ISSN: 0973-7510
E-ISSN: 2581-690X
An efficient and facile method for the synthesis of 2-hydroxy-N’-{(E)-[6′-(2H-1″,2″,3″-triazol-2″-yl)pyridin-3′-yl]methylidene} benzohydrazide via N-arylation is described. The synthesized ligand was further coordinated with a variety of organotin(IV) salts. Structural elucidations of ligand and the complexes thus formed were made by CHN, IR, NMR and MS. The newly synthesized compounds were also screened for antifungal, antibacterial, cytotoxic and antiurease activities. The compounds 3,7,9 showed best antifungal activity against various strains while diorganotin (IV) moieties (7,8,10) showed excellent antibacterial activities. Compound 3 and 9 remained good antiurease and cytotoxic agents.
N-arylation, organotin(IV), 1,2,3-triazol, 2-hydroxybenzohydrazide.
The ligand design, its synthesis and characterization plays a significant role in addressing and transporting the molecule to the target, interactions with biomolecules and pathogenic resistance1. This growing field has always been at the esteem of synthetic, biological and medicinal perspectives. Among the organic compounds/ligands, the Schiff bases particularly derived from aroylhydrazones are regarded as the most privileged class of ligands owing to their versatile synthesis, novel structural features & flexibility and superior metal chelating ability. Aroylhydrazones, mostly can exhibit keto-enol tautomerism (Figure-1), that is primarily due to the solvent/reagent effect, temperature change, UV irradiations or pH change. These isomers can impart diverse chelating properties2 and are proved to be the best antimicrobial, antioxidant, anti-inflammatory, antitumour, antitubercular, anticoagulants, anticonvulsant agents3-5. Also the metal complexes of hydrazones are proved to be the best luminescent probes6, nonlinear optics, catalysts7 and molecular sensors8 etc.
The biocidal activities of the ligands increases upon chelation with suitable metal ions as it promotes their target oriented nature2. In this regard, organometallic derivatives/complexes of p-block elements, preferentially belonging to group 14, (e.g. tin(IV) and silicon(IV)), have been of significant interests. Organotin(IV) complexes with Schiff base ligands have fascinating chemical behavior, kinetically stable, relatively lipophilic in nature and less toxic than that of platinum drugs. Also the phenyl- and n- butyltin(IV) complexes display a larger array of biological activities as compared to their methyl-, hexyl- or octyltin(IV) analogs9, 10. Although there are several limitations of tin metal complexes used as pharmacological agents, like high cost, toxicity, immunogenicity, low lipid solubility, little penetration into cells etc. But these therapeutic agents still include a plethora of compounds that are mostly antitumor, antimicrobial, antioxidant, antiflammatory and radioprotectants11.
On chelation the polarity of the tin metal declines to a much greater extent, this is due to the overlap of the ligand orbital/orbitals, partial sharing of the positive charges of the metal ions and delocalization This delocalization over the whole chelate ring enhances the lipophilic characters of the complexes. That actually promotes the penetration of the complexes into lipid layer of cell membrane and blocks the active sites on enzymes of microorganisms12, 13. As a result of which the macromolecular or DNA synthesis inhibition, reduction of mitochondrial energy metabolism or elevation in oxidative and DNA damage occurs. Therefore, the organotin(IV) compounds appear to be very promising as potential drugs particularly active against cancers14.
Chemicals and reagents used in this study were research grade products and purchased from the commercial sources like Merck and Aldrich. These were used either without any further purification or purified where necessary. Analytical grade solvents were dried via methods cited in the literature before use. To determine melting points of the synthesized moieties, GallenKemp melting point apparatus was used and are uncorrected. Bruker Tensor 27 Fourier Transform Infra-Red Spectrophotometer was used in the range of 4000-400 cm-1 to collect IR spectral data. The elemental analyses (CHNS) were carried out on Carlo Erba 2400 automatic analyzer. Multinuclear NMR (1H and 13C) spectra were recorded on Varian MR Instrument at 300 MHz and/or 400 MHz in DMSO, CDCl3 or deutrated acetone using tetramethylsilane (TMS) as internal standard. The mass spectra were recorded on JEOL JMS 600-H series (EBE) MS spectrometer.
Synthesis of 6-(2′H-1′,2′,3′-triazol-2′-yl)pyridine-3-carbaldehyde (1)
N-arylation of 2H-1,2,3-triazole was co-reacted with 6-chloropyridine-3-carbaldehyde (Scheme-1), via pre-described method in literature15. Briefly, a mixture of 2H-1,2,3-triazole (0.1 mmol), anhydrous potassium carbonate (0.1 mmol), 6-chloropyridine-3-carbaldehyde (0.1 mmol), hexadecyltrimethylammonium bromide (20 µg) and dioxin (50 ml) were refluxed for the period of 18-22 hours. The progress of the reaction was monitored with the help of TLC. The precipitates of the required product were obtained in crushed ice. That were filtered, dried and recrystallized in suitable solvents like n-hexane etc.
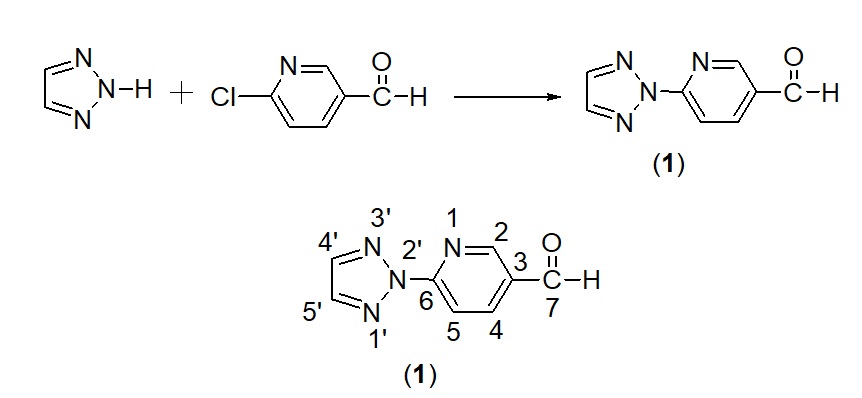
Scheme 1. Preparation and numbering scheme of 6-(2’H-1′,2′,3′-triazol-2′-yl)pyridine-3-carbaldehyde
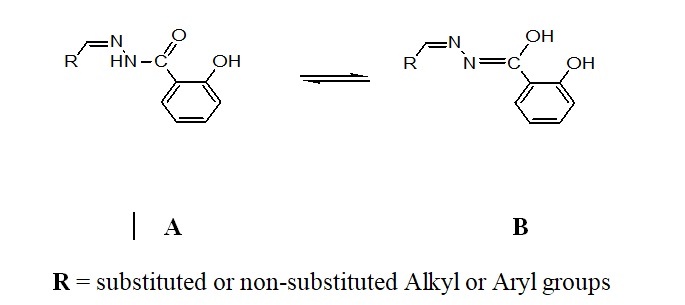 Fig. 1. Possible tautomeric forms of aroylhydrazones and E/Z configuration with respect to the C=N double bond
Fig. 1. Possible tautomeric forms of aroylhydrazones and E/Z configuration with respect to the C=N double bondSynthesis of 2-hydroxy-N’-{(E)-[6′-(2H-1″,2″,3″-triazol-2″-yl)pyridin-3′-yl]methylidene}benzohydrazide (2)
The aldehyde (1) thus formed via N-arylation was further reacted with 2-hydroxybenzohydrazide following a procedure quoted in literature16 (Scheme-2). Briefly, an equimolar (1mmol) mixture of aldehyde (1) and 2-hydroxybenzohydrazide in acetone was reflux for the period of 3-4 hours, few drops of acetic acid was added from the side wall. The reaction outcomes were monitored with TLC, the resulting solution was poured into crushed ice and left overnight. Fine crystalline product was obtained.
