ISSN: 0973-7510
E-ISSN: 2581-690X
Coprinus comatus (O.F. Mull.) Pers. is a medicinal and edible mushroom containing bioactive compounds known as antidiabetic and antioxidant agents. The pancreatic β-cells were sensitive to reactive oxygen species (ROS) attacks, which can cause necrosis and disrupt insulin production. Therefore, this research aimed to evaluate the C. comatus ethyl acetate extract for antidiabetic and antioxidant to decrease fasting blood glucose, dipeptidyl peptidase-4 enzyme (DPP-4) levels, and increase glutathione peroxidase (GPx). Subsequently, 24 male Wistar rats were divided into six groups, namely 2-5 streptozotocin (STZ)-induced 45 mg/kg, 1/HC (without any treatment), 2/NC (negative control), 3/PC (administered metformin 45 mg/kg Body weight (BW), 4-6 (T1: administered 250 mg, T2: administered 500 mg, and T3: administered 750 mg/kg BW extract). The parameters were analyzed using ANOVA, followed by Duncan’s multiple range test. According to the results, the extract significantly decreased DPP-4 levels, blood glucose and increased GPx (p ≤ 0.05). The 500 mg extract effectively reduced blood glucose levels and DPP-4 enzyme as well as increased levels of GPx.
Antidiabetic, Coprinus comatus, Enzymatic Antioxidant, Medicinal Mushroom, Reactive Oxygen Species
Diabetes mellitus (DM) is a degenerative and metabolic syndrome characterized by high blood glucose levels (hyperglycemia) due to insulin deficiency or resistance. The state of hyperglycemia in people with DM causes the formation of free radicals such as anion superoxide (O2-), radical hydroxyl (OH–), and hydrogen peroxide (H2O2). Furthermore, free radicals can be neutralized and degraded by antioxidants such as glutathione peroxidase (GPx) and catalase (CAT).1
In DM, hyperglycemia patients also experience decreased endogenous or enzymatic antioxidants. Superoxide dismutase (SOD) is the first line of defense to protect β-cells from radical attack and another harmful effect of reactive oxygen species (ROS), especially O2–. Subsequently, CAT converts these free radicals into H2O2, consisting of hydrogen (H2O) and oxygen (O2) by GPx.2
Catalase is an iron-dependent enzyme concentrated mainly in the liver and erythrocytes. Meanwhile, GPx is a selenium-dependent and cytosolic enzyme, which requires reduced glutathione (GSH) as a substrate of GPx and produces oxidized glutathione (GSSG) as a product. However, the pancreas, like the brain, heart, and skeletal muscle, contains only low amounts of CAT and GPx enzyme and is sensitive to ROS attack.3
Exogenous antioxidants are needed to overcome the deficiency of enzymatic antioxidants, including from mushroom C. comatus. The phytochemical compounds in this mushroom are quercetin and rutin flavonoids. These compounds can trap free radicals while preventing complications of DM and other diseases. The inhibition of ROS makes both quercetin and rutin the ideal bioactive compounds that may provide benefits in neutralizing free radicals and protective effects against cardiovascular, diabetes, and neurodegenerative diseases.4 Flavonoids have antioxidant activity that can protect the body against damage caused by ROS by donating hydrogen atoms and binding with free radicals to form more stable flavonoid radicals. Therefore, they can inhibit lipid peroxidation that damages cells and prevents degenerative diseases like DM5 Ergothioneine, found in C. comatus mushrooms, is similar to methionine, is required to synthesize GSH and is a substrate for the enzyme GPx. Furthermore, it contains iron (Fe2+), a CAT enzyme cofactor.2
Glucose in the intestine is broken down by glucagon-like peptide 1 or GLP-1 hormone, and the peak activity concentration is 15-30 minutes. Subsequently, GLP-1 will bind to the GLP-1-receptor (GLP-1R) on the surface of the β-cells to increase insulin biosynthesis and secretion, suppress apoptotic cell death and increase the pancreatic β-cell proliferation and biosynthesis. The dipeptidyl peptidase-4 enzyme (DPP-4) can immediately break down this hormone, resulting in 65% of the successful binding with GLP-1R. Under normal conditions, the DPP-4 enzyme will degrade GLP-1 for 2 minutes. However, in a hyperglycemia state, especially in people with DM, it is broken down in less than 1 minute. The binding of GLP-1 to its receptors on pancreatic β-cells is decreased, resulting in low secretion and release of insulin hormone while increasing the blood glucose levels.6
World Health Organization (WHO) has recommended using herbal medicines such as mushrooms. However, research on the activity test as an inhibitor of the DPP-4 enzyme is still scarce. This needs to be conducted considering that DPP-4 plays a vital role in metabolism within the intestinal mucosa and affects the hormone GLP-1 as an insulinotropic.7 Furthermore, ethyl acetate as a solvent in extracting fruit bodies of C. comatus and streptozotocin (STZ) induction to produce hyperglycemic rat permanent is still rare. Based on this, there is a need to determine the activity of the fruit body extract of C. comatus by using ethyl acetate solvent on Wistar rats (R. norvegicus) induced STZ. This research aims to evaluate the changes in DPP-4 enzyme and GPx levels as an enzymatic antioxidant to evaluate and determine the dose of C. comatus extract most effective in lowering the blood glucose levels and DPP-4 hormone while increasing GPx.
