ISSN: 0973-7510
E-ISSN: 2581-690X
Diabetes mellitus is a complex metabolic disorder that affects all body systems of an individual. Siddha system of medicine which has its origin in South India terms this multiorgan disorder as Madhumegam and has an enormous collection of antidiabetic drugs that are time-tested. Avaarai kudineer is one such polyherbal antidiabetic formulation that is safe and effective in the management of diabetes. Since Siddha system offer holistic approach in the management of diseases, the present work was carried out to evaluate the antimicrobial effect of this antidiabetic formulation. The results of phytochemical analysis and antimicrobial action of this formulation Avaarai kudineer (AK) is encouraging as it offers mutual benefit to the patients both in the management of diabetes and also in the prevention and treatment of diabetic complications that are due to infections.
Siddha, Diabetes mellitus, Avarai kudineer, Antimicrobial study, Phytochemicals.
Diabetes mellitus (DM) is considered as one of the largest emerging health threat to mankind. This metabolic disorder is estimated to increase to 380 million of world’s population by the year 20251. Though it is a non-communicable disease as such , the presence of decreased T cell–mediated immune response and impaired function of neutrophils is responsible for diabetes associated complications and infections2,3. Despite recent advances in the diabetic management, the increase in risk of infections is well established due to the relationship between glycemia and infections4-10. Previous studies show that diabetic population have an increased risk of urinary tract infection, skin and mucous membrane infections including diabetic foot ulcers and candida infections11. Since antibiotics can cause marked alteration in gut microbiota and further increases the risk of disturbance in glucose homeostasis the scenario has created a search for natural and alternative methods for the management of diabetic complications associated with infection12. The present study was proposed to carry out antimicrobial effect of a reputed polyherbal Siddha formulation Avaarai kudineer whose safety and efficacy are already proven through recent researches13.
Pathology of Infection in Diabetes
Neutrophils play an important role in host inflammatory response against infections. The chemotactic activity of neutrophils is found to be significantly lower in diabetics than in healthy individuals14. Hence hyperglycemic condition was found to have significant correlation with impaired phagocytosis, decrease in release of lysosomal enzymes, reduced production of reactive oxygen species 15.
The reduction in the polymorphonuclear leukocyte transmigration through the endothelium is attributed to the hyperglycemic environment which inhibits glucose-6-phosphate dehydrogenase (G6PD). Moreover, previous studies also reported that the increased glycation also inhibits the production of interferon gamma (IFN-g) and tumor necrosis factor (TNF)-a by T cells and the secretion of interleukin-1 (IL-1) and IL-6 in response to stimulation by lipopolysaccharides by the inflammatory cytokines Mononuclear cells and monocytes is lessened. All these factors play an important role in the sucsceptibility of infections16,17.
Aavirai Kudineer
Avirai Kudineer is one of the most widely used and time-tested Siddha anti-diabetic formulation in south India18. It is a polyherbal formulation consisting of seven herbal ingredients. The formulation has been taken from the classical Siddha Literature “Theraiyar Kudineer”19 and it is also found in the Siddha literature “Gunapadam mooligai vaguppu” (Siddha Materia medica)20.There are several well established research evidences of each of the ingredient of Aavaarai kudineer having antimicrobial activity. Hence the formulation was also analysed for antimicrobial effect so that this single formulation could target the issue of both diabetes and its associated infections.
Preparation of the Trial Drug – Aavirai kudineer (AK)
Ingredients:
1.Cassia auriculata (Aavirai)
2.Cassia fistula (Kondrai)
3.Syzygium jambos (Naval)
4.Olax scandens (Kadalazhinjil)
5.Saussurea lappa (Koshtam)
6.Terminalia arjuna (Marutham pattai)
7.Cyperus rotundus (Korai kizhangu)
Preparation of the Decoction(Kudineer)
The above ingredients 1-7 were dried in shade and coarsely grounded. 20gram of the above mixture was boiled in 80ml, 160ml and 320ml of water and each of them were reduced down to 20ml respectively and filtered. the above samples were named as AK-A, AK-B and AK-C respectively and were subjected for phytochemical and antimicrobial analysis.
