complex trade-offs. Studies suggest that while complete bans may reduce AMR development, they can initially increase therapeutic antibiotic usage and production costs. The EU experience indicates that savings from discontinued AGP use can offset decreased feed efficiency costs, though substantial investments in alternative production systems may be required.73,74 Global projections estimate that AMR could lead to a 3%-8% reduction in livestock production by 2050, with low-income countries potentially experiencing up to 11% reduction. For India’s rapidly growing poultry sector, valued at over USD 28 billion in 2021-22, such reductions could translate to significant economic losses.73,75
Antimicrobial resistance policy frameworks: fragmented enforcement in India and lessons from global models
Multiple agencies with overlapping mandates but fragmented implementation shape the regulatory framework for antimicrobial resistance (AMR) control in Indian poultry. FSSAI announced its AMR action plan in 2024, emphasizing farmer awareness on judicious antibiotic use and initiating surveillance of antimicrobial susceptibility in food matrices. It has issued directives restricting antibiotic use in livestock and introduced mandatory withdrawal periods for animal products.76,77 Parallel efforts are coordinated by the National Centre for Disease Control (NCDC), which launched the National Programme on AMR Containment in 2013 with the objectives of developing laboratory-based surveillance, monitoring antimicrobial usage patterns, strengthening infection control practices, and promoting awareness among healthcare providers. The program currently operates through ten network laboratories tracking four priority pathogens of public health concern.78 The Indian Council of Agricultural Research (ICAR) has also expanded its role through the All India Network Programme on AMR (AINP-AMR), which compiles surveillance data via the Indian Network for Fisheries and Animal Antimicrobial Resistance (INFAAR). In addition, ICAR-IVRI has applied machine learning approaches to identify critical drivers of AMR in poultry, including agro-climatic zones and pathogen phylotypes.79 Despite these initiatives, policy enforcement in India remains weak, marked by poor intersectoral coordination, absence of uniform standards on antibiotic usage in animals, and continued prophylactic application of antimicrobials in poultry production. Structural limitations such as inadequate surveillance systems, insufficient regulatory oversight, and limited resources for compliance further undermine effectiveness.80,81
In contrast, the European Union (EU) offers a more consolidated model of AMR control. The EU introduced a complete ban on antibiotic growth promoters (AGPs) in 2006, following incremental restrictions that began in the 1970s with the UK’s Swann Committee recommendations. This comprehensive approach contributed to a 53% reduction in antibiotic sales for livestock between 2011 and 2022.82 The regulatory framework integrates strict prescription requirements, veterinary supervision, and extensive monitoring. Denmark has pioneered targeted interventions, including the “yellow card” system that penalizes producers with antibiotic consumption exceeding twice the industry average, while the Danish Integrated Antimicrobial Resistance Monitoring Programme (DANMAP) provides systematic surveillance across animal, food, and human sectors.82 Economic analyses of the EU ban revealed only modest impacts, with costs estimated at EUR 0.008 per kilogram of live broiler weight, equivalent to approximately 16% of overall regulatory compliance expenses. Longitudinal assessments further demonstrated sustained reductions in antibiotic usage without adverse effects on productivity in Denmark, Norway, and Sweden.82,83 India, by comparison, has yet to implement a comprehensive prohibition on AGPs despite recommendations by the Bureau of Indian Standards in 2007 to discontinue antibiotics with systemic action for growth promotion. Existing measures remain fragmented, enforcement mechanisms are limited, and surveillance infrastructure is inadequate. The ongoing availability of AGPs in Indian markets despite regulatory advisories illustrates persistent gaps between stated policy and actual practice.80,81,84 The National Action Plan on AMR (2017-2021) endorsed a One Health framework, but operationalization has been constrained by weak coordination between human and animal health sectors. These limitations are compounded by India’s federal structure, where outcomes rely heavily on state-level administrative capacity and political will.80 Regulatory enforcement challenges persist even where guidelines exist: although FSSAI has mandated withdrawal periods and the Drug Controller General of India requires labeling of antibiotics with withdrawal information, monitoring and enforcement remain minimal.80 Economic incentives also remain misaligned, as small-scale farmers continue to depend on low-cost antibiotics in the absence of financial support for adopting improved biosecurity, husbandry, or alternative interventions. Without comprehensive cost-sharing mechanisms to ease the transition toward antibiotic-free systems, reliance on prophylactic use is unlikely to diminish. Moreover, the absence of integrated data systems represents a fundamental limitation to policy evaluation. Unlike Denmark’s DANMAP, which links antibiotic consumption with resistance trends across sectors, India lacks cross-sectoral databases, preventing evidence-based refinements and adaptive management of AMR policies.81