Material research
C. comatus mushroom fruiting bodies were cultivated and harvested from CV Asa Agro Corporation (AAC), Cianjur. Male Wistar Rat (Rattus norvegicus) were obtained from UD Wistar Yogyakarta, and STZ (Himedia), Glucometer (Gluco Dr, Germany), GPx kit, and Rat DPP-4 Elisa kit Cat. No. E1946Ra8 were used. This research was conducted in the laboratory of the Pharmacy Department, Health Sciences Faculty, Jenderal Soedirman University (UNSOED) for animal treatment and phytochemicals compound identification in Medico Labora Laboratory (GPx measurement), Integrated Laboratory of Faculty of Medicine, Jenderal Soedirman University (DPP-4 enzyme measurement/ Enzyme-linked Immunosorbent Assay (ELISA).
Research design and animal treatment
A completely randomized design and post-test-only approach were used with a control group. A simple random sampling technique was adopted, in which the twenty-four male Wistar rats (Rattus norvegicus) with 180-200 body weight (BW) were randomly divided into six treatment groups, resulting in 4 rats per group. Each group of rats was housed and treated in a polycarbonate cage with perforated stainless-steel wire as the cover and was placed inside a climate-controlled room. The rats were fed bio pellets twice at 07.00 am and 04.00 pm daily with ad libitum access to distilled water for drink. Every 2 days, the cages were cleaned, and the base was replaced to avoid diseases and smell. The six treatment groups are HC (received no treatment; healthy control), NC (STZ-induced 45 mg/kg BW; negative control), PC (administered metformin 45 mg/kg BW; positive control), T1 (administered 250 mg/kg BW of C. comatus extract), T2 (administered 500 mg/kg BW of C. comatus extract), and T3 group (administered 750 mg/kg BW of C. comatus extract). The extract of C. comatus and the metformin were administered for 14 days orally starting on day 5 after the induction of STZ to the rats.
Phytochemicals identification
The flavonoid test was performed by steaming an amount of 2 mL of C. comatus sample extract for 4-5 minutes. The addition of 0.10 mL of hydrochloric acid showed a yellow (+), orange (++), and red color (+++). The alkaloid and terpenoid tests were performed by adding a Mayer-dragendorff-bouchardat reagent and three drops of HCl before dropping 2.0 mL of Sulfuric acid (H2SO4) into C. comatus sample extract. The polyphenol investigation was conducted by adding 5.0 mL of distilled water (sterile) to C. comatus sample extract, then steaming for 240 seconds, before adding two drops of Iron lll Chloride Hexahydrate (FeCl3). Saponins were identified and tested in boiling distilled water containing the extracted sample with 2.0-3.0 mL methanol solution. The sample was cooled and shaken for about 15-30 seconds (observe the foam forming).
C. comatus extraction (maceration method)
C. comatus fruiting bodies were cut to ±5 cm and oven-dried at 45-55°C. A total of 200 g mushroom powder is added to a glass beaker plus 1:5 ethyl acetate solvent. This was allowed to stand for 24 hours, after which the macerate was filtered with a Millipore vacuum. The powder was macerated on the second (1:3) and third day (1:2). Subsequently, the total macerate was combined and evaporated on the rotary vacuum evaporator. The thick extract produced by evaporation is weighed following the dose to be administered and is ready for testing.
Streptozotocin induction
The experimental rats were acclimatized to the climate-controlled room for 7-10 days. The room’s temperature was maintained between 20-22°C with 60-65% humidity. Subsequently, 0.5 mL of STZ was administered intraperitoneally at a 45 mg/kg BW dose in buffer citrate solution 0.1M at pH 4.5. STZ induction was performed on all rats of the treatment groups, except the HC group as healthy control. The experimental animals classified as hyperglycemic, with fasting blood glucose levels of ≥250 mg/dL 7, were used as the treatment group. Before the treatment, the rats were confirmed and evaluated in good physical health condition.
Plasma blood sample collection and blood glucose measurement
A blood serum sample was obtained to measure the main parameters. Before the samples were collected, all experimental animals fasted for 12 hours. Initial/baseline blood glucose levels were obtained from the lateral veins on day four after induction of STZ. The blood directly dripped 1-2 µL onto the GlucoDr glucose strips. On day 15, after the treatment of C. comatus extract, a sample was obtained from the orbital veins of the rats. It was used to measure final blood glucose levels (using GlucoDr strips), and the plasma was obtained using a centrifuge machine at 6,000 rotations per minute (rpm). Subsequently, it was stored in a refrigerator with a temperature of 2-8°C.