Phytochemical screening
The above prepared decoction AK-A, AK-B and AK-C were analysed for the presence of various secondary metabolites such as tannins,alkaloid, flavanoid, steroid, glycosides, saponins, proteins, terpenoids and phenols using standard procedures ( Brain and Turner, 1975; Harborne, 1992)21,22.
Antimicrobial activity
The human pathogenic bacteria such as Bacillus subtilis , Staphylococcus aureus Enterococcus faecalis, Pseudomonas aeruginosa, Escherichia coli Klebsiella pneumoniae and fungal strains of Candida albicans and Aspergillus niger were obtained from CLATR, Sathyabama university,Tamilnadu and were maintained in Nutrient agar slant at 4oC for experimental studies.
Antimicrobial Screening of Plants’ Extracts by Disc diffusion method (zone of inhibition measurement)
The antibacterial activities of all the samples (AK-A, AK-Band AK-C) were carried out by disc diffusion method. The concentrations of the test compounds were used at the concentration of 100, 200, 300 µg. The target microorganisms were cultured in Mueller–Hinton broth (MHB). After 24 h the suspensions were adjusted to standard sub culture dilution. The Petri dishes containing Muller Hinton Agar (MHA) medium were cultured with diluted bacterial strain. Disc made of Whatman No.1, diameter 6 mm was pre-sterilized and was maintained in aseptic chamber. Each concentration was injected to the sterile disc papers. Then the prepared discs were placed on the culture medium. Standard drug streptomycin (20µg) was used as a positive reference standard to determine the sensitivity of each microbial species tested. Then the inoculated plates were incubated at 37 oC for 24 h. The diameter of the clear zone around the disc was measured and expressed in millimeters as its anti-microbial property. The results were depicted in Table.
Table (1):
Phytochemical Analysis of Avaarai kudineer (AK)
S.No |
Test Sample |
Steroids |
Alkaloids |
Flavonoid |
Glycosides |
Terpenoids |
Tannins |
Polyphenol |
Protein |
|---|---|---|---|---|---|---|---|---|---|
1. |
AK-A |
– |
+ |
+ |
+ |
+ |
+ |
+ |
+ |
2. |
AK-B |
– |
+ |
+ |
– |
+ |
+ |
+ |
– |
3. |
AK-C |
– |
+ |
+ |
– |
+ |
+ |
+ |
– |
(+) -> Presence (-) -> Absence
The Avarai kudineer decoction was prepared by extraction method in the serial dilutions of ¼,1/8 and 1/16 (AK-A,AK-B and AK-C) and three concentrations of 100 µg, 200 µg and 300 µg respectively. Among the tested dilutions, AK-A was found to have maximum zone of inhibition against gram positive bacteria Staphylococcus aureus (19mm at 300 µg) and Enterococcus faecalis (16mm at 300 µg) when compared to AK-B and AK-C which was almost equal to that of the standard drug. However, AK-B exhibited higher zone of inhibition against the gram positive microbe Bacilus subtilis (25mm) which was equal to that of the zone of inhibition of standard drug streptomycin. AK-B showed a moderate zone of inhibition against all the gram negative bacteria Pseudomonas aeruginosa ( 9mm at 300 µg), E.coli(15mm at 300 µg ) and Klebsiella pneumoniae (10mm at 300 µg ). The sample AK-A showed moderate zone of inhibition against both the tested fungal strains of Candida albicans (16mm at 300 µg)and Aspergillus niger( 10mm at 300 µg). The resultsof zone of inhibition are shown in (Table-2) and represented in figure 1&2.
Table (2):
Measurement of Zone of Inhibition data of Anti-bacterial activity.
