ISSN: 0973-7510
E-ISSN: 2581-690X
To generate oregano essential oil, the leaves and flowering tops of the Origanum vulgare plant go through the process known as steam distillation. This essential oil is known for its antibacterial activity. Bacterial biofilms are microbial communities attached to inert surfaces or tissues and encapsulated in complex matrices. Planktonic bacteria reversibly attach to surfaces, form microcolonies, and generate polymeric matrices around biofilms. Bacteria in biofilms provide bacteria with a safer way to reproduce and survive. This research tests the antibacterial activity and effect on the biofilm formation of Oregano essential oil. The antibacterial activity and effect on biofilm formation were tested against five bacterial strains, including Escherichia coli ATCC 14169, Escherichia coli ATCC 25922, Staphylococcus aureus NCTC 12393, Staphylococcus aureus ATCC 25923, and Staphylococcus aureus ATCC 6538. The concentrations of oil that were used in this research were 100%(v/v), 75%(v/v), 50%(v/v), and 25%(v/v). The best antibacterial effect was achieved against Staphylococcus aureus NCTC 12393 at 25%(v/v) of oil concentration. While performing the experiment, a variety of oregano oil concentrations had significant results for further tests to be performed.
Antibacterial Activity, Biofilm-forming Capacity, Oregano Essential Oil
Essential oil (EO) is an essence derived from plants with a characteristic odor. Volatile compounds, which are found in plants, form a complex mixture that makes an essential oil. These volatile compounds can be isolated from different parts of a plant, using distillation and compression. Terpenes are the main compounds found in essential oil, but others such as aldehydes, alcohols, and esters can also be found in smaller amounts.1
Oregano is used in diet as a spice and as an aromatic oil. People in the Mediterranean region have used it for centuries to treat many illnesses such as skin aches, muscle aches, asthma, cramps, diarrhea, indigestion, and colds, and to improve their overall health. It may help in fighting against bacteria, reduction of inflammation, regulating blood sugar and lipids, and fighting against cancer. Toxic substances and environmental stress can cause the formation of free radicals which are harmful to the human body. Oregano and other herbs provide antioxidants that can help the body eliminate free radicals. Oxidative stress may be caused by the accumulation of these free radicals. Oxidative stress can cause cell damage, and it can also be the reason for developing various diseases such as cancer and diabetes.2
Planktonic growth and biofilm
In the environment, multicellular communities of bacteria known as biofilms are most frequently seen in close relation to surfaces and interfaces. Bacterial cells can survive as single cells, but they typically coexist in multicellular communities, making them comparable to many other forms of living cells.3 Biofilms are held together by a self-produced extracellular matrix and the formation and maintenance of extracellular components that are crucial for the development of organized multicellular communities. While the ability to produce extracellular matrices seems to be a common trait of multicellular communities, the processes used to generate these matrices exhibit variations.4 Bacteria in a planktonic growth are both untethered to surfaces and each other. Because of that, at low cell densities in shear flow, they can move independently from each other.5 Typically, in antimicrobial and immunological tests, bacteria are grown using planktonic inoculums; however, in nature, the natural mode of bacterial growth is in the form of biofilm. Differing from biofilm, planktonic growth of bacteria results in colonized new niches which have less chance of survival. Planktonic growth is also an initial stage of bacterial growth which can then, by further developing, result in a biofilm.6,7
Biofilm formation occurs in several stages. Planktonic bacteria reversibly attach to surfaces, form microcolonies, and generate polymeric matrices around biofilms. Bacteria in biofilms provide themselves a safer way to reproduce and survive. This is partly explained by the increased tolerance of biofilms to antibiotics, disinfectants, and the immune system.6
