ISSN: 0973-7510
E-ISSN: 2581-690X
There has been a spreading attention in the present day to develop environmentally friendly materials such as biodegradable starch films with antimicrobial properties for food protection. Chitosan has exhibited antimicrobial activity against a wide range of microorganisms which provided great potential to be used as a packaging material to prolong the shelf life of food products. The antimicrobial properties of chitosan against some food-borne pathogens were investigated. Chitosan ranging from 2.5 to 10 mg ml-1 against some pathogenic bacteria (Escherichia coli TISTR 512, Bacillus cereus TISTR 035 Staphylococcus aureus TISTR 746 and Salmonella typhimurium TISTR 1470) and yeast (Candida albicans TISTR 5554) was determined. All tested bacteria could be inhibited by concentration of 10 mg ml-1 except C. albicans TISTR 5554. Cassava starch/chitosan films incorporated with lemongrass essential oil were examined in addition to its mechanical properties and antimicrobial stability during storage. Total bacteria and fungi counts were low during 12 weeks of storage and tensile strength decreased after 6 months of storage. However, elongation at break showed no significant difference during the storage process. The functional and structural groups of tested films did not change during 6 months of storage confirmed by fourier transform infrared (FTIR) analysis. Overall, these films have been recognized to be beneficial for application due to its stability, antimicrobial property and biodegradable nature.
The Mechanical Property, Antimicrobial Activity, Cassava Starch, Chitosan, Lemongrass Essential Oil, Storage
Plastic pollution has become one of the most serious environmental issues. Approximately 8 million tons of plastic waste escape into the oceans and it often contains additives that extend the life of products. Since the United Nations established the sustainable development goal 14 regarding life below water, all countries agreed to reduce global marine pollution, in particular plastics.1 Biodegradable packaging obtained from renewable sources such as starch offers an alternative option which has been widely used because of its low cost, good film-forming properties and environmental friendliness.2-6 Cassava is one of the most relatively inexpensive source to produce cassava starch that offers quite good film forming characteristics with similar mechanical properties with plastic, although the water vapor transmission rate is quite high.4,5,7,8 In addition, cassava is acknowledged one of Thailand’s most important economic crops and approximately 67% of global products was exported by Thailand in 2016.9 In 2018, Thailand was the world’s second biggest producer.10 To improve water vapor barrier, brittleness, and mechanical properties of the film, plasticizers such as glycerol and kaolinite clay were added to cassava starch films.5,6,11,12 Thus, the improved mechanical and barrier properties of starch film can act as a biodegradable package resulting in more sustainability that result in positive environmental impacts in consumer packaged goods.
The synthetic preservation has raised many health concerns and these have gained the interest for the use of natural antimicrobial compounds to improve food safety and shelf life.2 Chitosan has been investigated against a wide range of microorganisms for its antimicrobial activity.2,13-15 Chitosan with its cationic nature could interact with negatively charged microbial cell membranes causing cellular lysis.13,14,16 It has been shown that the antimicrobial activity of chitosan is more active in a coating solution than in a film matrix.17 The application of a chitosan coating showed the positive effect on the quality maintenance and extending the shelf life of postharvest blueberries18 and tomatoes.19 The addition of essential oils with chitosan coating were also effective in extending the shelf life of fresh blueberries20 and chilies.5 Moreover, the incorporated chitosan with starch film could enhance the mechanical properties and improve the surface structure.5,21 In addition, water vapor permeability and solubility of starch films are reduced with the addition of chitosan.17
The potential application of essential oils (EOs) as natural antimicrobial compounds has obtained attention as the demand increased for natural food preservatives.22-25 The antimicrobial effects of EOs against food-borne pathogens that cause food spoilage have been reported.5,22,25-27 The application of EOs as antimicrobials, besides being used as foods flavoring, have been demonstrated to increase the safety and shelf life of food products.23,25-27 It has been recorded that lemongrass EO (LEO) solution could reduce Salmonella enterica populations on leafy greens by dip treatment.27 Two major bioactive volatile components, terpene and aroma compounds, have been shown to correlate with antimicrobial or other biological activities.22,24 The antibacterial activity of cinnamon, oregano, thyme and clove EOs can be related to the principle bioactive constitutes notably carvacrol, cinnamaldehyde, eugenol and thymol.26 The general