| Sample code Klebsiellapn- |
Zone of inhibition (mm) | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bacillus | Staphylococcus | Enterococcus | Pseudomonas | Escherichia | ||||||||||||||
| subtilis | aureus | faecalis | aeruginosa | coli | eumoniae | |||||||||||||
| 100 | 200 | 300 | 100 | 200 | 300 | 100 | 200 | 300 | 100 | 200 | 300 | 100 | 200 | 300 | 100 | 200 | 300 | |
| µg | µg | µg | µg | µg | µg | µg | µg | µg | µg | µg | µg | µg | µg | µg | µg | µg | µg | |
| AK-A | 7 | 12 | 16 | 12 | 16 | 19 | – | 12 | 16 | – | – | 8 | – | 9 | 14 | – | – | 7 |
| AK-B | 10 | 15 | 25 | – | 7 | 14 | – | 10 | 14 | – | – | 9 | – | 8 | 15 | – | 7 | 10 |
| AK-C | – | – | 11 | – | 7 | 12 | – | – | 10 | – | – | 7 | – | 7 | 13 | – | – | – |
| Streptomycin 20 µg | 25 | 26 | 18 | 12 | 23 | 19 | ||||||||||||
– = Not active
Table (3):
Zone of Inhibition data of Anti-Fungal activity
| Sample code | Zone of inhibition (mm) | |||||
|---|---|---|---|---|---|---|
| Candida albicans | Aspergillusniger | |||||
| 100
µg |
200
µg |
300
µg |
100
µg |
200
µg |
300
µg |
|
| AK-A | 10 | 14 | 16 | – | – | 10 |
| AK-B | 8 | 10 | 10 | – | 10 | 10 |
| AK-C | 8 | 10 | 10 | 8 | 10 | 10 |
| Fluconazole (20µg) | 20 | 26 | ||||
- = Not active
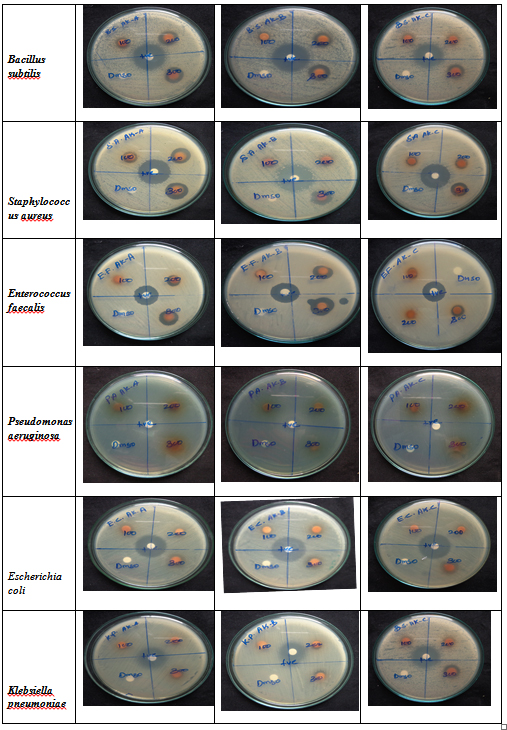
Fig. 1. Measurement of Zone of Inhibition showing Anti-bacterial activity
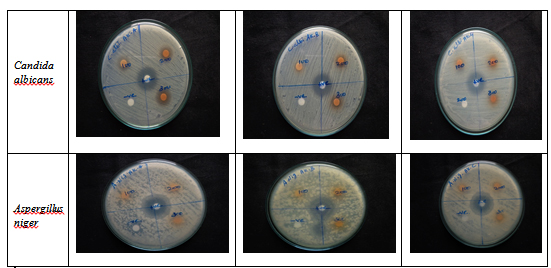
Fig. 2. Anti-fungal action of Avaarai kudineer
Antimicrobial activity of individual ingredients of the polyherbal formulation Aavarai kudineer has been extensively studied in previous researches in recent years. The ingredient Cassia auriculata is said to exhibit antibacterial action due to the presence of flavanoids which is a hydroxylated phenolic substitute23. Cassia fistula contains a rich source of tannin which has antimicrobial activity against various human pathogens24. Costunolide a sesquiterpene from costus speciosus , Mangiferin, kotanelol and salacinol from Salacia reticulata and the phytochemical luteolin fron Terminalia arjuna have been identified to possess significant antimicrobial action against various gram positive and gram negative bacteria and fungus25-29. As a consequence of these researches Avaarai kudineer which is a content of all these seven ingredients has been screened for antimicrobial activity was found to contain the phytochemicals such as alkaloids, flavanoids, saponins, tannins, protein , triterpenoids and phenolic compounds that are responsible for antimicrobial action.