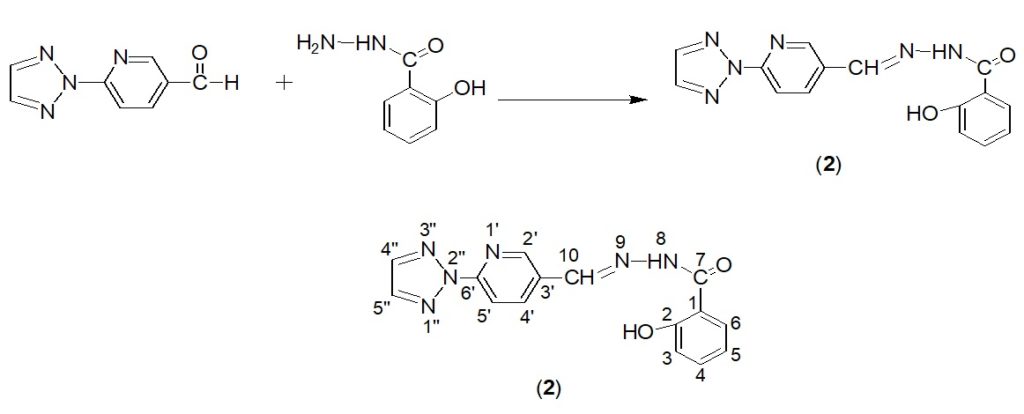 Scheme 2. Preparation and numbering scheme of 2-hydroxy-N’-{(E)-[6′-(2H-1″,2″,3″-triazol-2″-yl)pyridin-3’y]methylidene}benzohydrazide
Scheme 2. Preparation and numbering scheme of 2-hydroxy-N’-{(E)-[6′-(2H-1″,2″,3″-triazol-2″-yl)pyridin-3’y]methylidene}benzohydrazideGeneral procedure for the synthesis of complexes (3-11)
The Na-salts of the ligand i.e. 2-hydroxy-N’-{(E)-[6′-(2H-1″,2″,3″-triazol-2″-yl)pyridin-3′-yl]methylidene}benzohydrazide (2A) were further reacted with various organotin(IV) chlorides (Scheme-3) following method reported by Hussain et al.,17. Suitable dry solvents like toluene, methanol, n-hexane etc. were used for reactions to complete. All the complexes thus isolated were crystalline or amorphous solids, stable at room temperature, non-hygroscopic and having sharp melting points. The products so obtained were soluble in organic solvents like benzene, chloroform, DMF and DMSO. Further, 1H, 13C NMR, IR and mass spectrometry were carried out for their confirmation.
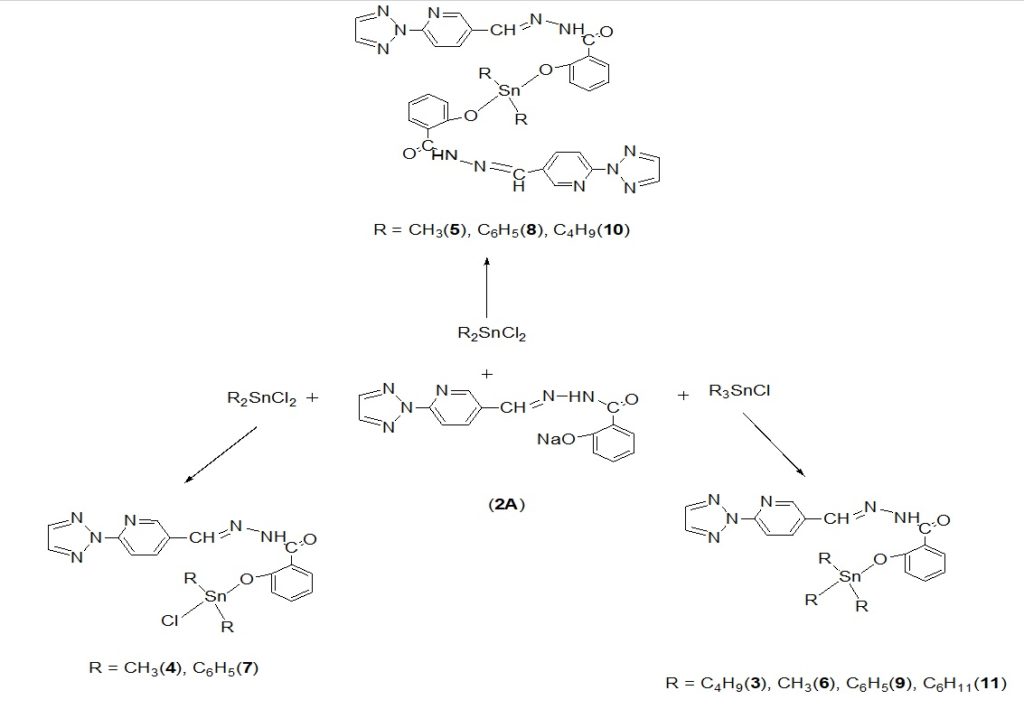
Scheme 3. Preparation of di- and tri-oraganotin(IV) carboxylates through Na-salt of 2-hydroxy-N’-{(E)-[6′-(2H-1″,2″,3″-triazol-2″-yl)pyridin-3′-yl] methylidene} benzohydrazide
Biological Activities
Pertinent literature confirmed that, the organotin(IV) complexes depict remarkable in vivo and in vitro biocidal and antimicrobial activities, also these are frequently be used in cancer chemotherapy because of their best apoptotic properties. Usually, in vitro antibacterial or fungicidal activities of the organotin(IV) follow the general order of activity as: RSnX3 < R2SnX2 < R4Sn – R3SnX, where the anionic X exhibit little influence on activity18, 19. Hubert et al. reported that the geometry and stereochemistry around tin atom while Hadjikakou et al., confirmed that it is the only ligand type not the geometry that is responsible for the antitumor activity of the organtin(IV) compounds20-22. Hence, to explore the therapeutic effects of the synthesized compounds various biological activities were performed.
Procedure for biological activities
Antifungal assay
Antifungal assay of the ligands and their organotin(IV) complexes was carried out against various fungal strains i.e. C. albicans, A. flavus, M. canis using Miconazole as a standard drug.
Procedure at 96 –Well Plates
Antifungal bioassay was performed by pre-described method15. Briefly, the test compounds were dissolved in 2% DMSO and 20µg/well of this were loaded into wells of 96 well plates. In each well, 180µl overnight preserved fungal culture was poured.A volume in each well was retained up to 200 µl. The whole mixture was then incubated at 25oC for 24 hours by covering micro-plate with a lid to ensure the aseptic environment. After incubation, the absorbance of each well was measured at 405 nm using ELISA micro-plate reader.