The development of bacterial biofilms is a multi-step process that begins with the cell’s temporary attachment to surfaces, which is facilitated by hydrophobicity and intermolecular forces, and then advances to the production of extracellular polymeric substances (EPS), which allows the cells to adhere to surfaces permanently. The creation of biofilms specifically involves five stages: reversible attachment, irreversible attachment, EPS synthesis, biofilm maturity, and dispersal/detachment.8 Proteins, DNA, and EPS make up around 90% of the structure of the biofilm. EPS gives the stability of the cell, controls surface adherence, and acts as a scaffold for the attachment of cells, enzymes, and antibiotics.9
The capacity of biofilms to resist antimicrobial treatments via multiple pathways, as opposed to a single pathway, is referred to as multifactorial biofilm resistance. The multifactorial resistance of biofilms is caused by many characteristics, including the EPS matrix, slow growth rate, diverse population, gene expression, and quorum sensing. It can be difficult to control biofilms by using standard antimicrobial treatments because of the multifactorial nature of biofilm resistance. As a result, novel approaches are being developed that specifically target the processes behind biofilm resistance.10
The reason why biofilms have become of great importance for public health is that microorganisms, which are capable of forming a biofilm, are less susceptible to antimicrobial agents. There are two different types of susceptibility, which are intrinsic, being a natural result of biofilm formation, or acquired, which suggest that it was gained by transferring extrachromosomal elements to susceptible organisms in biofilms.11
In response to environmental stressors such as ultraviolet (UV) radiation, desiccation, nutritional deprivation, extreme pH and temperature, high salt concentrations, high pressure, and antimicrobial agents, bacteria create biofilms.8 In the food industry, harmful bacteria can create biofilms inside processing equipment, causing food to deteriorate.12 It has been demonstrated that biofilms can continue to grow on the surfaces of medical equipment and the tissues of patients in hospitals, leading to lingering infections.13 There was a long focus on the prevention and management of hazardous biofilms due to the substantial impact that these biofilms have on human health and other aspects.14
In contrast, the modification of adhesion surfaces utilizing quorum sensing (QS) signals and environmental factors can promote the creation of advantageous biofilms. To facilitate cell-to-cell communication within the bacterial population, bacteria use QS to produce and release first messengers such as chemical signals, which are named autoinducers (AIs).15 Universal AIs may be used by both gram-negative and gram-positive bacteria.
Several processes, including signal synthesis, signal distribution, signal receptors, signal recognition, gene expression, and signaling response, are necessary for bacterial QS. Quorum-sensing inhibitors (QSIs) that obstruct these procedures may eventually prevent bacterial QS and biofilm development.16
Healthcare facilities, medicine, industrial membrane bioreactors, agricultural production, and aquaculture all benefit from the quorum-quenching strategy, which uses quorum-quenching enzymes to deactivate quorum-sensing signals.17 Numerous bacterial species have been shown to have the quorum-quenching enzymes lactonase, acylase, oxidoreductase, and paraoxonase.18 The inactivation of acyl-homoserine lactone molecules is the widely recognized mode of action of QQs. Another method is the suppression of Lux operon protein enzymatic activity by QSIs like N-octanoylL-HSL (C8-HSL), which hinders the creation of signal molecules (like AHLs), for example.19
Biofilm formation is important for cell-cell adhesion and attachment of cells to different surfaces. Pilli and fimbriae are cell appendages and other crucial proteinaceous structures for biofilm formation.20
The rapid development of bacterial resistance led to the problem that treatment options became limited and bacteria that were previously susceptible to a specific antibacterial agent, became resistant. The unnecessary usage of antibacterial agents had to be reduced to prevent the further spreading of antibiotic resistance. Further research was based on developing new antibacterial drugs which can inhibit the growth and spreading of pathogenic bacteria.21 Antibacterial agents are used to destroy bacteria or inhibit their growth and spreading. They can be classified into natural, semi-synthetic, or synthetic forms.22,23
Sample Collection
Oregano essential oil that has been used was from MARNYS® Martinez Nieto S.A., Cartagena, Spain Ingredients: Origanum vulgare leaf oil.