Infections caused by Bacillus species include wound and burn infections, ear infections, ophthalmitis, urinary tract infections and respiratory infections and most of these infections tend to occur as secondary infections in immunodeficient states30.In patients with normal anatomic structure and function more than 90% of acute UTI’s are caused by certain strains of E coli. The most common causes of complicated UTI resulting from anatomic obstructions, or from catheterization are E. coli, Klebsiella pneumoniae, Enterococcus sp., Pseudomonas aeruginosa. In rare cases Candida albicans can cause UTI in diabetic patients. Nearly 85% of diabetic patients with uncontrolled glycemic index are prone to superficial fungal foot infection (SFFI) which is a serious sequelae for the risk of lower limb cellulitis. Aspergillus niger was found to be the most common pathogen followed by Candida albicans for the cause of fungal foot infections in diabetics31.
Although diabetes is considered as a metabolic disorder and the modern day treatment of hyperglycemic drugs target on reducing the glycemic index, Siddha system of medicine has a distinct difference in the approach and treatment of diabetes. This can be attributed to the fact that Siddha medicines are designed in a highly specialized manner of not only treating a specific disease but they aim at having a totally rejuvenating effect on the entire body constituents. Therefore the Siddha antidiabetic formulations concentrate on healing the impaired function of liver which is the prime source of all metabolic functions and pacifies the altered pitha dosham which is also responsible for other associated diabetic complications such as infections.
In this study the Siddha formulation Avaarai kudineer was found to have antimicrobial action against the tested gram positive and gram negative pathogens . It was also found to have antifungal action against Aspergillus niger and candida albicans. The antimicrobial and antifungal action was more in AK-A(1/4 dilution) when compared with the other two samples of AK-B (1/8 dilution)and AK-C(1/16 dilution). It can also be concluded that the presence of phytochemicals such as flavanoids, tannins, saponins, triterpenoids, alkaloids etc., which were analysed through phytochemical analysis may be responsible for the anti diabetic action of this polyherbal formulation as well as its antimicrobial action. Hence this compound herbal formulation can act as a single medicine in targeting hyperglycemia along with the treatment and prevention of microbial infections associated with diabetes.
ACKNOWLEDGMENTS
This project is funded by The Tamilnadu state council for Science and Technology (DST), Chennai. The authors thankfully acknowledge this.
- Atkins RC, Zimmet P. Diabetic kidney disease: Act now or pay later Saudi J Kidney Dis Transpl 2010; 21:217-21.
- Shah BR, Hux JE. Quantifying the risk of infectious diseases forpeople with diabetes.Diabetes Care., 2003, 26: 510–3.
- Delamaire M, Maugendre D, Moreno M, Le Goff M-C, Allannic H, Genetet B. Impaired leucocyte functions in diabetic patients. Diabet Med., 1997, 14: 29–34.
- Balasoiu D, van Kessel KC, van Kats-Renaud HJ,ColletTJ,Hoepelman AIM. Granulocyte function in women with diabetes and asymptomatic bacteriuria. Diabetes Care., 1997, 20: 392–5.
- Calvet HM, Yoshikawa TT. Infections in diabetes.InfectDisClinNorth Am., 2001, 15: 407–21.
- Pozzilli P, Leslie RDG. Infections and diabetes: mechanisms and prospects for prevention. Diabet Med., 1994, 11: 935–41.
- Boyko EJ, Fihn SD, Scholes D, Chen CL, Normand EH, Yarbro P. Diabetes and the risk of urinary tract infection among postmenopausal women. Diabetes Care., 2002, 25: 1778–83.
- Hu KK, Boyko EJ, Scholes D, et al. Risk factors for urinary tract infections in postmenopausal women. Arch Intern Med., 2004, 164: 989–93.
- Patterson JE, Andriole VT. Bacterial urinary tract infections in diabetes. Infect Dis Clin North Am., 1997, 11: 735–50.
- Joshi N, Caputo GM, Weitekamp MR, Karchmer AW. Infections in patients with diabetes mellitus. N Engl J Med., 1999, 341: 1906–12.
- Kristian Hallundbæk Mikkelsen, Filip Krag Knop, Morten Frost, Jesper Hallas, and Anton Pottegård. Use of Antibiotics and Risk of Type 2 Diabetes: A Population-Based Case-Control Study. J Clin Endocrinol Metab., October 2015, 100(10): 3633–3640.