antimicrobial action of EOs and their bioactives, including lipophilic properties may be involved to the ability to penetrate bacterial membranes resulting in the inhibition of the functional contents of the cell.22,23 However, some limitations have been observed in their application due to the intense aroma which could result in sensory defects and the interaction of EOs constituents with food components.23 Therefore, the use of edible film to carry EOs would reduce the negative impact.7,8,15 Starch/chitosan film incorporated with thyme EO shows antioxidant and antibacterial properties with an increase in film yellowness.15 The cinnamon EO (CEO) incorporated into cassava starch film exhibits the antimicrobial activity and all added CEO is released in 2 h.8 The potential use of antimicrobial films for prolonging the shelf life of packed food has been suggested.7,8,15,25,28
Recently, cassava starch/chitosan/LEO films have been successfully made through the casting method.5 The application is successfully conducted using a coating system and the results show that it prolongs the chilies’ storage by lowering the total microbial growth and the chili’s weight loss. Moreover, the biodegradation of starch-based films using soil burial tests and study effects of biodegradation on soil microorganisms and growth of water convolvulus (Ipomoea aquatica) have been studied (unpublished data). The released chitosan significantly improves its shoot length and root weight (unpublished data). However, the shelf life of the films has not been studied. Therefore, this research aims to investigate the antimicrobial properties of chitosan against some food-borne pathogens and determine film mechanical properties and antimicrobial stability during storage.
Materials
The materials used in this research were cassava starch (Fish brand, Thailand), kaolinite clay (Amarin Ceramics Co., Ltd, Nonthaburi, Thailand), glycerol (Univar, Australia), lemongrass essential oil (AP Operation Co., Ltd, Chonburi, Thailand), chitosan (low molecular weight 75-85% deacetylated, Sigma Aldrich Co., Ltd, USA), Tween-80 (Univar, Australia).
Microorganisms and culture conditions
All microorganisms used in this research were previously listed.5 Bacterial strains were prepared in the nutrient broth (NB) and compared with 0.5 McFarland standard of 1 x 108 CFU ml-1. It was conducted by adding a bacterial suspension culture in 9 ml of sterilized NB media and repeated to achieve 1 x 108 CFU ml-1 for each strain respectively. These strains were then grown overnight at 37 °C and measured using spectrophotometry under the absorbance of 600 nm to determine its turbidity and compared with 0.5 McFarland standard equal turbidity to 1 x 108 CFU ml-1. Moreover, cell suspensions were diluted in its respective culture media to reach the concentration of 1 x 105 CFU ml-1 for antibacterial activity test. All fungal strains were grown in potato dextrose agar (PDA) media and incubated for 72 h at 30 °C.
Antimicrobial activity of chitosan
The stock chitosan solution (20 mg ml-1) was prepared according to Perdana et al.5 Chitosan was dissolved in 1% v/v of glacial acetic acid and pH was adjusted to 5.6 by adding 1 N NaOH.
Antibacterial activity of chitosan against bacterial strains was measured by the agar well diffusion method.29 The bacterial suspension of 100 µl was spread on the nutrient agar (NA). The 5 mm well were made on the agar and 50 µl chitosan solution with different concentrations of 10 mg ml-1, 5 mg ml-1 and 2.5 mg ml-1 were put aseptically on each well. In addition, the negative control (1% of acetic acid) and the positive control (tetracycline antibiotic 30 µg per disk) were put on the agar. Plates containing bacteria were incubated at 37 °C for 24 h. The inhibition zone was measured using a vernier caliper.
Antifungal activity was measured as described by Jiang et al.18 with modification and reported as the percent of inhibition of fungal growth. PDA medium mixed with different concentrations of chitosan was prepared. The plates were inoculated with 5 mm diameter plugs taken from 7 day old colonies, grown on PDA and incubated for 3 days at 30 °C. The plate without chitosan was used as control. The percent of inhibition was calculated by using the equation below:
![]() Dt is the diameter of growth area in the treatment plates and Dc is the diameter of growth in the control plate. The experiments were carried out in three replicates.
Dt is the diameter of growth area in the treatment plates and Dc is the diameter of growth in the control plate. The experiments were carried out in three replicates.
Films preparation
The starch solution was prepared using a solution-casting method described by Perdana et al.5 The gelatinized starch solution (3% w/v) was mixed with glycerol (20 wt% of starch) and kaolinite clay (4 wt% of starch). 10 mg ml-1 chitosan (40 wt% of starch and lemongrass EO (LEO) final concentration of 2% (v/v) were added to the solution to obtain cassava starch/chitosan/lemongrass essential oil (C/CS/LEO) film. The solutions mentioned above were cooled, casted and dried at 50 °C in an oven for two days.