DPP-4 Measurements
DPP-4 enzyme measurement followed the manufacturer’s procedure of BT Laboratory. Initially, 50 μL of the standard solution was added to the wells with a 40 μL sample and 10 μL of DPP-4 antibody. An amount of 50 μL streptavidin-HRP was added to all the wells and incubated for 60 minutes at a temperature of 37°C. The well plate was washed five times before soaking in a healthy plate containing 300 μL of wash buffer for 30-60 seconds. Consecutively, 50 μL of substrate solution A and B were added to each well. The plate was sealed with a new sealer for 10 minutes at 37°C in dark conditions. After incubation, 50 μL of stop solution changed the color from blue to yellow. The optical density (OD) measurement was taken using a microplate reader at a wavelength of 450 nanometers (nm) for 10 minutes.
GPx Measurement
GPx measurements were performed using 500 μL of PBS, 850 μL of sterile water, and 50 μL of homogenized Cety tri-methyl ammonium bromide (CTNB) placed into a cuvette. Subsequently, 50 μL of plasma samples were added to the cuvette, and the absorbance value was read at 340 nm wavelength in the first and second minute using a spectrophotometer.
Ethical Approval
The Committee approved the experimental research design, animal treatment and the procedures of Health Research Ethics, Faculty of Medicine (FK), Universitas Jenderal Soedirman (UNSOED), Indonesia (ethical approval number: 3798/KEPK/VIII/2019). The ethical requirements are based on principles of 3R: replacement, reduction and refinement. Furthermore, it considers the principles of 5F: freedom from discomfort, pain, injury and diseases, thirst and hunger, fear and distress, and the expression of the natural behavior of the rats. Following the recommendations of the Institutional Committee of Animal Care and Use (IACUC), the termination procedure was conducted to end the suffering of experimental animals.
Statistical analysis
All parameter data were presented as a mean ± standard of error (SE) and the independent sample. One-way variance analysis (ANOVA) and Duncan’s (multiple range tests) were used. The correlation test was conducted using the SPSS statistical software package (v.26.0), and the P values (p ≤ 0.05) were used as statistical significance different of data.
The results showed that the extraction process of 250 g of C. comatus fruiting bodies using 1,000 mL semi-polar ethyl acetate solvent yielded 2.25 g of thick extract. Table 1 shows that the phytochemical compounds of the mushroom of C. comatus extract used ethyl acetate solvent.
Table (1):
Qualitative identification of C. comatus phytochemicals compound.
No |
Phytochemicals Compounds |
Chemicals Reagent |
Result (Qualitative) |
|---|---|---|---|
1 |
Flavonoids |
Mg/Zn + HCl +Amyl Alcohol |
Reddish orange (++) |
2 |
Alkaloids |
Mayer, Dragendorff, Bouchardat dye |
Dark brown (+) |
3 |
Terpenoids |
HCl + H2SO4 |
Purplish black (+) |
4 |
Polyphenol |
FeCl3 |
Greenish Blue (+) |
3 |
Saponins |
Distilled water + boiling + methanol |
Formed Foam (++) |
Notes: Plus signs in table were indicate the following information: + (adequate concentration), ++ (moderate concentration), and +++ (strong concentration).
The ethyl acetate extract of C. comatus mushroom contained phytochemical compounds such as flavonoids, alkaloids, terpenoids, polyphenols, and saponins. The results of qualitative identification (Table 1) of flavonoids showed a moderate compound concentration with an indicator of a color change to reddish-orange (++). Meanwhile, the identification of alkaloids showed a color change to dark brown (+), indicating adequate alkaloids after adding Mayer, Dragendorff, and Bouchardat dye. The identification of terpenoids and polyphenols also showed similar values with a change in color of the terpenoids to blackish purple (+)and turquoise (+) for polyphenols indicating an adequate concentration of the compounds. Moderate saponin concentration was identified, where after heating the C. comatus extract sample and adding 3 drops of methanol, the shaking process formed a reasonably stable foam (++) for 1 minute.