Cultivation of strains
Staphylococcus aureus NCTC 12393, Staphylococcus aureus ATCC 25923, Staphylococcus aureus ATCC 6538, Escherichia coli ATCC 14169, Escherichia coli ATCC 25922 were grown on trypticase soy broth (TSB) broth and Mueller Hinton (MH) agar, both solid and liquid media. The turbidity of the bacterial density is adjusted to 0.5 McFarland standard during an overnight incubation at 37°C; this value was utilized for the following experiments.20
Microbroth Dilution Method
The oregano essential oil was used to evaluate its effect for Staphylococcus aureus NCTC 12393, Staphylococcus aureus ATCC 25923, Staphylococcus aureus ATCC 6538, Escherichia coli ATCC 14169, Escherichia coli ATCC 25922. All equipment, including a multichannel pipette, tips, gloves, and face mask, need to be sterile or cleaned with 70% ethanol to prevent any contamination while performing the dilutions. In a 96-well plate, the first column was used as a negative control, where only 130µL of TSB was added. The second column served as positive control where 100µL of TSB and 30µL of a specific bacteria were added. The rest of the plate, from the third column to the end, were dilutions that contained 100µLof TSB, 30µL of bacteria, and different dilutions of oregano essential oil. Dilutions went as follows: 100%, 50%, 25%, 12.5%, 6.25%, 3.12%, 1.56%, 0.78%, 0.39%, and 0.195%. The plates were incubated for 24 hours at 37°C in a CO2 incubator to get valid results. After 24h, the results of planktonic growth were read using an ELISA plate reader on a 595nm wavelength.23,24
Biofilm Forming Capacity
After reading on the ELISA plate reader, the plates were cleaned with distilled water, and they were stained for 10 minutes with 0.1% crystal violet solution. The plates are treated with 96% ethanol for 10 minutes after washing. The ELISA plate reader (Biochrom EZ Read 400) reads the absorbance values at a wavelength of 595 nm. From the obtained measurements of the absorbance values, the mean values are evaluated for all bacterial strains. The results of the biofilm formation are classified into four categories: non-adherent (NA), weakly adherent (W), moderately adherent (M), and strongly adherent (S). To classify each result, the optical density cut-off value (ODc) is calculated as three standard deviations of the negative control above the average OD of the negative control. The biofilm categories are determined as the following: OD ≤ ODc: non-adherent, ODc < OD ≤ 2 x ODc: weakly adherent, 2 x ODc < OD ≤ 4 x ODc: moderately adherent, and 4xODc < OD: strongly adherent.25
Determination of antibacterial activity
The antibacterial activity of the oregano essential oil was tested against five different bacterial strains, including Staphylococcus aureus NCTC 12393, Staphylococcus aureus ATCC 25923, Staphylococcus aureus ATCC 6538, Escherichia coli ATCC 14169, Escherichia coli ATCC 25922. The samples were tested against mentioned strains in triplicates. Sterile cotton swabs were used to inoculate bacteria on MH agar plates. Four wells were made on each plate for four different oil concentrations 25% (v/v), 50% (v/v), 75% (v/v), and, 100% (v/v). Using a micropipette, each oil concentration was added to its specific well on each plate. Plates were incubated for 24h at 37°C in a CO2 incubator. Lastly, after 24h of incubation, zones of inhibition were measured on each plate.26
Table 1. shows the antibacterial activity of 100%(v/v), 75%(v/v), 50%(v/v), and 25% (v/v) of the concentration of oregano essential oil against Escherichia coli ATCC 14169, Escherichia coli ATCC 25922, Staphylococcus aureus NCTC 12393, Staphylococcus aureus ATCC 25923, and Staphylococcus aureus ATCC 6538. Oregano essential oil showed antibacterial activity against E.coli ATCC 14169 with zones of inhibition of 24mm for 100% (v/v) oil concentration, 23mm for 75% (v/v), 21mm for 50% (v/v), and 19mm for 25% (v/v). The antibacterial activity was also observed against E.coli ATCC 25922 where zones of inhibition were 32mm for 100% (v/v) oil concentration, 33mm for 75% (v/v), 28mm for 50% (v/v), and 29mm for 25% (v/v). The Oregano essential oil also showed antibacterial activity against S.aureus NCTC 12393 with zones of inhibition of 31mm for 100% (v/v) of oil concentration, 29mm for 75% (v/v), 28mm for 50% (v/v), and 34mm for 25% (v/v). S. aureus ATCC 25923 also showed susceptibility to Oregano essential oil with zones of inhibition of 25mm at 100% (v/v) of oil concentration, 22mm at 75% (v/v), 18mm at 50% (v/v), and 18mm at 25% (v/v). Lastly, antibacterial activity was also observed against S.aureus ATCC 6538 with zones of inhibition of 28mm for 100% (v/v) of oil concentration, 25mm for 75% (v/v),21mm for 50% (v/v), and 23mm for 25% (v/v). Zones of inhibition are measured and represented in millimeters.