Film shelf life studies
The C/CS/LEO films were punched with the diameter of 5 mm using a punch plier. At least six samples were kept in petri dishes at room temperature for 6 months. Total microbial count, mechanical properties and FTIR analysis were carried out to investigate the film stability and durability.
Total microbial count
The total mesophilic bacteria were determined by spreading them in NA plates, while fungi were grown in PDA. The 5 mm diameter of C/CS/LEO film was added in the 1 ml of 0.1% peptone. Further decimal dilutions were made and 100 µl of film suspension was spread into NA and PDA. Total number of bacteria and fungi were observed every two weeks for 12 weeks.
Mechanical properties
Mechanical properties of C/CS/LEO films were measured using tensile testing machine, model Tinius Olsen Horison test according to ASTM D624 as previously described.5
Fourier transform infrared analysis
FTIR spectra of the film were determined every two weeks by Bruker Tensor 27 FT-IR spectrometer (Bruker, Germany) with attenuated total reflectance (ATR) mode. All spectra were scanned five times at a wave number ranging from 400 to 4000 cm-1.
Statistical analysis
All experiments were carried out in triplicate and the mean values ± standard deviations were calculated. The results were analyzed using a one-way analysis of variance (ANOVA). Multiple comparisons were performed using Duncan’s multiple range test (DMRT) to determine the significant variation (P < 0.05).
Antimicrobial activity of chitosan
The effect of different concentrations of chitosan ranging from 2.5 to 10 mg ml-1 against some pathogenic bacteria, both Gram-positive and Gram-negative, and yeast C. albicans TISTR 5554 were determined by the agar well diffusion method (Table). All tested bacteria could be inhibited by chitosan at the concentration of 10 mg ml-1. However, 2.5 mg ml-1 of chitosan did not show an inhibition zone similar to the control (acetic acid). S. aureus TISTR 746 and B. cereus TISTR 035 were sensitive to a chitosan concentration of 10 mg ml-1. In contrast, E. coli TISTR 512 and S. typhimurium TISTR 1470 were more sensitive toward chitosan as shown with its inhibition zone by 5 mg ml-1. The antimicrobial potency of chitosan depends on degree of deacetylation, forms, its molecular weight, concentrations and microorganism strains.13,30-33 Our results showed that chitosan had a higher activity on Gram-negative than Gram-positive bacteria. In contrast to data obtained for chitosan with medium molecular weight and 75-85% deacetylation, in gel form, it exhibited more activity against the Gram-positive S. aureus than Gram-negative E. coli based on turbidity and well inhibition zone.14 However, there is no conclusive data on whether the chitosan had higher activity in Gram-positive or Gram-negative bacteria.13 Zheng and Zhu33 have reported the antimicrobial activity of chitosan using different molecular weights and found that when increasing molecular weight of chitosan, the antimicrobial activity for S. aureus increased, while it decreased for E. coli. Chitosan used in our research was low molecular weight and it showed a stronger effect for Gram-negative bacteria. Liu et al.34 have reported the bactericidal activity of chitosan and showed that chitosan increases the permeability of the outer and inner membranes resulting in the disruption of bacterial cell membranes. Previous studies showed that the interaction between protonated amine groups and phosphoryl groups of the phospholipid components of cell membrane caused cell damage, therefore, high degree of deacetylation of chitosan is more effective than those with low degree.13,31,34,35 Chitosan did not inhibit C. albicans TISTR 5554 at all tested concentrations (Table). Chitosan starting at 666 µg ml-1 or more inhibited growth of C. albicans strain ATCC 10231.36 Therefore, the inhibition effects depend on the medium used in addition to the amount of cells.