Previous studies have shown qualitative identification of the fruiting bodies of C. comatus ethyl acetate extract containing a moderate concentration of flavonoids compound (++), low levels of alkaloids (+), and saponins (+).7 In the qualitative test of other studies, the mycelium extract of C. comatus using ethyl acetate solvent with a concentration of 100 parts per million (ppm) was identified to contain a strong concentration of flavonoids (+++), polyphenols (+++) and terpenoids (+).2 The same results were also shown from the qualitative identification of the C. comatus ethanol extract of fruiting bodies containing moderate and low flavonoids (++) and saponins (+).5 In another study, 70% and 95% ethanolic C. comatus extracts was found to contain phenolic compounds and have acted as an antioxidant in DPPH radicals.9
Flavonoids and polyphenols are the main compounds that act as antioxidants, while saponin and alkaloids serve as antidiabetic agents. Furthermore, alkaloids become inhibitors of the DPP-4 enzyme that degrades the GLP-1 hormone.10 Previous research showed that alkaloids had inhibition activity of a-amylase, a-glucosidase, aldose reductase, and DPP-4 enzyme. They also have a role in inhibiting advanced glycation end products (AGEs), increment of insulin release and secretion from pancreatic β-cell and its sensitivity, and the enhancement of glucose uptake from circulation to lower the levels.11 Additionally, saponins play a role in preventing excess glucose uptake in the intestinal mucosa to minimize catabolism by increasing the activity of adenosine monophosphate kinase (AMPK).7 Another role is increasing insulin signalling to maintain consistent production and reduce blood glucose levels. Previous research has also shown that saponins reduce and minimize complications due to obesity and inflammation and alleviate liver necrosis, injury, and pain in insulin hormone-resistant rats while exhibiting signal transduction.12 After the C. comatus extract was given to the diabetic rat, these bioactive compounds decreased the blood glucose level.
Other research has shown that the C. comatus fruiting body extract contains quinic acid. The activity of quinic acid compared to the C. comatus fruit body extract as an antioxidant was greater with 4 μmol/L inhibition. In contrast, the C. comatus fruit body extract was 2.3 μmol/L using the HydroxoPerhydroxo Mercury(II) Complex (HPMC) method.13 Two flavonoids with the highest amount are quinic acid at 14.60 mg/100 g and the quercetin compound at 3.010 mg/100 g.14 The activity as an antioxidant is essential to protect pancreatic β-cells from free radical attack. Another study stated that ethanol extract of C. comatus cap could ward off free radicals DPPH with an EC50 value of 0.860 mg/mL, against OH– radicals 3.230 mg/mL, and against O2- radicals 25.30 mg/mL.15 Therefore, C. comatus is effective in neutralizing OH– radicals.16
The measurements of blood glucose levels also showed that STZ induction after 96 hours increased fasting blood glucose levels >150 mg/dL (initial data). Similarly, the diabetogenic agents increased the fasting blood glucose to >150.0 mg/dL, and the highest was >185 mg/dL. The levels are presented in Figure 1, and the decreasing percentage is shown in 2.
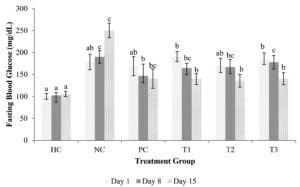
Figure 1. Fasting blood glucose levels after the administration of the C. comatus extract
Notes: Data is presented as mean±standard deviation (n=4). The columns marked with an identical letter indicate the difference is insignificant (p ≤ 0.05). The groups are Healthy control (HC), Negative control (STZ induction 45 mg/kg BW) (NC), Positive control (Metformin 45 mg) (PC), Administered C. comatus extract of 250 mg (T1), Administered C. comatus extract dose of 500 mg (T2), and Administered C. comatus extract dose of 750 mg (T3)
The highest and lowest blood glucose level on day 5 was in the T1 and HC group, with 190.6 mg/dL and 100.25 mg/dL. Blood glucose levels were decreased on day 8 after the administration of C. comatus ethyl acetate extract and metformin. The blood glucose levels of the group that was administered decreased significantly (p < 0.05) (Figure 1) in the T1 group by 15.46%. Meanwhile, the NC group has increased blood glucose levels (Figure 1) from 178.6 mg/dL to 190.6 mg/dL and 250.15 mg/dL on day 15.
The percentage decrease was measured by comparing the initial level with the value obtained at the measurement time. The PC group also showed a downward trend on days 8 and 15, from 168.8 mg/dL (day 1), 146.58 (day 8), and 140.5 mg/dL (day 15). On day 15 the T1 group showed the most significant decrease (p < 0.05) with 26.86% (190.6 mg/dL to 139 mg/dL), while in the T2 group with 20.53% (170 mg/dL to 135.5 mg/dL) and the T3 group 24.45% (185.57 mg/dL to 140.2 mg/dL) (Figure 1).
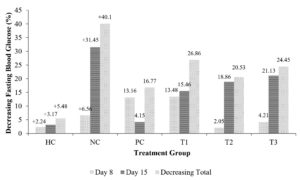
The results showed that administering C. comatus extract for 15 days significantly decreased blood glucose levels (Figure 2). Meanwhile, the NC group’s fasting blood glucose increased on the 8th and 15th days, while the highest decrease was observed in group T1. On the 8th and 15th days, it decreased by 15.46% and 26.86% compared to the first day. This study has shown that the C. comatus extract could lower and reduce fasting blood glucose levels by an average of 23.95% (Figure 2).