Table (1):
Antibacterial activity (mm) of oregano essential oil.
| Oil Concentration | E.coli ATCC 14169 | E.coli ATCC 25922 | S.aureus NCTC 12393 | S.aureus ATCC 25923 | S.aureus ATCC 6538 | |
|---|---|---|---|---|---|---|
| 100% | 24 mm | 32 mm | 31 mm | 25 mm | 18 mm | Zones of inhibition |
| 75% | 23 mm | 33 mm | 29 mm | 28 mm | 21 mm | |
| 50% | 21 mm | 28 mm | 28 mm | 22 mm | 18 mm | |
| 25% | 19 mm | 29 mm | 34 mm | 25 mm | 23 mm |
Table (2):
Obtained average and standard deviation values.
| Bacterial strain |
PC | Subinhibitory concentrations of oregano essential oil | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 100% | 50% | 25% | 12.50% | 6.25% | 3.12% | 1.56% | 0.78% | 0.39% | 0.19% | ||
| EC2 | 0.383 ±
0.008 |
3.044 ±
0.065 |
/ | 3.165 ±
0.287 |
1.815 ±
0.181 |
1.625 ±
0.159 |
1.349 ±
0.315 |
0.625 ±
0.129 |
0.372 ±
0.089 |
0.208 ±
0.063 |
0.164 ±
0.046 |
| EC1 | 0.324 ±
0.006 |
/ | / | 2.713 ± / | 2.22 ±
0.429 |
2.39 ±
0.238 |
2.324 ±
0.286 |
1.274 ±
0.268 |
0.561 ±
0.169 |
0.231 ±
0.063 |
0.168 ±
0.026 |
| SA1 | 0.175 ±
0.035 |
3.191 ±
0.171 |
/ | 3.169 ± / | 2.702 ±
0.546 |
2.324 ±
0.227 |
2.135 ±
0.610 |
1.171 ±
0.445 |
0.683 ±
0.173 |
0.309 ±
0.092 |
0.167 ±
0.069 |
| SA2 | 0.595 ±
0.062 |
3.338 ±
0.034 |
/ | 2.960 ±
0.328 |
2.296 ±
0.267 |
2.052 ±
0.211 |
1.269 ±
0.424 |
0.728 ±
0.256 |
0.485 ±
0.122 |
0.310 ±
0.044 |
0.257 ±
0.072 |
| SA6 | 1.150 ± 0.027 |
/ | / | / | 2.742 ±
0.1441 |
2.157 ±
0.336 |
2.328 ±
0.392 |
0.920 ±
0.139 |
0.576 ±
0.096 |
0.328 ±
0.074 |
0.202 ±
0.01 |
| PC – Positive Control; Bacterial strains: EC1 – E.coli ATCC 14169, EC2 – E.coli ATCC 25922, SA1 – S.aureus NCTC 12393, SA2 – S.aureus ATCC 25923, SA6 – S.aureus ATCC 6538 | |||||||||||
By treating five bacterial strains with 100% (v/v) concentration of oregano essential oil, their planktonic growth and biofilm-forming capacity were measured. Table 2 shows the obtained average and standard deviation values that were obtained from the effects of oregano essential oil on the biofilm formation of pathogenic bacteria. The positive control represents only the pathogenic bacteria that has been applied in the 96-well plate, while the further columns are the concentrations of oregano oil that were applied on the pathogenic bacteria. The 25% concentration of oregano oil has showed the minimum inhibitory concentration (MIC) has been showcased in the 25% oregano essential oil concentration for all pathogenic bacteria, except for SA6. For SA6, it has been noted at the concentration of 12.50%.
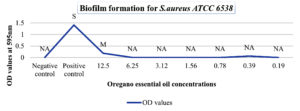
Positive control of Staphylococcus aureus ATCC 6538 showed strong biofilm-forming capacity. Concentrations 100% (v/v), 50% (v/v), and 25% (v/v) could not be measured at 595nm wavelength. Biofilm-forming capacity for the 12.5% (v/v) concentration was moderate, while for the concentrations from 6.25% (v/v) to 0.19% (v/v) oil concentration was non-adherent as shown in Figure 1.