Table:
Antimicrobial activity of chitosan (2.5, 5 and 10 mg ml-1) against different pathogenic microorganisms.
| Test Microorganisms | Inhibition zone (mm) | |||
|---|---|---|---|---|
| 0 | 2.5 mg ml-1 | 5 mg ml-1 | 10 mg ml-1 | |
| E. coli TISTR 512 | 0.00±0a | 0.00±0a | 3.13±0.55b | 5.30±0.53c |
| S. typhimurium TISTR 1470 | 0.00±0a | 0.00±0a | 3.53±0.31b | 4.33±0.85b |
| B. cereus TISTR 035 | 0.00±0a | 0.00±0a | 0.00±0a | 4.77±1.99b |
| S. aureus TISTR 746 | 0.00±0a | 0.00±0a | 0.00±0a | 3.33±0.21b |
| C. albicans TISTR 5554 | 0.00±0a | 0.00±0a | 0.00±0a | 0.00±0a |
Values are mean±SD of three independent replications. Mean values with the same letter in the same row are not significantly different from each other (ANOVA, DMRT, P > 0.05)
In Figure 1, antifungal activities of chitosan against the fungi Aspergillus niger TISTR 3130 and Mucor ruber TISTR 3006 were carried out. There was no significant difference among the concentrations of chitosan used for A. niger TISTR 3130 (Figure 1A). However, different concentrations of chitosan significantly increased the inhibition of M. ruber TISTR 3006 (Figure 1B). These results demonstrated that the antifungal effect of chitosan against M. ruber TISTR 3006 was concentration-dependent. Consistently, the dose-dependent inhibitory action of chitosan was previously reported by Hemalatha et al.37 and Raafat and Sahl.31 The study of antifungal activity for four important food pathogenic fungi, A. niger, A. flavus, Fusarium sp. and Penicillium sp. showed that chitosan at 0.1% exhibited inhibitory action on fungal growth from day 1 while 5% concentration resulted in growth arrest.37 In our study, at 10 mg ml-1, chitosan showed the most significant inhibition (Figure 1B). Therefore, this concentration was chosen to be incorporated with cassava starch film.
Figure 1. % Inhibition of chitosan at concentrations of 0, 2.5, 5 and 10 mg ml-1 for A. niger TISTR 3130 (A) and M. ruber TISTR 3006 (B) under radial growth method for 3 days of incubation on PDA agar. Each of the small letters (a,b,c,d) indicate statistical significance (P < 0.05), analyzed by Duncan’s Multiple Range Test. The values are given as mean ± SE
Film shelf life studies
Total microbial count
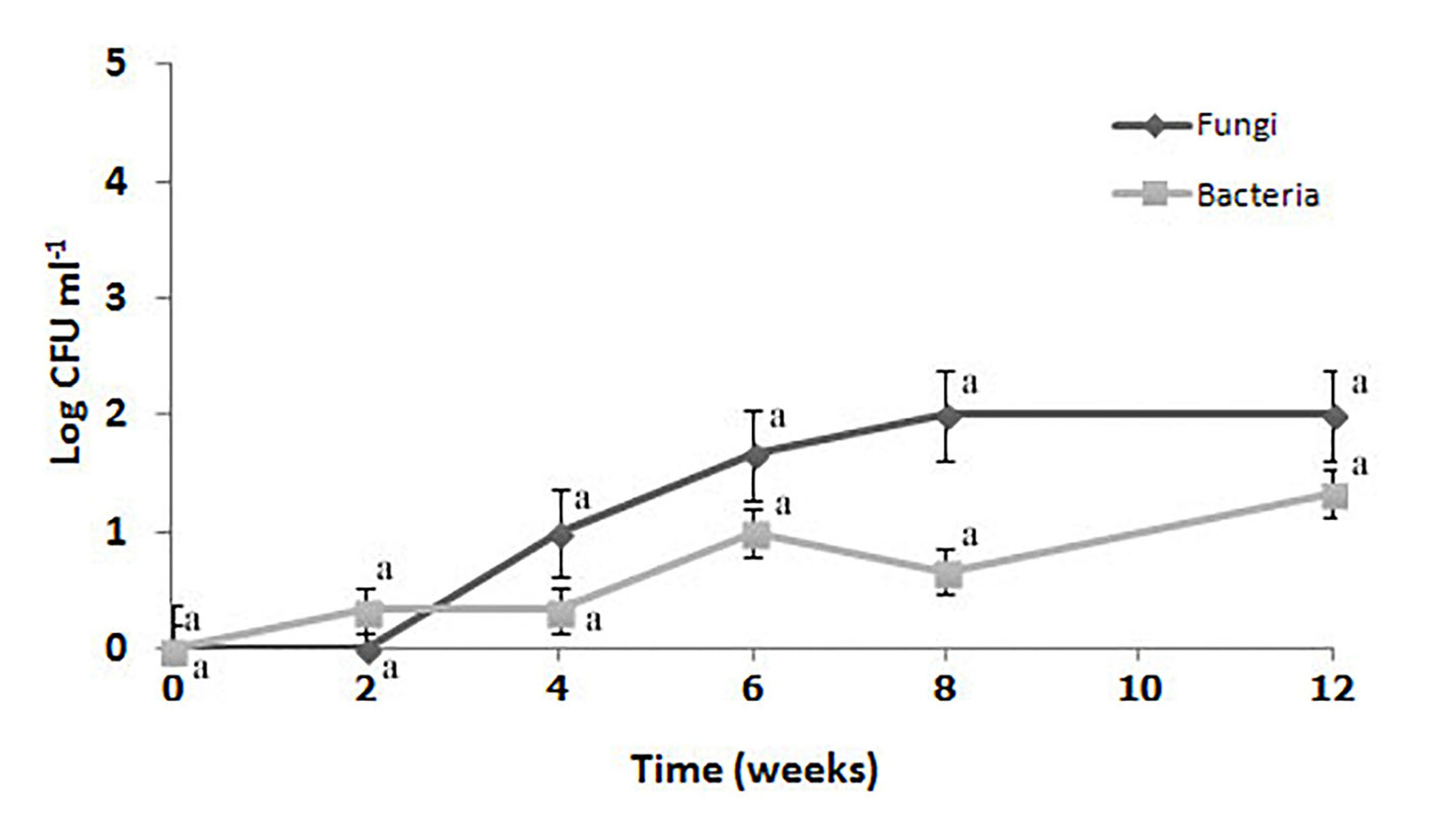
There have been several studies conducted on the antimicrobial activity of film incorporated with chitosan and/or EOs.5,28,38 Hernandez-Garcia et al.28 reported that antimicrobial activity of biodegradable films incorporated with antimicrobial compounds to control the growth of pathogenic or spoilage bacteria have been wildly studied. In addition, the application of antimicrobial films could extend the postharvest shelf life while maintaining the overall produce quality.5,19,25,39-41 Based on the current literature, this is the first report that presents the result of total microbial count on film during storage. The total microbial count has been carried out during 12 weeks of observation as shown by Figure 2. From week 3-12, the total number of bacteria was lower than the fungi number. However, bacterial and fungal counts were not significantly different (P > 0.05) during 12 weeks of storage. The number of microbial load on cassava starch film has not been determined.
