ISSN: 0973-7510
E-ISSN: 2581-690X
Endophytes are the mimicking the plant secondary metabolites as well as vast number chemical synthesizers inside plants and also have been extensively investigated for their endophytic microbial complement. Different parts of Rauwolfia tetraphylla L were subjected to the isolation of endophytic fungi. The isolated endophytic fungi were identified morphologically and, isolation, colonization rates and relative frequency of various isolates were calculated. The endophytic fungal isolates were screened for antimicrobial activity against Staphylococcus aureus, Bacillus cereus, Pseudomonas aeruginosa, Escherichia coli, Salmonella typhimurium and Candida albicans. The identification of the prospective endophytic fungi Curvularia sp. (RTFs-6) and Aspergillus sp. (RTL-6) were selected for the production of secondary metabolites which were grown in rice medium and extracted with ethyl acetate and were screened for their antimicrobial activity by agar well diffusion method and Phytochemical analysis. The extract of Curvularia sp. gave effective inhibition to the all the tested organisms whereas extract of Aspergillus sp. gave effective inhibition against tested organisms except P. aeruginosa. Phytochemical compounds screening of the extracts revealed the presence of alkaloids, terpenoids and tannin compounds.
Rauwolfia tetraphylla L, Endophytic fungi, Colonization rate, Isolation rate, Antimicrobial, Phytochemicals.
All plants are in natural ecosystem and emerge to be symbiotic with endophytic fungi. Endophytic fungi are endosymbionts residing in plants without causing adverse effects to the host plant and produce a vast range of secondary metabolites with medicinal properties 1. They exhibit mutualistic association with their host and enhance the ability of plants to tolerate abiotic 2 and biotic 3 stresses. It is very important to isolate novel endophytic microorganisms as well as novel bioactive compounds. A specific rationale selection of each plant for endophyte isolation and bioactive secondary metabolite discovery is used. The reasonable hypotheses are used for plant selection strategy 4. The number of plant species in the world produce bioactive metabolites, their traditional usage history (use by indigenous people) related to the specific uses or applications and significant technique used to search for endophytes displaying bioactivity 4. The endophytes of non-grass plants or trees should be screened for bioactive compounds which have rich sources of secondary metabolites 5 .
The demand and need for new antimicrobial agents is growing rapidly as the infectious diseases are still a worldwide problem and the development of drug resistance by the pathogens is a growing concern 6. The problem extends from the clinical application of antimicrobial drugs and many microorganisms of agricultural concern are also recognized to have acquired resistance to normally used antimicrobial chemicals 7, which indicates a growing desire for new bioactive compounds. Historically, a majority of the compounds have been isolated from the normal environment, predominantly plants, and have been used in the treatment of many diseases and illnesses. Many of the drugs obtainable commercially from these natural products and have become potential drug sources 8.
The member of Apocynaceae, Rauwolfia tetraphylla L. a medicinal plant traditionally used for treating various diseases; the seeds and leaves extracts of produces various medicinally important alkaloids 9 which are used as ethnopharmaceutical compounds 10 and in the treatment of snake bite as an antidote 11. It also widely used to encourage uterine expansion in case of difficult delivery and to treat high blood pressure, muscular and rheumatism pain10.
Recently, more attention is turned towards the endophytic fungi associated with the medicinal plants as they mimic to produce host plant’s secondary metabolites 12; easy to be grown in laboratory and subjected to large scale production. The crystalline compound mycophenolic acid from Penicillium glaucoma was the first anti microbial secondary metabolite, discovered in 1896 by Gosio 13. Therefore, it is believed that search for new compounds bound towards endophytic fungi for medicinal purpose and to save the mass utilization of plants to produce secondary metabolites. The bioactive metabolites from endophytic fungi include alkaloids, benzopyranones, benzoquinones, flavonoids, glycosides, steroids, saponins, tannins, and terpenoids, phenol, phenolic compounds, phenylpropanoids, tetralones, xanthones, and other compounds 14. The implication of biotechnological tools will enhance the production of fungal secondary metabolites at low cost and environment friendly 15. The most important quality of secondary metabolites is their unique chemical structures, frequent occurrence and biological activity 16. Biological activity will be used for interactions between chemicals and molecular targets of living organisms 13. Pathogenic microorganisms like Staphylococcus aureus, Bacillus cereus, Salmonella typhimurium, Escherichia coli, Pseudomonas aeruginosa and Candida albicans cause many infectious diseases which could be fatal. Staphylococcus aureus, gram positive coccus bacterium causes cellulitis (inflammation), mastitis in breast feeding women 17. Bacillus cereus is a Gram-positive rod-shaped, beta haemolytic bacterium which causes food borne illness 18. Pseudomonas aeruginosa is a Gram-negative rod-shaped bacteria cause infections of pulmonary tract, urinary tract, burns, wounds, and blood 19. Escherichia coli is a facultative anaerobic gram-negative rod-shaped bacteria and some virulent strains can cause food poisoning and urinary tract infections 20. Salmonella typhimurium, Gram-negative rod shaped bacteria predominately found in the intestinal lumen causing typhoid fever 21. Candida albicans is yeast and grows as filamentous cells causes opportunistic oral, genital infections leading to candidiasis 22.
In the present research an attempt was made to study the diversity of endophytic fungal isolates of R. tetraphylla, screen for antimicrobial activity against the pathogenic microorganisms. Also, phytochemical analysis was carried out for the potential endophytic fungal extract exhibiting good antimicrobial activity.
Collection of plant sample and sampling site
The medicinal plant Rauwolfia tetraphylla (Fig 1) was collected from Namachilume forest, Tumkur District, Karnataka. Different plant parts were cut from the medicinal plants with knife disinfected with 70% ethanol and was brought to the laboratory. The samples were collected every three months during year 2010- 2011 (July – Sep, Oct – Dec, Jan – Mar, Apr- June) and the collection was repeated.

Fig. 1. Medicinal plants selected for present study from Apocynaceae
Authentication of plant samples
The herbarium samples of R. tetraphylla was deposited in National Ayurveda Dietetics Research Institute, CCRA&S (Central Council for Research in Ayurveda and Siddha), Department of AYUSH, Ministry of Health and Family Welfare, Govt. of India, Jayanagar, Bangalore with the accession number RRCBI-15390.
Isolation and maintenance of the endophytic fungi
Endophytic fungi were isolated from fresh material of healthy medicinal plant R. tetraphylla. The collected samples were washed under running tap water and cut into 0.5cm2 segments and were surface disinfected using sodium hypochlorite – ethanol surface sterilization techniques 23. The effectiveness of the sterilization procedure was confirmed by the vitality test 24. Ten leaf segments from each individual part were placed in a Petri dish (9cm) containing Potato Dextrose Agar (PDA). Ten surface sterilized segments from each individual part were placed on PDA plates amended with 50mg/L tetracycline to suppress the bacterial growth and incubated at 28°C to 30°C for 2 to 3 days. The hyphal tip of endophytic fungi growing out from the plant tissue was transferred to fresh PDA plates. After incubation at 30°C for 7 to 14 days, purity of the culture was determined by colony morphology.
Morphological identification of endophytic fungi
The endophytic fungi were identified based on the cultural characteristics, the morphology of the fruiting bodies and spores using standard manuals 25, 26, 27. The isolates were induced for sporulation by culturing them on different media such as Potato Sucrose Agar (PSA), Potato Carrot Agar (PCA), Malt extract Agar (MEA), Sabouraud Dextrose Agar (SDA), Corn Meal Agar (CMA) and Tap Water Agar (TWA). Non sporulating cultures were distinguished from each other by their cultural characteristics such as colony morphology, hyphal mat characteristics and pigmentation of the colony in the medium and were grouped under Mycelia sterilia 28. The fungi were mounted on the clean glass slide with lactophenol, sterile distilled water or lactophenol cotton blue stain and edges were sealed with DPX mountant.
Identification of potential endophytic fungi by molecular methods
In addition to the morphological identification, molecular methods were carried out to confirm the identification of potential endophytic fungi exhibiting antimicrobial activity.