Figure 2. The decrease in the percentage of fasting blood glucose levels in the treatment group compared to the initial blood glucose levels
Notes: Data is presented as mean±standard deviation (n=4). The columns marked with an identical letter indicate the difference is insignificant (p ≤ 0.05). The groups are Healthy control (HC), Negative group (STZ induction 45 mg/kg BW) (NC), Positive control (Metformin 45 mg) (PC), Administered C. comatus extract of 250 mg (T1), Administered C. comatus extract dose of 500 mg (T2), and Administered C. comatus extract dose of 750 mg (T3)
A previous study has also shown that the ethyl acetate extract of C. comatus at 250 mg doses can reduce blood glucose levels by 23.92% compared to the initial levels after 15 days of treatment using the extract.7 In addition, the administration at a dose of 750 mg can reduce the level by 26.69%.5
STZ induction in all treatment groups of rats not treated showed an increase of >230 mg/dL of fasting blood glucose levels after 14 days.7 Another study that induced alloxan 200 mg administered a dose of C. comatus extract at 750 mg showed a 50.05% decrease after 14 days, while rats without the extract showed an increase of 47.25%.17
STZ directly affects pancreatic cells by entering through the glucose transporter (GLUT)-2 mechanism. This stimulated the formation of nitric oxide (NO-) and caused DNA alkylation. High NO- will cause an increase in xanthine oxidase (XOD) enzyme activity and triggers the biosynthesis of free radicals such as O2- and H2O2 that can lead to oxidative stress.18 The uptake of streptozotocin in pancreatic β-cells by GLUT-2 receptors caused oxidative stress reaction due to NO– production and release, and ROS production caused inflammation which damages pancreatic β-cell. The subsequent effects are oxidative stress, lipid peroxidation of cell membranes, and a decrease in ATP formation. Due to the damage to pancreatic β-cell, insulin production is decreased, leading to increased fasting blood glucose.19 The conditions under which STZ causes cell necrosis, membrane damage, and raises blood pressure vary according to physiology, biochemistry, and immunity.
The increased glycoxidation reaction produces free radicals (ROS) such as anion superoxide, hydroxyl radicals and hydrogen peroxide. This accelerates the formation and production of AGEs, which create more free molecules and can contribute to the necrosis of pancreatic β-cell. Crosslink formation caused the reaction and interactions between AGEs molecules with their receptors (RAGEs) and contributed to the development and progression of diabetes disease.20 Furthermore, flavonoids in C. comatus extract can suppress ROS formation and prevent the development of pancreatic β-cell damage. This is consistent with previous studies as evidenced by histology observations of pancreatic tissue, where the treatment of given extract doses of 500 and 750 mg showed fewer inflammatory reactions, pyknosis, and karyolitic of β-cells compared to the negative control. The insulin levels show that the pancreas is in good condition with >7.1 mIU/mL but was very low at 6.8 mIU/mL in the NC group.5
Glutathione peroxidase levels in diabetic rats were examined 15 days following ethyl acetate extract C. comatus and metformin administration (Table 2).
Table (2):
GPx levels in STZ-induced hyperglycemic rat model.
No |
Treatment groups |
GPx levels (U/mL) |
% Increase |
|---|---|---|---|
1 |
HC |
23.0 ± 5.94 ab |
160.7 |
2 |
NC |
8.82 ± 3.33 a |
-61.65 |
3 |
PC |
33.0 ± 16.73b |
274.41 |
4 |
T1 |
39.0 ± 4.94 b |
342.17 |
5 |
T2 |
38.5 ± 16.50 b |
336.51 |
6 |
T3 |
22.2 ± 8.06 ab |
151.70 |
Notes: Data is presented as mean±standard deviation (n=4). The columns marked with an identical letter indicate the difference is insignificant (p ≤ 0.05). The groups are Healthy control (HC), Negative control (STZ induction 45 mg/kg BW) (NC), Positive control (Metformin 45 mg) (PC), Administered C. comatus extract of 250 mg (T1), Administered C. comatus extract dose of 500 mg (T2), and Administered C. comatus extract dose of 750 mg (T3).