Table (6):
Knowledge Gaps & Future Directions
Knowledge Gap |
Research/Policy Recommendation |
|---|---|
Fragmented AMR surveillance |
Nationwide WGS-based AMR monitoring network |
Low research output on Salmonella/AMR |
Incentivize collaborative, multi-institutional projects |
Efficacy data for alternatives in India |
Fund RCTs and field trials in Indian poultry settings |
Regulatory gaps for feed additives |
Accelerate FSSAI guideline development & implementation |
Farmer/veterinarian education |
Launch national AMR stewardship & awareness campaigns |
Challenges and recommendation
Challenges in Addressing Salmonella and AMR in Indian Poultry (Table 6): The Indian poultry sector faces several challenges in addressing the issue of Salmonella and AMR:
Overuse and Misuse of Antibiotics: The widespread use of antibiotics in poultry for growth promotion and disease prevention has been a major driver of AMR. This practice is often unregulated and lacks proper oversight.85-87
Lack of Awareness and Education: Many poultry farmers and workers lack awareness about the risks of AMR and the proper use of antibiotics. This lack of knowledge contributes to the indiscriminate use of antibiotics.85,87,88
Inadequate Infrastructure and Biosecurity: Poor farm management practices, inadequate biosecurity measures, and insufficient infrastructure for waste management and disinfection contribute to the spread of AMR bacteria in poultry farms.85,87,89
Regulatory and Policy Gaps: Despite existing policies and regulations, their implementation is often weak. This lack of enforcement allows the continued misuse of antibiotics in the poultry sector.85-87
Emergence of MDR Salmonella Strains: The emergence of MDR Salmonella strains, such as Salmonella Infantis, poses a significant challenge to public health. These strains are often resistant to multiple antibiotics, making treatment difficult.90-92
Recommendations for Mitigating Salmonella and AMR in Indian Poultry (Table 6): To address the challenges posed by Salmonella and AMR in Indian poultry, the following recommendations are proposed:
Strengthening Regulatory Frameworks: There is a need for stricter enforcement of existing regulations on antibiotic use in poultry. This includes banning the use of antibiotics for growth promotion and ensuring that antibiotics are used only for therapeutic purposes under veterinary supervision.85-87
Improving Awareness and Education: Educational programs for poultry farmers, workers, and veterinarians on the risks of AMR and the proper use of antibiotics are essential. This can be achieved through workshops, training sessions, and awareness campaigns.85,87,88
Enhancing Biosecurity and Farm Management: Improving biosecurity measures, such as better waste management, disinfection protocols, and farm hygiene, can reduce the spread of AMR bacteria in poultry farms.85,87,89
Promoting Alternative Strategies: Encouraging the use of antibiotic alternatives, such as probiotics, prebiotics, bacteriophages, and vaccines, can reduce the reliance on antibiotics in poultry production.93-95
Strengthening Surveillance and Monitoring: Establishing a robust surveillance system to monitor the use of antibiotics and the spread of AMR bacteria in poultry is crucial. This includes regular testing of poultry products for antibiotic residues and AMR bacteria.85,86
Encouraging Public-Private Partnerships: Collaboration between government agencies, poultry industry stakeholders, and research institutions is essential for developing and implementing effective strategies to combat AMR in poultry.88,96,97
Artificial intelligence and machine learning applications offer transformative potential for AMR surveillance and prediction. AI-driven tools can automate resistance gene identification, predict resistance phenotypes, and optimize treatment strategies based on genomic data. Studies demonstrate that ML models can predict antibiotic resistance in E. coli with high accuracy using metagenomic data from livestock environments.98-100
Whole genome sequencing (WGS)-based surveillance represents the future of AMR monitoring, providing comprehensive characterization of resistance mechanisms and transmission dynamics. However, implementation requires substantial investments in infrastructure, training, and bioinformatics capabilities that many low-resource settings currently lack.98
The findings of this review highlight that Salmonella and AMR remain major threats to poultry production, food safety, and public health in India. The high prevalence of non-typhoidal Salmonella, coupled with increasing resistance to critically important antimicrobials, reflects systemic challenges related to biosecurity, antibiotic stewardship, and surveillance capacity within the poultry sector.