Isolation of the genomic DNA
The potential endophytic fungi were grown on 50mL PDB for 5-6 days at 28±2°C. The mycelia was harvested and washed with distilled water and ground with liquid nitrogen. Total genomic DNA was extracted using the cetyl trimetyl ammonium bromide (CTAB) method 29. The purity of extracted DNA was determined spectrophotometrically and the quality of the DNA was checked by electrophoresis using 1% Agarose gel.
ITS amplification
The ITS region of the rDNA was amplified using primers ITS1 and ITS4 29. Amplification reactions were performed in a total volume of 30µL containing 1X PCR buffer, 200µM each dNTPS, 1.4mM MgCl2, 1µL of each primer (100nmol), 1.0 U Taq DNA polymerase, and 20ng of genomic DNA. PCR amplification conditions included an initial denaturation step at 94°C for 4min, cycling conditions were 94°C for 45s, 56°C for 45s, 72°C for 1min (30 cycles), followed by a final extension at 72°C for 5 min. Each set of the experiment included negative controls (without template DNA) to test the presence of contaminating DNA in reagents. The amplicons were checked in 1% agarose gels run parallel to standard DNA molecular weight marker.
Purification and Sequencing of PCR products
The PCR products were purified using GenElute™ PCR clean-up kit according to the manufacturer’s instructions and sequencing was carried out in an ABI automated DNA sequencer. The sequencing PCR was set up using ABI-BigDye® Terminatorv3.1 Cycle Sequencing Kit. BLAST analysis was carried out for obtained sequences in the NCBI database and sequences are submitted to NCBI.
Phylogenetic tree construction
The sequence of the highly bioactive isolate was further compared by phylogenetic analysis. Multiple sequence alignments of the obtained sequence and reference sequences retrieved from GenBank were used to generate phylogenetic tree using the online software server phylogeny.fr. 30.
Data analysis Isolation rate
Isolation rate (IR) of the endophytic fungi was calculated as number of isolates obtained from tissue segments divided by total number of tissue segments 31.
Colonization rate
Colonization rate (CR) of endophytic fungi was expressed as percentage of total number of isolates obtained from different tissue segments divided by total number of isolates obtained from overall tissue segments incubated 32.
Relative percentage occurrence of each group of fungi
Relative frequencies (RF) of isolation, used to represent fungal species density was calculated as the number of isolates of each species of the endophytic fungi divided by the total number of isolates and they were expressed as percentage 31.
Screening of endophytic fungi for antimicrobial activity
Endophytic fungi isolated from medicinal plants of Rauwolfia tetraphylla, was screened for the antimicrobial activity against the human pathogenic bacteria (Staphylococcus aureus NCIM No. 2079, Bacillus cereus NCIM No. 2106, Pseudomonas aeruginosa NCIM No. 2200, Escherichia coli NCIM No. 2256, Salmonella typhimurium NCIM No. 2501) and yeast (Candida albicans NCIM No. 3471) procured from National collection of industrial microorganism (NCIM), NCL, Pune.
Agar plug method
The bacteria were grown in Nutrient Broth (NB) for 24hrs at 37°C and yeast was grown in Sabouraud Dextrose Broth (SDB) for 48hrs at 25°C and the turbidity was matched with 0.5 McFarland standards (106 CFU/mL). The microbial cultures were swabbed on to the respective media (Nutrient agar for bacteria, Sabouraud Dextrose agar for yeast). Five mm cylindrical pieces cut out from well grown culture of the endophytic fungi were placed on Petri dishes swabbed with the test microorganisms, incubated at 2 – 8ºC for 12hrs to allow the diffusion of antimicrobial substance. Thereafter, bacterial plates were incubated at 37ºC for 24hrs and Yeast plates were incubated at 25ºC for 48hrs. The antimicrobial activity was measured as diameter of inhibition zone in mm 33.
Production and extraction of secondary metabolites
The endophytic fungi exhibiting potent antimicrobial activity was subjected for mass production of secondary metabolites using Rice medium.
The fresh mycelia of endophytic fungi Curvularia sp. (RTFs-6) and Aspergillus sp. (RTL-6) were grown on PDA plates at 28 ± 2ºC for 3-6 days. They were further inoculated into 1000mL flasks containing 200g of unpolished rice, soaked in 200mL distilled water (autoclaved twice at 121ºC for 20 min), followed by incubation for 30 days at 28 ± 2ºC 34.
Extraction of secondary metabolites from Rice medium culture
The cultures of endophytic fungi grown in rice medium were filled with 300mL of ethyl acetate and allowed to stand for one day, shaken thoroughly and filtered. The following procedure was repeated until most of the metabolites were extracted. Then the ethyl acetate filtrate was extracted with pure distilled water to remove debris and other particles. Finally ethyl acetate extract was treated with anhydrous Sodium sulphate to remove the moisture content and dried using a rotary evaporator 35. All the crude extracts were weighed after concentration.
Antimicrobial activity of the crude endophytic fungal extract by agar well diffusion method
The extracted secondary metabolite was dissolved in DMSO at different concentrations of 10µg/mL, 20µg/mL, 40µg/mL, 60µg/mL, 80µg/mL and 100µg/mL; added into the 5mm diameter well bored in Petri dishes (NA for bacteria, SDA for yeast) inoculated with a fixed amount of test microorganisms (106 CFU/mL) so as to obtain a lawn culture. The plates were kept for 12hrs at 2-8°C for the antimicrobial metabolite diffusion and thereafter incubated at optimum temperature for growth. The zone of inhibition was measured in millimeter 36.
Statistical analysis
Analysis of variance (ANOVA) was used to determine the significance of difference between treatment groups (p < 0.05). Means between treatment groups were compared for significance using Duncan’s Multiple Range test.
Phytochemical analysis of ethyl acetate extract of endophytic fungi
The ethyl acetate extract of the potent endophytic fungi were subjected to chemical constituent analysis 37, 38. The test details are as follows:
Test for Alkaloids
A drop of extract was spotted on the pre coated TLC plate followed by spraying with Dragendorffs reagent. The orange red or brown coloration indicated presence of alkaloids.
Test for Steroids and Terpenoids
One mL of extract, 1mL of chloroform, 2-3 mL of acetic anhydride and 1 to 2 drops of concentrated sulfuric acid were added. The dark green coloration of solution indicated the presence of steroids and dark pink or red coloration of solution indicated presence of terpenoids.
Test for Phenols and phenolic compounds
A drop of extract was spotted initially on a filter paper followed by phosphomolybdic acid spot. The blue coloration indicated presence of phenols and phenolic compounds.
Test for Tannins
Two to three mL of extract, 10% alcoholic ferric chloride solution was added. A dark blue or greenish grey coloration of solution indicated the presence of tannins.
Test for Flavonoids
Two to three mL of the extract, a piece of magnesium strip and 1mL of concentrated hydrochloric acid were added. A pink red or red coloration of solution indicated the presence of flavonoids.
Test for the presence of proteins
Ninhydrin test: To the extract, 0.25% Ninhydrin reagent was added and boiled for few a minutes. Formation of blue color indicated the presence of amino acid.
Isolation of endophytic fungi
A total of 26 endophytic fungi were isolated from different parts of R. tetraphylla and named as RTS1-4 (R. tetraphylla stem isolates), RTFr1-8 (R. tetraphylla fruits isolates) and RTL1-14 (R. tetraphylla leaf isolates).
Identification of endophytic fungi
The identified endophytic fungi were Aspergillus sp., Cladosporium sp., Colletotrichum sp., Curvularia sp., Cylindrocephalum sp., Fusarium sp., Paecilomyces sp., Penicillium sp., Pestalotiopsis sp., Thielavia terricola and Mycelia sterilia were found to be associated with R. tetraphylla L. The percentage of Relative frequencies of endophytic fungi is shown in Table 1.