The percentage inhibition was calculated by employing formula,
Percentage inhibition = 100(X-Y)/X
X = Absorbance for control with fungal culture without any antifungal agent
Y= Absorbance for test sample with fungus
Cytotoxicity Assay
Cytotoxicity assay of the ligand (2) and its corresponding organotin(IV) complexes (3-11) was performed by evaluating inhibitory effects on the growth of Nauplii (brine shrimps), by using brine shrimp lethality method23.The method includes following steps. The hatching tray was filled with artificial sea water (i.e. salt solution 38g/l) and a porous membrane used to divide the tray into two halves. Brine shrimp eggs were sprinkled in one half of the tray and covered with a lid. The second half of the hatching tray was left uncovered, exposed under artificial light for 1-2days at 30 ±30C till the eggs hatched and the larvae moved towards the illuminated part of the tray via porous membrane. 10mM solutions of test compound were prepared in DMSO. From which, a specific volume of the test solution was poured in a tube and left overnight for evaporation. To this tube, the salt solution was added and the volume made up to 5ml. To this sample tube, 2-4 days old Shrimps, (10 in number) were added via Pasteur pipette. The tubes were incubated overnight and maintained 30 ±3 0C under illumination. Number of the survivors was recorded and the cytotoxicity was determined as the percentage of the dead larvae as given below:
Dead (%) = Number of larvae dead/Number of larvae added × 100
The salt solution served as the negative control and Etoposide (standard drug) act as positive control in this study. Results of the assay are given in Figure. 2.
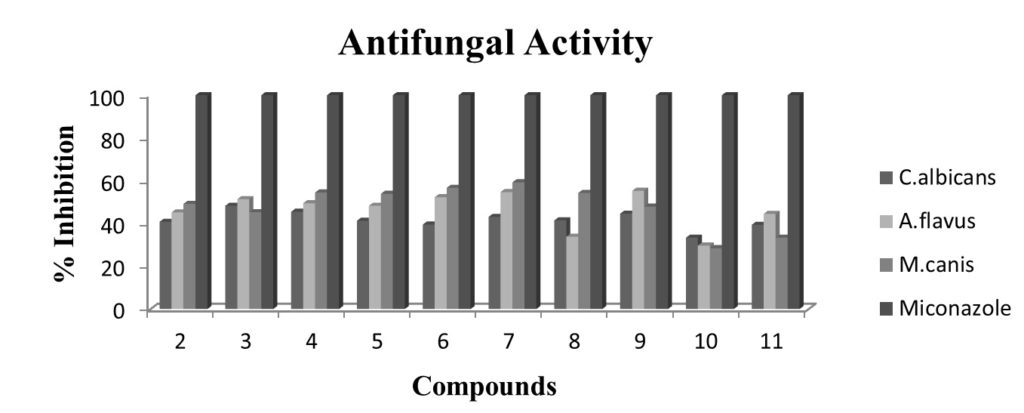
Fig. 2. Antifungal activity profile of ligand (2) and its corresponding organotin(IV) complexes by using 96 – well plates method. Each bar represents the percentage inhibition (mm) for compounds and reference drug (10mM test concentration)
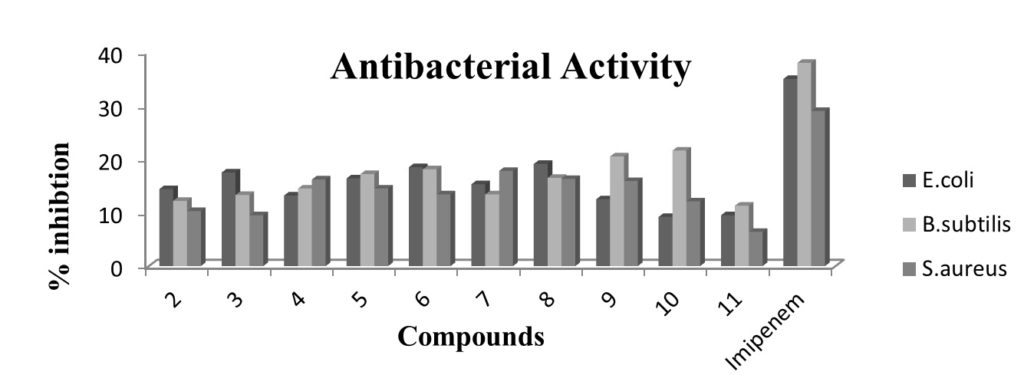
Fig. 3. Antibacterial assay profile of ligand (2) and its corresponding organotin(IV) complexes by using disc diffusion method. Each bar represents the percentage inhibition (mm) for compounds and reference drug (10mM test concentration)
Antiurease Activity
The antiurease activity test was modified from Berthelot assay24,25. Briefly explained here as, in each well of 96-well plate, 200µl assay volume containing 55µl of phosphate buffer (0.2 M) with pH 7 followed by the addition of 10µl test solution and 10µl enzyme mixture (0.015 units) was used. All the contents were incubated at 37ºC for 10 minutes. Then 10µl of urea stock solution (80mM) was added in each well and incubated at 37ºC for further 10 minutes. A pre-reading was taken at 630 nm. After that 115µl of phenol hypochlorite reagent was added in each well. The Phenol hypochlorite reagent was made freshly by combining 45µl phenol solution with 70µl alkali solution. After incubation at 37ºC for 10 minutes, the color appeared. And the absorbance was taken at 630 nm using 96-well plate reader.
Percentage Inhibition = {(O.D of positive control – O.D of Sample)/O.D of positive control} × 100
The solution having no sample served as a negative control. Wells containing thiourea served as positive control.
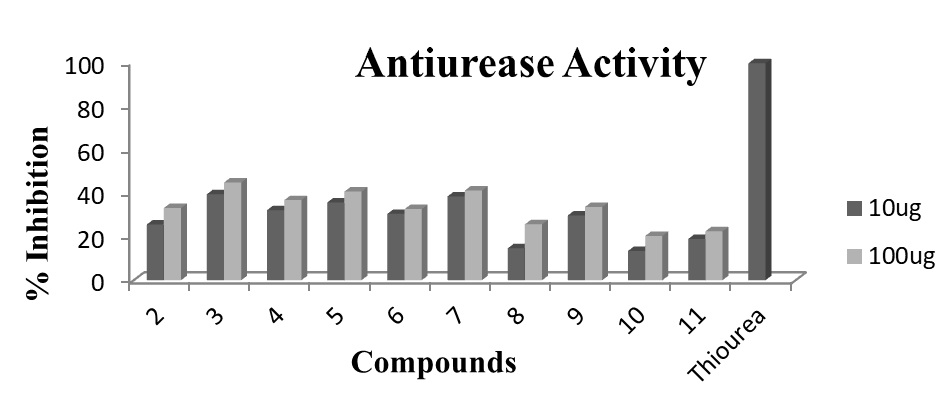 Fig. 4. Antiurease assay profile of ligand (2) and its corresponding organotin(IV) complexes by using Berthelot (phenol-hypochlorite) method via 96-well plates. Each bar depicts the percentage inhibition (mm) for compounds and reference drug (10mM test concentration)
Fig. 4. Antiurease assay profile of ligand (2) and its corresponding organotin(IV) complexes by using Berthelot (phenol-hypochlorite) method via 96-well plates. Each bar depicts the percentage inhibition (mm) for compounds and reference drug (10mM test concentration)Antibacterial Assay
Antibiotic sensitivity test was performed using a standard disc diffusion assay method by using E.coli, B. subtilis and S. aureus as indicator target strains [26] Briefly described, 50µl compound were loaded onto a sterile filter-paper disc (6 mm in diameter). A 50µl simple broth was used as a negative control, and imipenem (20µg/ml) was used as positive control. After application onto paper discs, the discs were air dried and placed onto the nutrient agar plate, which had been inoculated with a lawn of bacterial strain. After incubation for 24 h at 37oC, the antibacterial activity was evaluated by measuring the diameter of the growth-inhibition zones from the edge of each filter paper. The inhibitive radii mean (the clear zone radii in which the tested strains did not grow) was noted.