From a One Health perspective, these challenges highlight the urgent need for a holistic and integrated approach that recognizes the interconnectedness of animal, human, and environmental health in addressing Salmonella and AMR in Indian poultry systems. The widespread overuse and misuse of antibiotics in poultry production have significantly accelerated the emergence and dissemination of MDR Salmonella, exacerbating risks to food safety and public health. To mitigate these risks, alternative strategies such as bacteriophages, probiotics, prebiotics, phytobiotics, and vaccines have gained increasing attention as viable tools to reduce Salmonella colonization and lessen dependence on antibiotics. These approaches offer promising, sustainable options to curb AMR while maintaining productivity in poultry production.
This review further highlights that MDR Salmonella is becoming an increasingly serious concern in the Indian poultry industry, with regional variations in prevalence and resistance patterns posing direct threats to animal health, human health, and environmental safety. The fragmented nature of surveillance systems and India’s relatively low research output, when compared with global leaders, emphasize the need for a coordinated national response. Adoption of a comprehensive One Health strategy which encompasses strengthened surveillance, enforcement of antimicrobial stewardship frameworks such as the WHO AWaRe classification, closure of regulatory gaps, and accelerated implementation of antibiotic alternatives that is essential to safeguard public health and ensure the long-term sustainability of the poultry sector. Investment in research, cross-sectoral collaboration, and farmer-centered education will be critical to addressing existing gaps and building long-term resilience against AMR.
However, while alternative interventions show considerable promise, further research and development are required to optimize their efficacy, scalability, and cost-effectiveness to ensure widespread adoption within the poultry industry. By embedding One Health principles into policy, practice, and research, and by promoting responsible antibiotic use alongside validated alternatives, India can substantially reduce the burden of AMR and move toward a safer, more sustainable food production system.
While this review offers a comprehensive bibliometric and epidemiological synthesis, it is limited by potential publication bias (exclusion of non-English and gray literature), lack of access to certain proprietary data, and the absence of meta-analytical statistical pooling due to heterogeneity of methods in included studies. Additionally, the field is evolving rapidly, and surveillance data published after 2024 may alter key trends and recommendations.
Top five priorities
- Establishment of a national One Health AMR surveillance platform linking animal, food, and human sectors.
- Enforcing restrictions on non-therapeutic antibiotic use in poultry move toward prescription-only models.
- Accelerating field validation and approval of antibiotic alternatives, especially those showing robust efficacy.
- AI-powered surveillance systems capable of real-time AMR monitoring and outbreak prediction.
- Inclusion of low-cost Whole genome sequencing (WGS) to obtain comprehensive insights into AMR mechanisms and transmission.
- Farmer-centric intervention studies to identify effective behavior change strategies
- Investing in research consortia and capacity building (funding, training, equipment) for AMR/Salmonella research.
- Promoting cross-sectoral (public, private, academic) partnerships for innovation, education, and policy implementation.
ACKNOWLEDGMENTS
The authors are thankful to Dr. Gouri Shankar Koner, CEO, PBGSBS; Dr. Utpal Kumar Karmakar, Managing Director, WBLDCL; and Dr. Amar Krishna Das, In-Charge, Broiler Integration Programme, WBLDCL, for their guidance and support. Authors also acknowledge the Directorate of Animal Husbandry and Veterinary Services, Government of West Bengal, for institutional support.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
Not applicable.
- Gast RK, Porter RE Jr. Salmonella infections. In: Swayne DE, et al., eds. Diseases of Poultry. 14th ed. Hoboken, NJ: John Wiley & Sons, Inc.; 2020:717-753.
Crossref - Upadhyaya M, Poosaran N, Fries R. Prevalence and predictors of Salmonella spp. in retail meat shops in Kathmandu. J Agric Sci Technol B. 2012;2(9A)1094-1106.
- Ahmad MUD, Sarwar A, Najeeb MI, et al. Assessment of microbial load of raw meat at abattoirs and retail outlets. J Anim Plant Sci. 2013;23(3):745-748.
- Bhandari N, Nepali DB, Paudyal S. Assessment of bacterial load in broiler chicken meat from the retail meat shops in Chitwan, Nepal. Int J Infect Microbiol. 2013;2(3):99-104.
- World Health Organization. Salmonella (non-typhoidal). 2021. https://www.who.int/news-room/fact-sheets/detail/salmonella-(non-typhoidal). Accessed 20 Jan 2021.
- Centers for Disease Control and Prevention (CDC). Antibiotic Resistance Threats in the United States. Atlanta, GA: U.S. Department of Health and Human Services, CDC. 2019. Accessed on 11 April 2025.
- EFSA and ECDC. The European Union One Health 2018 zoonoses report. EFSA J. 2019;17(12):e05926.
Crossref - Hatha AM, Lakshmanaperumalsamy P. Prevalence of Salmonella in fish and crustaceans from markets in Coimbatore, South India. Food Microbiol, 1997;14(2):111-116.