Figure 2. Total microbial count on C/CS/LEO film during 12 weeks of storage at room temperature. The values are mean ± SE. The same vertical letters are not significantly different from each other (Duncan’s Multiple Range Test, P < 0.05)
Mechanical properties stability during storage
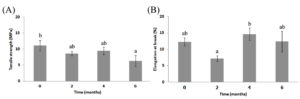
During storage, the shelf life of film was promptly examined by changes in their mechanical properties. The tensile strength and elongation at break during 6 months of storage varied between 6.29±1.72 to 11.06 ± 1.61 MPa and 7.18 ± 0.81 to 12.36 ± 3.14 % respectively (Figure 3). The film’s tensile strength gradually decreased during longer storage times. After storage for 6 months, tensile strength was reduced significantly 43.13% (P < 0.05) compared to 0 month (Figure 3A). However, there was no significant difference between 2 and 4 months of storage. Elongation at break also showed no significant difference during the storage (Figure 3B). The additives in the film affected the film stability during storage.42,43 Therefore, it is a major challenge to retain the good functional properties of film during storage. Previous studies showed that the appearance or the mechanical properties of whey protein isolate based films plasticized with glycerol did not changed during storage for 30 weeks.43 In contrast, films plasticized with sorbitol showed lower moisture content when compared to films plasticized with glycerol and resulted in films being harder and less flexible.43 Mechanical properties of corn-wheat starch/zein bilayer films were studied during storage for 150 days.42 Both tensile strength and elongation at break decreased, while storage temperature and moisture are important factors, affecting film’s mechanical properties.42
Figure 3. Mechanical properties of C/CS/LEO film on tensile strength (A) and elongation at break (B) during 6 months of storage. Values shown are mean ± SE. Bars with different letters show significantly different (P < 0.05) when analyzed by Duncan’s Multiple Range Test
Fourier transform infrared (FTIR) analysis
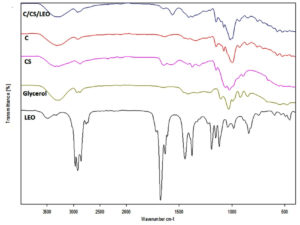
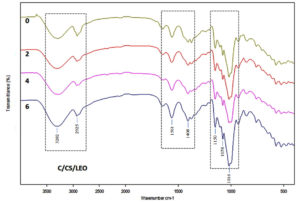
The ATR/FTIR spectra of different films (C, CS and C/CS/LEO), glycerol and LEO are presented in Figure 4. Similar trends in FTIR spectra of C/CS/LEO were observed during storage of 6 months (Figure 5). The peak around 3300 cm-1 and 1100-1000 cm-1 corresponded to the stretching of -OH group and C-OH belonging to starch, glycerol and water.44 Recently, studies on soil burial degradation of C/CS/LEO reported that the peak around 3300 cm-1 gradually disappeared in 20 days (unpublished data). The peak around 1150 cm-1, which was associated with the C-O-C in the starch’s glycosidic linkage, was decreased by the activity of a-amylase.45 Three major peak regions, highlighted with the dashed lines in Figure 5, gradually disappeared in 20 days due to soil burial degradation (unpublished data). However, C/CS/LEO film showed less reduction than C/CS, while C film was degraded entirely within 5 days (unpublished data). The addition of essential oil in film formulation could reduce water vapor permeability, possess antimicrobial activity and contribute to the film network compactness.4,5,27,46 Our FTIR analysis confirmed that the functional and structural groups in C/CS/LEO film did not change during 6 months of storage.