The percentage inhibition was calculated by the following formula
Percentage Inhibition = [(Rradii of positive control – radii of Sample)/ radii positive of control)] × 100
Spectral Data of Compounds 1-11
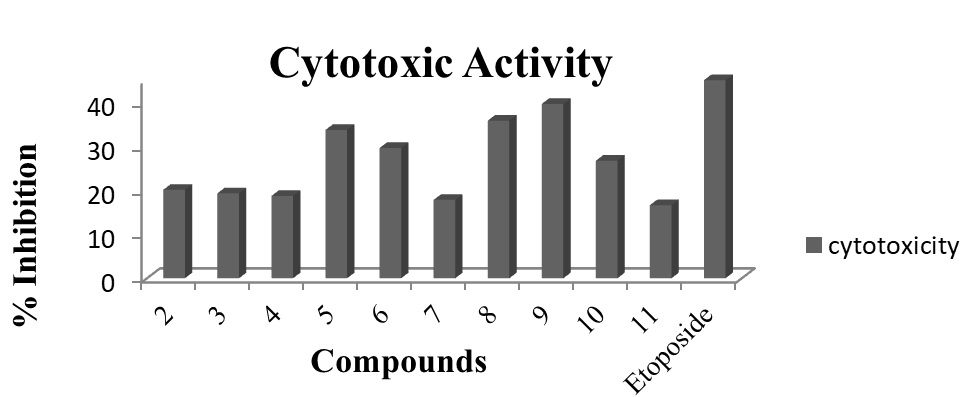 Figure 5: Cytotoxicity assay profile of ligand (2) and its corresponding organotin(IV) complexes by using brine shrimp lethality method. Each bar reveals the percentage inhibition (mm) of compounds and reference drug (10mM test concentration).
Figure 5: Cytotoxicity assay profile of ligand (2) and its corresponding organotin(IV) complexes by using brine shrimp lethality method. Each bar reveals the percentage inhibition (mm) of compounds and reference drug (10mM test concentration).6-(2′H-1′,2′,3′-triazol-2′-yl)pyridine-3-carbaldehyde (1)
White amorphous solid, 63.3% yield; 1H-NMR (400 MHz, DMSO): ´ 9.21 (s, 1H, H-2), 8.32 (d, 1H, J = 7.7Hz, H-4), 7.42 (d, 1H, J = 7.3Hz, H-5), 9.83 (s, H-7), 7.94 (d, 2H, J = 6.1Hz, H-4′,5′),; 13C-NMR (100 MHz, CDCl3): ´ 153.5 (C-2), 129.7 (C-3), 133.04 (C-4), 120.2 (C-5), 157 (C-6), 192.4 (C-7), 134.1 (C-4′,5′), IR (KBr, 4000-400cm-1): v 1279(C=O), 2985(aromatic ring), 1579(C=C), 1342 (C-N)
M.P. 150-152oC. Anal. Calcd. For C8H6N4O : C, 55.17; H, 3.47; N, 32.17; O, 9.19 Found: C, 53.67; H, 5.1; N, 32.03; O, 9.19.
2-hydroxy-N’-{(E)-[6′-(2H-1″,2″,3″-triazol-2″-yl)pyridin-3′-yl]methylidene}benzohydrazide (2)
Yellow crystalline solid, 47.5% yield; 1H-NMR (400 MHz, DMSO): ´ 5.6 (s, 1H, OH-2), 6.1 (d, 1H, J = 7.1Hz, H-3), 7.21 (t, 2H, J = 7.8Hz, H-4,5), 7.62 (d, 1H, J = 7.1Hz, H-6), 7.31 (s, 1H, H-8), 8.01 (s, 1H, H-10), 9.01 (s, 1H, H-2′), 8.3 (d, 1H, J = 9.1Hz, H-4′), 7.51 (d, 1H, J = 7.7Hz, H-5′), 8.4 (d, 2H, J = 8.7Hz, H-4’’,5’’); 13C-NMR (100 MHz, CDCl3): ´ 120.3 (C-1), 160.2 (C-2), 115.4 (C-3), 135.4 (C-4), 124.3 (C-5), 126.6 (C-6), 165.1 (C-7), 145.2 (C-10), 154.7 (C-2′), 128.1 (C-3′), 133.7 (C-4′), 125.4 (C-5′), 156.1 (C-6′), 131.2 (C-4’’,5’’); IR (KBr, 4000-400cm-1): v 3342 (OH), 1617 (-C=N), 1279(C=O), 2985(aromatic ring), 1579(C=C); M.P. 184-185oC. Anal. Calcd. For C15H12N6O2 : C, 58.44; H, 3.92; N, 27.2; O, 10.38 Found: C, 59.75; H, 2.87; N, 26.96; O, 10.38.