Table (1):
Relative frequency (%) of different endophytic fungal taxa isolated from four medicinal plants of Apocynaceae..
| Sl. No. | Endophytic fungi | Rauwolphia tetraphylla | Total (%) | ||
|---|---|---|---|---|---|
| Stem (%) | Leaf (%) | Fruit (%) | |||
| 1 | Aspergillus sp. | 0 | 11.53846 | 3.846154 | 15.3846154 |
| 2 | Beltrania sp. | 3.846154 | 0 | 0 | 3.84615385 |
| 3 | Cladosporium sp. | 0 | 0 | 3.846154 | 3.84615385 |
| 4 | Colletotrichum sp. | 3.846154 | 7.692308 | 3.846154 | 15.3846154 |
| 5 | Curvularia sp. | 0 | 0 | 3.846154 | 3.84615385 |
| 6 | Cylindrocephalum sp. | 0 | 3.846154 | 0 | 3.84615385 |
| 7 | Fusarium sp. | 0 | 7.692308 | 3.846154 | 11.5384615 |
| 8 | Mycelia sterilia sp. | 3.846154 | 11.53846 | 0 | 15.3846154 |
| 9 | Paecilomyces sp. | 0 | 3.846154 | 0 | 3.84615385 |
| 10 | Penicillium sp. | 0 | 3.846154 | 3.846154 | 7.69230769 |
| 11 | Pestalotiopsis sp. | 0 | 3.846154 | 3.846154 | 7.69230769 |
| 12 | Thielavia terricola | 3.846154 | 0 | 3.846154 | 7.69230769 |
| Total | 15.38462 | 53.84615 | 30.76923 | 100 | |
All the plant samples were found to be associated with various endophytic fungi with different isolation rates (IR) and colonization rates (CR). The IR value for endophytic fungi of R. tetraphylla L. was found to be highest in the leaf samples (0.7) followed by in fruit samples (0.4), whereas lowest in stem samples (0.2) (Fig 2). The CR of endophytic fungi was maximum in the leaf samples (53.38%) followed by fruit samples (30.76%), whereas lower in stem (15.38%) (Fig 3).
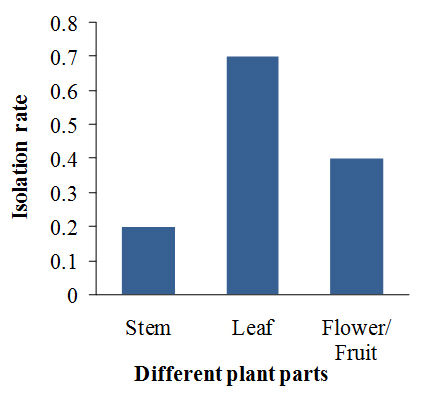
Fig. 2. Isolation rate of endophytic fungi of four medicinal plant parts of Apocynaceae
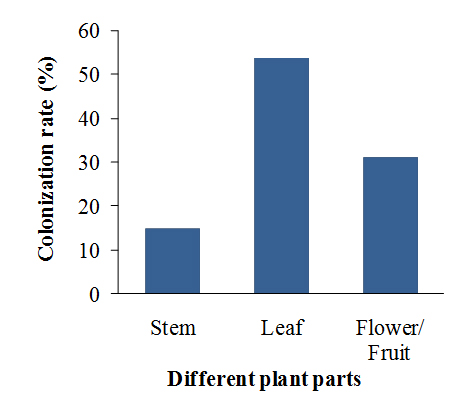
Fig. 3. Colonization rate of endophytic fungi isolated from four medicinal plant parts of Apocynaceae
Identification of endophytic fungi by molecular methods
The molecular identification of potential isolates exhibiting antimicrobial activity i.e., RTFs-6 were carried out. Based on the BLAST analysis for sequence of ITS region from 18S rDNA of selected isolates were identified with reference to GenBank NCBI. Five hundred and thirty two (532) bases of RTFs -6 showed the 100% homology with Curvularia sp. The ITS rDNA sequence of the strain Curvularia sp., (RTFs-6) has been deposited in the NCBI GenBank database with the accession number KF864556.
Screening for antimicrobial activity of endophytic fungi isolated from R. tetraphylla
Twenty six (26) endophytic fungi were isolated from R. tetraphylla and screened for the antimicrobial activity. Of these, 73% confirmed the antimicrobial activity and rest of the isolates did not exhibit the activity. Out of 19 isolates, 17 isolates showed potential inhibition against S. aureus and only 4 isolates inhibited the growth of B. cereus. Seven isolates exhibited inhibition against E. coli and six isolates showed activity against S. typhimurium. Only RTFs-6 isolate suppressed the growth of P. aeruginosa and three fungal isolates were effective against C. albicans (Table 2). Among the tested isolates, endophytic fungal isolates Curvularia sp. (RTFs-6) and Aspergillus sp. (RTL-6) revealed better antimicrobial activity was further subjected for production of secondary metabolites (Fig 4).
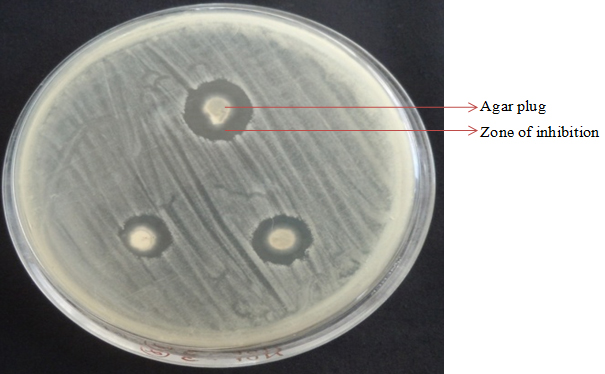
Fig. 4. Antimicrobial activity of endophytic fungi was determined by agar plug method
Table (2):
Screening for antimicrobial activity of endophytic fungi from R. tetraphylla by agar plug method..
| Endophytic fungi | Antimicrobial activity (Diameter zone of inhibition in mm) | |||||||
|---|---|---|---|---|---|---|---|---|
| Sl. No | Code | Endophytic fungi | Staphylococcus aureus | Bacillus cereus | Pseudomonas aeruginosa | Escherichia coli | Salmonella typhimurium | Candida albicans |
| 1. | RTS-1 | Mycelia sterilia sp. 1 | 0a | 0a | 0a | 0a | 0a | 0a |
| 2. | RTS-2 | Colletotrichum sp. | 9±1g | 0a | 0a | 0a | 0a | 0a |
| 3. | RTS-3 | Beltrania sp. | 0a | 0a | 0a | 0a | 0a | 0a |
| 4. | RTS-4 | Thielavia terricola | 0a | 0a | 0a | 0a | 0a | 0a |
| 5. | RTFs-1 | Cladosporium sp. | 7.33± 0.58cd | 0a | 0a | 0a | 0a | 0a |
| 6. | RTFs-2 | Pestalotiopsis sp. | 0a | 0a | 0a | 0a | 0a | 0a |
| 7. | RTFs-3 | Fusarium sp. | 0a | 0a | 0a | 0a | 0a | 0a |
| 8. | RTFs-4 | Penicillium sp. | 7.33±0.58cd | 0a | 0a | 0a | 0a | 0a |
| 9. | RTFs-5 | Aspergillus sp. | 9±0g | 0a | 0a | 0a | 0a | 0a |
| 10. | RTFs-6 | Curvularia sp. | 8.33±0.58efg | 7.67±0.58b | 6.67±0.58 b | 9±0e | 7.67±0.58d | 10.33±0.58d |
| 11. | RTFs-7 | Thielavia terricola | 0a | 0a | 0a | 0a | 0a | 0a |
| 12. | RTFs-8 | Colletotrichum sp. | 7.33±0.58cd | 0a | 0a | 7.33±0.58c | 6.33±1.15b | 0a |
| 13. | RTL-1 | Fusarium sp. | 7.33±0.58cd | 0a | 0a | 0a | 0a | 0a |
| 14. | RTL-2 | Mycelia sterilia sp.2 | 6±1b | 0a | 0a | 0a | 0a | 0a |
| 15. | RTL-3 | Pestalotiopsis sp. | 10±0h | 0a | 0a | 0a | 0a | 0a |
| 16. | RTL-4 | Colletotrichum sp. | 7.33±0.58 cd | 0a | 0a | 0a | 0a | 0a |
| 17. | RTL-5 | Mycelia sterilia sp.3 | 6±0b | 10.33±1.15c | 0a | 0a | 0a | 0a |
| 18. | RTL-6 | Aspergillus sp. | 6.67±1.52bc | 7.33±0.58b | 0a | 8.33±1.15d | 7±0c | 8.67±1.15c |
| 19. | RTL-7 | Colletotrichum sp. | 12±0i | 0a | 0a | 7.33±0.58c | 0a | 0a |
| 20. | RTL-8 | Fusarium sp. | 7.33±1.15cd | 7.67±0.58b | 0a | 0a | 8.33±0.58e | 0a |
| 21. | RTL-9 | Cylindrocephalum sp. | 7.67±0.58de | 0a | 0a | 6±0b | 0a | 0a |
| 22. | RTL-10 | Penicillium sp. | 8.33±1.15efg | 0a | 0a | 0a | 0a | 0a |
| 23. | RTL-11 | Aspergillus sp. | 10.33±0.58h | 0a | 0a | 12±1f | 8.33±0.58e | 0a |
| 24. | RTL-12 | Paecilomyces sp. | 0a | 0a | 0a | 0a | 0a | 0a |
| 25. | RTL-13 | Mycelia sterilia sp.4 | 0a | 0a | 0a | 5.67±0.58b | 6±1b | 0a |
| 26. | RTL-14 | Aspergillus sp. | 0a | 0a | 0a | 0a | 0a | 7.67±0.58b |
RTS- Rauwolfia tetraphylla stem isolates, RTFs- Rauwolfia tetraphylla fruit isolates, RTL- Rauwolfia tetraphylla leaf isolates.Values represent mean ± SD of three parallel experiments. In each column, mean values followed by the same letter are not significantly different according to DMRT at p < 0.05.