Escherichia coli ATCC 14169when treated with different concentrations of oregano essential oil showed non-adherent biofilm-forming capacity. The positive control showed strong biofilm-forming capacity. Concentrations of 100% (v/v), 50% (v/v), and 25% (v/v)could not be measured at 595nm wavelength. Biofilm-forming capacity remained non-adherent from 12.5% (v/v) to 0.19% (v/v) oil concentration as shown in Figure 2.
The positive control of Staphylococcus aureus NCTC 12393 showed moderate biofilm-forming capacity. The 100% (v/v), 50% (v/v), and 25% (v/v) oregano oil concentrations could not be measured at 595nm wavelength. Biofilm-forming capacity remained non-adherent from 12.5% (v/v) to 0.19% (v/v) oil concentration as shown in Figure 3.
Staphylococcus aureus ATCC 25923 showed similar biofilm-forming capacities when compared to Staphylococcus aureus NCTC 12393. The positive control showed strong biofilm-forming capacity. Concentrations 100% (v/v), 50% (v/v), and 25% (v/v) could not be measured at 595nm wavelength. Biofilm-forming capacity remained non-adherent from 12.5% (v/v) to 0.19% (v/v) oil concentration as shown in Figure 4.
Escherichia coli ATCC 25922 showed biofilm-forming capacity which was non-adherent from the 12.5% (v/v) to the 0,19% (v/v) oregano oil concentration as shown in Figure 5. The positive control showed weak biofilm-forming capacity. Concentrations 100% (v/v), 50% (v/v), and 25% (v/v) could not be measured at 595nm wavelength.
Earlier it was mentioned that the growth of the bacteria tested was effectively inhibited by oregano essential oil most likely as a result of the presence of thymol and carvacrol, two substances recognized for their antibacterial characteristics. These substances can damage bacterial cell membranes and other essential parts, which results in bacterial death or growth suppression.27 The antibacterial activity of the tested oregano essential oil was observed against S.aureus ATCC 6538, S.aureus NCTC 12393, S.aureus ATCC 25923, E.coli ATCC 14169, and E.coli ATCC 25922 in this study. The results revealed that in the case of E.coli ATCC 14169, a decrease in oil concentration led to a decrease in the ability to inhibit the growth of this bacteria. On the other hand, for E.coli ATCC 25922, the highest antibacterial activity was observed at 25% (v/v) oil concentration, while the lowest activity was observed at 50% (v/v). The results for S.aureus NCTC 12393 showed that the highest antibacterial activity was at 25% (v/v) oil concentration, surpassing even that at 100% (v/v) concentration. Similar to E.coli ATCC 14169, a gradual decrease in the antibacterial activity of the oil was observed for S.aureus ATCC 25923 due to the decrease in oil concentration. Lastly, as seen in the case of E.coli ATCC 25922, S.aureus ATCC 6538 exhibited an increase in antibacterial activity at 25% (v/v) concentration and the lowest antibacterial activity was at 50% oil concentration.
It has been demonstrated that oregano essential oil may be effective in preventing the formation of biofilms and treating infections caused by biofilms.28,29 When treated with 12.5% (v/v) to 0,19% (v/v) oregano oil concentration, the biofilm-forming capacity of E. coli strains reduced from weak or strong to non-adherent. Also, for S. aureus strains, the biofilm-forming capacity was found to decrease from strong or moderate to non-adherent when exposed to oregano oil concentrations ranging from 12.5% (v/v) down to 0.19% (v/v). In a similar study30 was performed using oregano oil in different concentrations when performing MIC. In this study, the bacterial concentration that was added was smaller than the concentration that we used, and instead of TSB, dimethyl sulfoxide (DMSO) was used as a control, in which the oregano oil was prepared. 40mg/ml was diluted. Antibiofilm activity, the bacteria in comparison were incubated in TSB that contained 0.1% glucose, and the procedure was performed on the 96 well plates. Instead of analyzing the results using crystal violet, the plates were washed in phosphate buffer saline (PBS), and the viability of the bacteria was observed using the Alamar Blue assay. Both results in the paper and this research have shown positive effects of oregano oil, where there were no signs of severe resistance.