The C. comatus extract at 250 mg and 500 mg doses administered to hyperglycemic rats significantly increased enzymatic antioxidant levels of GPx (p < 0.05). The group that received ethyl acetate extract with doses of 250 and 500 mg had GPx activity of 39.025 U/mL and 38.5 U/mL, respectively, which were higher than the NC group. Meanwhile, GPx levels in the NC group were below the normal limit of 8.82 U/mL. Normal GPx levels in experimental animal DM conditions are 20-25 U/mL5 since C. comatus contains a common type of flavonoid compound. Previous research has shown that administration of C. comatus ethanol extract 250 mg and 500 mg increased the enzymatic antioxidant of GPx levels of 39 U/mL and 38.5 U/mL.2
The previous study shows flavonoid compounds can inhibit lipid peroxidation by donating hydrogen ions (H+) and reducing tissue damage by free radicals.2 The hydroxyl and carboxyl of C. comatus polysaccharide were the groups that acted as electronic donors.21 Furthermore, C. comatus contains the ergothioneine compound, similar to methionine, a substrate of GPx. However, the GSH substrate regenerates with ergothioneine, and GPx activity returns to normal.17
The GPx level in the negative control group induced with STZ is at the lowest level. Streptozotocin that enters pancreatic β-cells through the GLUT2 transporter causes hyperglycemia, affecting ROS production and lipid peroxidation. Meanwhile, lipid peroxidation will damage pancreatic β-cells and cause necrosis because endogenous antioxidants such as GPx and catalase are lesser than in other organs. This increases blood glucose due to decreased insulin production and GPx levels.22
C. comatus contains flavonoid compounds like rutin and tocopherol (vitamin E), capable of breaking down the lipid peroxidation chain reaction. Flavonoids with many OH- groups can directly counteract free radicals by donating H+ and reducing the activity. They change radical molecules into flavonoid radicals, unreactive compounds against cells.23 Previous research showed that the content in the ethyl acetate extract was 16.40 mg/L or about 32.8%, which enabled the effectiveness of C. comatus in suppressing free radicals. Therefore, insulin production remained optimal, and homeostasis of blood glucose levels was maintained in experimental animals induced by STZ7. The extract also contains a Fe mineral of 1471 mg/Kg and a catalase enzyme cofactor. The catalase activity can reduce peroxide compounds in cells, such as H2O2, and convert them into H2O and O2. In addition, it contains cofactors that support the biosynthesis of Cu-SOD, Mg-SOD, and Zn-SOD, because the trace element content of Cu 10.17 mg/Kg, Mg 1334 mg/Kg, and Zn 31.73 mg/Kg allows the process to increase. and indirectly increase cell defense from oxidative stress.24
Dipeptidyl peptidase-4 levels in diabetic rats were measured 15 days following ethyl acetate extract C. comatus and metformin administration.
Table (3):
DPP-4 levels in STZ-induced hyperglycemic rat model.
No |
Treatment group |
DPP4 level (U/mL) |
% Decrease of DPP-4 Compared to NC Group |
|---|---|---|---|
1 |
HC |
78.35± 2.8a |
55.59 |
2 |
NC |
176.43± 3.5c |
+125.18 |
3 |
PC |
96.21± 4.7b |
47.74 |
4 |
T1 |
97.38± 5.6b |
44.81 |
5 |
T2 |
83.29± 4.4a |
52.79 |
6 |
T3 |
83.27± 3.9a |
53.37 |
Notes: Data is presented as mean±standard deviation (n=4). The columns marked with an identical letter indicate the difference is insignificant (p ≤ 0.05). The groups are Healthy control (HC), Negative control (STZ induction 45 mg/kg BW) (NC), Positive control (Metformin 45 mg) (PC), Administered C. comatus extract of 250 mg (T1), Administered C. comatus extract dose of 500 mg (T2), and Administered C. comatus extract dose of 750 mg (T3).
Table 3 shows the results of DPP-4 before and after treatment with C. comatus extract. Furthermore, 250 mg (T1), 500 mg (T2), and 750 mg (T3) doses were compared to the PC and NC group of rats. The highest level of DPP-4 enzyme was in the NC group with 176.43 U/mL, and the lowest was in the HC group with 78.35 U/mL. The treatment of C. comatus extract was generally lower than that of the NC group. Administration of 250 mg extract showed DPP-4 levels of 97.38 U/mL, while at a dose of 500 mg, there was a decrease in DPP-4 levels of 83.29 U/mL, comparable to a dose of 750 mg of 83.27 U/mL. The percentage decrease in DPP-4 enzyme levels compared to the NC group showed that the group of T3 had the highest decrease, with 53.37%.
According to the results, the average level of DPP-4 hormone in the hyperglycemic rat group decreased. The ANOVA analysis shows that the results were significant at p ≤ 0.05, and doses of T2 and T3 are at the same level of DPP-4. A substantial decrease in the levels of the DPP-4 enzyme in the hyperglycemic treatment group was possible. This is because of the serine enzyme endovascular inhibitory mechanism, such as DPP-4, by bioactive compounds in the form of comatin detected in the C. comatus extract. Comatin will stick to the DPP-4 enzyme to deactivate DPP-4 and promote GLP-1 activity.5
Previous studies demonstrated that comatin acts as a DPP-4 enzyme inhibitor by blocking the enzyme from connecting to and interacting with the GLP-1 hormone. It replaces DPP-4, which can attach to the active site of the GLP-1 hormone. Hence, DPP-4 can no longer attach to GLP-1, and the inactivation effect and the degradation of DPP-4 are minimized. Furthermore, comatin does not affect GLP-1, and its insulinotropic activity can normally run.6