Tributyltin (IV)-2-hydroxy-N’-{(E)-[6′-(2″H-1″,2″,3″-triazol-2″-yl)pyridin-3′-yl]methylidene}benzohydrazide (3)
1H-NMR (400 MHz, DMSO):´ 6.52 (d, 1H, J = 7.1Hz, H-3), 7.12 (t, 2H, J = 7.6Hz, H-4,5), 7.81 (d, 1H, J = 7.9Hz, H-6), 7.3 (s, 1H, H-8), 7.62 (s, 1H, H-10), 9.51 (s, 1H, H-2′), 8.6 (d, 1H, J = 9.1Hz, H-4′), 7.5 (d, 1H, J = 7.4Hz, H-5′), 7.92 (d, 2H, J = 8.2Hz , H-4’’,5’’), 1.43 (t, 6H, J = 2.3Hz, H±,±’,±”), 1.37 (m, 6H, J = 1.9Hz, H²,²’,²” ), 1.41 (m, 6H, J = 1.8Hz, HÓ,Ó ‘,Ó “ ), 0.73 (t, 9H, J = 1.8Hz, H´,´’,´”);
13C-NMR (100 MHz, CDCl3): ´ 120.2 (C-1), 168.6 (C-2), 114.2 (C-3), 134.1 (C-4), 120.7 (C-5), 130.1 (C-6), 165.3 (C-7), 145.1 (C-10), 154.2 (C-2′), 126.1 (C-3′), 137.3 (C-4′), 125.2 (C-5′), 154.2 (C-6′), 133.6 (C-4’’,5’’), 18.4 (C ±,±’,±”), 26.2 (C ²,²’,²”), 29.3 (C Ó,Ó ‘,Ó “), 15.4 (C ´,´’,´”); IR (KBr, 4000-400cm-1): v 1622 (-C=N), 1285 (C=O), 2989 (aromatic ring), 451 (Sn-O), 494(Sn-C), 1589 (C=C);
Chlorodimethyltin (IV)-2-hydroxy-N’-{(E)-[6′-(2″H-1″,2″,3″-triazol-2″-yl)pyridin-3′-yl]methylidene}benzohydrazide (4)
1H-NMR (400 MHz, DMSO):´ 6.73 (d, 1H, J = 6.9Hz, H-3), 7.51 (t, 2H, J = 7.6Hz, H-4,5), 7.82 (d, 1H, J = 8.2Hz, H-6), 8.01 (s, 1H, H-8), 7.34 (s, 1H, H-10), 9.6 (s, 1H, H-2′), 8.2 (d, 1H, J = 8.5Hz, H-4′), 7.63 (d, 1H, J = 7.8Hz, H-5′), 7.86 (d, 2H, J = 7.5Hz, H-4’’,5’’), 2.2 (s, 6H, H±,±’);
13C-NMR (100 MHz, CDCl3): ´ 121.2 (C-1), 168.4 (C-2), 119.2 (C-3), 134.3 (C-4), 123.2 (C-5), 131.1 (C-6), 166.1 (C-7), 141 (C-10), 153.5 (C-2′), 127 (C-3′), 134.2 (C-4′), 125.3 (-5′), 155.1 (C-6′), 131.7 (C-4’’,5’’), 1.8 (C±,±’);
IR (KBr, 4000-400cm-1): v 1626 (-C=N), 1279 (C=O), 2991 (aromatic ring), 446 (Sn-O), 489 (Sn-C), 1571 (C=C);
Dimethyltin (IV) bis [2-hydroxy-N’-{(E)-[6′-(2″H-1″,2″,3″-triazol-2″-yl)pyridin-3′-yl]methylidene}benzohydrazide] (5)
1H-NMR (400 MHz, DMSO):´ 6.82 (d, 1H, J = 7.0Hz, H-3), 7.12 (t, 2H, J = 7.6Hz, H-4,5), 7.83 (d, 1H, J = 7.7Hz, H-6), 7.3 (s, 1H, H-8), 7.61 (s, 1H, H-10), 9.5 (s, 1H, H-2′), 8.6 (d, 1H, J = 8.9Hz, H-4′), 7.42 (d, 1H, J = 7.6Hz, H-5′), 9.1 (d, 2H, J = 9.4Hz. H-4’’,5’’), 1.8 (s, 6H, H±);
13C-NMR (100 MHz, CDCl3): ´ 121.3 (C-1), 167.4 (C-2), 117.2 (C-3), 134.3 (C-4), 123.2 (C-5), 129 (C-6), 165.2 (C-7), 144 (C-10), 154.2 (C-2′), 125 (C-3′), 134.1 (C-4′), 125.3 (C-5′), 151 (C-6′), 136.2 (C-4’’,5’’), 2.4 (C±,±’); IR (KBr, 4000-400cm-1): v 1619 (-C=N), 1284 (C=O), 2995 (aromatic ring), 457 (Sn-O), 492 (Sn-C), 1584 (C=C);
Trimethyltin (IV)-2-hydroxy-N’-{(E)-[6′-(2″H-1″,2″,3″-triazol-2″-yl)pyridin-3′-yl]methylidene}benzohydrazide (6)
1H-NMR (400 MHz, DMSO):´ 6.82 (d, 1H, J = 6.6Hz, H-3), 7.41 (t, 2H, J = 7.6Hz, H-4,5), 7.82 (d, 1H, J = 8.1Hz, H-6), 7.21 (s, 1H, H-8), 7.61 (s, 1H, H-10), 9.5 (s, 1H, H-2′), 8.5 (d, 1H, J = 8.2Hz, H-4′), 7.4 (d, 1H, J = 7.7Hz, H-5′), 8.1 (d, 2H, J = 8.6Hz, H-4’’,5’’), 2.1 (s, 9H, H±, ±’, ±’’);
13C-NMR (100 MHz, CDCl3): ´ 122 (C-1), 168.3 (C-2), 117.2 (C-3), 135.5 (C-4), 124.3 (C-5), 130.1 (C-6), 165.1 (C-7), 145.2 (C-10), 153.2 (C-2′), 128 (C-3′), 137.1 (C-4′), 126.2 (C-5′), 154.3 (C-6′), 135.1 (C-4’’,5’’), 3.2 (C±, ±’, ±’’); IR (KBr, 4000-400cm-1): v 1610 (-C=N), 1273 (C=O), 2995 (aromatic ring), 446 (Sn-O), 487 (Sn-C), 1573 (C=C);
Chlorodiphenyltin (IV)-2-hydroxy-N’-{(E)-[6′-(2″H-1″,2″,3″-triazol-2″-yl)pyridin-3′-yl]methylidene}benzohydrazide (7)
1H-NMR (400 MHz, DMSO):´ 6.52 (d, 1H, J = 6.1Hz, H-3), 7.42 (t, 2H, J = 7.7Hz, H-4,5), 7.81 (d, 1H, J = 8.2Hz, H-6), 7.2 (s, 1H, H-8), 7.61 (s, 1H, H-10), 9.42 (s, 1H, H-2′), 8.6 (d, 1H, J = 8.4Hz, H-4′), 7.1 (d, 1H, J = 7.2Hz, H-5′), 8.2 (d, 2H, J = 8.4Hz, H-4’’,5’’), 7.3 (m, 10H, J = 7.7Hz, Ph-H);
13C-NMR (100 MHz, CDCl3): ´ 122.1 (C-1), 164.3 (C-2), 117.3 (C-3), 136.3 (C-4), 124.2 (C-5), 130.2 (C-6), 161 (C-7), 146 (C-10), 154.2 (C-2′), 125 (C-3′), 138.1 (C-4′), 121.3 (C-5′), 154.3 (C-6′), 137.3 (C-4’’,5’’), 130.2 (C±), 135.1 (C²,²’), 131 (CÓ,Ó’); IR (KBr, 4000-400cm-1): v 1623 (-C=N), 1280 (C=O), 2987 (aromatic ring), 444 (Sn-O), 493 (Sn-C), 1588 (C=C);
Diphenyltin (IV) bis [2-hydroxy-N’-{(E)-[6′-(2″H-1″,2″,3″-triazol-2″-yl)pyridin-3′-yl]methylidene}benzohydrazide] (8)