Figure 4. FTIR spectra of lemongrass EO (LEO), glycerol, cassava starch/chitosan film (CS), cassava starch film (C) and cassava starch/chitosan/lemongrass essential oil (C/CS/LEO)
Our investigation has confirmed that chitosan, as a natural antimicrobial agent, has the potential for future applications. Film shelf life stability studies have indicated that film mechanical properties did not change during 6 months of storage. The antimicrobial agents incorporated in starch film potentially extended the film’s shelf life. Our studies that investigated film stability, durability and shelf life provide important data on the potential application of the films. Therefore, C/CS/LEO films which have at least 6 months of shelf life, can be easily degraded in compost and has shown great promise for food packaging application.
ACKNOWLEDGMENTS
The authors would like to thank Graduate School of Prince of Songkla University, Thailand for their support. Authors are also thankful to Sukallaya Hemmanee for technical support, Dr. Christina C. Spevak and Ana Luiza Ramos Crawford for language correction.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
MIP carried out the experiments. SP, JR and ML designed and supervised the experiments. ML drafted and revised the manuscript. All authors read and approved the final manuscript for publication.
FUNDING
This research was supported by Thailand’s Education Hub for Asian Countries contract No. TEH-AC 079 /2017 and research grant for thesis, Graduate School, Prince of Songkla University to MIP.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This article does not contain any studies with human participants or animals performed by any authors.
- Conserve and sustainably use the oceans, seas and marine resources for sustainable development. https://sdgs.un.org/goals/goal14. Accessed November 21, 2021.
- Dutta PK, Tripathi S, Mehrotra GK, Dutta J. Perspectives for chitosan based antimicrobial films in food applications. Food Chem. 2009;114(4):1173-1182.
Crossref - Fonseca-Garcia F, Jimenez-Regalado EJ, Aguirre-Loredo RY. Preparation of a novel biodegradable packaging film based on corn starch-chitosan and poloxamers. Carbohydr Polym. 2021;251:1117009.
Crossref - Jimenez A, Fabra MJ, Talens P, Chiralt A. Edible and biodegradable starch films: A Review. Food Bioprocess Technol. 2012;5(6):2058-2076.
Crossref - Perdana MI, Ruamcharoen J, Panphon S, Leelakriangsak M. Antimicrobial activity and physical properties of starch/chitosan film incorporated with lemongrass essential oil and its application. LWT – Food Sci Technol. 2021;141:110934.
Crossref - Ruamcharoen J, Munlee R, Ruamcharoen P. Improvement of water vapor barrier and mechanical properties of sago starch-kaolinite nanocomposites. Polym Compos. 2019:1-9.
Crosref - Resianingrum R, Atmaka W, Khasanah LU, Utami R, Praseptiangga D. Characterization of cassava starch-based edible film enriched with lemongrass oil (Cymbopogon citratus). Nusantara Bioscie. 2016;8(2):278-282.