Another research study was done on more S. aureus strains where the antibiofilm analysis was performed, and the oregano essential oil had positive effects on the pathogenic bacteria, mildly preventing biofilm formation. The procedure was similar to the one performed in our experiment. In this experiment on the 96-well plate, the initial bacterial that was added was 10μL into 90μL of TSB, and TSB contained additionally 0.25% of glucose. The biofilm treatment was similar during the final reading and the staining. By adding 99% methanol to the wells and letting them sit at room temperature for 15 minutes, adherent bacteria were eliminated. After that, the wells were drained and allowed to dry. 200 L of 2% crystal violet dye was added to the biofilm to stain it for 5 minutes. The experimental method was similar to the method that we performed, making sure that all observations could be read successfully. In this investigation, oregano oil had a potent antibacterial action on strains of Staphylococci, including S. aureus, and it was showcased in the experiment that Staphylococci and E. coli produced fewer biofilms when oregano EO was present in subinhibitory amounts.31
The findings of this study show that oregano essential oil has the potential to be a powerful antibacterial agent against several strains of Escherichia coli and Staphylococcus aureus. At a concentration of 25%, the oil exhibited substantial antibacterial action, and at concentrations as low as 0.19%, it was able to stop the growth of biofilm. These results are in line with those of earlier research that examined the antibacterial and antibiofilm capabilities of oregano oil and found similar encouraging outcomes. Overall, our findings imply that oregano oil may represent a promising natural substitute for chemical antimicrobials in the treatment of bacterial infections and the avoidance of biofilm development. To fully explore this essential oil’s potential in clinical applications, more study is required and the analysis method is to be repeated in a variety of laboratories.
ACKNOWLEDGMENTS
The authors would like to thank the Department of Genetics and Bioengineering at International Burch University for providing support to complete this study.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
AUTHORS’ CONTRIBUTION
All authors listed have made substantial, direct, and intellectual contributions to the work, and approved it for publication.
FUNDING
None.
DATA AVAILABILITY
All datasets generated or analyzed during this study are included in the manuscript.
ETHICS STATEMENT
Not applicable.
- Leyva-López N, Gutiérrez-Grijalva EP, Vazquez-Olivo G, Heredia JB. Essential oils of oregano: Biological activity beyond their antimicrobial properties. Molecules. 2017;22(6):989.
- Gutiérrez-Grijalva EP, Picos-Salas MA, Leyva-López N, Criollo-Mendoza MS, Vazquez-Olivo G, Heredia JB. Flavonoids and Phenolic Acids from Oregano: Occurrence, Biological Activity and Health Benefits. Plants. 2018;7(1):2.
Crossref - Pecoraro C, Carbone D, Deng D, Cascioferro SM, Diana P, Giovannetti E. Biofilm Formation as Valuable Target to Fight against Severe Chronic Infections. Current Medicinal Chemistry. 2022;29(25):4307-4310.
Crossref - Branda SS, Vik Å, Friedman L, Kolter R. Biofilms: the matrix revisited. Trends in Microbiology. 2005;13(1):20-26.
Crossref - Stepanović S, Vuković D, Hola V, et al. Quantification of biofilm in microtiter plates: overview of testing conditions and practical recommendations for assessment of biofilm production by staphylococci. Apmis. 2007;115(8):891-9.
- Hernández-Jiménez E, Del Campo R, Toledano V, et al. Biofilm vs. planktonic bacterial mode of growth: which do human macrophages prefer?. Biochemical and biophysical research communications. 2013;441(4):947-52.
- Bassi T. Ventilation-associated brain injury. summit.sfu.ca. Published December 10, 2021. Accessed April 14, 2023. https://summit.sfu.ca/item/35553
- Hall-Stoodley L, Costerton JW, Stoodley P. Bacterial biofilms: from the Natural environment to infectious diseases. Nature Reviews Microbiology. 2004;2(2):95-108.
Crossref - Algburi A, Comito N, Kashtanov D, Dicks LMT, Chikindas ML. Control of Biofilm Formation: Antibiotics and Beyond. Pettinari MJ, ed. Applied and Environmental Microbiology. 2017;83(3).
Crossref - Flemming HC, Wingender J. The biofilm matrix. Nature reviews Microbiology. 2010;8(9):623-633.
Crossref - Chmielewski RA, Frank JF. Biofilm formation and control in food processing facilities. Comprehensive reviews in food science and food safety. 2003;2(1):22-32.
- Galie S, García-Gutiérrez C, Miguélez EM, Villar CJ, Lombó F. Biofilms in the food industry: health aspects and control methods. Frontiers in microbiology. 2018;9:898.