Production and extraction of secondary metabolites from potent endophytic fungi
The selected potential endophytic fungi were subjected to production and extraction of secondary metabolites. The crude extracts were obtained in rice media from the isolates of Curvularia sp. (RTFs-6) and Aspergillus sp. (RTL-6) yielded 936mg/100g of rice and 975mg/100g of rice respectively.
Antimicrobial activity of secondary metabolite extract isolated from potential endophytic fungi
The crude extract of the potential endophytic fungi Curvularia sp. (RTFs-6) and Aspergillus sp. (RTL-6) exhibited a broad spectrum of antimicrobial activity against the test pathogens (Fig 5) when compared with the positive control tetracycline for bacteria and fluconazole for yeast. The zone of inhibition of test pathogens ranged from 6.33mm to 19.67mm at concentrations of 20-100µg/mL of tested crude extracts. The extract of Curvularia sp. (RTFs-6) and Aspergillus sp. (RTL-6) exhibited antimicrobial activity at the concentration of 20µg/mL, however, there was no inhibition at 10µg/mL. Hence, the MIC was determined to be 20µg/mL. Both the extracts showed inhibition against S. aureus and B. cereus. The fungal extracts of Curvularia sp. (RTFs-6) inhibited E. coli and P. aeruginosa at different concentrations ranging from 20-100µg/mL. The fungal extract of Curvularia sp. (RTFs-6) and Aspergillus sp. (RTL-6) exhibited inhibitory activity against S. typhimurium at all the tested concentrations. The secondary metabolites of Curvularia sp. (RTFs-6) and Aspergillus sp. (RTL-6) showed the antifungal activity against C. albicans at the concentrations of 20-100µg/mL. These fungal crude extracts exhibited both antibacterial and antifungal activities similar to the positive controls used; tetracycline for bacteria and fluconazole for fungi (Table 3).
Table (3):
Antimicrobial activity of crude extracts of endophytic fungi isolated from medicinal plants of Apocynaceae.
| Endophytic fungi | Concentration of extract in µg/mL | Antimicrobial activity (Diameter of inhibition zone in mm) | |||||
|---|---|---|---|---|---|---|---|
| Staphylococcus aureus | Bacillus cereus | Pseudomonas aeruginosa | Escherichia coli | Salmonella typhimurium | Candida albicans | ||
| Curvularia sp. (RTFs-6) | 20 | 8.33±1.52ab | 8.33±1.15bc | 6.67±0.57bcd | 7±0bc | 7.67±0.57bc | 6.33±0.57b |
| 40 | 9.67±0.57bcde | 10.33±0.57efghi | 7.67±0.57ef | 10.33±0.57fgh | 8±1bc | 9.33±0.57def | |
| 60 | 10.33±0.57bcde | 11±0fghi | 8.67±0.57gh | 12.33±0.57ijk | 9±0cde | 10.33±1.52ghi | |
| 80 | 11±1cdef | 11.33±0.57ghij | 9.33±0h | 13.33±0.57kl | 10.33±0.57efg | 13.33±0.57jk | |
| 100 | 12.67±0.57fg | 12.67±0.57ijkl | 10.67±0.33i | 15.33±0.57mn | 12±1hi | 15.67±1.52l | |
| Aspergillus sp. (RTL-6) | 20 | 11.33±0.57defg | 6.67±0.57a | 0a | 7±1bc | 7±0.57b | 7.67±0.57c |
| 40 | 13±1fgh | 7.67±0.57ab | 0a | 9.67±0.57efg | 9±0cde | 9.67±1.15fg | |
| 60 | 15±1ijk | 9.33±0.57bcdef | 0a | 10.67±0.57fgh | 13±1hi | 10±0fgh | |
| 80 | 18±1lm | 11±0fghi | 0a | 12±0ghi | 15±1jk | 12.67±0.57jk | |
| 100 | 19.67±0.66mno | 12±1hijk | 0a | 13.33±0.57kl | 16.67±0.57k | 15±1l | |
| Tetracycline | 20 | 27.33±1.2r | 15.33±0.66m | 8.66±0.33gh | 16.33±0.33no | 11±0.57fgh | – |
| Fluconazole | 20 | – | – | – | – | – | 13.66±0.33k |
Values represent mean ± SD of three parallel experiments. In each column, mean values followed by the same letter are not significantly different according to DMRT at p < 0.05. “ –’’ Not determined.
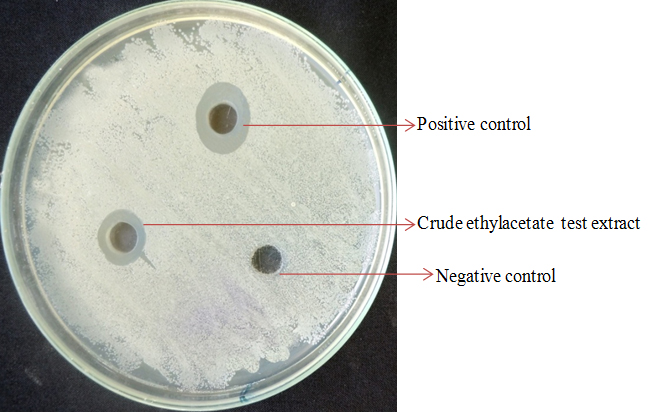 Fig. 5. Antibacterial activity of crude extract of endophytic fungi was determined by agar well diffusion method
Fig. 5. Antibacterial activity of crude extract of endophytic fungi was determined by agar well diffusion methodPhytochemical analysis of secondary metabolite extract from endophytic fung
Phytochemical analysis of the isolated endophytic fungal extracts of Curvularia sp. (RTFs-6) and Aspergillus sp. (RTL-6) which showed potential antibacterial and antifungal activity was carried out. The phytochemical analysis was performed to determine the presence of chemical components as a prospective source for medicinal and industrial use. Analysis of ethyl acetate extracts of Curvularia sp. (RTFs-6) indicated the presence of alkaloids whilst, the secondary metabolites of Aspergillus sp. (RTL-6) revealed the presence of alkaloids and tannins (Table 4).