1H-NMR (400 MHz, DMSO):´ 6.7 (d, 1H, J = 6.8Hz, H-3), 7.21 (t, 2H, J = 7.4Hz, H-4,5), 7.62 (d, 1H, J = 8.1Hz, H-6), 7.11 (s, 1H, H-8), 7.52 (s, 1H, H-10), 9.5 (s, 1H, H-2′), 8.61 (d, 1H, J = 8.8Hz, H-4′), 7.7 (d, 1H, J = 7.5Hz, H-5′), 8.01 (d, 2H, J = 8.3Hz, H-4’’,5’’), 7.23 (m, 10H, J = 7.9Hz, Ph-H);
13C-NMR (100 MHz, CDCl3): ´ 131.2 (C-1), 165.2 (C-2), 114.3 (C-3), 135.2 (C-4), 123.2 (C-5), 129.7 (C-6), 162.6 (C-7), 145.1 (C-10), 154.2 (C-2′), 127.1 (C-3′), 138.1 (C-4′), 126.3 (C-5′), 151 (C-6′), 136.6 (C-4’’,5’’), 130.2 (C±), 136.5 (C²,²’), 124.7 (CÓ,Ó’); IR (KBr, 4000-400cm-1): v 1621 (-C=N), 1276 (C=O), 3004 (aromatic ring), 458 (Sn-O), 488 (Sn-C), 1576 (C=C);
Triphenyltin (IV)-2-hydroxy-N’-{(E)-[6′-(2″H-1″,2″,3″-triazol-2″-yl)pyridin-3′-yl]methylidene}benzohydrazide (9)
1H-NMR (400 MHz, DMSO):´ 6.9 (d, 1H, J = 7.3Hz, H-3), 7.51 (t, 2H, J = 7.9Hz, H-4,5), 7.71 (d, 1H, J = 7.4Hz, H-6), 7.02 (s, 1H, H-8), 7.83 (s, 1H, H-10), 9.4 (s, 1H, H-2′), 8.7 (d, 1H, J = 8.2Hz, H-4′), 7.2 (d, 1H, J = 7.4Hz. H-5′), 8.3 (d, 2H, J = 8.8Hz, H-4’’,5’’), 7.8 (m, 15H, J = 8.3Hz, Ph-H);
13C-NMR (100 MHz, CDCl3): ´ 117.9 (C-1), 163.2 (C-2), 114.1 (C-3), 138.4 (C-4), 122.5 (C-5), 126.5 (C-6), 165.1 (C-7), 145.6 (C-10), 150.5 (C-2′), 128.3 (C-3′), 135.2 (C-4′), 124.5 (C-5′), 156.1 (C-6′), 133.6 (C-4’’,5’’), 130.2 (C±), 139.4 (C²), 123.1 (CÒ); IR (KBr, 4000-400cm-1): v 1615 (-C=N), 1270 (C=O), 2982 (aromatic ring), 442 (Sn-O), 480 (Sn-C), 1582 (C=C);
Dibutyltin (IV) bis [2-hydroxy-N’-{(E)-[6′-(2″H-1″,2″,3″-triazol-2″-yl)pyridin-3′-yl]methylidene}benzohydrazide] (10)
1H-NMR (400 MHz, DMSO):´ 6.52 (d, 1H, J = 7.1Hz, H-3), 7.12 (t, 2H, J = 7.7Hz, H-4,5), 7.81 (d, 1H, J = 7.9Hz, H-6), 6.98 (s, 1H, H-8), 7.46 (s, 1H, H-10), 9.5 (s, 1H, H-2′), 8.6 (d, 1H, J = 8.9Hz, H-4′), 7.53 (d, 1H, J = 7.7Hz, H-5′), 8.22 (d, 2H, J = 8.6Hz, H-4’’,5’’), 1.42 (t, 4H, J = 1.7Hz. H±,±’), 1.48 (m, 4H, J = 2.1Hz, H²,²’), 1.5 (m, 4H, J = 2.2Hz, HÓ,Ó’), 0.77 (t, 6H, J = 1.4Hz, H´,´’);
13C-NMR (100 MHz, CDCl3): ´ 117.8 (C-1), 165.6 (C-2), 119.2 (C-3), 136.5 (C-4), 123.2 (C-5), 129.5 (C-6), 161.2 (C-7), 146.4 (C-10), 154.5 (C-2′), 127.3 (C-3′), 138.2 (C-4′), 122.5 (C-5′), 155.1 (C-6′), 131.3 (C-4’’,5’’), 27.4 (C±,±’), 24.7 (C²,²’), 29.3 (CÓ,Ó’), 16.4 (C´,´’);
IR (KBr, 4000-400cm-1): v 1627 (-C=N), 1283 (C=O), 2988 (aromatic ring), 450 (Sn-O), 499 (Sn-C), 1574 (C=C);
Tricyclohexyltin (IV)-2-hydroxy-N’-{(E)-[6′-(2″H-1″,2″,3″-triazol-2″-yl)pyridin-3′-yl]methylidene} benzohydrazide (11)
1H-NMR (400 MHz, DMSO):´ 6.77 (d, 1H, J = 6.8Hz, H-3), 7.31 (t, 2H, J = 7.8Hz, H-4,5), 7.81 (d, 1H, J = 8.2Hz, H-6), 7.1 (s, 1H, H-8), 7.62 (s, 1H, H-10), 9.6 (s, 1H, H-2′), 8.61 (d, 1H, J = 8.3Hz, H-4′), 7.53 (d, 1H, J = 7.7Hz, H-5′), 8.3 (d, 2H, J = 8.5Hz. H-4’’,5’’), 1.5 (m, 3H, H±), 1.6;1.4 (m, 12H, H²,²’), 1.73;1.12 (m, 12H, HÓ,Ó’), 1.42;1.41 (m, 6H, H´);
13C-NMR (100 MHz, CDCl3): ´ 119.6 (C-1), 167.6 (C-2), 118.2 (C-3), 134.6 (C-4), 122.4 (C-5), 127.8 (C-6), 165 (C-7), 144.2 (C-10), 152.7 (C-2′), 125 (C-3′), 136.7 (C-4′), 120.4 (C-5′), 156 (C-6′), 132.7 (C-4’’,5’’), 9.2 (C±), 29.4 (C²,²’), 33.2 (CÓ,Ó’), 27.4 (C´);
IR (KBr, 4000-400cm-1): v 1611 (-C=N), 1286 (C=O), 3003 (aromatic ring), 449 (Sn-O), 495 (Sn-C), 1580 (C=C);
IR (4000-400 cm-1, KBr)
In free ligand 2, OH group is observed at 3342 cm-1 as a characteristic broad band. But the band vanished in the complexes (3-11) due to the deprotonation of the ligand. This gives a strong indication of the phenolic oxygen participation in coordination with the central tin atom.
Moreover, the stretching vibration of azomethine (-HC=N) for hydrazine appeared at 1617 cm-1 for ligand 2, as confirmed from the previous reports [27]. There is no significant decrease or increase in frequency (1610-1627 cm-1) of azomethine in almost all the spectra of complexes (3-11), this confirms the non-participation of nitrogen in complexation.
The carbonyl (-C=O) group showed its presence at 1279 cm-1 in 2, No significant change is detected in all the complexes (1270-1286 cm-1), it also confirms the non-participation of carbonyl oxygen in complexation.
Sn-C absorption bands appeared in the range of 487-499 cm-1 and an absorption band in the range of 442-458 cm-1 indicates the presence of a Sn-O bond in all the coordination compounds.