- Percival SL, Suleman L, Vuotto C, Donelli G. Healthcare-associated infections, medical devices and biofilms: risk, tolerance and control. Journal of Medical Microbiology. 2015;64(Pt_4):323-334.
Crossref - Muhammad MH, Idris AL, Fan X, Guo Y, Yu Y, Jin X, Qiu J, Guan X, Huang T. Beyond risk: bacterial biofilms and their regulating approaches. Frontiers in microbiology. 2020;11:928.
- Papenfort K, Bassler BL. Quorum sensing signal–response systems in Gram-negative bacteria. Nature Reviews Microbiology. 2016;14(9):576-588.
Crossref - Rémy B, Mion S, Plener L, Elias M, Chabrière E, Daudé D. Interference in bacterial quorum sensing: a biopharmaceutical perspective. Frontiers in pharmacology. 2018;9:203.
- Fong J, Zhang C, Yang R, et al. Combination therapy strategy of quorum quenching enzyme and quorum sensing inhibitor in suppressing multiple quorum sensing pathways of P. aeruginosa. Scientific reports. 2018;8(1):1-1.
- Chen F, Gao Y, Chen X, Yu Z, Li X. Quorum Quenching Enzymes and Their Application in Degrading Signal Molecules to Block Quorum Sensing-Dependent Infection. International Journal of Molecular Sciences. 2013;14(9):17477-17500.
Crossref - Sadekuzzaman M, Yang S, Mizan MFR, Ha SD. Current and Recent Advanced Strategies for Combating Biofilms. Comprehensive Reviews in Food Science and Food Safety. 2015;14(4):491-509.
Crossref - Lopez D, Vlamakis H, Kolter R. Biofilms. Cold Spring Harbor Perspectives in Biology. 2010;2(7):a000398-a000398.
Crossref - Barzic AI, Ioan S. Antibacterial Drugs — from Basic Concepts to Complex Therapeutic Mechanisms of Polymer Systems. IntechOpen; 2015. Accessed October 10, 2021. https://www.intechopen.com/chapters/48837.
- Biemer JJ. Antimicrobial susceptibility testing by the Kirby-Bauer disc diffusion method. Annals of Clinical & Laboratory Science. 1973;3(2):135-40.
- O’Toole GA. Microtiter Dish Biofilm Formation Assay. Journal of Visualized Experiments. 2011;30(47):2437.
Crossref - Soliman SSM, Semreen MH, El-Keblawy AA, Abdullah A, Uppuluri P, Ibrahim AS. Assessment of herbal drugs for promising anti-Candida activity. BMC Complementary and Alternative Medicine. 2017;17(1).
Crossref - Avdić M, Džuzić N, Hasanić O, et al. Development of a novel biofilm classification tool and comparative analysis of result interpretation methodologies for the evaluation of biofilm forming capacity of bacteria using tissue culture plate method. Med Glas (Zenica). 2019;16(1):13-21.
Crossref - Klančnik A, Piskernik S, Jeršek B, Možina SS. Evaluation of diffusion and dilution methods to determine the antibacterial activity of plant extracts. Journal of microbiological methods. 2010;81(2):121-6.
- Faleiro ML. The mode of antibacterial action of essential oils. Science against microbial pathogens: communicating current research and technological advances. 2011;2:1143-56.
- Nostro A, Procopio F, Pizzimenti FC, et al. Effects of oregano, carvacrol and thymol on Staphylococcus aureus and Staphylococcus epidermidis biofilms. Journal of Medical Microbiology. 2007;56(4):519-523.
Crossref - Turek C, Stintzing FC. Stability of Essential Oils: A Review. Comprehensive Reviews in Food Science and Food Safety. 2013;12(1):40-53.
Crossref - Lu M, Dai T, Murray CK, Wu MX. Bactericidal Property of Oregano Oil Against Multidrug-Resistant Clinical Isolates. Frontiers in Microbiology. 2018;9.
Crossref - Bilge Oral N, Sezer Ç, Baser KHC, et al. Effect of Oregano Essential Oil on Biofilms Formed By Staphylococci and Escherichia coli. Journal of the Faculty of Veterinary Medicine. 2009.
Crossref
© The Author(s) 2023. Open Access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License which permits unrestricted use, sharing, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.