Table (4):
Phytochemical analysis of ethylacetate extract of endophytic fungi isolated from medicinal plants of Apocynaceae.
| Endophytic fungal extract | Phytochemical analysis | ||||||
|---|---|---|---|---|---|---|---|
| Alkaloids | Steroids | Terpenoids | Phenol and phenolic compounds | Tannins | Flavonoids | Proteins and Amino acids | |
| Curvularia sp. (RTFs-6) | + | – | + | – | – | – | – |
| Aspergillus sp. (RTL-6) | + | – | – | – | + | – | – |
Note: + indicates presence and – indicates absence
Association of endophytic fungi varies from plant to plant, geographical distribution and also different seasons 39. The Isolation Rate (IR), Colonization Rate (CR) and Relative Frequency (RF) of the endophytic fungi vary with different medicinal plants 31. In present study, the IR, CR and RF of endophytic fungi varied with different tissue segments of the medicinal plants studied.
The IR and CR of endophytic fungi of R. tetraphylla L. was found to be highest in the leaf samples (0.7 and 53.38%) followed by fruit samples (0.4 and 30.76%), and lowest in stem samples (0.2 and 15.38%). Among the total isolates of R. tetraphylla, RF value of the endophytic fungi in stem, leaf and fruits of R. tetraphylla L. was found to be 15.38%, 53.84% and 30.76% respectively. Endophytic fungi such as Beltrania sp., Colletotrichum sp., Thielavia terricola, and Mycelia sterilia were found in stem. Likewise, Aspergillus sp., Colletotrichum sp., Cylindrocephalum sp., Fusarium sp., Paecilomyces sp., Penicillium sp., Pestalotiopsis sp. and Mycelia sterilia were associated with the leaf segments. Aspergillus sp., Cladosporium sp., Colletotrichum sp., Curvularia sp., Fusarium sp., Penicillium sp., Pestalotiopsis sp., and Thielavia terricola were found in fruit samples. There are no reports on endophytic fungi from R. tetraphylla L. to compare their geographical distribution. Qadri et al. 40 found Alternaria brassicae, Cladosporium cladosporioides, Alternaria alternata, Diaporthe helianthi, Fusarium proliferatum, Alternaria spp. Lasiodiplodia theobromae and Glomerella sp. from Rauwolphia serpentine.
Host plants of endophytic fungi harbors mutual or commensal symbionts, typically providing nourishment and shelter. A host plant is one that supplies food resources and substrate for certain fungi 41. The tissue specificity is usually defined by the part of the plant that is colonized with specific fungi 42. Various factors such as environmental conditions, different plant tissues are involved in host and tissue specificity of endophytic fungi 43, 44. In the present study, endophytic fungal distribution in leaves, stems and fruits were investigated in R. tetraphylla. Endophytic fungal composite and abundance varied with the different tissues of R. tetraphylla.
The present investigation has not showed any host and tissue specificity for Aspergillus sp. which was found in the entire test samples of different part of plants. Many of the previous research also have reported Aspergillus sp. as an endophyte from different plants 31, 45, 46. The genus Beltrania was found only in stem tissues of R. tetraphylla but, Cannon and Simmons 47 found this genus only in leaf segments of Carapa guianensis, Catostemma fragrans and Chlorocardium rodiei.
In the present study, Cladosporium sp. was found in fruit samples of R. tetraphylla. This result agrees with Huang et al. 48 who reported Cladosporium sp. in stem samples of N. oleander. Species of Cladosporium were also reported from various other plants like Embelia tsjeriam and Ocimum sanctum 49, 50. In this study Curvularia sp. were associated with stem parts of R. tetraphylla. Previous researchers also have reported Curvularia sp. associated with root tissues of C. roseus and different segments of Alstonia scholaris 32, 51.
The present study revealed tissue specificity of Cylindrocephalum sp. which varied with different plants. Cylindrocephalum sp. was found in the leaf segments of R. tetraphylla, where as Sunitha et al. 52 reported this genus in Alpinia calcarata. The current study revealed no tissue specificity for Fusarium spp., which were found fruit samples of R. tetraphylla. Zhang et al. 53 also has isolated Fusarium oxysporum from the phloem (inner bark) of C. roseus. Huang et al. 31 also reported Fusarium sp. from different Chinese medicinal plants. This study revealed the occurrence of Paecilomyces sp. in leaf segments of R. tetraphylla. Garcia et al. 54 has reported Paecilomyces sp. in leaf of Sapindus saponaria L. and Tunali et al. 55 reported Paecilomyces sp. from Turkish Grass seeds.
In the present investigation, Penicillium sp. did not exhibit host specificity, this agrees with many previous research reports 56, 57, 58, 59. Pestalotiopsis sp. was reported in Catharanthus roseus, Terminalia morobensis, Mangifera indica 60, 61, 62. There are no reports on Thielavia terricola as an endophyte but, the present study revealed its presence in stem and fruit samples of R. tetraphylla. In the current study there was no observation for tissue and host specificity for Mycelia sterilia spp. whereas, Huang et al. 48 reported 50% of Mycelia sterilia spp. in N. oleander.
The broad spectrum novel antibiotics are frequently required for treatment due to increase in the existence of naturally resistant bacteria, progression of new diseases caused by pathogenic microorganisms and toxicity of some of the current synthetic or natural compounds 63. Even though there are many antibiotics that have been effectively produced, new screening techniques are extensively required to isolate fresh and novel bioactive secondary metabolites from different sources in nature. The endophytic fungi are rich sources of secondary metabolites which kill wide variety of harmful disease causing agents 5. Therefore, the utilization of endophytic fungi as sources of antimicrobial agents leads up to new areas for biotechnological exploitations, which open the isolation and cultivation of these organisms. In previous studies many endophytic fungi have been identified from different plants species belonging to Apocynaceae which have shown potential antimicrobial properties 48, 64. In the present study, the endophytic fungi isolated from R. tetraphylla exhibited broad spectrum of antimicrobial activity against the test microorganisms.
Twenty six endophytic fungi of R. tetraphylla were tested for antimicrobial activity, among them 19 isolates produced inhibition zone of 6mm to 10.33mm and Curvularia sp. (RTFs-6) exhibited antimicrobial activity against all the tested pathogens. The endophytic fungi Curvularia sp. (RTFs-6) and Aspergillus sp. (RTL-6) also exhibited potential antimicrobial activity. Curvularia sp. (RTFs-6) showed antimicrobial activity against S. aureus, B. cereus, P. aeruginosa, E.coli, S. typhimurium and C. albicans with inhibition zone of 8.33mm, 7.67mm, 6.67mm, 9mm, 7.67mm and 10.33mm respectively. Whereas, Aspergillus sp. (RTL-6) suppressed the growth of S. aureus, B. cereus, E.coli, S. typhimurium and C. albicans with inhibition zone of 6.67mm 7.33mm, 8.33mm, 7mm and 8.67mm respectively. To the best of our knowledge, there are no previous reports on antimicrobial activity of endophytic fungi isolated from R. tetraphylla but, endophytic fungi F. proliferatum, Cladosporium cladosporioides and Glomerella acutata isolated from R. serpentina exhibited activity against E. coli and S. aureus; Alternaria spp. showed activity only for S. aureus 40.
The selected endophytic fungi, Curvularia sp. (RTFs-6) and Aspergillus sp. (RTL-6) in rice media it gave yield of 936mg/L and 975mg/L respectively. The secondary metabolite biosynthesis in microbes is sometimes controlled by regulatory mechanisms to avoid over production and these regulatory mechanisms process to undesirably low levels 65.
The crude extract of the potential endophytic fungal isolates, exhibited a broad spectrum of antimicrobial activity against the pathogens, when compared with that of positive control tetracycline for bacteria and fluconazole for yeast. The zone of inhibition ranged from 6.33 mm to 19.67 mm at concentrations of 20-100µg/mL. According to Rios and Recio 36 extracts of natural origin showing antimicrobial activity above 100µg/mL concentration should be avoided, hence in the present study, the concentrations of crude extract were limited to 10-100µg/mL. Both the fungal extracts of R.tetraphylla showed no inhibition at concentrations less than 20µg/mL and hence, MIC was determined to be 20µg/mL against test pathogens. Whereas, Huang et al. 48 reported the MIC to be 1.25-10mg/mL for S. aureus, 5-25mg/mL for B. cereus, 5-12.5mg/mL for E.coli when tested with the crude extracts of endophytic fungi isolated from N. oleander L. These results proved that endophytes from different host conditions exhibited varied antimicrobial activity 66.