C. comatus contains rutin which can increase antioxidant defenses in pancreatic cells and prevent cell damage due to oxidative stress. Rutin activity as antioxidant defense is imperative to prevent deterioration of pancreatic cells, hence the binding of GLP-1 hormone with GLP-1R remains optimal.7 A previous study showed that the decrease of the DPP-4 enzyme corresponds with the increasing levels of GLP-1 hormone after the oral treatment of C. comatus extract. The administration of 750 mg of C. comatus ethanol extract exhibited the most effective effect of decreasing DPP-4 enzyme levels in diabetic rats models induced with STZ 45 mg.5
This DPP-4 enzyme is essential because an increase directly affects the incretin hormone. Previous studies have shown that inhibition of DPP-4 can reduce glycosylated haemoglobin (HbA1c), affecting the function of pancreatic cells in producing insulin and blood pressure.25 The incretin hormones GIP and GLP-1 contribute 50-70% of the insulin hormone response in controlling blood glucose levels. Low GIP and GLP-1 hormones can lead to type 2 DM.26 Furthermore, the GLP-1 hormone affects the neogenesis, proliferation, and differentiation of pancreatic β-cells, modulated by a protein homeodomain (PDX-1), in the mature pancreas. The expression of PDX-1 is limited to insulin-releasing and production from pancreatic β-cells.27 Therefore, the role of C. comatus bioactive compounds containing flavonoids as exogenous antioxidants and the effect on increasing GPx can provide pancreatic cell defense from free radicals. Insulin biosynthesis and secretion will continue to run normally with the inhibitory effects of C. comatus against DPP-4, which are implicated in increased insulinotropic activity.7
The results demonstrated that C. comatus ethyl acetate extract in diabetic rats induced by streptozotocin at different doses could lower the value of GPx and DPP-4 in diabetic rats. Furthermore, the extract at 500 mg/kg BW effectively reduces fasting blood glucose and DPP-4 levels while increasing GPx as enzymatic antioxidants.
ACKNOWLEDGMENTS
The authors would like to thank Directorate General of Higher Education, Ministry of Education and Culture, Republic of Indonesia (DIRJEN-DIKTI) for its Kedaireka Matching Fund Program which has assisted in financing the publication of this research, and Jenderal Soedirman University (UNSOED) for supporting the research. The authors are also thankful to the late Ir. Triono Untung Priyadi for providing samples of mushroom fruiting bodies, and Muhammad Faishal Dzaky, who has helped with corrections and writing assistance, and laboratory assistants of Mycology Laboratory and Research Center Laboratory, Faculty of Medicine for assisting in tool preparation and analysing parameters.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
NIR, ESW, FH designed the study and carried out the laboratory work. NIR, FH, ARH, DS, AA worked on analyzing the data. FH, NIR, and DS wrote the manuscript. All authors have read and approved the final manuscript for publication.
FUNDING
This study was supported by the Institute of Research and Community Service (LPPM) through a Business Service Agency (BLU) Research Proposal under contract P/308/UN23/PN/2019.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
This study was approved by the Institutional Ethics Committee, Universitas Jenderal Soedirman (UNSOED), Indonesia with ethical approval number 3798/KEPK/VIII/2019.
- Ratnaningtyas NI, Hernayanti, Ekowati N, Husen F. Nephroprotective and antioxidant effects of ethanol extract of Coprinus comatus mushroom fruit-bodies on streptozotocin-induced diabetic rat models. In: The 4th International Conference on Biosciences (ICoBio 2021). IOP Conference Series: Earth and Environmental Science; 2021:948(1-13).
Crossref - Ratnaningtyas NI, Hernanyanti, Ekowati N, et al. Antioxidant activities and properties of Coprinus comatus mushroom both mycelium and fruiting body extracts in streptozotocin-induced hyperglycemic rats model. Biosaintifika Journal of Biology & Biology Education. 2022;14(1):9-21.
Crossref - Dworzański J, Strycharz-Dudziak M, Kliszczewska E, et al. Glutathione peroxidase (GPx) and superoxide dismutase (SOD) activity in patients with diabetes mellitus type 2 infected with Epstein-Barr virus. PLoS One. 2020;15(3):e0230374.
Crossref - Bhat IUH, Bhat R. Quercetin: A bioactive compound imparting cardiovascular and neuroprotective benefits: Scope for exploring fresh produce, their wastes, and by-products. Biology (Basel). 2021;10(7):1-34.
Crossref - Ratnaningtyas NI, Hernayanti H, Ekowati N, Husen F. Ethanol extract of the mushroom Coprinus comatus exhibits antidiabetic and antioxidant activities in streptozotocin-induced diabetic rats Nuniek. Pharm Biol. 2022;60(1):1126-1136.
Crossref - Kaneto H, Kimura T, Shimoda M, et al. Favorable effects of GLP-1 receptor agonist against pancreatic β-cell glucose toxicity and the development of arteriosclerosis: “the earlier, the better” in therapy with incretin-based medicine. Int J Mol Sci. 2021;22(15):7917.
Crossref - Husen F, Hernayanti H, Ekowati N, Sukmawati D, Ratnaningtyas NI. Antidiabetic effects and antioxidant properties of the saggy ink cap medicinal mushroom, Coprinus comatus (Agaricomycetes) on streptozotocin-induced hyperglycemic rats. Int J Med Mushrooms. 2021;23(10):9-21.