Crossref - Suresh T, Srinivasan D, Hatha AAM, Lakshmanaperumalsamy P. The incidence, antibiotic resistance and survival of Salmonella and Escherichia coli isolated from broiler chicken retail outlets. Microbes Environ. 2000;15(3):173-181.
Crossref - Sati H, Carrara E, Savoldi A, et al. The WHO Bacterial Priority Pathogens List 2024: a prioritisation study to guide research, development, and public health strategies against antimicrobial resistance. Lancet Infect Dis. 2025;25(9):1033-1043.
Crossref - van Eck NJ, Waltman L. Software survey: VOSviewer, a computer program for bibliometric mapping. Scientometrics. 2010;84(2):523-538.
Crossref - Singh S, Yadav AS, Singh SM, Bharti P. Prevalence of Salmonella in chicken eggs collected from poultry farms and marketing channels and their antimicrobial resistance. Food Res Int. 2010;43(8):2027-2030.
Crossref - Sharma J, Kumar D, Hussain S, et al. Prevalence, antimicrobial resistance and virulence genes characterization of non-typhoidal Salmonella isolated from retail chicken meat shops in Northern India. Food Control. 2019;102:104-111.
Crossref - Mir IA, Kashyap SK, Maherchandani S. Isolation, serotype diversity and antibiogram of Salmonella enterica isolated from different species of poultry in India. Asian Pac J Trop Biomed. 2015;5(7):561-567.
Crossref - Sain A, Sharma DK, Singathia R, et al. Antibiotic resistance and virulence genes profile of non-typhoidal Salmonella species isolated from poultry enteritis in India. Trop Anim Health Prod. 2024;56(2):91.
Crossref - Ganjeer T, Patyal A, Shakya S, et al. Antibiotic resistance pattern and distribution of resistance genes in Salmonella isolated from chicken and duck eggs in Chhattisgarh, India. Asian J Dairy Food Res. 2023;42(2):89-96.
Crossref - Sannat C, Patyal A, Rawat N, et al. Characterization of Salmonella gallinarum from an outbreak in Raigarh, Chhattisgarh. Vet World. 2017;10(2):144-148.
Crossref - Samanta I, Joardar SN, Das PK, et al. Prevalence and antibiotic resistance profiles of Salmonella serotypes isolated from backyard poultry flocks in West Bengal, India. J Appl Poult Res. 2014;23(3):536–545.
Crossref - Chakraborty S, Roychoudhury P, Samanta I, et al. Molecular detection of biofilm, virulence and antimicrobial resistance associated genes of Salmonella serovars isolated from pig and chicken of Mizoram, India. Indian J Anim Res. 2020;54(5):608-613.
- Chowdhury M, Bardhan R, Pal S, et al. Comparative occurrence of ESBL/AmpC beta lactamase producing Escherichia coli and Salmonella in contract farm and backyard broilers. Lett Appl Microbiol. 2022;74(1):53-62.
Crossref - Gautam RK, Kakatkar AS, Karani MN, Shashidhar R, Bandekar JR. Salmonella in Indian ready-to-cook poultry: Antibiotic resistance and molecular characterization. Microbiol Res. 2017;8(1):6882.
Crossref - Waghamare RN, Paturkar AM, Vaidya VM, et al Phenotypic and genotypic drug resistance profile of Salmonella serovars isolated from poultry farm and processing units located in and around Mumbai city, India. Vet World. 2018;11(12):1682-1688.
Crossref - Kolhe RP, Waskar VS, Mehere PV. Antimicrobial resistance pattern of Salmonella species recovered from livestock and poultry. Asian J Dairy Food Res. 2022;41(3):259-262.
Crossref - Babar A, Suryawanshi R, Deshmukh O, et al. Occurrence, characterization and antimicrobial resistance pattern of pathogenic Salmonella isolated from chicken and chicken meat products. Indian J Vet Sci Biotechnol. 2023;19(4):11-15.
Crossref - Sayed F, Kaur R, Jha V, Quraishi UA. Prevalence, antibiotic resistance, and implications for public health due to Salmonella contamination in food products. South Asian J Res. Microbiol. 2024;18(5):18-29.
Crossref - Dhanarani TS, Shankar C, Park J, Dexilin M, Kumar RR, Thamaraiselvi K. Study on acquisition of bacterial antibiotic resistance determinants in poultry litter. Poult Sci. 2009;88(7):1381-1387.