- V.Bhavapriya, S.Kalpana, S.Govindasamy, T.Apparanantham. Biochemical studies on hypoglycemic effect of Aavarai kudineer A Herbal formulation in alloxan diabetics rats, Indian journal of experimental biology., 2001, 39: 925-928.
- Tan JS, Anderson JL, Watanakunakorn C, Phair JP. Neutrophil dysfunction in diabetes mellitus. J Lab Clin Med., 1975, 85: 26-33.
- Bagdade JD, Nielson KL, Bulger RJ. Reversible abnormalities in phagocytic function in poorly controlled diabetic patients. Am J Med Sci., 1972, 263: 451-456.
- Mowat A, Baum J. Chemotaxis of polymorphonuclear leukocytes from patients with diabetes mellitus. N Engl J Med., 1971, 284: 621627.
- Nielson CP, Hindson DA. Inhibition of polymorphonuclear leukocyte respiratory burst by elevated glucose concentrations in vitro. Diabetes., 1989; 38: 1031-1035.
- Rajalakshmi K, Christian GJ, Shanmuga Priya P, Jeeva Gladys R. Validation of Anti-diabetic Potential of Avirai kudineer a Siddha herbal formulation-A Review. IOSR Journal of Dental and Medical Sciences (14)7; 7-15.
- 18. Veluchamy, S.Jagajothipandian, .K. Meenakshi sundaramoorthy, Theraiyar kudineer (CCRAS, 2nd edition, 1996), 40.
- K.S. Murugesa mudhaliyar , Siddha materia medica, Medicinal plant division ((Directorate ISM&H publications), 2006; 86.
- Brain KR, Turner TD. Wright – Scientechnica. 1st Ed. Bristol: 1975. Practical evaluation of phytopharmaceuticals; p. 144.
- Harborne JB. A guide to modern technique of plant analysis. London: Chapman and Hil., 1992 ; p. 279.
- T. Murugan, J. Albino Wins, and M. Murugan. Antimicrobial Activity and Phytochemical Constituents of Leaf Extracts of Cassia auriculata. Indian J Pharm Sci., 2013; 75(1): 122–125.
- N. R. Bhalodia, P. B. Nariya, R. N. Acharya, and V. J. Shukla. In vitro antibacterial and antifungal activities of Cassia fistula Linn. fruit pulp extracts. Ayu., 2012, 33(1): 123–129.
- G. P. Choudhary and M. S. Vijay Kanth. Antimicrobial Activity of Root bark of Salacia reticulate. Ancient Science of Life., 2005, Vol : XXV (1).
- Veeramuthu Duraipandiyan, Naif Abdullah Al-Harbi, Savarimuthu Ignacimuthu and Chinnasamy Muthukumar1. Antimicrobial activity of sesquiterpene lactones isolated from traditional medicinal plant, Costus speciosus (Koen ex.Retz.) BMC Complementary and Alternative Medicine., 2012, 12:13.
- Ahmed S. Kabbashi, Seif Eldeen A. Mohammed, Aisha Z. Almagboul and Ibrahim F. Ahmed. Antimicrobial activity and Cytotoxicity of Ethanolic Extract of Cyperus rotundus L. American Journal of Pharmacy and Pharmaceutical Sciences., 2015, 2(1): pp. 1– 13.
- Aparna pareek, Rishi kesh meena. Antimicrobial activity of Syzygium cumini. Indian Journal of applied research., 2015, 5(9):Pg64-66.
- Kamal Rai Aneja, Chetan Sharma, Radhika Joshi. Antimicrobial activity of Terminalia arjuna Wight & Arn An ethnomedicinal plant against pathogens causing ear infection. Brazilian Journal of otorhinolaryngology., 2012; 78(1) .
- Harold E. Pearson M.D.Human. Infections Caused by Organisms of the Bacillus Species.American. Journal of clinical pathology., 1970, 53(4).
- T. M. Wijesuriya. Proportion of lower limb fungal foot infections in patients with type 2 diabetes at a tertiary care hospital in Sri Lanka. Indian J Endocrinol Metab., 2014; 18(1): 63–6.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