Crossref - Souza AC, Goto GEO, Mainardi JA, Coelho ACV, Tadini CC. Cassava starch composite films incorporated with cinnamon essential oil: Antimicrobial activity, microstructure, mechanical and barrier properties. LWT – Food Sci Technol. 2013;54(2):346-352.
Crossref - Thailand Board of Investment. Leading the world in cassava production. Thailand Investment Review. 2017. https://www.boi.go.th/upload/content/TIR-SEP2017_81650.pdf. Accessed November 26, 2021.
- Sowcharoensuk C. Industry outlook 2020-2022: Cassava industry. Updated May 11, 2020. https://www.krungsri.com/en/research/industry/industry-outlook/agriculture/cassava/IO/io-cassava-20. Accessed November 26, 2021.
- Basiak E, Lenart A, Debeaufort F. How glycerol and water contents affect the structural and functional properties of starch-based edible films. Polymers. 04/08 2018;10(4):412.
Crossref - Souza AC, Benze R, Ferrao ES, Ditchfield C, Coelho ACV, Tadini CC. Cassava starch biodegradable films: Influence of glycerol and clay nanoparticles content on tensile and barrier properties and glass transition temperature. LWT – Food Sci Technol. 2012;46(1):110-117.
Crossref - Goy RC, de Britto D, Assis OBG. A review of the antimicrobial activity of chitosan. Polimeros. 2009;19(3):241-247.
Crossref - Goy RC, Morais STB, Assis OBG. Evaluation of the antimicrobial activity of chitosan and its quaternized derivative on E. coli and S. aureus growth. Rev Bras Farmacogn. 2016;26(1):122-127.
Crossref - Shaaban HA, Mahmoud KF. In-vitro antibacterial and antioxidant properties of starch/chitosan edible composite film incorporated with thyme essential oil. J Arab Soc Med Res. 2014;9(2):54-61.
- Shapi’i RA, Othman SH, Nordin N, Basha RK, Naim MN. Antimicrobial properties of starch films incorporated with chitosan nanoparticles: In vitro and In vivo evaluation. Carbohydr Polym. 2020;230:115602.
Crossref - Vasconez MB, Flores SK, Campos CA, Alvarado J, Gerschenson LN. Antimicrobial activity and physical properties of chitosan-tapioca starch based edible films and coatings. Food Res Int. 2009;42(7):762-769.
Crossref - Jiang H, Sun Z, Jia R, Wang X, Huang J. Effect of chitosan as an antifungal and preservative agent on postharvest blueberry. J Food Qual. 2016;39(5):516-523.
Crossref - Mior Azmai WNS, Abdul Latif NS, Md Zain N. Efficiency of edible coating chitosan and cinnamic acid to prolong the shelf life of tomatoes. J Trop Resour Sustain Sci. 2019;7(1):47-52.
Crossref - Sun X, Narciso J, Wang Z, Ference C, Bai J, Zhou K. Effects of chitosan-essential oil coatings on safety and quality of fresh blueberries. J Food Sci. 2014;79(5):955-960.
- Shapi’i RA, Othman SH. Effect of concentration of chitosan on the mechanical, morphological and optical properties of tapioca starch film. Int Food Res J. 2016;23:S187-S193.
- Calo JR, Crandall PG, O’Bryan CA, Ricke SC. Essential oils as antimicrobials in food systems – A review. Food Control. 2015;54:111-119.
Crossref - Khorshidian N, Yousefi M, Khanniri E, Mortazavian AM. Potential application of essential oils as antimicrobial preservatives in cheese. Innov Food Sci Emerg Technol. 2018;45:62-72.
Crossref - Phanthong P, Lomarat P, Chomnawang MT, Bunyapraphatsara N. Antibacterial activity of essential oils and their active components from Thai spices against foodborne pathogens. Science Asia. 2013;39:472-476.
Crossref - Shen Y, Zhou J, Yang C, et al. Preparation and characterization of oregano essential oil-loaded Dioscorea zingiberensis starch film with antioxidant and antibacterial activity and its application in chicken preservation. Int J Biol Macromol. 2022;212:20-30.
Crossref - Mith H, Dure R, Delcenserie V, Zhiri A, Daube G, Clinquart A. Antimicrobial activities of commercial essential oils and their components against food-borne pathogens and food spoilage bacteria. Food Sci Nutri. 2014;2(4):403-416.