Crossref - Harsha HT, Reshmi R, Varghese R, Divya PS, Rahiman KM, Hatha AM. Prevalence and antibiotic resistance of Salmonella from eggs of commercial samples. J Microbiol Infect Dis. 2011;1(3):93-100.
Crossref - Bangera SR, Umakanth S, Chowdhury G, Saha RN, Mukhopadhyay AK, Ballal M. Poultry: A receptacle for non-typhoidal Salmonellae and antimicrobial resistance. Iran J Microbiol. 2019;11(1):31-37.
Crossref - Sohail MN, Rathnamma D, Priya SC, et al. Salmonella from farm to table: Isolation, characterization, and antimicrobial resistance of Salmonella from commercial broiler supply chain and its environment. Biomed Res Int. 2021;2021:3987111.
Crossref - Anbazhagan PV, Thavitiki PR, Varra M, et al. Evaluation of efflux pump activity of multidrug-resistant Salmonella Typhimurium isolated from poultry wet markets in India. Infect Drug Resist. 2019;12:1081-1088.
Crossref - Singh R, Yadav AS, Tripathi V, Singh RP. Antimicrobial resistance profile of Salmonella present in poultry and poultry environment in north India. Food Control. 2013;33(2):545-548.
Crossref - Arora D, Kumar S, Singh D, Jindal N, Mahajan NK. Isolation, characterization and antibiogram pattern of Salmonella from poultry in parts of Haryana, India. Adv Anim Vet Sci. 2013;1(5):161-163.
- Singh A, Singh M, Malik MA, Padha S. Is there a shift in Salmonella diversity among poultry in Northern India? Avian Dis. 2023;67(1):108-113.
Crossref - Geyi D, Thomas P, Prakasan L, et al. Salmonella enterica serovars linked with poultry in India: Antibiotic resistance profiles and carriage of virulence genes. Braz J Microbiol. 2024;55(1):969-979.
Crossref - Rasve R, Waghamare R, Siddiqui F, Jadhav N, Khose K, Kuntawar N. Antimicrobial resistance profile of Salmonella enterica isolated from improved poultry breed farming chain, Maharashtra, India. Microbiol Res J Int. 2024;34(12):76-90.
Crossref - Maripandi A, Al-Salamah AA. Multiple-antibiotic resistance and plasmid profiles of Salmonella enterica serovars isolated from food sources. Am J Food Technol. 2010;5(4):260-268.
Crossref - Rajagopal R, Mini M. Outbreaks of salmonellosis in three different poultry farms of Kerala, India. Asian Pac J Trop Biomed. 2013;(6):496-500.
Crossref - Adesiji YO, Deekshit VK, Karunasagar I. Antimicrobial-resistant genes associated with Salmonella spp. isolated from human, poultry, and seafood sources. Food Sci Nutr. 2014;2(4):436-442.
Crossref - Saravanan S, Purushothaman V, Murthy TRGK, et al. Molecular epidemiology of non-typhoidal Salmonella in poultry and poultry products in India: Implications for human health. Indian J Microbiol. 2015;5(3):319-326.
Crossref - Hasan K, Rathnamma D, Narayanaswamy HD, Malathi V, Gupta S, Singh SV. Isolation of bacterial pathogens associated with broiler mortality in Kolar. Adv Anim Vet Sci. 2017;5(7):312-315.
Crossref - Pragasam AK, Anandan S, John J, et al. An emerging threat of ceftriaxone-resistant non-typhoidal Salmonella in South India: Incidence and molecular profile. Indian J Med Microbiol. 2019;37(2):198-202.
Crossref - Karabasanavar NS, Madhavaprasad CB, Gopalakrishna SA, Hiremath J, Patil GS, Barbuddhe SB. Prevalence of Salmonella serotypes S. Enteritidis and S. Typhimurium in poultry and poultry products. J Food Saf. 2020;40(6):e12852.
Crossref - Kalapriya B, Thirunavukarasu ES, Muninathan N, Baskaran K, Suresh A. Cross-sectional study of antibiotic residues and antimicrobial-resistant pathogens from raw meat in and around Kanchipuram, Tamil Nadu. Pravara Med Rev. 2024;6(4):83-95.
Crossref - Arora D, Kumar S, Jindal N, Narang G, Kapoor PK, Mahajan NK. Prevalence and epidemiology of Salmonella enterica serovar Gallinarum from poultry in some parts of Haryana, India. Vet World. 2015;8(3):1300-1304.
Crossref - Kumar T, Mahajan NK, Rakha NK. Isolation and prevalence of Salmonella serovars from poultry in different parts of Haryana, India. Indian J Anim Sci. 2012;82(6):557-559.