In this study, extracts of Curvularia sp. (RTFs-6) exhibited MIC at 20µg/mL against P. aeruginosa. The extracts of Curvularia sp. (RTFs-6) and Aspergillus sp. (RTL-6) inhibited S. typhimurium at MIC 20µg/mL. The activity of crude extracts was found to be similar to that of reference positive control – tetracycline for bacteria and flucanazole for C. albicans. These findings strongly suggest that the metabolites screened for antimicrobial activity have broad spectrum activity and can be successfully used as potent antimicrobial agents 67.
In previous report, the antimicrobial activity of crude extract of A. alternata isolated from Coffea arabica L against S. aureus and E. coli showed the MIC at a range of 50-100µg/mL and 400-800µg/mL respectively. However, it did not show any activity against C. albicans at all the tested concentrations 68. The extracts of endophytic fungi isolated from N. oleander showed MIC in the range of 1.25-10mg/mL for C. albicans 48. Lu et al. 69 found that ergosterol derivatives of Colletotrichum sp. isolated from Artemissia annua inhibited S. aureus and B. substillis and Pseudomonas sp. at the range of 25-75µg/mL and 50-100µg/mL for C. albicans. Endophytic Fusarium sp. isolated from plant Selaginella pollescens collected in the Guanacaste conservation area of Costa Rica also showed potent activity against Candida albicans in agar diffusion assay 4. The extract of endophytic fungus Cochliobolus intermedius isolated from Sapindus saponaria showed antibacterial activity against Escherichia coli, Staphylococcus aureus, Salmonella typhimurium, Micrococcus luteus and Enterococcus hirae 54. According to Huang et al. 48 most of the extracts of endophytic fungi from N. oleander possessed better antibacterial and antifungal activities when compared to the extracts of host plant. The secondary metabolite of endophytic fungus Curvularia lunata isolated from Catharanthus roseus exhibited antimicrobial activity against B. subtilis, S. paratyphi, P. vulgaris, V. cholerae, S. aureus and E. coli 70. The ethyl acetate extract of Aspergillus sp. isolated from Ficus carica showed significant antimicrobial activity against Pseudomonas aeruginosa 71.
In the present study, antimicrobial activity of crude extracts of Curvularia sp. (RTFs-6) and Aspergillus sp. (RTL-6) varied statistically with the zone of inhibition produced by the standard drug tetracycline (20µg/mL) and fluconazole (20µg/mL). The crude extract of Curvularia sp. (RTFs-6) and Aspergillus sp. (RTL-6) at 100µg/mL was in par with the positive control against B. cereus. Ethyl acetate extract of Aspergillus sp. (RTL-6) was inactive against P. aeruginosa. However, the antibacterial activity of Curvularia sp. (RTFs-6) and Aspergillus sp. (RTL-6) against E. coli was almost similar and higher than that of positive control. Inhibition zone of crude extracts of Curvularia sp. (RTFs-6) and Aspergillus sp. (RTL-6) against C. albicans of 100µg/mL was either near or higher than that of fluconazole. The endophytic fungi thus screened for antimicrobial activity exhibited potent activity against the test pathogens and proved to be promising as a drug in near future. The present results correlated with the previous findings of endophytes as potent antimicrobial agents 72.
Phytochemical analysis was carried out for endophytic fungal extracts to find the presence of chemical components as a prospective source for medicinal and industrial use. Their presence is an indicator so that, can be exploited as precursors in the development and advancement of synthetic drugs 73. Phytochemical analysis has been carried out in several plant species but very few reports are available on endophytes 48.
The active metabolites of fungal extracts contain chemical groups such as phenols, steroids, flavonoids, quinines, terpenoids, xantones, peptides, cytocatalasins, alkaloids, aliphatic compounds, and phenyl propanoids 74. In the current study, phytochemical analysis of extract Curvularia sp. (RTFs-6) revealed the presence of alkaloids and terpenoids. Similarly, the terpenoid was identified from extract of endophytic fungus C. lunata isolated from Catharanthus roseus 70. Extract of Aspergillus sp. (RTL-6) showed presence of alkaloids and tannins. Our results are well in accordance with previous reports of Lai et al. 75 who have reported the presence of different phytochemicals viz alkaloids, steroids, tannins 76; phenolic compounds and flavonoids 77 and are also known to possess strong antimicrobial activities.
The present study concludes that, the 26 endophytic fungi associated with R. tetraphylla, Curvularia sp. (RTFs-6) and Aspergillus sp. (RTL-6) are effective alternative sources of antimicrobial drugs and can be further commercially exploited.
ACKNOWLEDGMENTS
The authors are thankful to Dr. Siddamallaya for authenticating the plant sample and maintaining the herbarium in National Ayurveda Dietetics Research Institute. (Central Council for Research in Ayurveda and Siddha, Department of AYUSH, Ministry of Health and Family Welfare. Govt. of India, New Delhi) Jayanagar, Bangalore.
- Schulz, B., Boyle, C. The endophytic continuum. Mycol. Res., 2005; 109(6): 661-86.
- Bae, H., Soo-Hyung, K., Moon, S.K., Richard, C.S., David, L., Mary, D.S., Savithiry, N., Bryan, A.B. The drought response of Theobroma cacao (cacao) and the regulation of genes involved in polyamine biosynthesis by drought and other stresses. Plant Physiol Biochem. 2008; 46(2):174-188.
- Arnold, A.E., Mejía, L.C., Kyllo, D., Rojas, E.I., Maynard, Z., Robbins, N., Herre, E.A. Fungal endophytes limit pathogen damage in tropical tree. Proc. Natl. Acad. Sci., 2003; 100(26): 15649-15654.
- Strobel, G., Daisy, B. Bioprospecting for microbial endophytes and their natural products. Microbiol. Mol. Biol. Rev., 2003; 67(4): 491-502.
- Tan, R.X., Zou, W.X. Endophytes: a rich source of functional metabolites. Nat. Prod. Rep., 2001; 18(4): 448-459.
- Prakash, P., Gnanaprakasam, P., Emmanue, R., Arokiyaraj, S., Saravanan, M. Green synthesis of silver nanoparticles from leaf extract of Mimusops elengi Linn. for enhanced antibacterial activity against multi drug resistant clinical isolates. Colloids surf. B Biointerfaces, 2013; 1(108):255-259.
- Amiri, A., Dugas, R., Pichot, A.L., Bompeix, G. In vitro and in vivo activity of eugenol oil (Eugenia caryophylata) against four important postharvest apple pathogens. Int. J. Food Microbiol., 2008; 126(1-2):13-19.
- Daniel, A.D., Sylvia, U., Ute, R. A historical overview of natural products in drug discovery. Metabolites. 2012; 16; 2(2): 303-336.
- Srivastav, S.K., Agrawal, A.K., Srivastava, S.K., Agrawal, A.K., Singh, S.C., Khanna,V.K., Singh,J., Nath, C., Gupta, M.M., Gupta, S., Verma, R.K., Pal, A., Bawankule, D.U., Saikia, D., Gupta, A.K., Maurya, A., Khanuja, S.P.S. Antipsychotic agents and standardized antipsychotic fractions from Rauwolfia tetraphylla and process of their isolation. 2012; US20120184576 A1.
- Amjad, A.M.I., Firoz, A.K.K., Mohib, K. Ethno-Phyto-Pharmacological overview on Rauwolfia tetraphylla L., Int. J. Pharm. Phytopharmacol. Res., 2013; 2(4): 247-251.
- Shreevanitha, Anitha, A survey on snake bite management by folklore practioners. Int. Ayur. Med. J., 2013; 1(5): 1-4.