Antifungal activity profile of compound 2 and its corresponding organotin(IV) complexes is graphed in figure-2. Ligand (2) reveals percent inhibition against all fungal strains in the range of 40.8-49.2%. Among its chelates, compound 3 displayed highest value of 48.3% against C. albicans and the compound 9 (55.3%) exhibits highest inhibition against A. flavus. While the compound 7 (59.3%) has shown highest value against M. canis. The result shows that the activity of the ligand enhanced upon chelation that is in accordance to the previous reports28. From the results, it is also clear that ll the compounds are moderately antifungal in nature.
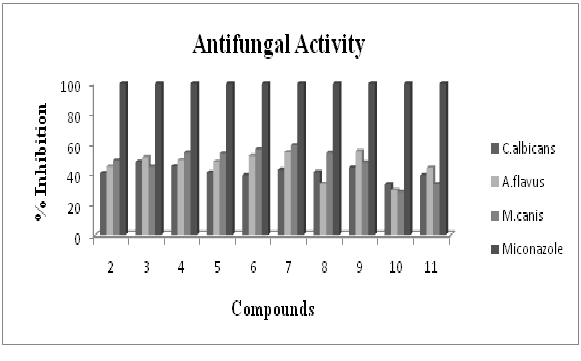 Figure 2: Antifungal activity profile of ligand (2) and its corresponding organotin(IV) complexes by using 96 – well plates method. Each bar represents the percentage inhibition (mm) for compounds and reference drug (10mM test concentration)
Figure 2: Antifungal activity profile of ligand (2) and its corresponding organotin(IV) complexes by using 96 – well plates method. Each bar represents the percentage inhibition (mm) for compounds and reference drug (10mM test concentration)Antibacterial assay profile of ligand (2) and its corresponding organotin(IV) complexes is represented in figure-3. The ligand (2) has shown percent inhibition of 12.2-14.4 against E. coli, B. subtilis and S. aureus. Among tin complexes, compound 8 depicted maximum inhibitions of 19.1% against E. coli and the compound 10 exhibited 21.6% against B. subtilis. While the compound 7 remained best inhibitor against S. aureus (17.8%). Compared with the inhibitory zones of the standard, the synthesized compounds are good to best antibacterial in nature. Also the results confirm the previous reports that the diorganotin (IV) complexes (7,8,10) are good antibacterial as compared to triorganotin (IV) complexes[29].
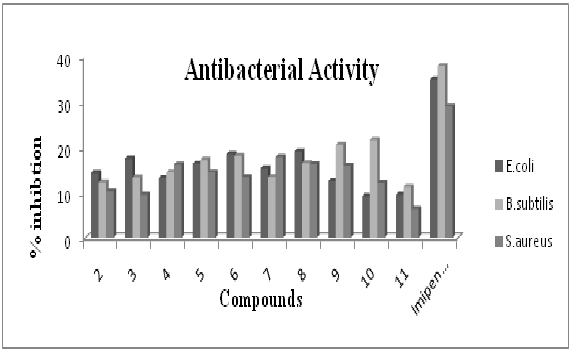
Figure 3: Antibacterial assay profile of ligand (2) and its corresponding organotin(IV) complexes by using disc diffusion method. Each bar represents the percentage inhibition (mm) for compounds and reference drug (10mM test concentration)
Figure-4 indicates that, compound 3 (tributyltin(IV)) has shown maximum antiurease inhibition of 39.4% for 10µg and 44.8% for 100µg concentration. Khan et al., (2007) also confirmed that tributyltin(IV) complexes are usually moderate antiurease agents.
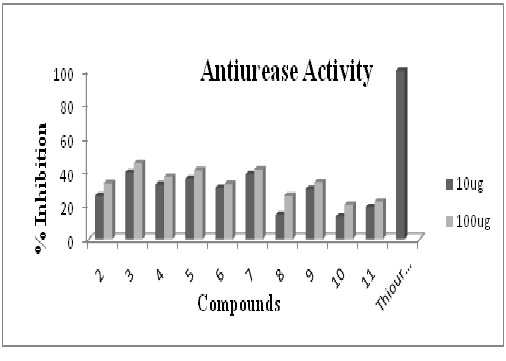 Figure 4: Antiurease assay profile of ligand (2) and its corresponding organotin(IV) complexes by using Berthelot (phenol-hypochlorite) method via 96-well plates. Each bar depicts the percentage inhibition (mm) for compounds and reference drug (10mM test concentration)
Figure 4: Antiurease assay profile of ligand (2) and its corresponding organotin(IV) complexes by using Berthelot (phenol-hypochlorite) method via 96-well plates. Each bar depicts the percentage inhibition (mm) for compounds and reference drug (10mM test concentration)Figure-5 confirms that, the compound 9 (triphenyltin (IV)) has shown maximum cytotoxicity of 39.6%. The literature also confirms its worth i.e. to be the best inhibitor29.
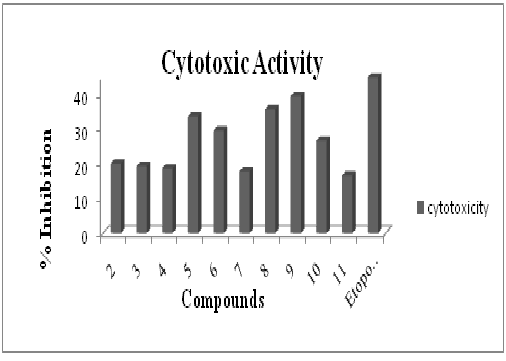 Figure 5: Cytotoxicity assay profile of ligand (2) and its corresponding organotin(IV) complexes by using brine shrimp lethality method. Each bar reveals the percentage inhibition (mm) of compounds and reference drug (10mM test concentration)
Figure 5: Cytotoxicity assay profile of ligand (2) and its corresponding organotin(IV) complexes by using brine shrimp lethality method. Each bar reveals the percentage inhibition (mm) of compounds and reference drug (10mM test concentration)ACKNOWLEDGMENTS
The authors are highly thankful to The Islamia University of Bahawalpur and HEC Pakistan for financial support.
- Alama, A..,Tasso, B..,Novelli, F.; Sparatore, F., Organometallic compounds in oncology: implications of novel organotins as antitumor agents. Drug discovery today 2009, 14(9), 500-508.
- Ferrari, M. B..,Capacchi, S..,Bisceglie, F..,Pelosi, G.; Tarasconi, P., Synthesis and characterization of square planar nickel (II) complexes with p-fluorobenzaldehyde thiosemicarbazone derivatives. Inorganica Chimica Acta 2001, 312(1), 81-87.
- Rollas, S.; Küçükgüzel, S. G., Biological activities of hydrazone derivatives. Molecules 2007, 12(8), 1910-1939.
- Vicini, P..,Incerti, M..,Doytchinova, I. A..,La Colla, P..,Busonera, B.; Loddo, R., Synthesis and antiproliferative activity of benzo [d] isothiazole hydrazones. European journal of medicinal chemistry 2006, 41(5), 624-632.
- Bezerra-Netto, H. J..,Lacerda, D. I..,Miranda, A. L. P..,Alves, H. M..,Barreiro, E. J.; Fraga, C. A., Design and synthesis of 3, 4-methylenedioxy-6-nitrophenoxy-acetylhydrazone derivatives obtained from natural safrole: new lead-agents with analgesic and antipyretic properties. Bioorganic & medicinal chemistry 2006, 14(23), 7924-7935.