- Naik VK, Shakya S, Patyal A, Gade NE, Bhoomika B. Isolation and molecular characterization of Salmonella spp. from chevon and chicken meat collected from different districts of Chhattisgarh, India. Vet World. 2015;8(10):1235-1239.
Crossref - Kaushik P, Anjay A, Kumari S, Bharti SK, Dayal S. Isolation and prevalence of Salmonella from chicken meat and cattle milk collected from local markets of Patna, India. Vet World. 2014;7(2):62-65.
Crossref - Raju R, O’Neil L, Kerr C, et al. Non-typhoidal Salmonella in humans in India, Vietnam, Bangladesh and Sri Lanka: A systematic review. JAC Antimicrob Resist. 2024;6(6):dlae190.
Crossref - Antony B, Scaria B, Dias M, Pinto H. Salmonella Wien from gastroenteritis cases encountered in Mangalore, India: A report of 10 cases and review of the literature. Indian J Med Sci. 2009;63(5):195-197.
- Jain P, Nandy S, Bharadwaj R, Niyogi SK, Dutta S. Salmonella enterica serovar Weltevreden ST1500 associated foodborne outbreak in Pune, India. Indian J Med Res. 2015;141(2):239-241.
Crossref - Deepanjali S, Jharna M, Chanaveerappa B, Sarumathi D, Gopichand P, Anupriya K. An outbreak of Salmonella Enteritidis food poisoning following consumption of chicken shawarma: A brief epidemiological investigation. F1000Research. 2022;10:851.
Crossref - Sumana MN, Rao MR, Rajshekar D, et al. The first-ever encounter with Salmonella enterica serovar Hvittingfoss causing acute gastroenteritis in India: A case report. Infect Dis Rep. 2024;16(6):1191-1196.
Crossref - Donthu N, Kumar S, Mukherjee D, Pandey N, Lim WM. How to conduct a bibliometric analysis: An overview and guidelines. J Bus Res. 2021;133:285-229.
Crossref - Van Boeckel TP, Brower C, Gilbert M, et al. Global trends in antimicrobial use in food animals. Proc Natl Acad Sci USA. 2015;112(18):5649-5654.
Crossref - Woc-Colburn L, Bobak DA. The expanding spectrum of disease due to Salmonella: An international perspective. Curr Infect Dis Rep. 2009;11(2):120-124.
Crossref - Suresh G, Das RK, Brar SK, et al. Alternatives to antibiotics in poultry feed: Molecular perspectives. Crit Rev Microbiol. 2018;44(3):318-335.
Crossref - Debnath BC, Choudhary KBD, Ravikanth K, Thakur A, Maini S. Comparative efficacy of natural growth promoter (AV/AGP/10) with antibiotic growth promoter on overall growth performance and intestinal morphometry in broiler birds. Int J Pharm Sci Health Care. 2014;2(4):155-168.
- Ahmad SM, Saleem A, Nazir J, et al. Synthesis and pharmacological evaluation of Andrographolide and Ajwain as promising alternatives to antibiotics for treating Salmonella Gallinarum infection in chicken. Int Immunopharmacol. 2024;142(Pt B):113163.
Crossref - Patel AP, Bhagwat SR, Pawar MM, Prajapati KB, Chauhan HD, Makwana RB. Evaluation of Emblica officinalis fruit powder as a growth promoter in commercial broiler chickens. Vet World. 2016;9(2):207-210.
Crossref - Arora SS, Debnath A, Dhara AK, Haldar S, Moreno RC, Riahi I. The efficacy of a novel water-soluble anti-mycotoxin solution in improving broiler chicken performance under mycotoxin challenge. Toxins (Basel). 2025;17(5):212.
Crossref - Saiyed MA, Joshi RS, Savaliya FP, Patel AB, Mishra RK, Bhagora NJ. Study on inclusion of probiotic, prebiotic and its combination in broiler diet and their effect on carcass characteristics and economics of commercial broilers. Vet World. 2015;8(2):225-231.
Crossref - Jayaraman S, Das PP, Saini PC, Roy B, Chatterjee PN. Use of Bacillus subtilis PB6 as a potential antibiotic growth promoter replacement in improving performance of broiler birds. Poult Sci. 2017;96(8):2614-2622.
Crossref - Thomas KS, Jayalaitha V. Effect of targeted delivery of Probeads-EC® on the production performance of Aseel cross chicks. J Vet Anim Sci. 2021;53(1):94-97.
Crossref - Soren S, Mandal GP, Mondal S, et al. Efficacy of Saccharomyces cerevisiae fermentation product and probiotic supplementation on growth performance, gut microflora and immunity of broiler chickens. Animals. 2024;14(6):866.