- Kaul, S., Ahmed, M., Zargar, K., Sharma, P., Dhar, M.K. Prospecting endophytic fungal assemblage of Digitalis lanata Ehrh.(foxglove) as a novel source of digoxin: a cardiac glycoside. 3 Biotech, 2013; 3(4): 335-340.
- Janos, B. Bioactive Microbial Metabolites. J. Antibiot., 2005; 58(1): 1-26.
- Eldeen, I.M., Effendy, M. A. Antimicrobial agents from mangrove plants and their endophytes. A. Méndez-Vilas, (Ed.) Microbial pathogens and strategies for combating them: science, technology and education. 2013; pp 872-882.
- Tran, H.B.Q., McRae, J.M., Lynch, F., Palombo, E.A. Identification and bioactive properties of endophytic fungi isolated from phyllodes of Acacia species. In: Current research, technology and education topics in applied microbiology and microbial biotechnology (Mendez-Vilas A, ed). Spain: Zurbaran-Formatex Research Center, 2010; pp 377-382.
- Pereira, D.M., Valentao, P., Pereira, J.A., Andrade, P.B. Phenolics: From chemistry to biology. Molecules, 2009; 14(6): 2202-2211.
- Cimolai, N. MRSA and the environment: implications for comprehensive control measures. Eur. J. Clin. Microbiol. Infect. Dis., 2008; 27(7): 481-493.
- Vila, B., Fontgibell, A., Badiola, I., Esteve-Garcia, E., Jimenez, G., Castillo, M., Brufau, J. Reduction of Salmonella enterica var. Enteritidis colonization and invasion by Bacillus cereus var. toyoi inclusion in poultry feeds. Poult, Sci., 2009; 88(5): 975-979.
- Wright, A., Hawkins, C., Anggard, E., Harper, D. A controlled clinical trial of a therapeutic bacteriophage preparation in chronic otitis due to antibiotic-resistant Pseudomonas aeruginosa; a preliminary report of efficacy. Clin. Otolaryngol., 2009; 34(4): 349-357.
- Lodinova, Z.R., Cukrowska, B., Tlaskalova-Hogenova, H. Oral administration of probiotic Escherichia coli after birth reduces frequency of allergies and repeated infections later in life (after 10 and 20 years). Int. Arch. Allergy Immunol., 2003; 131(3): 209-211.
- Surette, M.G., Miller, M.B., Bassler, B.L. Quorum sensing in Escherichia coli, Salmonella typhimurium, and Vibrio harveyi: a new family of genes responsible for autoinducer production. Proc. Natl. Acad. Sci. U.S.A., 1999; 96(4): 1639-1644.
- Yehuda, Z., Burnstein, S., Derazne, E., Sandler, V., Ianculovici, C., Halperin, T. Colonization of Candida: prevalence among tongue-pierced and non-pierced immunocompetent adults. Oral Dis., 2010; 16(2): 172-175.
- Nithya, K., Muthumary, J. Secondary metabolite from Phomopsis sp. isolated from Plumeria acutifolia Poiret. Rec. Res. Sci. Tech., 2010; 2(4): 99-103.
- Schulz, B., Wanke, U., Draeger, S., Aust, H.J. Endophytes from herbaceous plants and shrubs, effectiveness of surface sterilization methods. Mycol. Res., 1993; 97(12): 1447- 1450.
- Sutton, B.C. The Coelomycetes. Commonwealth Mycological Institute, Kew, Surrey, England. 1980.
- Barnett HL, Hunter BB. – Illustrated Genera of Imperfect Fungi. APS Press, St. Paul, Minnesota, USA; 1998.
- Ellis, M.B. Hyphomycetes. Commonwealth Mycological Institute, Cambrian News: Aberystwyth, U.K. 1971.
- Frohlich, J., Hyde, K.D., Petrini, O. Endophytic fungi associated with palms. Mycol. Res., 2000; 104(10): 1202-1212.
- O’Donnell, K., Cigelnik, E., Weber, N.S., Trappe, J.M. Phylogenetic relationships among ascomycetous truffles and the true and false morels inferred from 18S and 28S ribosomal DNA sequence analysis. Mycologia, 1997; 89: 48-65.
- Dereeper, A., Guignon, V., Blanc, G., Audic, S., Buffet, S., Chevenet, F., Dufayard, J.F., Guindon, S., Lefort, V., Lescot, M., Claverie, J.M., Gascuel, O. Phylogeny.fr: robust phylogenetic analysis for the non-specialist. Nucleic Acids Res., 2008; 1(36):W465-469.
- Huang, W.Y., Cai, W.Z., Hyde, K.D., Corke, H., Sun, M. Biodiversity of endophytic fungi associated with 29 traditional Chinese medicinal plants. Fungal Divers., 2008; 33: 61-75.
- Mahapatra, S., Banerjee, D. Diversity and screening for antimicrobial activity of endophytic fungi from Alstonia scholaris. Acta Microbiol. Immunol. Hung., 2010; 57(3): 215-223.
- Denitsa, N., Mariana, N. Screening the antimicrobial activity of Actinomycetes strains isolated from Antarctica. J. Cul. Coll., 2004-2005; 4: 29-35.
- Atalla, M.M., Hamed, E.R., El-Shami, A.R. Optimization of a culture medium for increased mevinolin production by Aspergillus terreus strain. Mal. J. Microbiol., 2008; 4(2): 6-10.
- Yuan-Chi, S., Jyh-Jye, W., Tzu-Tsen, L.T., Ming, P. Production of the secondary metabolites c-aminobutyric acid and monacolin K by Monascus. J. Ind. Microbiol. Biotechnol., 2003; 30(1): 41-46.
- Rios, J.L., Recio, M.C. Medicinal plants and antimicrobial activity. J. Ethnopharmacol., 2005; 100(1-2):80-84.
- Dipali, O.S., Vilas, A.K. Phytochemical screening of ethanolic extracts of stem, leaves, flower and seed kernel of Mangifera indica L. Int. J. Pharma Bio Sci., 2013; 4(2): 383-389.
- Harisaranraj, R., Babu, S.S., Suresh, K. Antimicrobial properties of selected indian medicinal plants against acne – inducing bacteria. Ethnobot. Leaflets, 2010; 14: 84- 94.
- Fernanda, L.S.S., Aline S.R., Paulo, T.L., Ricardo, H., Joa O.A., Itamar, S.M., Aline, A.P.K., Species diversity of culturable endophytic fungi from Brazilian mangrove forests. Curr. Genet., 2013; 59(3):153-166.
- Qadri, M., Sarojini, J., Bhahwal, A.S., Anamika, K., Tabasum, S., Surrinder, K.L., Malik, Z.A., Syed, R.U.H. Identification and bioactive potential of endophytic fungi isolated from selected plants of the Western Himalayas. Springer Plus, 2013; 2:8.
- Peter, S.D. Principles of genetics. 1st edn. eISBN- 9781467297875. 2013.
- Dufresne, D., Osbourn, A. E. Definition of Tissue-Specific and General Requirements for Plant Infection in a Phytopathogenic Fungus. Mol. Plant Microbe Interact., 2001; 14(3): 300-307.
- Qi, F., Jing, T., Zhan, Y. Characterization of Endophytic Fungi from Acer ginnala Maxim. in an Artificial Plantation: Media Effect and Tissue-Dependent Variation. Plos one, 2012; 7(10): e46785.
- Persoh, D., Melcher, M., Flessa, F., Rambold, G., First fungal community analyses of endophytic ascomycetes associated with Viscum album ssp. austriacum and its host Pinus sylvestris. Fungal Biol., 2010; 114(7): 585-596.
- Qiu, M., Rui-sheng, X., Yu, S., Haihua, Z., Hai-min, C. Isolation and identification of two flavonoid-producing endophytic fungi from Ginkgo biloba L. Ann. Microbiol., 2010; 60(1): 143-150.
- Raghunath, R, Radhakrishna, Angayarkanni, J., Palaniswamy, M. Production and cytotoxicity studies of lovastatin from Aspergillus niger PN2 an endophytic fungi isolated from Taxus baccata. Int. J. Appl. Biol. Pharm. 2012; 3(3): 342-351.