Crossref - Bioassay Technology Laboratory (BT. Laboratory). Rat Dipeptidyl Peptidase IV (DPP IV) ELISA KIT Procedure. Biotech. Co., Ltd.; 2018.
- Nowakowski P, Markiewicz-Zukowska R, Gromkowska-Kępka K, et al. Mushrooms as potential therapeutic agents in the treatment of cancer: Evaluation of anti-glioma effects of Coprinus comatus, Cantharellus cibarius, Lycoperdon perlatum and Lactarius deliciosus extracts. Biomed Pharmacother. 2021;133:111090.
Crossref - Shukla A, Srinivasan BP. 16,17-dihydro-17b-hydroxy isomitraphylline alkaloid as an inhibitor of DPP-IV, and its effect on incretin hormone and β-cell proliferation in diabetic rat. Eur J Pharm Sci. 2012;47(2):512-519.
Crossref - Adhikari B. Roles of Alkaloids from Medicinal Plants in the Management of Diabetes Mellitus. J Chem. 2021:1-10.
Crossref - Feng M, Liu F, Xing J, Zhong Y, Zhou X. Anemarrhena saponins attenuate insulin resistance in rats with high-fat diet-induced obesity via the IRS-1/PI3K/AKT pathway. J Ethnopharmacol. 2021;277:114251.
Crossref - Karaman M, Tesanovic K, Gorjanovic S, et al. Polarography as a technique of choice for the evaluation of total antioxidant activity: The case study of selected Coprinus Comatus extracts and quinic acid, their antidiabetic ingredient. Nat Prod Res. 2019;35(10):1711-1716.
Crossref - Tesanovic K, Pejin B, Sibul F, et al. A comparative overview of antioxidative properties and phenolic profiles of different fungal origins: fruiting bodies and submerged cultures of Coprinus comatus and Coprinellus truncorum. J Food Sci Technol. 2017;54(2):430-438.
Crossref - Li B, Lu F, Suo X, Nan H, Li B. Antioxidant properties of cap and stipe from Coprinus comatus. Molecules. 2010;15(3):1473-1486.
Crossref - Mwangi RW, Macharia JM, Wagara IN, Bence RL. The antioxidant potential of different edible and medicinal mushrooms. Biomed Pharmacother. 2022;147:112621.
Crossref - Ratnaningtyas NI, Hernayanti, Ekowati N, Sukmawati D, Widianti H. Chicken drumstick mushroom (Coprinus comatus) ethanol extract exerts a hypoglycaemic effect in the Rattus norvegicus model of diabetes. Biocatal Agric Biotechnol. 2019;19:101050.
Crossref - Hasanuzzaman M, Bhuyan MHMB, Zulfiqar F, et al. Reactive oxygen species and antioxidant defense in plants under abiotic stress: Revisiting the crucial role of a universal defense regulator. Antioxidants. 2020;9(8):681.
Crossref - Thabet Al-Nahdi A, John A, Raza H. Streptozotocin-induced molecular and metabolic targets in pancreatic beta-cell toxicity. Hamdan Med J. 2019;12(2):65-71.
Crossref - Rungratanawanich W, Qu Y, Wang X, Essa MM, Song BJ. Advanced glycation end products (AGEs) and other adducts in aging-related diseases and alcohol-mediated tissue injury. Exp Mol Med. 2021;53(2):168-188.
Crossref - Govindan S, Jayabal A, Shanmugam J, Ramani P. Antioxidant and hepatoprotective effects of Hypsizygus ulmarius polysaccharide on alcoholic liver injury in rats. Food Sci Hum Wellness. 2021;10(4):523-535.
Crossref - Goud BJ, Dwarakanath V, Chikka BK. Streptozotocin-a diabetogenic agent in animal models. Int J Pharm Pharm Res. 2015;3(1):253-269.
- Jamshidi-Kia F, Wibowo JP, Elachouri M, et al. Battle between plants as antioxidants with free radicals in human body. J HerbMed Pharmacol. 2020;9(3):191-199.
Crossref - Stilinovic N, Capo I, Vukmirovic S, et al. Chemical composition, nutritional profile and in vivo antioxidant properties of the cultivated mushroom Coprinus comatus: Comprimus comatus properties. R Soc Open Sci. 2020;7(9):1-18.
Crossref - Duez H, Cariou B, Staels B. DPP-4 inhibitors in the treatment of type 2 diabetes. Biochem Pharmacol. 2012;83(7):823-832.
Crossref - Ovlund T, Pacini G, Ahren B. Impact of Incretin Hormone Receptors on Insulin-Independent Glucose Disposal in Model Experiments in Mice. Front Endocrinol (Lausanne). 2021;12:680153.
Crossref - Kaneto H, Kimura T, Shimoda M, et al. Molecular Mechanism of Pancreatic β-Cell Failure in Type 2 Diabetes Mellitus. Biomedicines. 2022;10(4):818.
Crossref
© The Author(s) 2022. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.