Crossref - Bhagwat VG, Tattimani SVG, Baig MR. Dietary supplementation of synbiotic formulation with phytoactives on broiler performance, relative ready-to-cook weight, health, nutrient digestibility, gut health, and litter characteristics. J Appl Biol Biotechnol. 2023;11(5):126-132.
Crossref - Bagal VL, Khatta VK, Tewatia BS, Sangwan SK, Raut SS. Relative efficacy of organic acids and antibiotics as growth promoters in broiler chicken. Vet World. 2016;9(4):377. Crossref
- Sonalika J, Srujana AS, Akhila DS, Juliet MR, Santhosh KS. Application of bacteriophages to control Salmonella Enteritidis in raw eggs. Iran J Vet Res. 2020;21(3):221-225.
- Maiti S, Halder P, Banerjee S, et al Development of a novel trivalent invasive non-typhoidal Salmonella outer membrane vesicles based vaccine against salmonellosis and fowl typhoid in chickens. Immunobiology. 2022;227(2):152183.
Crossref - Vaani ST, Karunakaran R, Raja P, Bandeswaran C. Effects of antimicrobial peptide supplementation on immune response and immune organ index in broiler chicks. Asian J Dairy Food Res. 2025.
Crossref - Gomaa AA, Klumpe HE, Luo ML, Selle K, Barrangou R, Beisel CL. Programmable removal of bacterial strains by use of genome-targeting CRISPR-Cas systems. MBio.0 2014;5(1):e00928-13.
Crossref - Basit A, Tahir H, Haider Z, Tariq H, Ullah A, Rehman SU. CRISPR/Cas9-based deletion of SpvB gene from Salmonella gallinarum leads to loss of virulence in chicken. Front Bioeng Biotechnol. 2022;10:885227.
Crossref - Luber P. Cross-contamination versus undercooking of poultry meat or eggs—which risks need to be managed first?. International journal of food microbiology, 2009;134(1-2):21-28.
Crossref - Teillant A. Costs and benefits of antimicrobial use in livestock. AMR Control. 2015;2015:116-122.
- Lhermie G, Ndiaye Y, Rushton J, Raboisson D. Economic evaluation of antimicrobial use practices in animal agriculture: a case of poultry farming. JAC Antimicrob. Resist. 2022;4(6):dlac119.
Crossref - FAIRR. AMRmageddon: the Material Risks of AMR to Animal Health and the Livestock Sector. 2022. https://www.fairr.org/news-events/insights/amr-material-risks-animal-health. Accessed on 1 October, 2025.
- PIB. Central Advisory Committee of FSSAI unveils Action Plan on Anti-Microbial Resistance; Press Release 2024. https://www.pib. gov.in/PressReleasePage.aspx?PRID=2015026. Accessed on 5 October, 2025.
- World Health Organization. (CDN) Antimicrobial resistance and its containment in India; 2016. https://cdn.who.int/media/docs/default-source/searo/india/antimicrobial-resistance/amr-containment.pdf?sfvrsn=2d7c49a2_2. Accessed on 5 October, 2025
- NCDC. National Action Plan on Antimicrobial Resistance (NAP-AMR) 2017–2021. 2017. https://ncdc.mohfw.gov.in/wp-content/uploads/2024/03/File645 .pdf Accessed on 5 October, 2025
- PIB. Use Of Antibiotics In Poultry; Press release 2025. https://www.pib.gov.in/PressReleasePage.aspx?PRID=2117764®=3&lang=2. Accessed on 10 October, 2025
- Walia K, Sharma M, Vijay S, Shome BR Understanding policy dilemmas around antibiotic use in food animals & offering potential solutions. Indian J Med Res. 2019;149(2):107-118.
Crossref - ReAct. Antibiotic Use in Food Animals: India Overview. https://www.reactgroup.org/wp-content/uploads/2018/11/Antibiotic_Use_in_Food_Animals_India_LIGHT_2018_web.pdf. Accessed on 10 October, 2025
- Zheng S, Li Y, Chen C, Wang N, Yang F. Solutions to the dilemma of antibiotics use in livestock and poultry farming: Regulation policy and alternatives. Toxics. 2025;13(5):348.
Crossref - Feed Strategy. EU antibiotic growth promoter ban not most costly regulation. 2016. https://www.feedstrategy.com/animal-nutrition/poultry/article/15438494 /eu-antibiotic -growth-promoter-ban-not-most-costly-regulation (Accessed: 29 Mar 2016).