- Basu, C..,Chowdhury, S..,Banerjee, R..,Evans, H. S.; Mukherjee, S., A novel blue luminescent high-spin iron (III) complex with interlayer O–Hï” Cl bridging: Synthesis, structure and spectroscopic studies. Polyhedron 2007, 26(14), 3617-3624.
- Pouralimardan, O..,Chamayou, A.-C., Janiak, C.; Hosseini-Monfared, H., Hydrazone Schiff base-manganese (II) complexes: Synthesis, crystal structure and catalytic reactivity. Inorganica chimica acta 2007, 360(5), 1599-1608.
- Bakir, M..,Green, O.; Mulder, W. H., Synthesis, characterization and molecular sensing behavior of [ZnCl 2 (ç 3-N, N, O-dpkbh)](dpkbh= di-2-pyridyl ketone benzoyl hydrazone). Journal of Molecular Structure 2008, 873, (1), 17-28.
- Shang, X..,Cui, J..,Wu, J..,Pombeiro, A. J.; Li, Q., Polynuclear diorganotin (IV) complexes with arylhydroxamates: Syntheses, structures and in vitro cytotoxic activities. Journal of inorganic biochemistry 2008, 102(4), 901-909.
- Baul, T. S. B..,Masharing, C..,Basu, S..,Rivarola, E..,Holèapek, M..,Jirásko, R..,Lyèka, A..,de Vos, D.; Linden, A., Synthesis, characterization, cytotoxic activity and crystal structures of tri-and di-organotin (IV) complexes constructed from the â-{[(E)-1-(2-hydroxyaryl) alkylidene] amino} propionate and â-{[(2Z)-(3-hydroxy-1-methyl-2-butenylidene)] amino} propionate skeletons. Journal of organometallic chemistry 2006, 691(5), 952-965.
- De Vizcaya-Ruiz, A..,Rivero-Muller, A..,Ruiz-Ramirez, L..,Kass, G..,Kelland, L..,Orr, R.; Dobrota, M., Induction of apoptosis by a novel copper-based anticancer compound, casiopeina II, in L1210 murine leukaemia and CH1 human ovarian carcinoma cells. Toxicology in vitro 2000, 14 (1), 1-5.
- Begum, T. N.; Sreeramulu, J., Synthesis And Spectroscopic Characterization Of New Azomethine Metal Complexes: Biological Activity.
- Baul, T. S. B..,Basu, S..,de Vos, D.; Linden, A., Amino acetate functionalized Schiff base organotin (IV) complexes as anticancer drugs: synthesis, structural characterization, and in vitro cytotoxicity studies. Investigational new drugs 2009, 27(5), 419.
- Leovac, V. M..,Bogdanoviæ, G. A..,Èešljeviæ, V. I..,Jovanoviæ, L. S..,Novakoviæ, S. B.; Vojinoviæ-Ješiæ, L. S., Transition metal complexes with Girard reagent-based ligands. Structural Chemistry 2007, 18(1), 113-119.
- Hussain, T..,Siddiqui, H. L..,Zia-ur-Rehman, M..,Yasinzai, M. M.; Parvez, M., Anti-oxidant, anti-fungal and anti-leishmanial activities of novel 3-[4-(1H-imidazol-1-yl) phenyl] prop-2-en-1-ones. European journal of medicinal chemistry 2009, 44(11), 4654-4660.
- Vigato, P. A.; Tamburini, S., The challenge of cyclic and acyclic Schiff bases and related derivatives. Coordination Chemistry Reviews 2004, 248, (17), 1717-2128.
- Hussain, M..,Rauf, A..,Tahir, A. A.; Ali, S., New tetrahedral, square-pyramidal, trigonal-bipyramidal and octahedral organotin (IV) 4-ethoxycarbonylpiperazine-1-carbodithioates: Synthesis, structural properties and biological applications. Journal of Organometallic Chemistry 2010, 695(10), 1526-1532.
- Kerk, V..,Der, G.; Luijten, J., Investigations on organo tin compounds. III. The biocidal properties of organo tin compounds. Journal of Chemical Technology and Biotechnology 1954, 4(6), 314-319.
- Xanthopoulou, M. N..,Hadjikakou, S. K..,Hadjiliadis, N..,Milaeva, E. R..,Gracheva, J. A..,Tyurin, V. Y..,Kourkoumelis, N..,Christoforidis, K. C..,Metsios, A. K.; Karkabounas, S., Biological studies of new organotin (IV) complexes of thioamide ligands. European Journal of Medicinal Chemistry 2008, 43(2), 327-335.
- Saxena, A.; Huber, F., Organotin compounds and cancer chemotherapy. Coordination chemistry reviews 1989, 95(1), 109-123.
- Xanthopoulou, M. N..,Hadjikakou, S. K..,Hadjiliadis, N..,Kubicki, M..,Karkabounas, S..,Charalabopoulos, K..,Kourkoumelis, N.; Bakas, T., Synthesis and characterization of a new chloro-di-phenyltin (IV) complex with 2-mercapto-nicotinic acid: study of its influence upon the catalytic oxidation of linoleic acid to hydroperoxylinoleic acid by the enzyme lipoxygenase. Journal of Organometallic Chemistry 2006, 691(8), 1780-1789.
- Balas, V. I..,Hadjikakou, S. K..,Hadjiliadis, N..,Kourkoumelis, N..,Light, M. E..,Hursthouse, M..,Metsios, A. K.; Karkabounas, S., Crystal Structure and Antitumor Activity of the Novel Zwitterionic Complex of tri-. Bioinorganic chemistry and applications 2008.
- Tariq, M..,Muhammad, N..,Sirajuddin, M..,Ali, S..,Shah, N. A..,Khalid, N..,Tahir, M. N.; Khan, M. R., Synthesis, spectroscopic characterization, X-ray structures, biological screenings, DNA interaction study and catalytic activity of organotin (IV) 3-(4-flourophenyl)-2-methylacrylic acid derivatives. Journal of Organometallic Chemistry 2013, 723, 79-89.
- Searle, P. L., The Berthelot or indophenol reaction and its use in the analytical chemistry of nitrogen. A review. Analyst 1984, 109(5), 549-568.
- Watson, C. J. In Urease activity and inhibition-principles and practice, Proceedings-International Fertiliser Society, 2000; International Fertiliser Society: pp 1-40.
- Kaspady, M..,Narayanaswamy, V. K..,Raju, M.; Rao, G. K., Synthesis, antibacterial activity of 2, 4-disubstituted oxazoles and thiazoles as bioisosteres. Letters in Drug Design & Discovery 2009, 6(1), 21-28.
- Alpert, N. L..,Keiser, W. E.; Szymanski, H. A., IR: Theory and practice of infrared spectroscopy. Springer Science & Business Media: 2012.
- James, B. D..,Gioskos, S..,Chandra, S..,Magee, R. J.; Cashion, J. D., Some triphenyltin (IV) complexes containing potentially bidentate, biologically active anionic groups. Journal of organometallic chemistry 1992, 436 (2), 155-167.
- Zuckerman, J. J., Organotin Compounds: New Chemistry and Applications. ACS Publications: 1976.
© The Author(s) 2017. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.