Crossref - Moore-Neibel K, Gerber C, Patel J, Friedman M, Ravishankar S. Antimicrobial activity of lemongrass oil against Salmonella enterica on organic leafy greens. J Appl Microbiol. 2012;112(3):485-492.
Crossref - Hernandez-Garcia E, Vargas M, Gonzalez-Martinez C, Chiralt A. Biodegradable antimicrobial films for food packaging: Effect of antimicrobials on degradation. Foods. 2021;10(6):1256.
Crossref - Harrigan WF, MaCance EM. Laboratory Methods in Food and Dairy Microbiology. 3rd Edition ed. Academic Press; 1986.
- No HK, Park NY, Lee SH, Meyers SP. Antibacterial activity of chitosans and chitosan oligomers with different molecular weights. Int J Food Microbiol. 2002;74(1-2):65-72.
Crossref - Raafat D, Sahl HG. Chitosan and its antimicrobial potential-a critical literature survey. Microb Biotechnol. 2009;2(2):186-201.
Crossref - Savard T, Beaulieu C, Boucher I, Champagne CP. Antimicrobial action of hydrolyzed chitosan against spoilage yeasts and lactic acid bacteria of fermented vegetables. J Food Prot. 2002;65(5):828-833.
Crossref - Zheng L-Y, Zhu J-F. Study on antimicrobial activity of chitosan with different molecular weights. Carbohydr Polym. 2003;54(4):527-530.
Crossref - L Liu H, Du Y, Wang X, Sun L. Chitosan kills bacteria through cell membrane damage. Int J Food Microbiol. 2004;95(2):147-155.
Crossref - Tsai GUO, Su W-H, Chen H-C, Pan C-L. Antimicrobial activity of shrimp chitin and chitosan from different treatments. Fish Sci. 2002;68(1):170-177.
Crossref - Pena A, Sanchez NS, Calahorra M. Effects of chitosan on Candida albicans: conditions for its antifungal activity. BioMed Res Int. 2013;2013:527549.
Crossref - Hemalatha T, UmaMaheswari T, Senthil R, Krithiga G, Anbukkarasi K. Efficacy of chitosan films with basil essential oil: perspectives in food packaging. J Food Meas Charact. 2017;11:2160-2170.
Crossref - Syafiq R, Sapuan SM, Zuhri MRM. Antimicrobial activity, physical, mechanical and barrier properties of sugar palm based nanocellulose/starch biocomposite films incorporated with cinnamon essential oil. J Mater Res Technol. 2021;11:144-157.
Crossref - Perdones A, Escriche I, Chiralt A, Vargas M. Effect of chitosan-lemon essential oil coatings on volatile profile of strawberries during storage. Food Chem. 2016;197:979-986.
Crossref - Valdes A, Ramos M, Beltran A, Jimenez A, Garrigos MC. State of the art of antimicrobial edible coatings for food packaging applications. Coatings. 2017;7(4):56.
Crossref - Yuan G, Chen X, Li D. Chitosan films and coatings containing essential oils: The antioxidant and antimicrobial activity, and application in food systems. Food Res Int. 2016;89:117-128.
Crossref - Chen C, Du Y, Zuo G, Chen F, Liu K, Zhang L. Effect of storage condition on the physico-chemical properties of corn-wheat starch/zein edible bilayer films. Royal Soc Open Sci. 2020;7(2):191777.
Crossref - Oses J, Fernandez-Pan I, Mendoza M, Mate JI. Stability of the mechanical properties of edible films based on whey protein isolate during storage at different relative humidity. Food Hydrocoll. 2009;23(1):125-131.
Crossref - Medina Jaramillo C, Gutierrez TJ, Goyanes S, Bernal C, Famá L. Biodegradability and plasticizing effect of yerba mate extract on cassava starch edible films. Carbohydr Polym. 2016;151:150-159.
Crossref - Torres FG, Troncoso OP, Torres C, Diaz DA, Amaya E. Biodegradability and mechanical properties of starch films from Andean crops. Int J Biol Macromol. 2011;48(4):603-606.
Crossref - Ojagh SM, Rezaei M, Razavi SH, Hosseini SMH. Development and evaluation of a novel biodegradable film made from chitosan and cinnamon essential oil with low affinity toward water. Food Chem. 2010;122(1):161-166.
Crossref
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.