- Deccan Herald. Alarming levels of antibiotic resistance found in poultry environment in Andhra Pradesh. 2024. https://www.deccanherald.com/india /andhra-pradesh/alarming-levels-of-antibiotic-resistance-found-in-poultry-environment-in-andhra-3035919 (Accessed: 24 May 2024).
- Chakraborty D, Debnath F, Giri S, et al. Contribution of veterinary sector to antimicrobial resistance in One Health compendium: An insight from available Indian evidence. Front Vet Sci. 2024;11:1411160.
Crossref - Bhoi B, Tiwari R, Kumar M. Impact of poultry farming on antibacterial drug resistance. Vet World. 2024;17(2):327-336.
Crossref - Noor AUZ, Kabir H, Chowdhury MAA, Ather MF, Hasan MK. An emerging route to antibiotic resistance in South Asia: A correspondence. Ann Med Surg (Lond.). 2023;85(2):335-336.
Crossref - Sharma SK, Chatterjee D, Mehrotra V. Zoonoses in the Indian poultry sector and prospect of One Health approach as a sustainable solution: A review. J Adv Biol Biotechnol. 2024;27(9):84-97.
Crossref - Anyanwu MU, Jaja IF, Okpala COR, Njoga EO, Okafor NA, Oguttu JW. Mobile colistin resistance (mcr) gene-containing organisms in poultry sector in low- and middle-income countries: Epidemiology, characteristics, and One Health control strategies. Antibiotics. 2023;12(7):1117.
Crossref - Mattock J, Chattaway MA, Hartman H, et al. A One Health perspective on Salmonella enterica serovar Infantis, an emerging human multidrug-resistant pathogen. Emerg Infect Dis. 2024;30(4):701.
Crossref - Lamichhane B, Mawad AM, Saleh M, et al. Salmonellosis: An overview of epidemiology, pathogenesis, and innovative approaches to mitigate the antimicrobial resistant infections. Antibiotics. 2024;13(1):76.
Crossref - Rahman MM, Hossain H, Chowdhury MSR, et al. Molecular characterization of multidrug-resistant and extended-spectrum β-lactamases-producing Salmonella enterica serovars Enteritidis and Typhimurium isolated from raw meat in retail markets. Antibiotics. 2024;13(7):120.
Crossref - Ruvalcaba-Gómez JM, Villagrán Z, Valdez-Alarcón JJ, et al. Non-Antibiotics strategies to control salmonella infection in poultry. Animals. 2022;12(1):102.
Crossref - Rahman MRT, Fliss I, Biron E. Insights in the development and uses of alternatives to antibiotic growth promoters in poultry and swine production. Antibiotics. 2022;11(6):766.
Crossref - Sevilla-Navarro S, Torres-Boncompte J, Garcia-Llorens J, Bernabeu-Gimeno M, Domingo-Calap P, Catala-Gregori P. Fighting Salmonella infantis: Bacteriophage-driven cleaning and disinfection strategies for broiler farms. Front Microbiol. 2024;15:1223456.
Crossref - Dutta S, Chowdhury G, Mukhopadhyay AK, Ramamurthy T. Toward One Health Approach: Linking Enteric Pathogens from Diverse Sources with Widespread Dissemination of Antimicrobial Resistance. In: Mothadaka MP, Vaiyapuri M,
Rao Badireddy M, Nagarajrao Ravishankar C, Bhatia R, Jena J. (eds) Handbook on Antimicrobial Resistance. Springer, Singapore. 2023
Crossref - Dutta S, Chowdhury G, Mukhopadhyay AK, Ramamurthy T. Toward One Health Approach: Linking Enteric Pathogens from Diverse Sources with Widespread Dissemination of Antimicrobial Resistance. In: Mothadaka MP, Vaiyapuri M,
Rao Badireddy M, Nagarajrao Ravishankar C, Bhatia R, Jena J. (eds) Handbook on Antimicrobial Resistance. Springer, Singapore. 2023
Crossref - Sherry NL, Lee JY, Giulieri SG, et al. Genomics for antimicrobial resistance—progress and future directions. Antimicrob Agents Chemother. 2025;69(5):e01082-24.
Crossref - Baker M, Zhang X, Maciel-Guerra A, et al. Machine learning and metagenomics reveal shared antimicrobial resistance profiles across multiple chicken farms and abattoirs in China. Nat Food. 2023;4(8):707-720.
Crossref - Arnold A, McLellan S, Stokes JM. How AI can help us beat AMR. NPJ Antimicrob. Resist. 2025;3(1):18.
Crossref
© The Author(s) 2026. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.