- Cannon, P.F., Simmons, C.M. Diversity and host-pre-ference of leaf endophytic fungi in the Iwokrama Forest Reserve, Guyana. Mycologia, 2002; 94(2): 210-220.
- Huang, W.Y., Yi-Zhong, C., Kevin, D.H., Harold, C., Mei, S. Endophytic fungi from Nerium oleander L (Apocynaceae): main constituents and antioxidant activity. World J. Microbiol. Biotechnol., 2007; 23(9): 1253-1263.
- Chandrappa, C.P., Anitha, R., Jyothi, P., Rajalakshmi, K., Seema Mahammadi, H., Govindappa, M., Sharanappa, P. Phytochemical analysis and antibacterial activity of endophytes of Embelia tsjeriam cottam Linn. Int. J. Pharm. Biol. Sci. 2013; 3(1): 467-473
- Gurupavithra, S., Jayachitra, A. Isolation and identification of endophytic fungi from Ocimum sanctum and analyse its antioxidant properties. Int. J. Resear. Pharmaceut. Biomed. Sci., 2013; 4(4): 1120-1125.
- Kharwar, R.N., Verma, V.C., Strobel, G., Ezra, D. The endophytic fungal complex of Catharanthus roseus (L.) G. Don. Curr. Sci., 2008; 95(2): 228-233.
- Sunitha, V.H., Ramesha, A., Savitha, J., Srinivas, C. Amylase production by endophytic fungi Cylindrocephalum sp. isolated from medicinal plant Alpinia calcarata (Haw.) Roscoe. Braz. J. Microbiol., 2012; 43(3): 1213-1221.
- Zhang, L., Guo, B., Li, H., Zeng, S., Shao, H., Gu, S., Wei, R., Preliminary study on the isolation of endophytic fungus of Catharanthus roseus and its fermentation to produce products of therapeutic value. Chin. Tradit. Herbal Drugs, 2000; 31(11): 805–807.
- Garcia, A., Rhoden, S.A., Rubin-Filho, C.J., Nakamura, C.V., Pamphile, J.A. Diversity of foliar endophytic fungi from the medicinal plant Sapindus saponaria L. and their localization by scanning electron microscopy. Biol. Res., 2012; 45(2):139-148.
- Tunali, B., Shelby, R. A., and Kodan, M. Endophytic fungi and ergot alkaloids in native Turkish grasses. Phytoparasitica, 2000; 28(4):375-377.
- Zhuang, H., Wen-Li, M., Hai-Bin, C., Yan-Bo, Z., Hai-Peng, L., Kui, H. Antibacterial constituents from the endophytic fungus Penicillium sp of mangrove plant Cerbera manghas. Che. J. Chinese U., 2008; 29(4): 749-752.
- Yi, Z., Jun, M., Yan, F., Yue, K., Jia, Z., Peng-Juan, G., Yu, W., Li-Fang, M., Yan-Hua, Z. Broad-spectrum antimicrobial epiphytic and endophytic fungi from marine organisms: isolation, mioassay and taxonomy. Mar. Drugs, 2009; 7(2): 97-112.
- Devi, N.N., Prabakaran, J.J., Wahab, F., 2012. Phytochemical analysis and enzyme analysis of endophytic fungi from Centella asiatica. Asian Pac. J. Trop. Biomed., 2012; 2(3): 1280-1284.
- Gherbawy, Y.A., Gashgari, R.M. Molecular characterization of fungal endophytes from Calotropis procera plants in Taif region (Saudi Arabia) and their antifungal activities. Plant Biosyst., 2014; 148(6):1085-1092.
- Harper, J.K., Ford, E.J., Strobel, G.A., Arif, A., Grant, D.M., Porco, J., Tomer, D.P., Oneill, K. Pestacin: a 1,3,-dihydro isobenofuran from Pestalotipsis microspora possessing antioxidant and antimycotic activities. Tetrahedron, 2003; 59: 2471-2476.
- Subban, K., Ramesh, S., Muthumary, J. A novel antibacterial and antifungal phenolic compound from the endophytic fungus Pestalotiopsis mangiferae. Nat. Prod. Res., 2013; 27(16): 1445-1449.
- Srinivasan, K., Muthumary, J. Taxol production from Pestalotiopsis sp an endophytic fungus Isolated from Catharanthus roseus. J. Ecobiotechnol., 2009; 1(1): 028- 031.
- Rouveix, B. Antibiotic safety assessment. Int. J. Antimicrob. Agents, 2003; 21(3): 215-221.
- Gangadevi, V., Muthumary, J. Isolation of Colletotrichum gloeosporioides, a novel endophytic taxol-producing fungus from the leaves of a medicinal plant, Justicia gendarussa. Mycol. Balc., 2008; 5: 1-4.
- Suja, M., Vasuki, S., Sajitha, N. Optimization and antimicrobial metabolite production from endophytic fungi Aspergillus terreus KC 582297. Eur. J. Exp. Biol., 2013; 3(4):138-144.
- Petrini, O. Taxonomy of endophytic fungi of aerial plant tissues. In: Fokkema NJ, van den Huevel J, eds. Microbiology of the phyllosphere. Cambridge, UK: Cambridge University Press, 1986; pp175-187.
- Sibi, G., Kalpana, K., Dhananjaya, K., Ravikumar, K.R., Mallesha, H. Antibacterial activity of Sechium edule (Jacq.) Swartz against gram negative food borne bacteria. Adv. Appl. Sci. Res., 2013; 4(2):259-261.
- Fernandes, M.R.V., Costa S.T.A., Pfenning, L.H., Costa-Neto, C.M., Heinrich, T.A., Alencar, S.M. De Lima, M.A., Ikegaki, M. Biological activities of the fermentation extract of the endophytic fungus Alternaria alternata isolated from Coffea arabica L. Braz. J. Pharm. Sci., 2009; 45(4): 678-685.
- Lu, H., Wen, X.Z., Jun, C.M., Jun, H., Ren, X.T. New bioactive metabolites produced by Colletotrichum sp., an endophytic fungus in Artemisia annua. Plant Sci., 2000; 151(1): 67-73.
- Parthasarathy, R., Sathiyabama, M. Screening and characterzation of antimicroial compound from endophytic fungus Curvularia lunata, isolated from Catharanthus roseus. World J. Pharm. Res., 2013; 2(6): 3078-3086.
- Prabavathy, D., Valli Nachiyar, C. Cytotoxic potential and phytochemical analysis of Justicia beddomei and its endophytic Aspergillus sp. Asian J. Pharm. Clin. Res., 2013; 6(5): 159-161.
- Li, H.Q., Li, X.J., Wang, Y.L., Zhang, Q., Zhang, A.L., Gao, J.M., Zhang, X.C. Antifungal metabolites from Chaetomium globosum, an endophytic fungus in Ginkgo biloba. Biochem. Syst. Ecol., 2011; 39(4-6): 876-879.
- Segismundo, A.B., Florendo, P.E., Roman, P.A. In vitro antifungal activity and phytochemical screening of Gouania javanica Miq. leaves. UNP Res. J., 2008; 17: 1-10.
- Aly, A.H., Debbab, A., Kjer, J., Proksch, P. Fungal endophytes from higher plants: a profilic source of phytochemicals and other bioactive natural products, Fungal Divers., 2010; 41(1):1-16.
- Lai, H.Y., Yau, Y.Y., Kim, K.H. Blechnum orientale Linn – a fern with potential as antioxidant, anticancer and antibacterial agent. BMC Complement. Altern. Med., 2010; 10(15): 1-8.
- Rekha, B., Bhattacharaya, S. Effect of various extracts of Desmodium gangeticum on Streptozotocin-nicotinamide induced type-2 diabetes. Asian J. Plant Sci. Res., 2013; 3(3):28-34.
- Poonam, C.P., Varsha, D. J., Shivprasad, D.M. Pharmacognostical studies on leaf of Antidesma ghaesembilla Gaertn, A promising wild edible plant. Der Pharmacia Sinica, 2013; 4(3):136-142.
© The Author(s) 2018. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.


